Submitted:
27 April 2026
Posted:
28 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Methods
2.1. Patients and Samples
2.2. Assay of SARS-CoV-2 Spike Specific IgG, IgA and SIgA Antibodies by ELISA
2.3. Avidity Assay for IgG and IgA Antibodies
2.4. CD4 T Cell Exhaustion / Senescence Using Flow Cytometry
2.5. Cytokine Assays
2.6. Statistical Analysis
3. Results
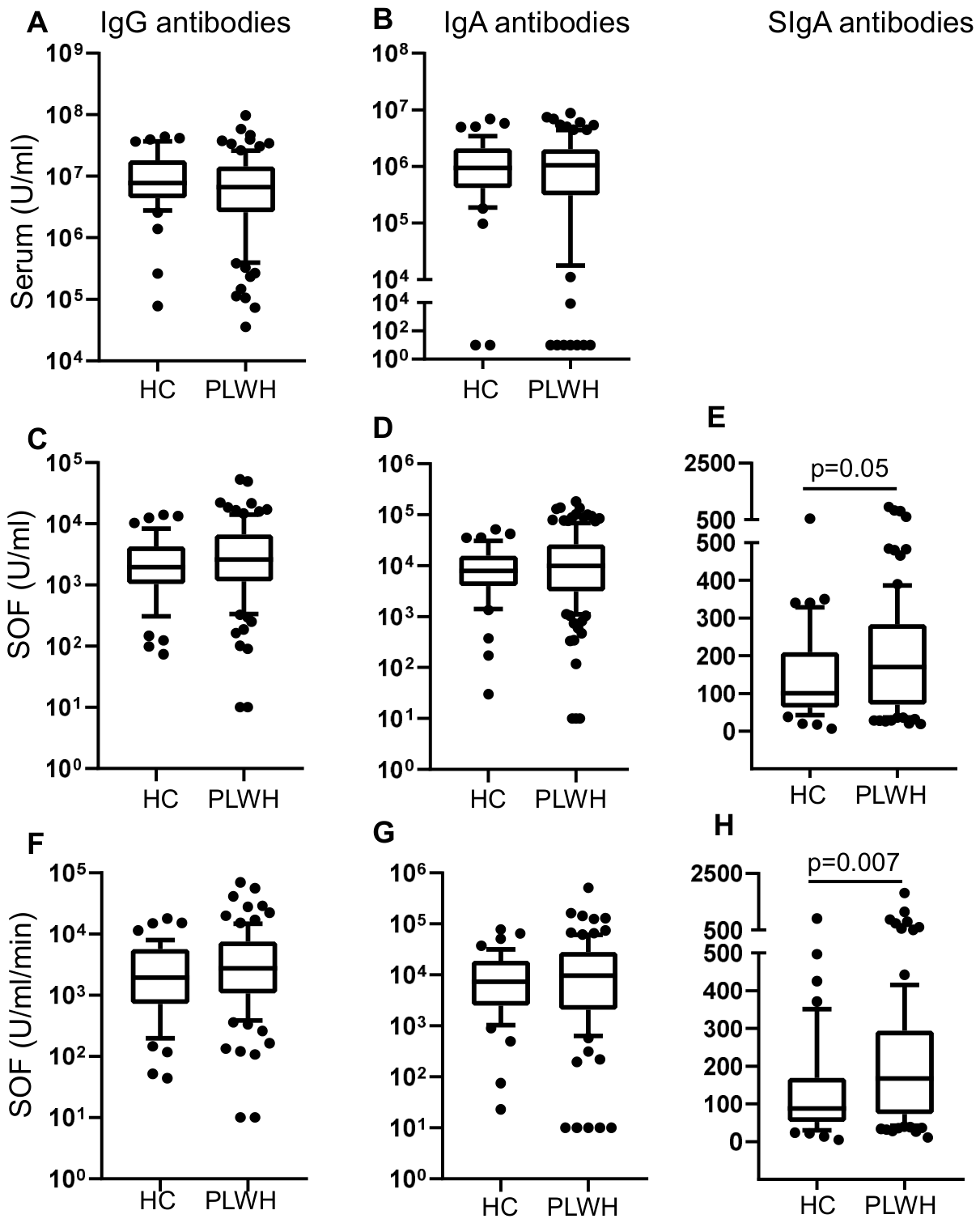
3.1. PLWH Elicited Higher Anti-SARS-CoV-2 Spike SIgA Antibodies
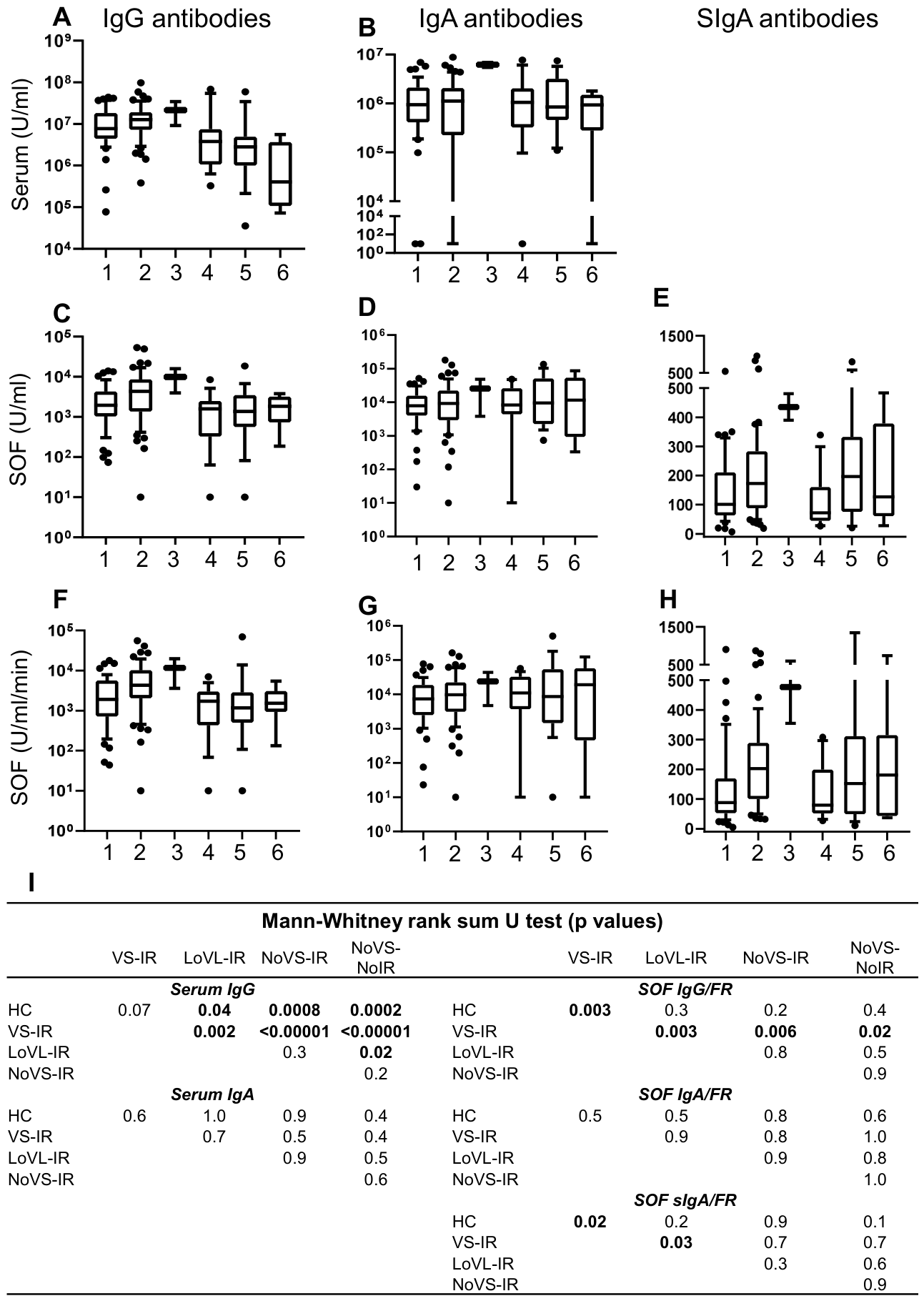
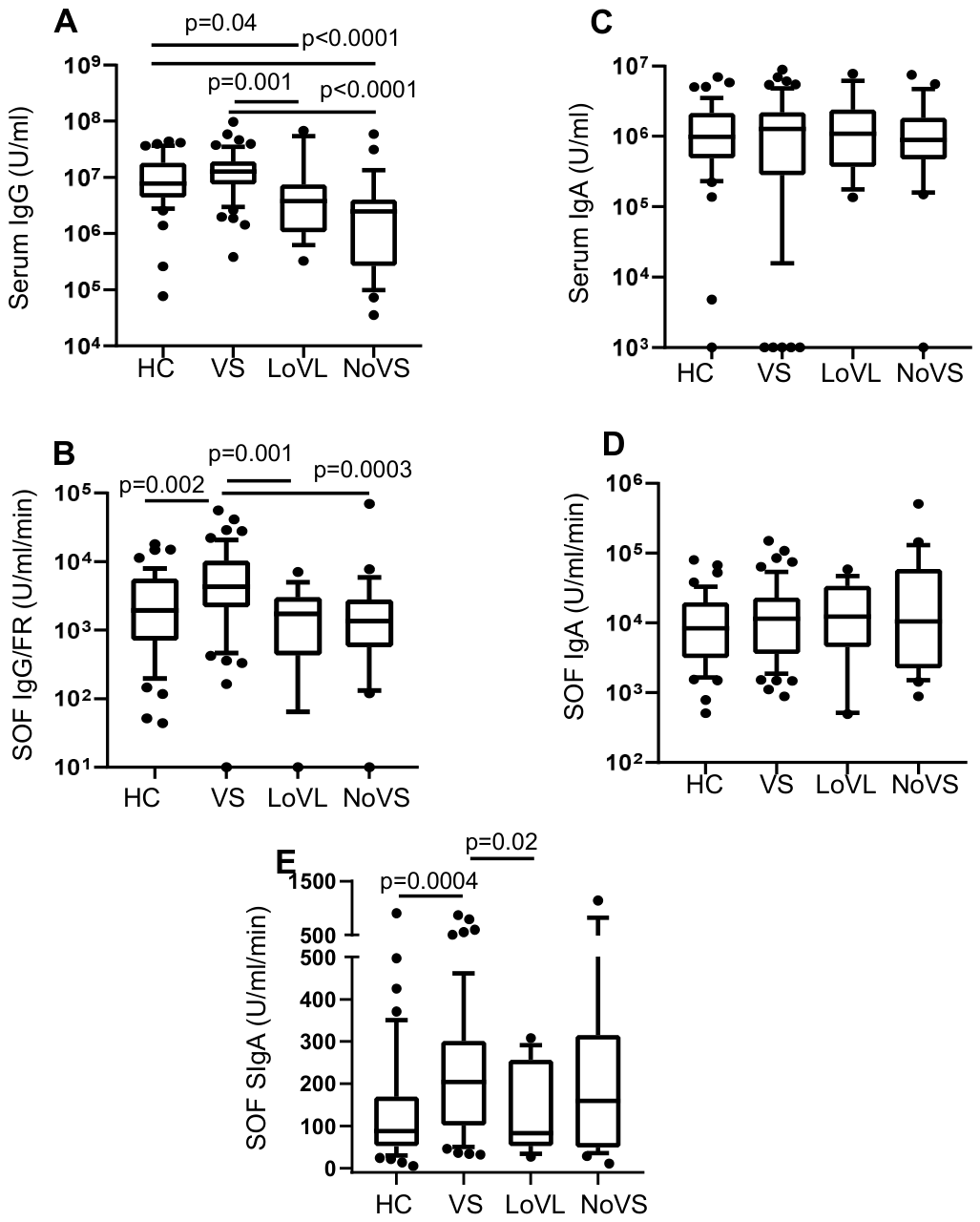
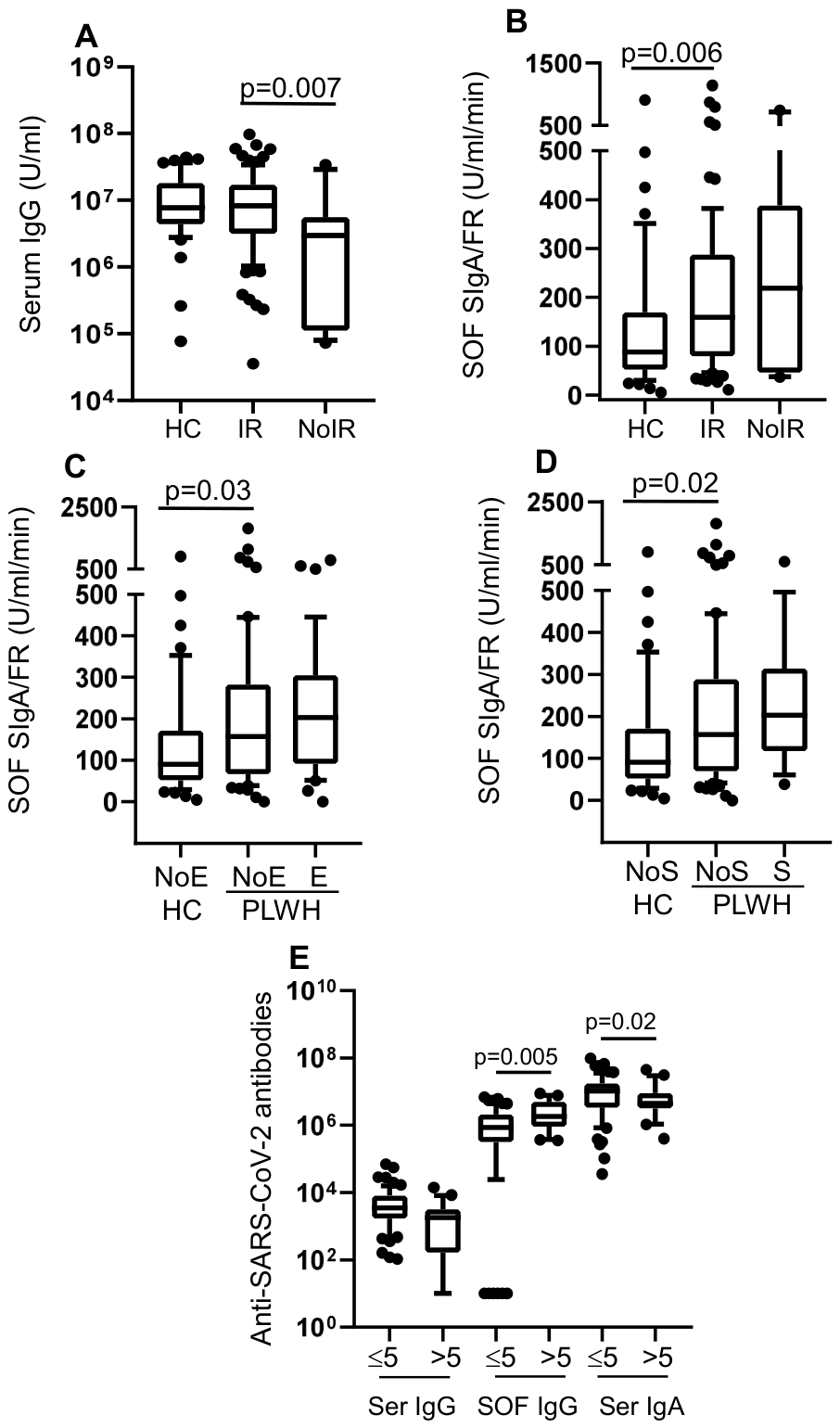
3.2. Viral Suppression (VS) in PLWH Has a Greater Impact on Antibody Production than Immune Reconstitution (IR)
3.3. Factors Possibly Influencing SARS-CoV-2 Specific Antibody Production
3.4. IgA and SIgA Antibodies Are Elicited in the Oral Mucosa of PLWH upon Vaccination
3.5. Differential IgG Avidities and Similar IgA Avidities
3.6. Higher Expression of Innate Immunity Cytokines in the Oral Mucosa of PLWH
4. Discussion
Author Contributions
Funding
References
- Muthukrishnan, J.; Vardhan, V.; Mangalesh, S.; Koley, M.; Shankar, S.; Yadav, A.K.; Khera, A. Vaccination status and COVID-19 related mortality: a hospital based cross sectional study. Med. J. Armed Forces 2021, 77(S2), S278–S282. [Google Scholar] [CrossRef]
- Kannian, P.; Eniya, M.L.; Mahanathi, P.; Gracemary, A.; Kumarasamy, N.; Challacombe, S.J. Impact of COVID-19 on mucosal immunity and antibody responses in COVID-19 vaccinees. Vaccines 2025, 13(9), 967. [Google Scholar] [CrossRef]
- Inan, A.; Barkay, O.; Karapinar, A.; Yilmaz-Karadag, F.; Aktas, S.; Bolukcu, S. COVID-19 incidence and factors influencing infection risk among people living with HIV in Turkiye: is current issue the vaccine hesitancy – opposition? Can. J. Infect. Dis. Med. Microbiol. 2025, 6767853. [Google Scholar] [CrossRef]
- Inzaule, S.; Silva, R.; Ford, N.; Thwin, S.S.; Wassila, J.; Zumla, A.; Doherty, M.; Diaz, J.; Bertagnolio, S. Comparative analysis of COVID-19 in-hospital mortality in people living with HIV during SARS-CoV-1 pre-delta, delta and Omicron waves: data from the WHO global clinical platform. AIDS 2025, 39(14), 2114–2122. [Google Scholar] [CrossRef]
- Siegrist, C.A. Vaccine Immunology. In Plotkin’s Vaccines; 2018; pp. 16–34.e7. [Google Scholar] [CrossRef]
- Fichtenbaum, C.J.; Malvestutto, C.D.; Watanabe, M.G.; Smith, E.D.; Ribaudo, H.J.; McCallum, S.; Fitch, K.V.; Currier, J.S.; Diggs, M.R.; Chu, S.M.; et al. Effects of antiretrovirals on major adverse cardiovascular events in the REPRIEVE trial: a longitudinal cohort analysis. Lancet HIV. 2025, 12(7), e496–e505. [Google Scholar] [CrossRef] [PubMed]
- Jaimsakul, A.; Rupasinghe, D.; Woolley, I.; Choi, J.Y.; Templeton, D.J.; Widhani, A.; Petoumenos, K.; Tanuma, J. Treat Asia HIV Observational Database (TAHOD) and Australian HIV Observational Database (AHOD) of IeDEA Asia-Pacific. HIV treatment outcomes after 10 years on ART in the TREAT Asia observational database and Australian HIV observational database. J. Acquir Immune Defic. Syndr. 2024, 97(5), 460–470. [Google Scholar] [CrossRef] [PubMed]
- Vangelov, D.; Emilova, R.; Todorova, Y.; Yancheva, N.; Dimitrova, R.; Grigorova, L.; Alexiev, I.; Nikolova, M. T-lymphocyte phenotypic and mitochondrial parameters as markers of incomplete immune restoration in people living with HIV+ on long-term cART. Biomedicines 2025, 13(8), 1839. [Google Scholar] [CrossRef]
- Watanabe, M.; Jergovic, M.; Davidson, L.; La Fleur, B.J.; Castaneda, Y.; Martinez, C.; Smithey, M.J.; Stowe, B.J.; Haddad, E.K.; Nikolich-Zugich, J. Inflammatory and immune markers in HIV-infected older adults on long-term antiretroviral therapy: persistent elevation of sCD14 and of proinflammatory effector memory T cells. Aging Cell. 2022, 21(9), e13681. [Google Scholar] [CrossRef]
- Zhang, W.; King, D.; Zhang, X.; Hu, L.; Nian, Y.; Shen, Z. T cell aging and exhaustion: mechanisms and clinical implications. Clin. Immunol. 2025, 110486. [Google Scholar] [CrossRef] [PubMed]
- Tugizov, S.M. Molecular pathogenesis of human immunodeficiency virus-associated disease of oropharyngeal mucosal epithelium. Biomedicines 2023, 11(5), 1444. [Google Scholar] [CrossRef]
- Hickman, D.; Moutsopoulos, N.M. Viral infection and antiviral immunity in the oral cavity. Nat. Rev. Immunol. 2025, 25(4), 235–249. [Google Scholar] [CrossRef]
- Bellocchio, L.; Dipalma, G.; Inchingolo, A.M.; Inchingolo, A.D.; Ferrante, L.; Vecchio, G.D.; Malcangi, G.; Palermo, A.; Qeadro, A.; Inchingolo, F. COVID-19 on oral health: a new bilateral connection for the pandemic. Biomedicines 2023, 12(1), 60. [Google Scholar] [CrossRef]
- Xu, N.; Shen, Y.; Huang, W.; Nie, J. The current status in terms of vaccination for individuals infected with human immunodeficiency virus. Viruses 2025, 17(2), 171. [Google Scholar] [CrossRef]
- Chapman, A.; Berenbaum, F.; Curigliano, G.; PliakasT; Shiekh, A.; Abduljawad, S. Risk of severe outcomes from COVID-19 in immunocompromised people during the Omicron wave era: a systematic review and meta-analysis. S0149-2918(25)00243-7; Clin Ther. 2025. [Google Scholar] [CrossRef]
- Kausalya, B.; Saravanan, S.; Pallikkuth, S.; Pahwah, R.; Saini, S.R.; Iqbal, S.; Solomon, S.; Murugavel, K.G.; Poongulali, S.; et al. Immune correlates of cardiovascular co-morbidity in HIV infected participants from South India. BMC Immunol. 2022, 23(21), 24. [Google Scholar] [CrossRef]
- Balagopal, A.; Asmuth, D.M.; Yang, W.; Campbell, T.B.; Gupte, N.; Smeaton, L.; Kanyama, C.; Grinsztein, B.; Santos, B.; Supparatpinyo, K.; et al. Pre-cART elevation of CRP and CD4+ T-cell immune activation associated with HIV clinical progression in a multinational case-cohort study. J. Acquir Immune Defic. Syndr. 2015, 70(2), 163–71. [Google Scholar] [CrossRef]
- Spinelli, M.A.; Jones, B.L.H.; Gandhi, M. COVID-19 outcomes and risk factors among people living with HIV. Curr. HIV/AIDS Rep. 2022, 19(5), 425–432. [Google Scholar] [CrossRef] [PubMed]
- Ssentongo, P.; Heilbrunn, E.S.; Ssentongo, A.E.; Advani, S.; Chinchilli, V.M.; Nunez, J.J.; Du, P. Epidemiology and outcomes of COVID-19 in HIV-infected individuals: a systematic review and meta-analysis. Sci. Rep. 2021, 11(1), 6283. [Google Scholar] [CrossRef] [PubMed]
- Lombardi, A.; Butta, G.M.; Donnici, L.; Bozzi, G.; Oggioni, M.; Bono, P.; Matrea, M.; Consonni, D.; Ludovisi, S.; Muscatello, A.; et al. Anti-spike antibodies and neutralizing antibody activity in people living with HIV vaccinated with COVID-19 mRNA-1273 vaccine: a prospective single-centre cohort study. Lancet Reg. Health Eur. 2022, 13, 100287. [Google Scholar] [CrossRef] [PubMed]
- Halenmubieke, S.; Nueraimaiti, A.; Liu, X.; Chang, L.; Ji, H.; Sun, H.; Yam, Y.; Xu, J.; Wang, L. Dynamic monitoring of antibody titres in people living with HIV during Omicron epidemic: comparison between unvaccinated and vaccinated individuals. BMC Infect. Dis. 2025, 25(1), 962. [Google Scholar] [CrossRef]
- Nasab, S.D.S.; Eniya, M.L.; Judith, A.; Clasen, F.; Faith, B.; Poongulali, S.; Gita, J.B.; Ashok, C.; Raghavi, V.; Vedavalli, S.; et al. Detection and consistency of mucosal fluid T lymphocyte phenotypes and their relationship with blood, age and gender. J. Immunol. Methods 2024, 532, 113731. [Google Scholar] [CrossRef]
- Matveev, V.A.; Michelic, E.Z.; Benko, E.; Budylowski, P.; Grocott, S.; Lee, T.; Korosec, C.S.; Colwill, K.; Stephenson, H.; Law, R.; et al. Immunogenicity of COVID-19 vaccines and their effect on HIV reservoir in older people with HIV. iScience 2023, 26, 107915. [Google Scholar] [CrossRef] [PubMed]
- Murata, M.; Matsumoto, Y.; Shimino, N. Comparison of SARS-CoV-2 antibody responses following the second dose of BNT162b2 and mRNA-1273 vaccines in people living with HIV-1. Vaccine 2025, 62, 127457. [Google Scholar] [CrossRef] [PubMed]
- Lahdentausta, L.; Kivimaki, A.; Oksanen, L.; Tallgren, M.; Oksanen, S.; Sanmark, E.; Salminen, A.; Geneid, A.; Sairanen, M.; Paju, S.; et al. Blood and saliva SARS-CoV-2 antibody levels in self-collected dried spot samples. Med. Microbiol. Immunol. 2022, 211, 173–183. [Google Scholar] [CrossRef] [PubMed]
- Cerutti, A. The regulation of IgA class switching. Nat. Rev. Immunol. 2008, 8, 421–434. [Google Scholar] [CrossRef]
- Twigg, H.L., 3rd; Spain, B.A.; Soliman, D.M.; Bowen, L.K.; Heidler, K.M.; Wilkes, D.S. Impaired IgG production in the lungs of HIV infected individuals. Cell. Immunol. 1996, 170(1), 127–133. [Google Scholar] [CrossRef]
- Cognasse, F.; Beniguel, L.; El Habib, R.; Sabido, O.; Chavarin, P.; Genin, C.; Garraud, O. HIV-gp160 modulates differentially the production in vitro of IgG, IgA and cytokines by blood and tonsil B lymphocytes from HIV-negative individuals. Clin. Exp. Immunol. 2003, 132(2), 304–308. [Google Scholar] [CrossRef]
- Spickett, G.P.; Dalgleish, A.G. Cellular immunology of HIV-infection. Clin. Exp. Immunol. 1988, 71, 1–7. [Google Scholar]
- Navas, A.; dos Santos, J.C.; van Cranenbroek, B.; Vadaq, N.; Groenendijk, A.L.; Vos, W.A.J.W.; Blaauw, M.J.T.; van Eekeren, L.; Rokx, C.; Stalenhoef, J.E.; et al. Circulating immune landscape and immune signatures in spontaneous HIV controllers. Front Immunol. 2025, 16, 1642482. [Google Scholar] [CrossRef]
- Lofstrom, E.; Eringfalt, A.; Kotz, A.; Weckbom, F.; Tham, J.; Lingman, M.; Nygren, J.M.; Unden, J. Dynamics of IgG avidity and antibody levels after COVID-19. J. Clin. Virol. 2021, 144, 104986. [Google Scholar] [CrossRef]
- Manuylov, V.; Burgasova, O.; Borisova, O.; Smetanina, S.; Vasina, D.; Grogoriev, I.; Kudryashova, A.; Semashko, M.; Cherepovich, B.; Kharchenko, O.; et al. Avidity of IgG to SARS-CoV-2 RBD as a prognostic factor for the severity of COVID-19 reinfection. Viruses 2022, 14, 617. [Google Scholar] [CrossRef]
- Lara-Aguilar, V.; Llamas-Adan, M.; Brochado-Kith, O.; Crespo-Bermejo, C.; Grande-Garcia, S.; Arca-Lafuente, S.; Santos, I.I.; Prado, C.; Alia, M.; Sainz-Pinos, C.; et al. Low-level HIV-1 viremia affects T cell activation and senescence in long-term treated adults in the INSTI era. J. Biomed. Sci. 2024, 31, 80. [Google Scholar] [CrossRef]
- Shive, C.L.; Freeman, M.L.; Younes, S.; Kowal, C.M.; Cannaday, D.H.; Rodriguez, B.; Lederman, M.M.; Anthony, D.D. Markers of T cell exhaustion and senescence and their relationship to plasma TGF-β levels in treated HIV+ immune non-responders. Front Immunol. 2021, 12, 638010. [Google Scholar] [CrossRef]
- Naismith, E.; Pangrazzi, L.; Grasse, M.; Keller, M.; Miggitsch, C.; Weinberger, B.; Trieb, K.; Grubeck-Loebenstein, B. Peripheral antibody concentrations are associated with highly differentiated T cells and inflammatory processes in the human bone marrow. Immun. Ageing 2019, 16, 21. [Google Scholar] [CrossRef]
- Moosa, A.; Gengiah, T.N.; Lewis, L.; Naidoo, K. Long-term adherence to antiretroviral therapy in a South African adult patient cohort: a retrospective study. BMC Infect. Dis. 2019, 19, 775. [Google Scholar] [CrossRef]
- Bijker, R.; Kiertiburanakul, S.; Kumarasamy, N.; Pujari, S.; Sun, L.P.; Ng, O.T.; Lee, M.P.; Choi, J.Y.; Nguyen, K.V.; Chan, Y.J.; et al. Survival after long-term ART exposure: findings from an Asian patient population retained in care beyond five years on ART. Antivir. Ther. 2020, 25(3), 131–142. [Google Scholar] [CrossRef]
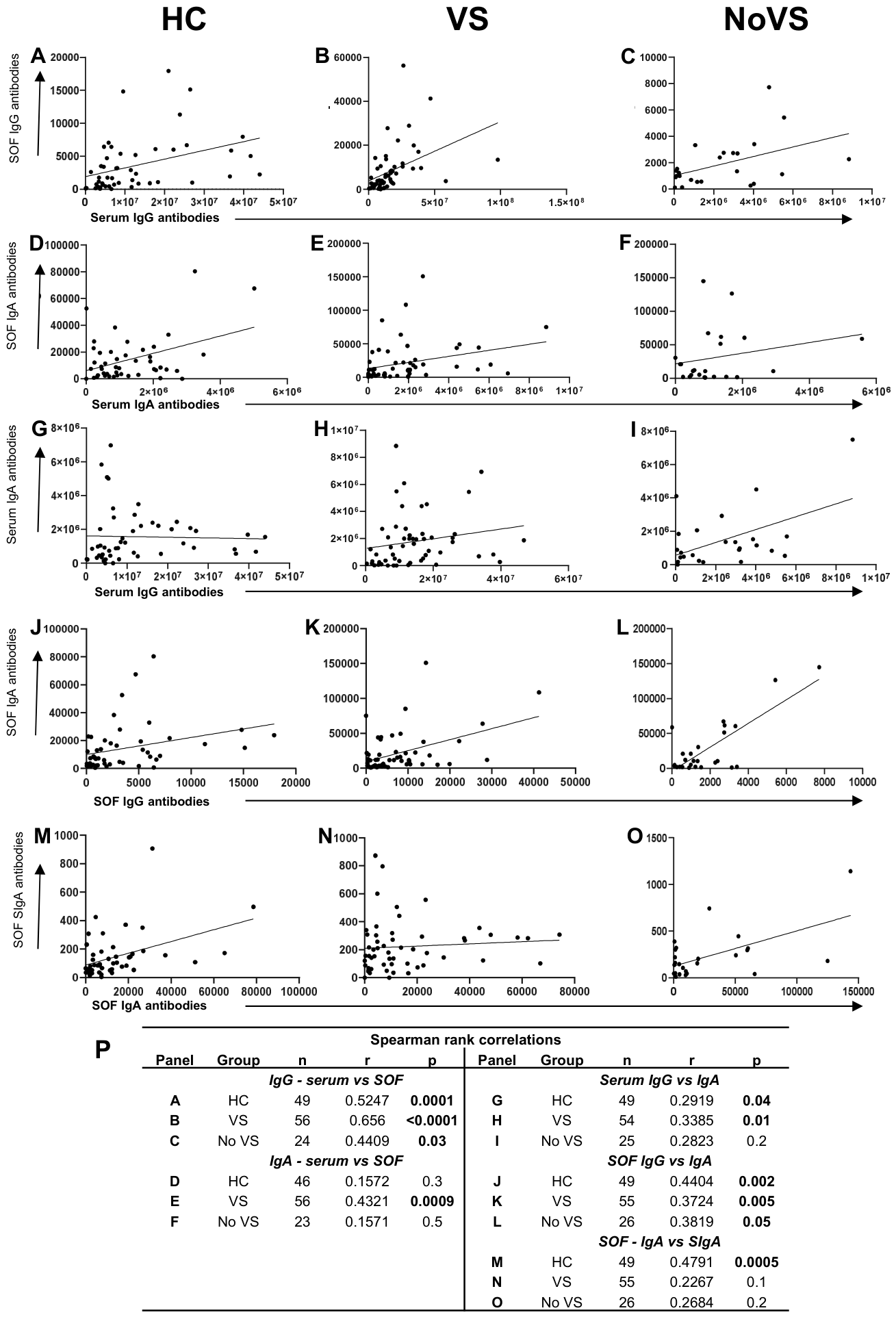
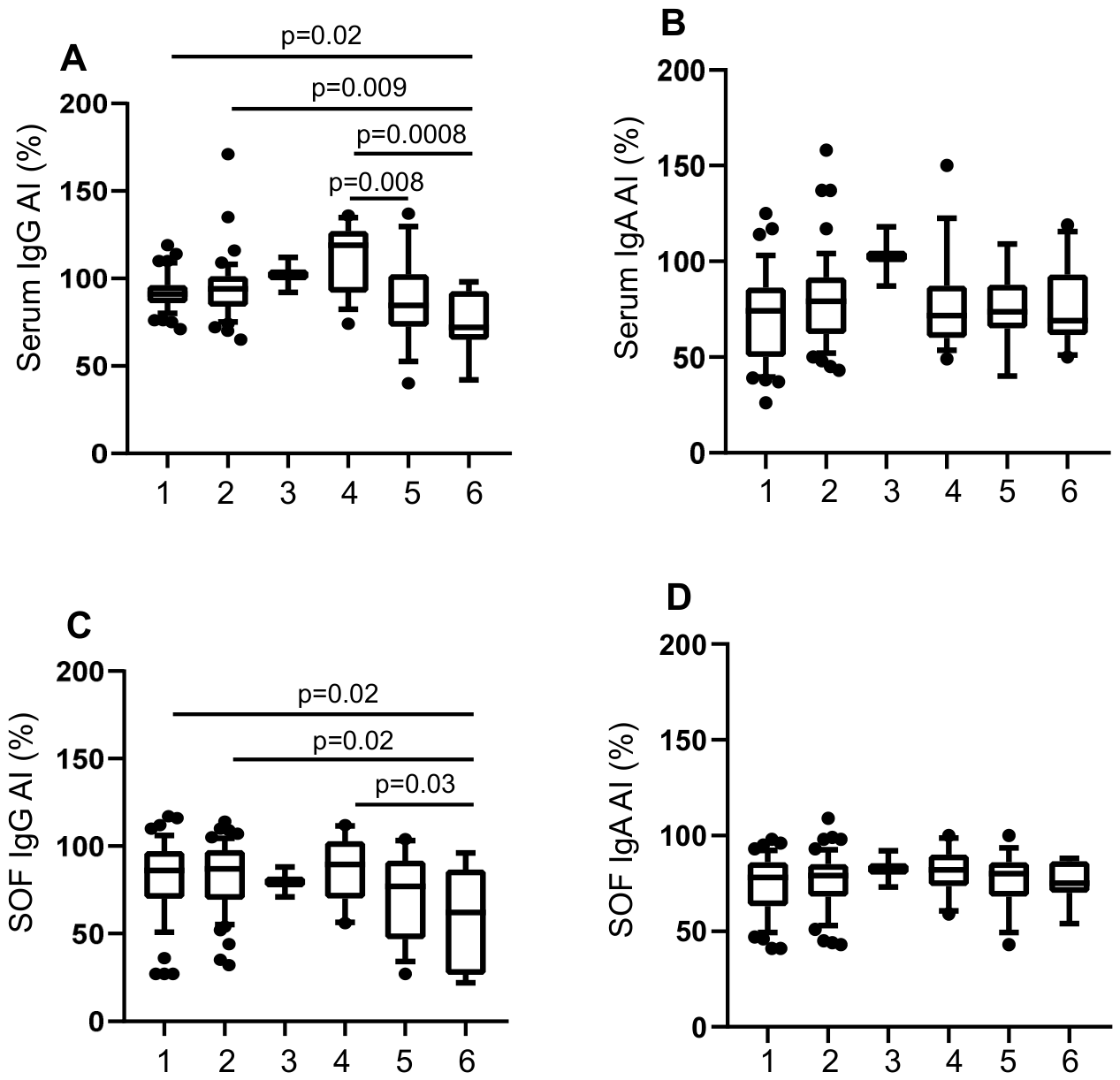
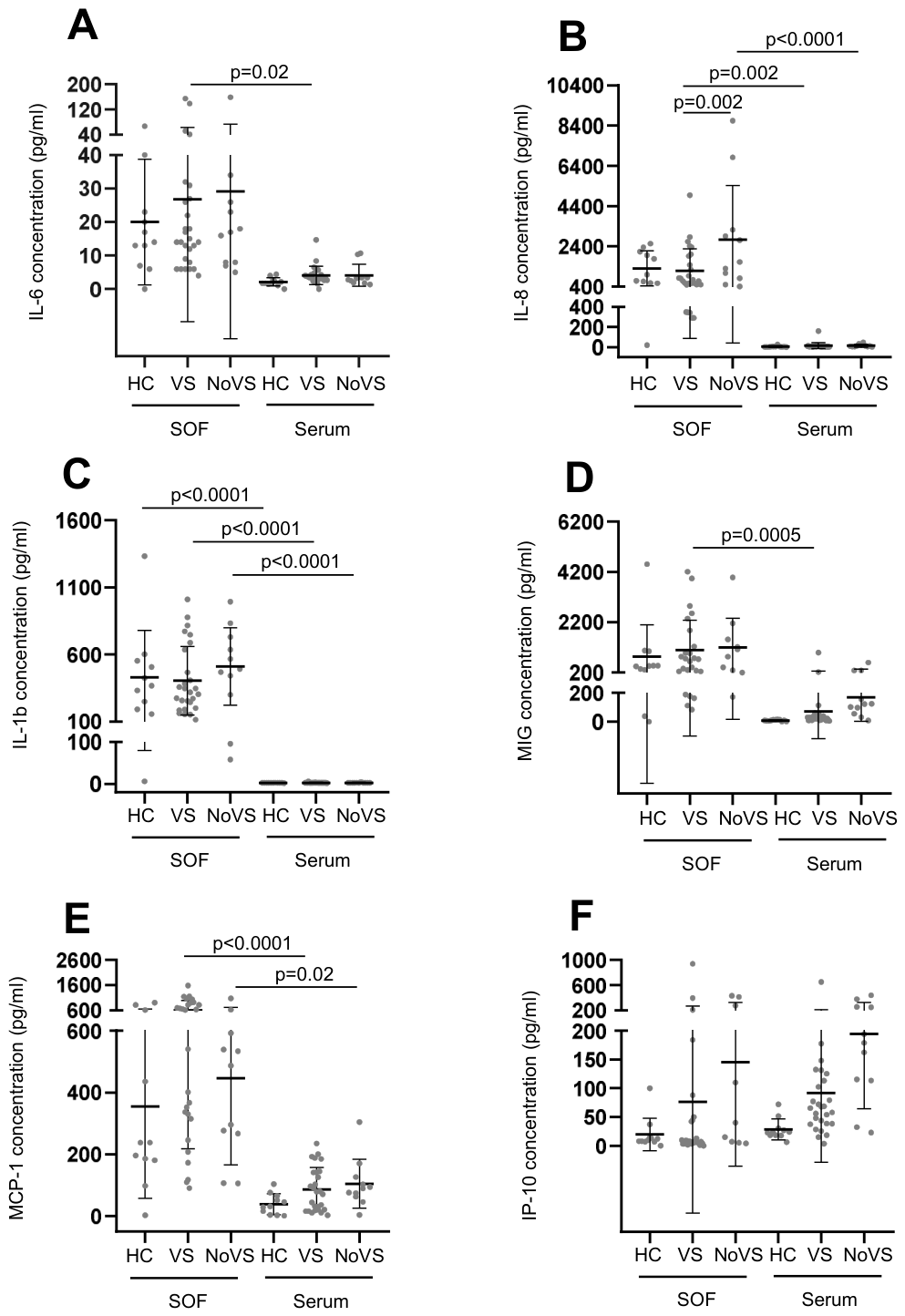
| Overall | HC | PLWH | NoVS-IR | NoVS-NoIR | VS-IR | VS-NoIR | LowVL-IR | |
|---|---|---|---|---|---|---|---|---|
| N= | 49 | 94 | 16 | 9 | 54 | 2 | 13 | |
| Serum IgG pos | 49 (100) | 94 (100) | 16 (100) | 9 (100) |
54 (100) | 2 (100) |
13 (100) |
|
| Gender | Males | 33 (67) | 72 (77) | 13 (81) | 6 (67) | 39 (72) | 2 (100) | 12 (92) |
| Females | 14 (29) | 22 (23) | 3 (19) | 3 (33) | 15 (28) | 0 | 1 (8) | |
| Age (yrs) | <60 | 48 (98) | 81 (86) | 14 (88) | 8 (89) | 48 (89) | 1 (50) | 10 (77) |
| >=60 | 1 (2) | 13 (14) | 2 (12) | 1 (11) | 6 (11) | 1 (50) | 3 (23) | |
| Vaccine type | Covishield | 37 (76) | 86 (91) | 16 (100) | 9 (100) |
47 (87) | 2 (100) |
12 (92) |
| Covaxin | 12 (24) | 7 (7) | 0 | 0 | 6 (11) | 0 | 1 (8) | |
| Sputnik | 0 | 1 (1) | 0 | 0 | 1 (2) | 0 | 0 | |
|
COVID-19 BTI |
Total | 8 (16) | 1 (1) | 0 | 1 (11) | 0 | 0 | 0 |
| Gender | Males | 6 (75) | 1 (100) | 1 (100) | ||||
| Females | 2 (25) | 0 | 0 | |||||
| Age (yrs) | <60 | 8 (100) | 1 (100) | 1 (100) | ||||
| >=60 | 0 | 0 | 0 | |||||
| Vaccine type | Covishield | 7 (19) | 1 (1) | 1 (11) | ||||
| Covaxin | 1 (8) | 0 | 0 |
| Sample / Antibody type | HC | PLWH | |||
|---|---|---|---|---|---|
| Overall | VS | LoVL | NoVS | ||
| Antibody levels - median | |||||
| Serum IgG (U/ml) | 7.8x106 | 6.6x106 | 1.3x107 | 3.8x106 | 2.5x106 |
| SOF IgG (U/ml/min) | 1935 | 2757 | 4325 | 1729 | 1349 |
| Serum IgA (U/ml) | 9x105 | 1x106 | 1.3x106 | 1.1x106 | 8.9x105 |
| SOF IgA (U/ml/min) | 7373 | 9576 | 11505 | 12364 | 1.630 |
| SOF SIgA (U/ml/min) | 88 | 168 | 204 | 83 | 159 |
| Antibody median ratios | |||||
| Serum IgG/IgA | 8* | 6 | 10#^ | 3# | 2*^ |
| SOF IgG/IgA | 0.3 | 0.4 | 0.6 | 0.1 | 0.1 |
| SOF IgG/SIgA | 22* | 20 | 27^ | 16 | 11*^ |
| SOF IgA/SIgA | 79 | 64 | 41 | 117 | 118 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




