Submitted:
27 April 2026
Posted:
28 April 2026
You are already at the latest version
Abstract
Keywords:
1. Background
2. Methodology and Sources of Information
3. Aetiology
4. Epidemiology
5. Transmission and Spread
6. Clinical Signs and Symptoms
7. Diagnosis of Malaria and COVID-19
8. Malaria and COVID-19 Comprehensive Integrated Management Approaches
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- World malaria report 2018. 2018.
- Wang, M.Y.; Zhao, R.; Gao, L.J.; et al. SARS-CoV-2: Structure, Biology, and Structure-Based Therapeutics Development. Front Cell. Infect. Microbiol. 2020, 10, 1–17. [Google Scholar] [CrossRef]
- Park, J.; Kang, S.; Seok, D.; et al. Barriers against and strategies for malaria control during the COVID-19 pandemic in low- and middle-income countries: a systematic review. Malar. J. 2023, 22, 1–16. [Google Scholar] [CrossRef]
- Barbosa, D.S. Effects of public health emergencies of international concern on disease control : a systematic review 2023, 1–9.
- Heuschen, A.K.; Abdul-Mumin, A.; Abubakari, A.; et al. Effects of the COVID-19 pandemic on general health and malaria control in Ghana: a qualitative study with mothers and health care professionals. Malar. J. 2023, 22, 1–18. [Google Scholar] [CrossRef]
- Okunade, K. Profiles of Sugar Fermenting Bacteria of the Oral Cavity among Children with Dental Caries Attending Stomatology Services at Ruhengeri Referral Hospital in Musanze District, Northern Rwanda; 2018; pp. 19–26. [Google Scholar]
- Sanyaolu, A.; Okorie, C.; Hosein, Z.; et al. Global Pandemicity of COVID-19: Situation Report as of June 9, 2020. Infect. Dis. Res. Treat. 2021, 14, 117863372199126. [Google Scholar] [CrossRef]
- Shen, J.; Duan, H.; Zhang, B.; et al. Prevention and control of COVID-19 in public transportation : Experience from China. 266 2020. [Google Scholar] [CrossRef] [PubMed]
- Mumali, R.K.; Okolimong, C.; Kabuuka, T.; et al. Health workers’ adherence to the malaria test, treat and track strategy during the COVID-19 pandemic in malaria high transmission area in Eastern Uganda. Malar. J. 2023, 22, 3–11. [Google Scholar] [CrossRef] [PubMed]
- UCG 2016. Uganda Clinical Guidelines 2016. Minist. Heal Uganda 2016 Retrieved on. 2018, 1–1142. [Google Scholar]
- Additional guidelines for management of COVID-19 response Fund Donations.pdf.
- Namuyinga, R.J.; Mwandama, D.; Moyo, D.; et al. Health worker adherence to malaria treatment guidelines at outpatient health facilities in southern Malawi following implementation of universal access to diagnostic testing. Malar. J. 2017, 1–14. [Google Scholar] [CrossRef]
- MOH UMRSP. THE UGANDA MALARIA REDUCTION STRATEGIC PLAN 2014-2020. 2020, 1–83. [Google Scholar]
- Musoke, D.; Nalinya, S.; Lubega, G.B.; et al. PLOS GLOBAL PUBLIC HEALTH The effects of COVID-19 lockdown measures on health and healthcare services in Uganda; 2023; pp. 1–16. [Google Scholar]
- Kessel, B.; Heinsohn, T.; Ott, J.J.; et al. Impact of COVID-19 pandemic and anti-pandemic measures on tuberculosis, viral hepatitis, HIV/AIDS and malaria–A systematic review. PLoS Glob. Public Heal 2023, 3, e0001018. [Google Scholar] [CrossRef]
- Lone, S.A.; Ahmad, A. COVID-19 pandemic – an African perspective. Epub Ahead Print. 2020. [Google Scholar] [CrossRef]
- Rajvanshi, H.; Saha, K.B.; Shukla, M.M.; et al. Assessment of ASHA for knowledge, diagnosis and treatment on malaria in Mandla district of Madhya Pradesh as part of the malaria elimination demonstration project. Malar. J. 2021, 1–8. [Google Scholar] [CrossRef] [PubMed]
- WHO. Word Malaria Report 2021. 2021. Available online: https://www.who.int/teams/global-malaria-programme/reports/world-malaria-report-2021.
- World malar ia report 2022. 2022.
- Hák, T.; Janoušková, S.; Moldan, B. Sustainable Development Goals: A need for relevant indicators. Ecol. Indic. 2016, 60, 565–573. [Google Scholar] [CrossRef]
- Initiative, M.; President US. PRESIDENT ’ S MALARIA INITIATIVE SIERRA LEONE Malaria Operational Plan FY 2017.
- Namayanja, C.; Emma, E.; Eregu, I.; et al. Unusual clinical spectra of childhood severe malaria during malaria epidemic in eastern Uganda : a prospective study. Malar. J. 2023, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Naigino, R.; Katamba, A.; Babikako, H.M.; et al. Utilization of Malaria Diagnostic Tests and Receipt of Anti-Malarial Drugs by Febrile Patients Attending Outpatient Clinics of Health Centre IV Facilities in Mukono District, Uganda; 2014; pp. 1628–1642. [Google Scholar]
- The Global Fund. The Impact of Covid-19 on Hiv, Tb and Malaria Services and Systems for Health : a Snapshot From 502 Health Facilities. In Glob Fund to Fight AIDS Malaria, Tuberc; 2021; Available online: https://reliefweb.int/sites/reliefweb.int/files/resources/covid-19_2020-disruption-impact_report_en.pdf.
- Kizza, J.; Kamaduuka, R.Z.; Arineitwe, J.; et al. The Economic Effects of COVID-19 Pandemic on the Market Vendors in Kampala City in Uganda; 2023; pp. 0–16. [Google Scholar]
- Miller, L.M.S.; Gee, P.M.; Katz, R.A.; et al. The Importance of Understanding COVID-19 : The Role of Knowledge in Promoting Adherence to Protective Behaviors. 2021, 9, 1–8. [Google Scholar] [CrossRef]
- Akanteele Agandaa, S.; Kweku, M.; Agboli, E.; et al. Implementation and challenges of test, treat and track (T3) strategy for malaria case management in children under five years in the Bongo District, Ghana. Clin. Res. Trials 2016, 2, 235–241. [Google Scholar] [CrossRef]
- Jowsey, T.; Yen, L.; Mathews, W. P. Time spent on health related activities associated with chronic illness: A scoping literature review. BMC Public Health 2012, 12, 1. [Google Scholar] [CrossRef] [PubMed]
- Adhikari, S.P.; Meng, S.; Wu, Y.; et al. Epidemiol. Causes Clin. Manif. Diagn. Prev. Control Coronavirus Dis. ( COVID-19 ) Dur. Early Outbreak Period A Scoping Rev. 2020, 1–12.
- Chen, C.Y.; Chang C ke; Chang, Y.W.; et al. Structure of the SARS Coronavirus Nucleocapsid Protein RNA-binding Dimerization Domain Suggests a Mechanism for Helical Packaging of Viral RNA. J. Mol. Biol. 2007, 368, 1075–1086. [Google Scholar] [CrossRef]
- Chen, J.; Wang, R.; Hozumi, Y.; et al. Emerging Dominant SARS-CoV-2 Variants. J. Chem. Inf. Model 2023, 63, 335–342. [Google Scholar] [CrossRef]
- Harvey, W.T.; Carabelli, A.M.; Jackson, B.; et al. SARS-CoV-2 variants, spike mutations and immune escape. Nat. Rev. Microbiol. 2021, 19, 409–424. [Google Scholar] [CrossRef]
- Fontanet, A.; Autran, B.; Lina, B.; et al. SARS-CoV-2 variants and ending the COVID-19 pandemic. Lancet 2021, 397, 952–954. [Google Scholar] [CrossRef]
- Tao, K.; Tzou, P.L.; Nouhin, J.; et al. The biological and clinical significance of emerging SARS-CoV-2 variants. Nat. Rev. Genet 2021, 22, 757–773. [Google Scholar] [CrossRef]
- Ajari, E.E.; Kanyike, A.M.; Abdulbasit, I.O. Review Article ___________________________________________________________ COVID-19 in Uganda : Epidemiology and Response.
- Mcintosh, A.K.; Editor, M.; Hirsch, M.S.; et al. COVID-19 : Epidemiology, virology, and prevention. 2.
- Sato, S. Correction to: Plasmodium—a brief introduction to the parasites causing human malaria and their basic biology (Journal of Physiological Anthropology, (2021), 40, 1, (1), 10.1186/s40101-020-00251-9). J. Physiol. Anthropol. 2021, 40, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Florey, L. Measures of Malaria Parasitemia Prevalence in National Surveys : DHS ANALYTICAL.
- Zhou, G.; Githure, J.; Lee, M.C.; et al. Malaria transmission heterogeneity in different eco-epidemiological areas of western Kenya: a region-wide observational and risk classification study for adaptive intervention planning. Malar. J. 2024, 23, 1–11. [Google Scholar] [CrossRef]
- Chamchoy, K.; Sudsumrit, S.; Thita, T.; et al. Cytochrome P450 2D6 (CYP2D6) and glucose-6-phosphate dehydrogenase (G6PD) genetic variations in Thai vivax malaria patients: Implications for 8-aminoquinoline radical cure. PLoS Negl. Trop. Dis. 2022, 16, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Meibalan, E.; Marti, M. Biology of malaria transmission. Cold Spring Harb. Perspect. Med. 2017, 7, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Indicator, M. Malaria Indicator Survey 2014-2015.
- Chu, C.S.; White, N.J. Management of relapsing Plasmodium vivax malaria. Expert Rev. Anti Infect. Ther. 2016, 14, 885–900. [Google Scholar] [CrossRef] [PubMed]
- Delves, M.; Plouffe, D.; Scheurer, C.; et al. The activities of current antimalarial drugs on the life cycle stages of plasmodium: A comparative study with human and rodent parasites. PLoS Med. 2012, 9. [Google Scholar] [CrossRef]
- Howick, V.M.; Russell, A.J.C.; Andrews, T.; et al. The malaria cell atlas: Single parasite transcriptomes across the complete Plasmodium life cycle. Science (80- ) 2019, 365. [Google Scholar] [CrossRef]
- Moumaris, M.; Bretagne, J.-M.; Abuaf, N. Biological Membranes and Malaria-Parasites. Open. Parasitol. J. 2019, 7, 1–18. [Google Scholar] [CrossRef]
- Dvorin, J.D.; Goldberg, D.E. Plasmodium Egress Across the Parasite Life Cycle. Annu Rev. Microbiol. 2022, 76, 67–90. [Google Scholar] [CrossRef]
- Matteelli, A.; Castelli, F. Chapter -2 Life cycle of malaria parasites.
- Siciliano, G.; Alano, P. Enlightening the malaria parasite life cycle: Bioluminescent Plasmodium in fundamental and applied research. Front Microbiol. 2015, 6, 1–8. [Google Scholar] [CrossRef]
- Ceusters, W.; Smith, B. Malaria Diagnosis and the Plasmodium Life Cycle: the BFO Perspective. Nat. Preced 2010. [Google Scholar] [CrossRef]
- Brief, W. AIRA Infodemic Trends Report Top trends. 2023, 2023, 1–7. [Google Scholar]
- Bell, D.; Hansen, K.S. Relative burdens of the COVID-19, malaria, tuberculosis, and HIV/AIDS epidemics in sub-Saharan Africa. Am. J. Trop. Med. Hyg. 2021, 105, 1510–1515. [Google Scholar] [CrossRef] [PubMed]
- Reza, N.; Defilippis, E.M.; Jessup, M. Secondary Impact of the COVID-19 Pandemic on Patients With Heart Failure; 2020; pp. 11–13. [Google Scholar]
- Bell, D.; Hansen, K.S.; Kiragga, A.N.; et al. Predicting the Impact of COVID-19 and the Potential Impact of the Public Health Response on Disease Burden in Uganda. 2020, 103, 1191–1197. [Google Scholar] [CrossRef] [PubMed]
- Characteristics C, Respiratory SA, Consortium EI. Letters 2022, 19, 2100–2104.
- Apiyo, M.; Olum, R.; Kabuye, A.; et al. Clinical Characteristics and Outcomes of Patients Hospitalized with COVID-19 at Case Hospital, Uganda. Epub ahead of print. 2022. [CrossRef]
- Tumwesigye, N.M.; Denis, O.; Kaakyo, M.; et al. Effects of the COVID-19 Pandemic on Health Services and Mitigation Measures in Uganda Working Paper. 571 March 2021. [Google Scholar]
- Musoke, D.; Nalinya, S.; Lubega, G.B.; et al. The impact of the COVID-19 lockdown on social and economic welfare in Uganda. In Arch Public Heal; 2024; p. 82. [Google Scholar] [CrossRef]
- Nachega, J.B.; Kapata, N.; Sam-Agudu, N.A.; et al. Minimizing the impact of the triple burden of COVID-19, tuberculosis and HIV on health services in sub-Saharan Africa. Int. J. Infect. Dis. 2021, 113, S16–S21. [Google Scholar] [CrossRef]
- Agbemafle, E.E.; Addo-Lartey, A.; Odikro, M.A.; et al. Adherence to the test, treat and track strategy for malaria control among prescribers, Mfantseman Municipality, Central Region, Ghana. 2023. [Google Scholar] [CrossRef]
- David, A.; Swalehe, O.; Amour, J.D. Accessibility of malaria commodities in Geita District Council, mainland Tanzania : the experiences from healthcare providers and clients. 17. [CrossRef]
- WHO. Guidelines for t h e t r e at m en t of m alar i a.
- Usman, Rabi. Predictors of malaria Rapid Diagnostic Tests ’ utilisation among healthcare workers in Zamfara State. 2019, 198, 1–10. [Google Scholar] [CrossRef]
- Bamiselu, O.F.; Ajayi, I.; Fawole, O.; et al. Adherence to malaria diagnosis and treatment guidelines among healthcare workers in Ogun State, Nigeria. BMC Public Health 2016, 1–10. [Google Scholar] [CrossRef]
- Kigozi, S.P.; Kigozi, R.N.; Sebuguzi, C.M.; et al. Spatial-temporal patterns of malaria incidence in Uganda using HMIS data from 2015 to 2019. 2020, 1–14. [Google Scholar] [CrossRef]
- Epstein, A.; Namuganga, J.F.; Nabende, I.; et al. Mapping malaria incidence using routine health facility surveillance data in Uganda. Epub Ahead Print. 2023. [Google Scholar] [CrossRef]
- Malaria, N.; Program, C. Uganda national malaria control policy.
- Ayouni, I.; Maatoug, J.; Dhouib, W.; et al. Effective public health measures to mitigate the spread of COVID-19: a systematic review. BMC Public Health 2021, 21, 1–14. [Google Scholar] [CrossRef]
- Asia, S.E.; Africa, S.; Covid-, T. International Journal of Infectious Diseases COVID-19 and malaria : A symptom screening challenge for malaria endemic countries. 2020, 94, 151–153. [Google Scholar]
- Indicator, M. Malaria Indicator Survey (MIS) 2014-15.
- Gondwe, G. Assessing the Impact of COVID-19 on Africa’s Economic Development. In United Nations Conf Trade Dev; 2020; pp. 1–21. [Google Scholar]
- Weiss, P.; Murdoch, D.R. COVID-19 : towards controlling of a pandemic. 2020, 6736, 1015–1018. [Google Scholar]
- Allam, M.; Cai, S.; Ganesh, S.; et al. COVID-19 Diagnostics, Tools, and Prevention; pp. 1–33.
- FİRA, Ş. COVID-19 : Prevention and control measures in community. 50. 2020. [CrossRef]
- Aarø, L.E.; Mathews, C.; Kaaya, S.; et al. Promoting sexual and reproductive health among adolescents in southern and eastern Africa ( PREPARE ): project design and conceptual framework. [CrossRef]
- UNICEF. Malaria Rapid Diagnostic Tests An implementation guide. 2013. [Google Scholar]
- Agbemafle, E.E.; Addo-lartey, A.; Id, A.O.; et al. Adherence to the test, treat and track strategy for malaria control among prescribers, Mfantseman Municipality, Central Region, Ghana. 2023, 47, 1–15. [Google Scholar] [CrossRef]
- Kamya, M.R.; Arinaitwe, E.; Wanzira, H.; et al. Malaria Transmission, Infection, and Disease at Three Sites with Varied Transmission Intensity in Uganda : Implications for Malaria Control. 2015, 92, 903–912. [Google Scholar] [CrossRef] [PubMed]
- Cohen, J. Prices, Diagnostic Tests and the Demand for Malaria Treatment : Evidence from a Randomized Trial.
- Asingizwe, D.; Murindahabi, M.M.; Poortvliet, M. Co-Designing a Citizen Science Program for Malaria Control in Rwanda sustainability Co-Designing a Citizen Science Program for Malaria Control in Rwanda. Epub Ahead Print. 2019. [Google Scholar] [CrossRef]
- Kankpetinge, C.; Kweku, M.; Baiden, F.; et al. Clinicians’ Adherence to Implementation of Test, Treat and Track Strategy for Malaria Control among Children Under-five Years in Ho Municipality, Volta Region, Ghana. Int. J. Trop. Dis. Heal 2016, 20, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Tetteh, M.; Dwomoh, D.; Asamoah, A.; et al. Impact of malaria diagnostic refresher training programme on competencies and skills in malaria diagnosis among medical laboratory professionals : evidence from Ghana 2015 – 2019. Malar. J. 2021, 1–12. [Google Scholar] [CrossRef]
- Ocen, E.; Opito, R.; Tegu, C.; et al. Severe malaria burden, clinical spectrum and outcomes at Apac district hospital, Uganda : a retrospective study of routine health facility- based data. Malar. J. 2023, 1–8. [Google Scholar] [CrossRef]
- World Malaria Report Malaria 7.1. 2014.
- Romero-Castro, N.S.; Colín-Hernández, I.; Godoy-Reyes, M.E.; et al. Clinical Signs and Symptoms Associated with COVID-19: A Cross Sectional Study. Int. J. Odontostomatol. 2022, 16, 112–119. [Google Scholar] [CrossRef]
- Struyf, T.; Deeks, J.J.; Dinnes, J.; et al. Signs and symptoms to determine if a patient presenting in primary care or hospital outpatient settings has COVID-19. Cochrane Database Syst. Rev. 2022. Epub ahead of print 2022. [Google Scholar] [CrossRef]
- Baguma, S.; Okot, C.; Alema, N.O.; et al. Factors Associated With Mortality Among the COVID-19 Patients Treated at Gulu Regional Referral Hospital : A Retrospective Study. 2022, 10, 1–11. [Google Scholar] [CrossRef]
- Burchett, H.E.D.; Leurent, B.; Baiden, F.; et al. Improving prescribing practices with rapid diagnostic tests (RDTs): Synthesis of 10 studies to explore reasons for variation in malaria RDT uptake and adherence. BMJ Open. 2017, 7. [Google Scholar] [CrossRef]
- Jatau, S.S.; Ishaku, S.G.; Dauda, M.; et al. Knowledge, use of mRDT, and malaria control practice among health care workers in three Local Government areas of Kano. 2021, 20, 56–65. [Google Scholar]
- Ndewa, K.; Kihamia, C.M.; Mubi, M.; et al. Performance Characteristics of Malaria Rapid Diagnostic Test and Its Utilization in Management of Febrile Patients in Korogwe District (Tanga) Tanzania. Univers J. Med. Sci. 2016, 4, 81–87. [Google Scholar] [CrossRef]
- Di, Gennaro F; Marotta, C.; Locantore, P.; et al. Malaria and COVID-19 : Common and Di ff erent Findings.
- Diaz, J. Clinical Management for COVID-19 Advancing the care pathway.
- Wu, W.; Cheng, Y.; Zhou, H.; et al. The SARS-CoV-2 nucleocapsid protein: its role in the viral life cycle, structure and functions, and use as a potential target in the development of vaccines and diagnostics. Virol. J. 2023, 20, 1–16. [Google Scholar] [CrossRef]
- Levine, R.; Caputo, N. CT SCAN OF A COVID POSITIVE PATIENT; pp. 2–5.
- Li, T. Diagnosis and clinical management of severe acute respiratory syndrome Coronavirus 2 (SARS-CoV-2) infection: an operational recommendation of Peking Union Medical College Hospital (V2.0): Working Group of 2019 Novel Coronavirus, Peking Union Medical Colle. Emerg. Microbes Infect. 2020, 9, 582–585. [Google Scholar] [CrossRef]
- RBM. Best practices in mitigating the effect of COVID-19 on malaria; 2021; pp. 1–40.
- Rahi, M.; Das, P.; Sharma, A. COVID-19 mitigation steps provide a blueprint for malaria control and elimination. Am. J. Trop. Med. Hyg. 2020, 103, 28–30. [Google Scholar] [CrossRef]
- Uwaezuoke, S.N. Strengthening health systems in Africa: The COVID-19 pandemic fallout. J. Pan Afr. Thorac. Soc. 2020, 1, 15–19. [Google Scholar] [CrossRef]
- Rogerson, S.J.; Beeson, J.G.; Laman, M.; et al. Identifying and combating the impacts of COVID-19 on malaria. BMC Med. 2020, 18, 1–7. [Google Scholar] [CrossRef]
- WHO. Tailoring malaria interventions in the COVID-19 response. Glob Malar Program, 2020; 34. [Google Scholar]
- Rahi, M.; Baharia, R.K.; Das, P.; et al. Overlaying COVID-19 mitigation plans on malaria control infrastructures. Trans. R. Soc. Trop. Med. Hyg. 2021, 115, 6–8. [Google Scholar] [CrossRef]
- Malaria and COVID-19. 2000; 19.
- Ajayi, I.O.; Ajumobi, O.O.; Falade, C. Malaria and COVID-19: commonalities, intersections and implications for sustaining malaria control. Pan Afr. Med. J. 2020, 37, 1–10. [Google Scholar] [CrossRef]
- COVID19 vaccines 101 – to be or not to be ( jabbed ).
- USAID / PMI. Presidential ’s Malaria Initiative Uganda Malaria Operational Plan FY 2017. 2017, 10. [Google Scholar]
- Watson, O.J.; Sumner, K.M.; Janko, M.; et al. False-negative malaria rapid diagnostic test results and their impact on community-based malaria surveys in sub-Saharan Africa. Epub Ahead Print. 2019. [Google Scholar] [CrossRef] [PubMed]
- Habimana, A.; Harerimana, A.; Asingizwe, D.; et al. Community Health Workers ’ Knowledge, Attitudes and Practices about Malaria Prevention in Gicumbi District, Rwanda. 2016, 3, 27–35. [Google Scholar] [CrossRef]
- Mbacham, W.F.; Mangham-jeff, L.; Cundill, B.; et al. Basic or enhanced clinician training to improve adherence to malaria treatment guidelines : a cluster-randomised trial in two areas of Cameroon 2014, 346–358.
- Ai, chanda P; et al. Prescriber and patient-oriented behavioural interventions to improve use of malaria rapid diagnostic tests in Tanzania: Facility-based cluster randomised trial. Cundill Al 2015, 13, 1–16. [Google Scholar]
- Asingizwe, D.; Poortvliet, P.M.; Koenraadt, C.J.M.; et al. Role of individual perceptions in the consistent use of malaria preventive measures : mixed methods evidence from rural Rwanda. Malar. J. 2019, 1–19. [Google Scholar] [CrossRef]
- Manuscript, A. NIH Public Access. 2013, 121, 184–195. [Google Scholar]
- Mathieu, E.; Ritchie, H.; Ortiz-Ospina, E.; et al. A global database of COVID-19 vaccinations. Nat. Hum. Behav. 2021, 5, 947–953. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Jing, R.; Lai, X.; et al. Acceptance of covid-19 vaccination during the covid-19 pandemic in china. Vaccines 2020, 8, 1–14. [Google Scholar] [CrossRef]
- Lokhandwala, S.; Waghela, S.D.; Bray, J.; et al. Induction of robust immune responses in swine by using a cocktail of adenovirus-vectored African swine fever virus antigens. Clin. Vaccine Immunol. 2016, 23, 888–900. [Google Scholar] [CrossRef]
- Mboowa, G.; Musoke, D.; Bulafu, D.; et al. Face-masking, an acceptable protective measure against COVID-19 in ugandan high-risk groups. Am. J. Trop. Med. Hyg. 2021, 104, 502–513. [Google Scholar] [CrossRef] [PubMed]
- Pietersen, T. Zertifikat Certificat Certificado Certificate. 2016, 20140328. [Google Scholar]
- Midzi, N.; Mutsaka-Makuvaza, M.J.; Charimari, L.S.; et al. Factors affecting hand hygiene practice during the COVID-19 pandemic in the Zimbabwean population: a qualitative study. BMC Infect. Dis. 2024, 24, 1–12. [Google Scholar] [CrossRef]
- Alwan, N.; Safwan, J.; Kerek, R.; et al. Hand hygiene during the spread of COVID-19: a cross-sectional study of awareness and practices among academic institutions in Lebanon. Front Public Heal 2023, 11, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Graves, P.; Gelband, H. Vaccines for preventing malaria (blood-stage). Cochrane Database Syst. Rev. Epub Ahead Print. 2006. [Google Scholar] [CrossRef]
- Boyce, R.M.; Muhindo, E.; Baguma, E.; et al. Permethrin - treated baby wraps for the prevention of malaria : results of a randomized controlled pilot study in rural Uganda. Malar. J. 2022, 1–12. [Google Scholar] [CrossRef]
- Maiteki-sebuguzi, C.; Gonahasa, S.; Kamya, M.R.; et al. Effect of long-lasting insecticidal nets with and without piperonyl butoxide on malaria indicators in Uganda ( LLINEUP ): final results of a cluster-randomised trial embedded in a national distribution campaign. Lancet Infect. Dis. 2023, 23, 247–258. [Google Scholar] [CrossRef]
- Kukundakwe, C.; Nankabirwa, C.M.; Katushabe, C.; et al. Articles The effect of intermittent preventive treatment for malaria with dihydroartemisinin – piperaquine on vaccine-specific responses among schoolchildren in rural Uganda ( POPVAC B ) A Double-Blind Randomised Control. Trial 1838–1848.
- Ali, A.M.; Savic, R.M. Interplay among malnutrition, chemoprevention, and the risk of malaria in young Ugandan children : Longitudinal pharmacodynamic and growth analysis 2023, 656–667.
- Bosco, A.B.; Anderson, K.; Gresty, K.; et al. Genetic diversity and genetic relatedness in Plasmodium falciparum parasite population in individuals with uncomplicated malaria based on microsatellite typing in Eastern and Western regions of Uganda, 2019–2020. Malar. J. 2021, 20, 1–16. [Google Scholar] [CrossRef]
- Castelli, F.; Odolini, S.; Autino, B.; et al. Malaria prophylaxis: A comprehensive review. Pharmaceuticals 2010, 3, 3212–3239. [Google Scholar] [CrossRef]
- Consortium, M. Integrating mRDTs into the health system in Uganda.
- Road, L. The Uganda Malaria Reduction Strategic Plan 2014-2020; 2020; pp. 1–83. [Google Scholar]
- Yeka, et al. Malaria Indicator Survey (MIS) 2014-15. 2014. [Google Scholar]
- WHO. World malaria World report. 2023. Available online: https://www.wipo.int/amc/en/mediation/%0Ahttps://www.who.int/teams/global-malaria-programme/reports/world-malaria-report-2023.
- Kayentao, K.; Florey, L.S.; Mihigo, J.; et al. Impact evaluation of malaria control interventions on morbidity and all-cause child mortality in Mali, 2000-2012. Malar. J. 2018, 17, 1–15. [Google Scholar] [CrossRef]
- Elmardi, K.A.; Adam, I.; Malik, E.M.; et al. Impact of malaria control interventions on malaria infection and anaemia in low malaria transmission settings: a cross-sectional population-based study in Sudan. BMC Infect. Dis. 2022, 22, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Prosper, O.; Martcheva, M. Impact of enhanced malaria control on the competition between Plasmodium falciparum and Plasmodium vivax in India. Math. Biosci. 2013, 242, 33–50. [Google Scholar] [CrossRef] [PubMed]
- Payne, D.; Grab, B.; Fontaine, R.E.; et al. Impact of control measures on malaria transmission and general mortality. Bull. World Health Organ 1976, 54, 369–377. [Google Scholar]
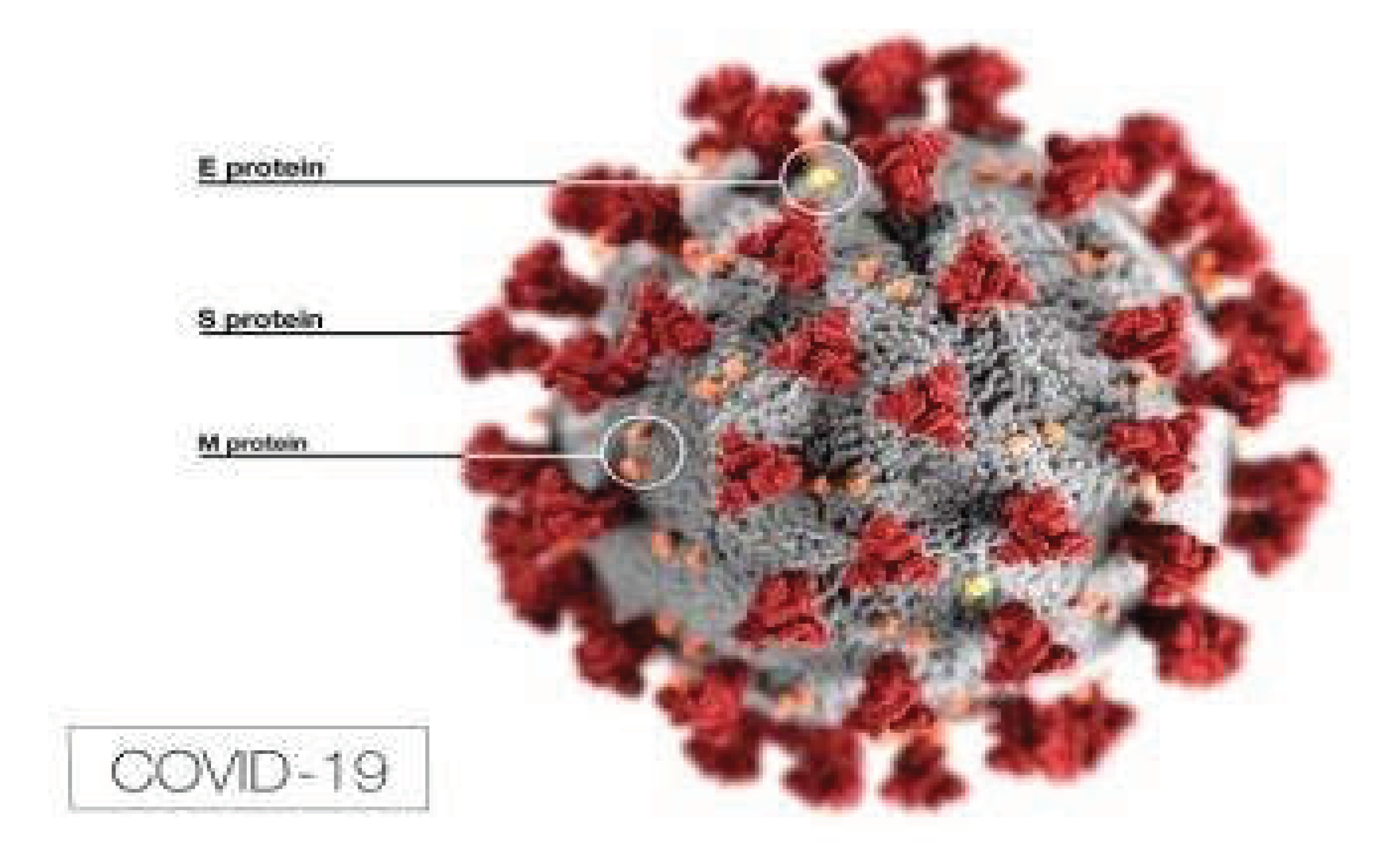
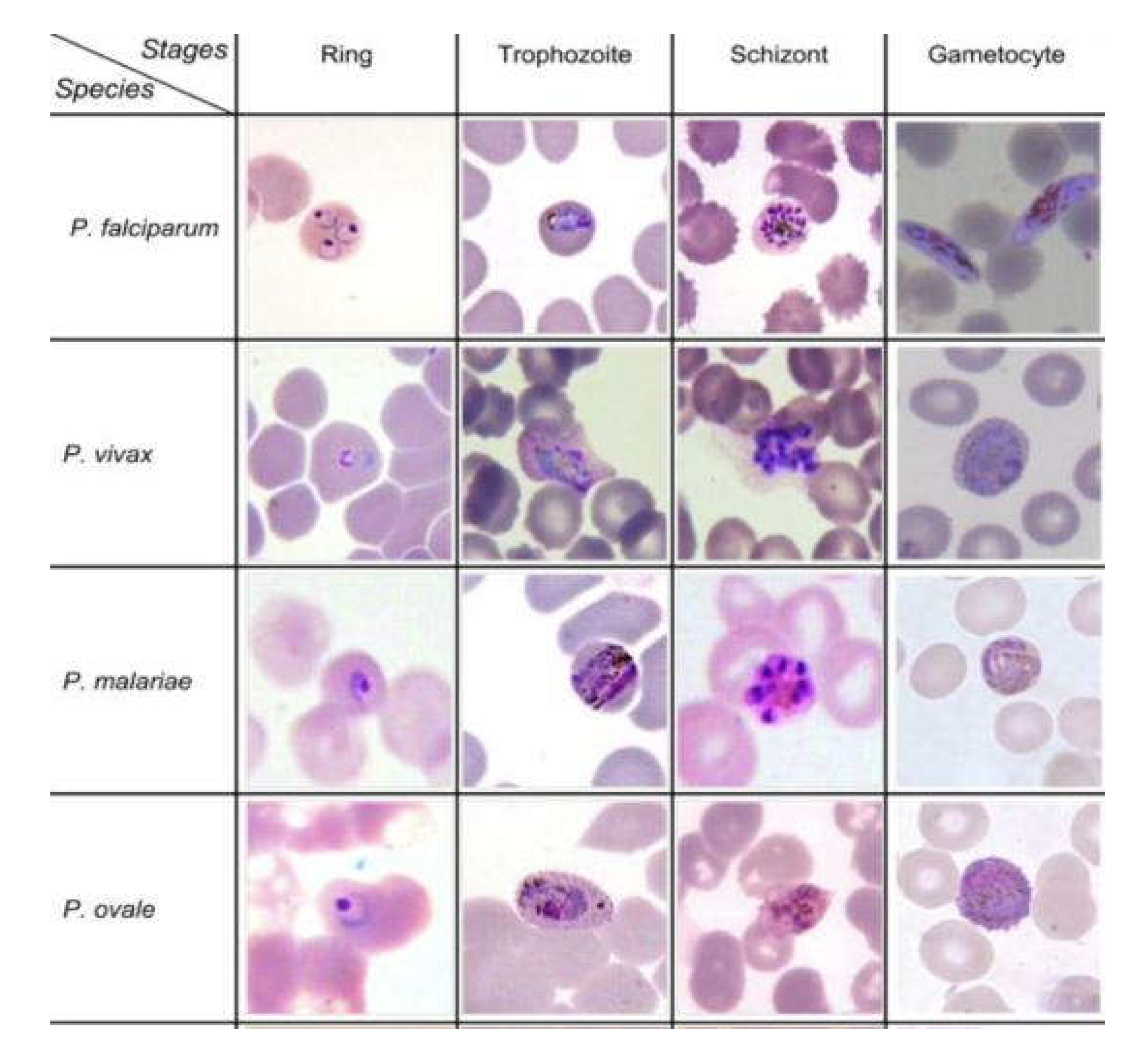
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.



