Submitted:
27 April 2026
Posted:
28 April 2026
You are already at the latest version
Abstract
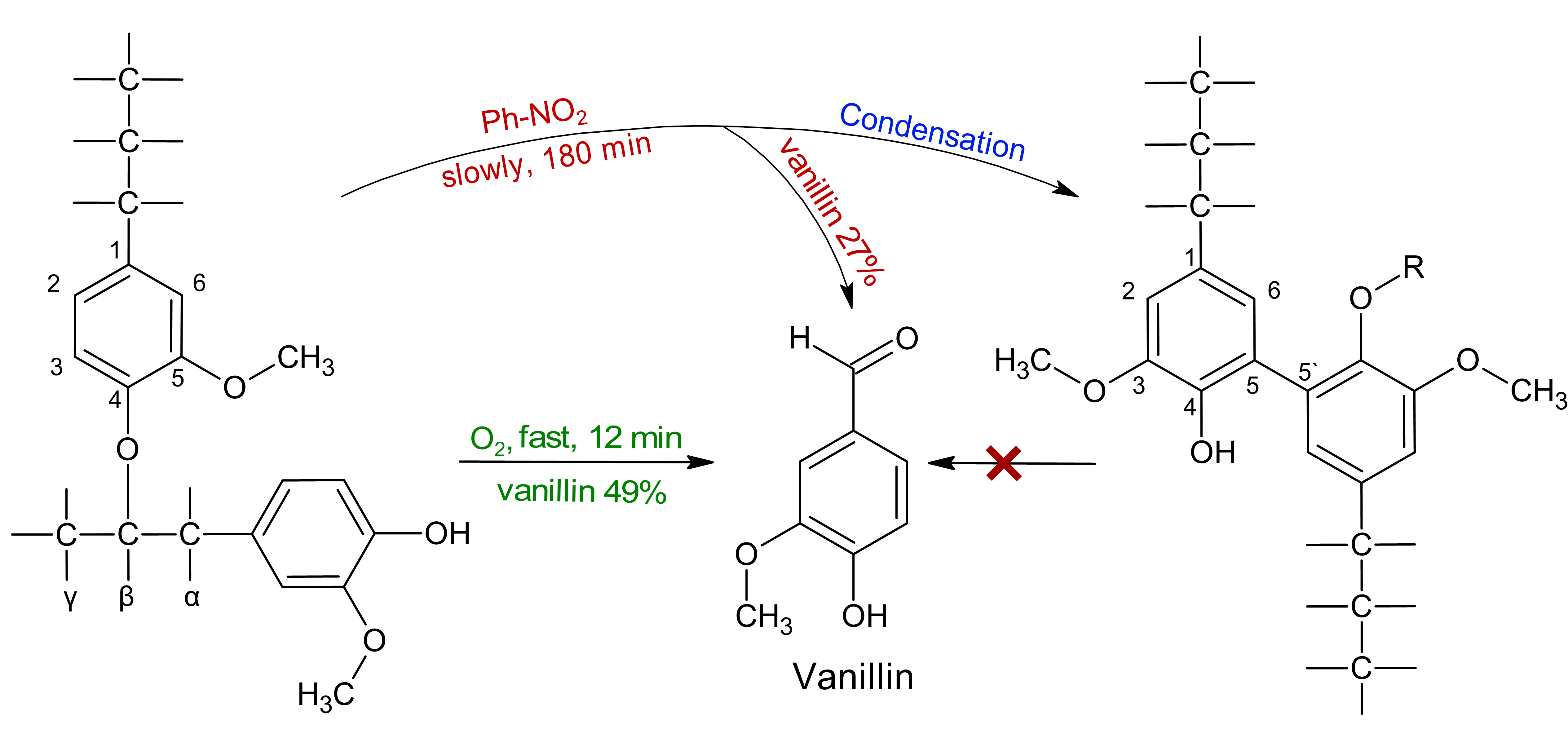
Keywords:
1. Introduction
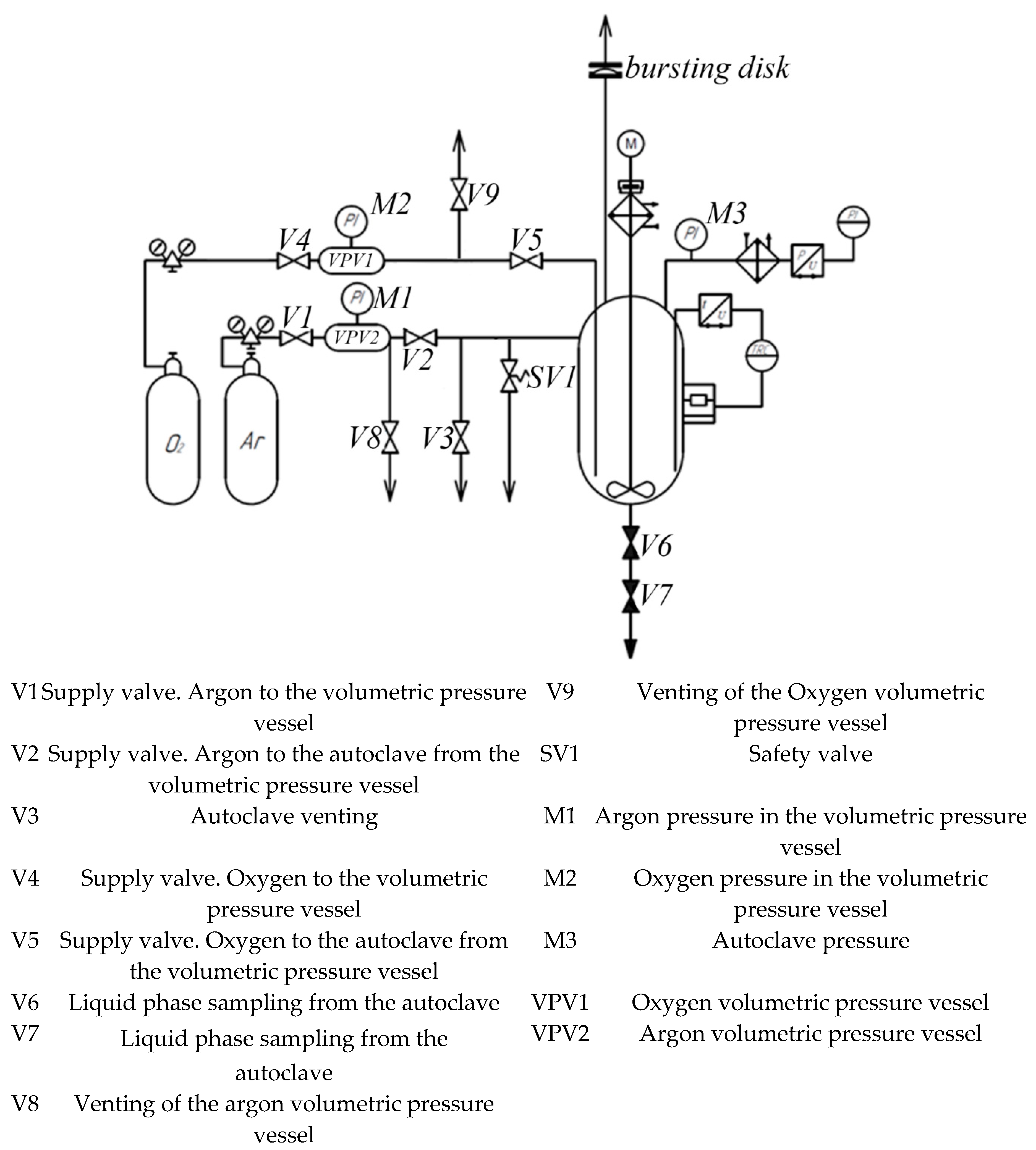
2. Materials and Methods
3. Results and Discussion
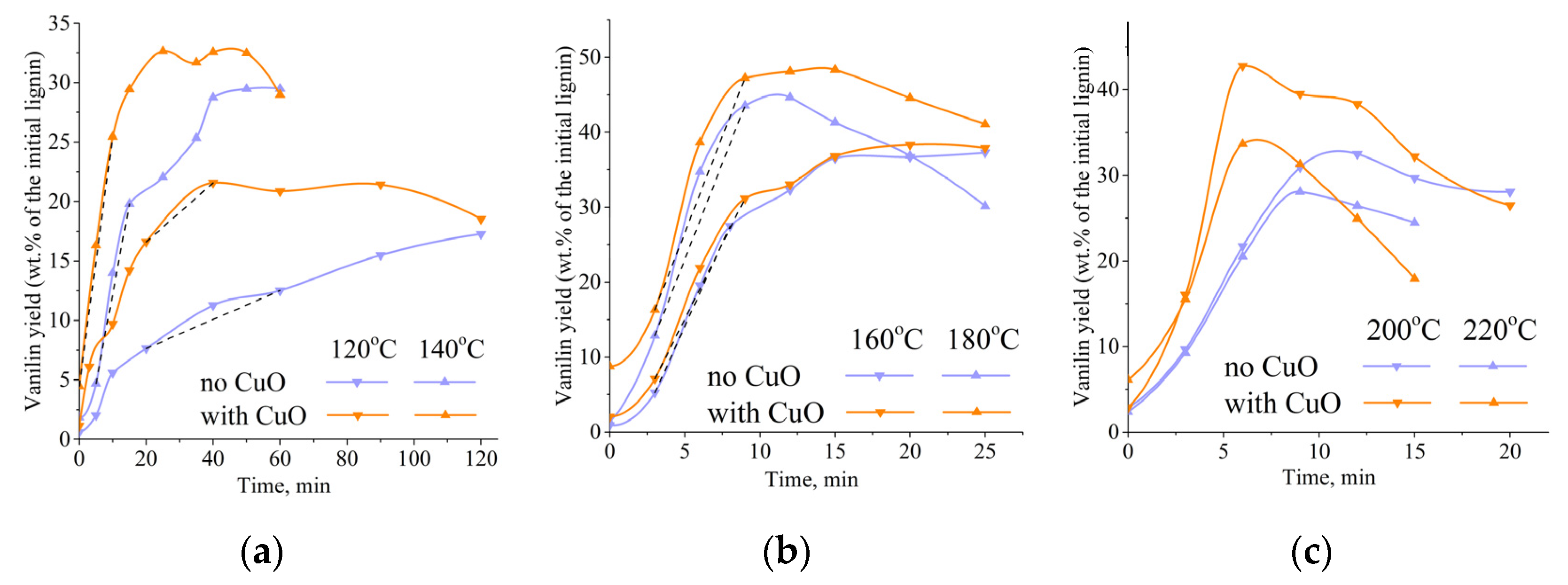
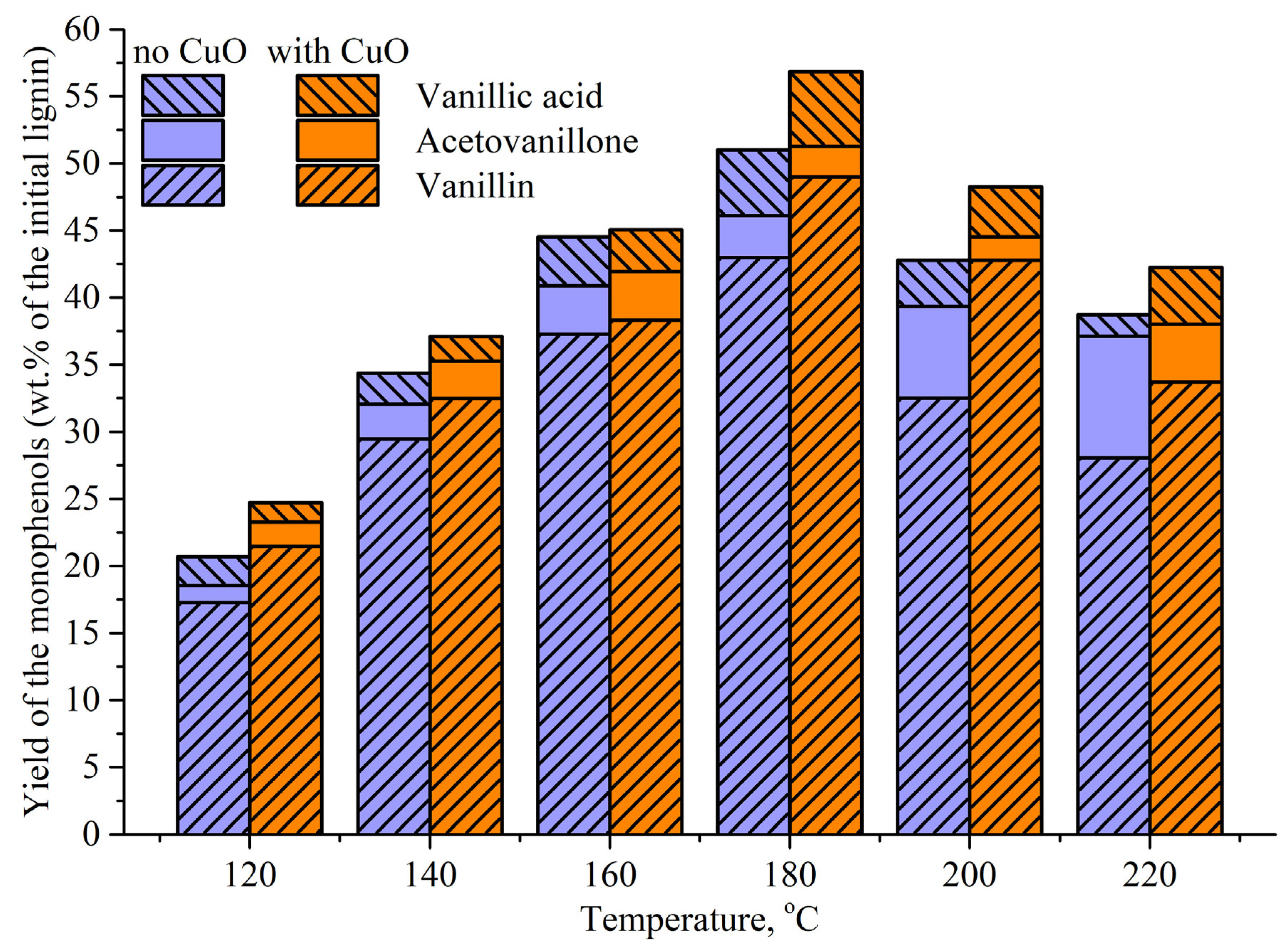
3.1. Influence of Temperature on Vanillin Formation Rate and Its Maximum Yield
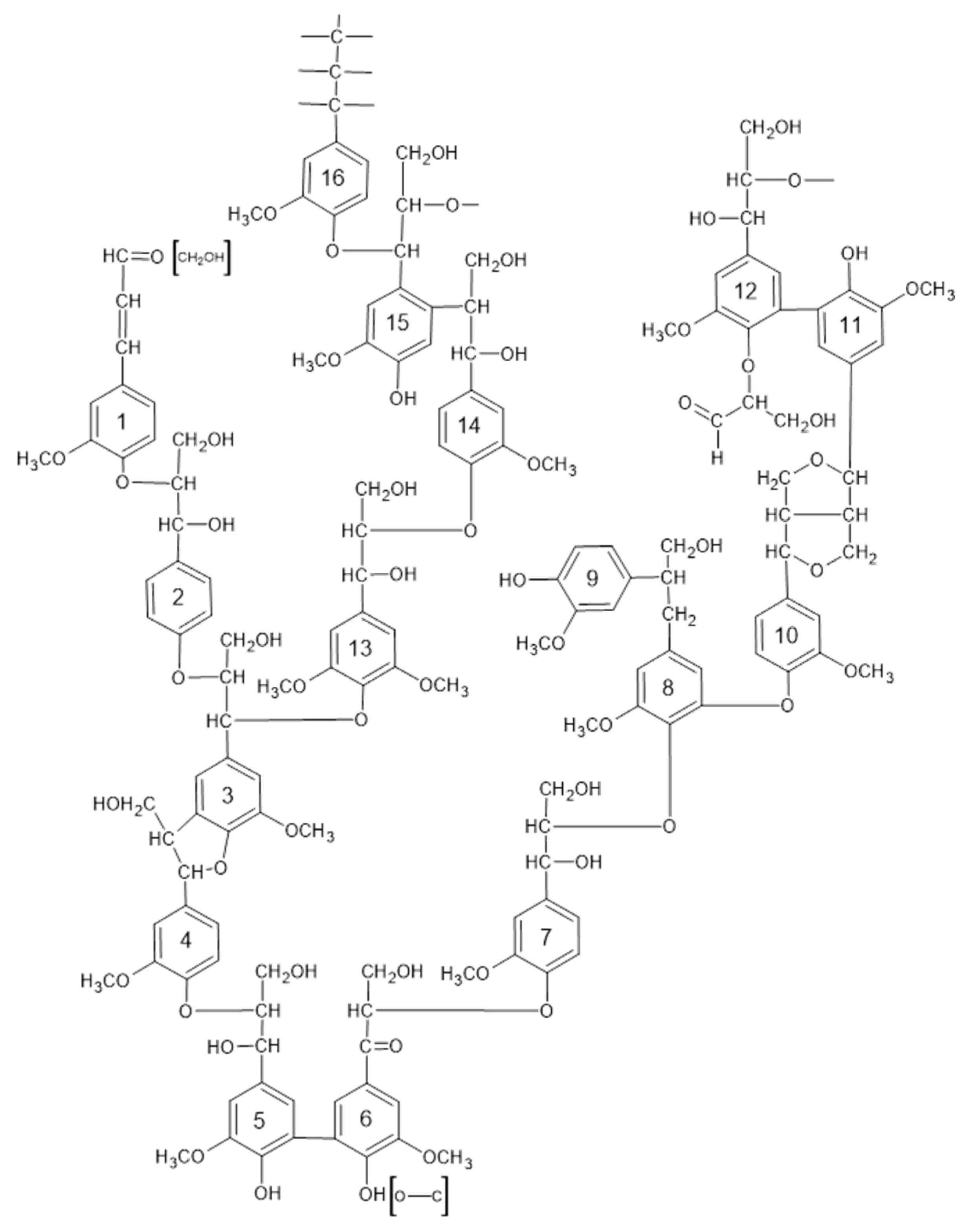
3.2. Mechanisms for Influence of Temperature and the Process Rate on the Yield of Vanillin and the Monophenols
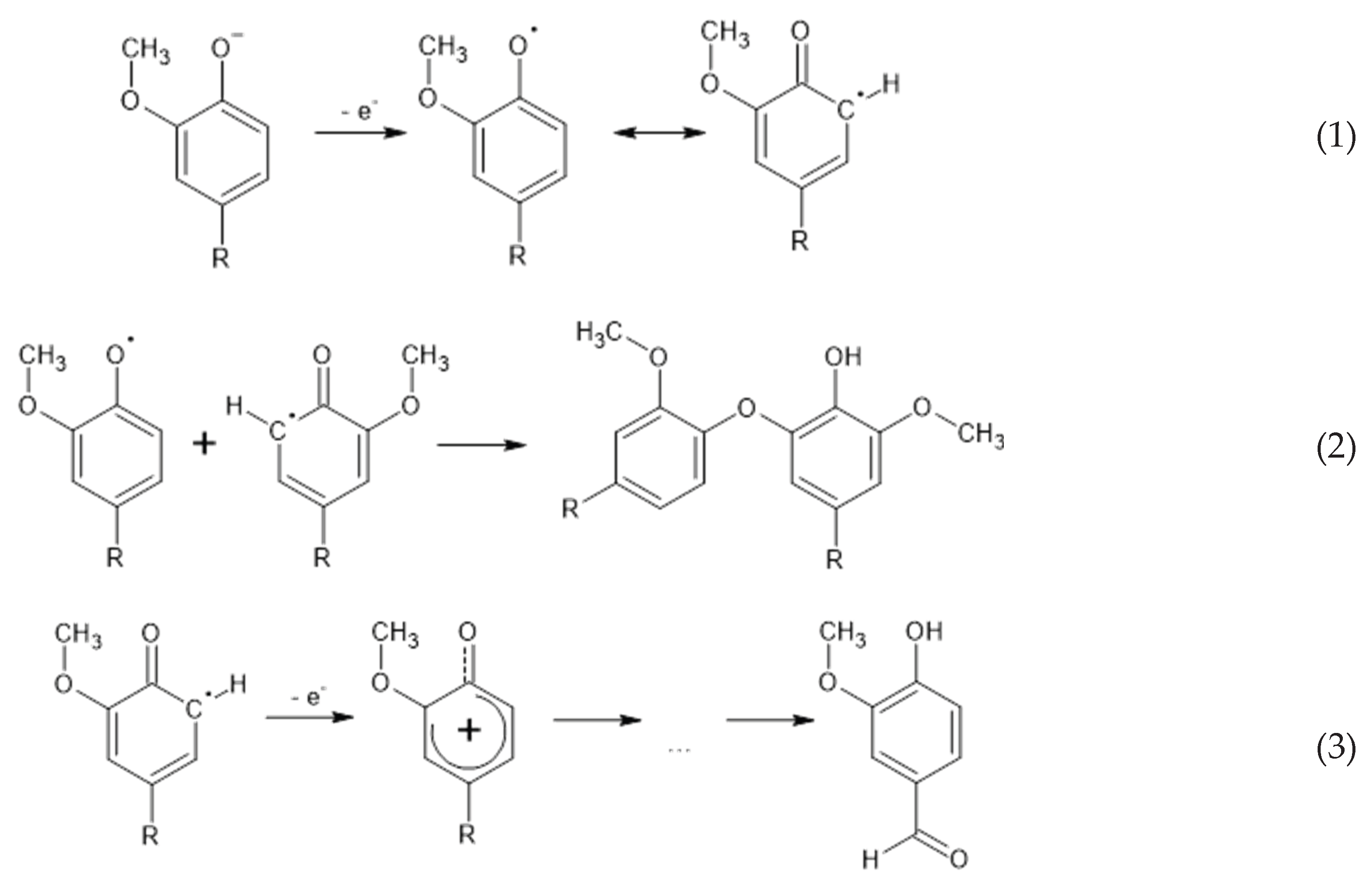
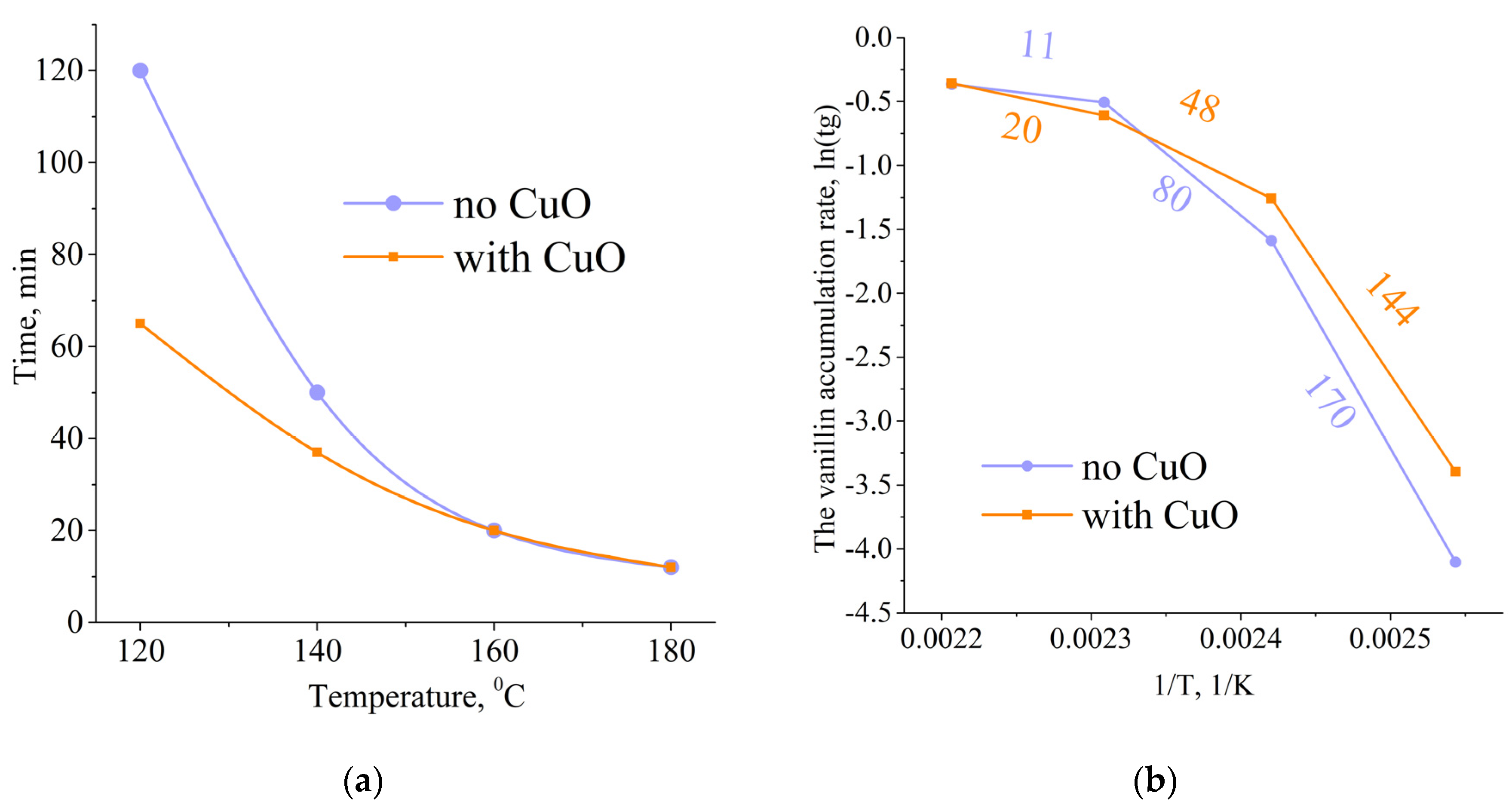
3.3. Influence of a Catalyst on the Oxidation Rate and the Vanillin Yield
3.4. Influence of Temperature on the Apparent Activation Energy of Pine Native Lignin Oxidation
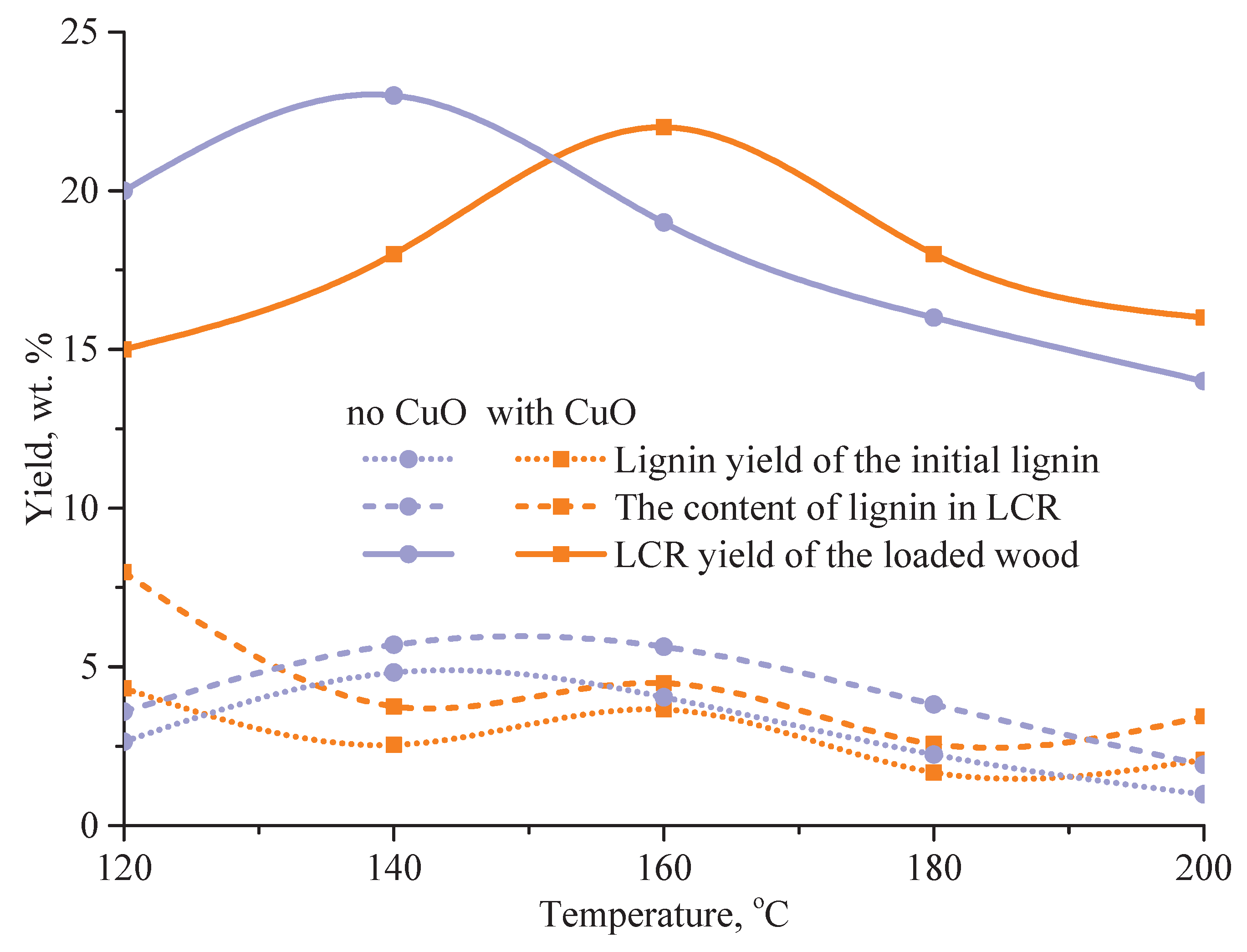
3.5. Influence of Temperature on Cellulose Yield in Oxidation of Pine Wood Lignin
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Song, J.X.; Zhang, H.J.; Niu, M.H.; Guo, Y.Z.; Li, H.M. Research progress on vanillin synthesis by catalytic oxidation of lignin: A review. Ind. Crop. Prod. 2024, 214, 118443. [Google Scholar] [CrossRef]
- Tarabanko, V.E.; Tarabanko, N. Catalytic oxidation of lignins into the aromatic aldehydes: general process trends and development prospects. Int. J. Mol. Sci. 2017, 18, 2421. [Google Scholar] [CrossRef]
- Zhu, Y., Liao, Y., Lv, W., Liu, J., Song, X., Chen, L., ... Ma, L. Complementing vanillin and cellulose production by oxidation of lignocellulose with stirring control. ACS Sustain. Chem. Eng. 2020, 8, 2361–2374. [CrossRef]
- Zhu, Y., Liao, Y., Lu, L., Lv, W., Liu, J., Song, X., ... Sels, B. F. Oxidative catalytic fractionation of lignocellulose to high-yield aromatic aldehyde monomers and pure cellulose. Acs Catal. 2023, 13, 7929–7941. [CrossRef]
- Koropachinskaya, N.V.; Tarabanko, V.E.; Chernyak, M.Y. Catalytic oxidation of birch wood (Betula Pendula Roth.) with oxygen in syringaldehyde and vanillin. Khimiya Rastit. Syr’ya 2003, 2, 9–14. [Google Scholar]
- Pacek, A.W.; Ding, P.; Garrett, M.; Sheldrake, G.; Nienow, A.W. Catalytic conversion of sodium lignosulfonate to vanillin: engineering aspects. Part 1. Effects of processing conditions on vanillin yield and selectivity. Ind. Eng. Chem. Res. 2013, 52, 8361–8372. [Google Scholar] [CrossRef]
- Leopold, B.E.N.G.T.; Malmstrom, L.L. Studies on lignin. IV. Investigation on the nitrobenzene oxidation products of lignin from different woods by paper partition chromatography. 1951. [Google Scholar] [CrossRef]
- Gogotov, A.F.; Rybal'chenko, N.A.; Makovskaya, T.I.; Babkin, V.A. Catalytic nitrobenzene oxidation of lignins. Russ. Chem. Bull. 1996, 45, 2854–2857. [Google Scholar] [CrossRef]
- Tarabanko, V.E.; Kaygorodov, K.L.; Vigul, D.O.; Tarabanko, N.; Chelbina, Y.V.; Smirnova, M.A. Influence of acid prehydrolysis on the process of wood oxidation into vanillin and pulp. J. Wood Chem. Technol. 2020, 40, 421–433. [Google Scholar] [CrossRef]
- Schoefel, E.W. Manufacture of vanillin. US Pat. 2598311, 27.05.1952.
- Smirnova, M.A.; Taraban'ko, V.E.; Chelbina Yu, V.; Kajgorodov, K.L.; Oreshina, I.D. Rostostimuliruyushhaya aktivnost` lignokislot–pobochnogo produkta okisleniya rastitel`nogo sy`r`ya v vanilin i cellyulozu. Khimiya Rastit. Syr’ya 2024, (4), 427–437. [Google Scholar] [CrossRef]
- Smirnova, M.A.; Taraban'ko, V.E.; Kajgorodov, K.L.; Korsakov, A.V.; Golubkov, V.A.; Chelbina Yu, V. Vliyanie lignokislot, pobochnogo produkta okislitelnoj pererabotki sosnovoj i osinovoj drevesiny v vanilin i cellylozu, na prorastanie semyan redisa raphanus sativus var radicula. Khimiya Rastit. Syr’ya 2025, (4), 456–465. [Google Scholar] [CrossRef]
- Taraban'ko, V.E.; Koropatchinskaya, N.V.; Kudryashev, A.V.; Kuznetsov, B.N. Influence of lignin origin on the efficiency of the catalytic oxidation of lignin into vanillin and syringaldehyde. Russ. Chem. Bull. 1995, 44, 367–371. [Google Scholar] [CrossRef]
- Tarabanko, V.E.; Vigul, D.O.; Kaygorodov, K.L.; Kosivtsov, Y.; Tarabanko, N.; Chelbina, Y.V. Influence of mass transfer and acid prehydrolysis on the process of flax shives catalytic oxidation into vanillin and pulp. Biomass Convers. Biorefinery 2024, 14, 489–499. [Google Scholar] [CrossRef]
- Tarabanko, V.E.; Kaygorodov, K.L.; Kazachenko, A.S.; Smirnova, M.A.; Chelbina, Y.V.; Kosivtsov, Y.; Golubkov, V.A. Mass transfer in the processes of native lignin oxidation into vanillin via oxygen. Catalysts 2023, 13, 1490. [Google Scholar] [CrossRef]
- Buranov, A.U.; Mazza, G.J.I.C. Lignin in straw of herbaceous crops. Ind. Crop. Prod. 2008, 28, 237–259. [Google Scholar] [CrossRef]
- Wu, G.; Heitz, M.; Chornet, E. Improved alkaline oxidation process for the production of aldehydes (vanillin and syringaldehyde) from steam-explosion hardwood lignin. Ind. Eng. Chem. Res. 1994, 33, 718–723. [Google Scholar] [CrossRef]
- Min, D.; Xiang, Z.; Liu, J.; Jameel, H.; Chiang, V.; Jin, Y.; Chang, H.M. Improved protocol for alkaline nitrobenzene oxidation of woody and non-woody biomass. J. Wood Chem. Technol. 2015, 35, 52–61. [Google Scholar] [CrossRef]
- Zhu, Y.; Li, N.; Liu, H.; Cai, C.; Wang, Y.; Mu, J.; Wang, F. Oxidative catalytic fractionation of lignocellulose enhanced by copper–manganese-doped CeO2. ACS Catal. 2024, 14, 16872–16884. [Google Scholar] [CrossRef]
- Tamai, A.; Goto, H.; Akiyama, T.; Matsumoto, Y. Revisiting alkaline nitrobenzene oxidation: quantitative evaluation of biphenyl structures in cedar wood lignin (Cryptomeria japonica) by a modified nitrobenzene oxidation method. Holzforschung 2015, 69, 951–958. [Google Scholar] [CrossRef]
- Tarabanko V.E., e.a. Khimiya Rastitel’nogo Syr’ya, 2026, in press.
- Freudenberg, K. Lignin: Its Constitution and Formation from p-Hydroxycinnamyl Alcohol: Lignin is duplicated by dehydrogenation of these alcohols; intermediates explain formation and structure. Science 1965, 148, 595–600. [Google Scholar] [CrossRef] [PubMed]
- Song, Q.; Wang, F.; Cai, J.; Wang, Y.; Zhang, J.; Yu, W.; Xu, J. Lignin depolymerization (LDP) in alcohol over nickel-based catalysts via a fragmentation–hydrogenolysis process. Energy Environ. Sci. 2013, 6, 994–1007. [Google Scholar] [CrossRef]
- Evstigneyev, E.I. Selective depolymerization of lignin: Assessment of yields of monomeric products. J. Wood Chem. Technol. 2018, 38, 409–415. [Google Scholar] [CrossRef]
- Adler, E. Lignin chemistry—past, present and future. Wood Sci. Technol. 1977, 11, 169–218. [Google Scholar] [CrossRef]
- Abu-Omar, M. M.; Barta, K.; Beckham, G. T.; Luterbacher, J. S.; Ralph, J.; Rinaldi, R.; Wang, F. Guidelines for performing lignin-first biorefining. Energy Environ. Sci. 2021, 14, 262–292. [Google Scholar] [CrossRef]
- Tarabanko, V.E.; Petukhov, D.V.; Selyutin, G.E. New mechanism for the catalytic oxidation of lignin to vanillin. Kinet. Catal. 2004, 45, 569–577. [Google Scholar] [CrossRef]
- Nonhebel, D.C.; Walton, J.C. Free-radical chemistry: structure and mechanism; CUP Archive, 1974. [Google Scholar]
- Garcia-Rollan, M., Rivas-Marquez, M. N., Bertran-Llorens, S., Deuss, P. J., Ruiz-Rosas, R., Rosas, J. M., ... Cordero, T. Biobased vanillin production by oxidative depolymerization of kraft lignin on a nitrogen-and phosphorus-functionalized activated carbon catalyst. Energy Fuels 2024, 38, 7018–7032. [CrossRef]
- Abdelaziz, O.Y.; Clemmensen, I.; Meier, S.; Bjelić, S.; Hulteberg, C.P.; Riisager, A. Oxidative depolymerization of Kraft Lignin to Aromatics over bimetallic V–Cu/ZrO2 catalysts. Top. Catal. 2023, 66, 1369–1380. [Google Scholar] [CrossRef]
- Schutyser, W., Kruger, J. S., Robinson, A. M., Katahira, R., Brandner, D. G., Cleveland, N. S., ... Beckham, G. T. Revisiting alkaline aerobic lignin oxidation. Green. Chem. 2018, 20, 3828–3844. [CrossRef]
- Pen, R.Z.; Shapiro, I.L. Kinetics of soda cooking of coniferous wood in wide temperature range. Russ. J. Appl. Chem. 1995, 68, 1536–1538. [Google Scholar]
- Pen, R.Z.; Shapiro, I.L.; Pen, V.R. Kinetics of soda-anthraquinone pulping of pine wood. 1989. [Google Scholar]
- Buwa, V.V.; Roy, S.; Ranade, V.V. Three-phase slurry reactors. Multiph. Catal. React. Theory Des. Manuf. Appl. 2016, 132–155. [Google Scholar] [CrossRef]
- Steinfeld, J.I.; Francisco, J.S.; Hase, W.L. Chemical kinetics and dynamics; Prentice Hall: Upper Saddle River, NJ, 1999; Vol. 2. [Google Scholar]
- Hafezisefat, P.; Rahic, E.; Brown, R.C. Oxidative fractionation of biomass to produce phenolic monomers and processable carbohydrate pulp. React. Chem. Eng. 2025, 10, 2644–2656. [Google Scholar] [CrossRef]
- Tarabanko, V. E., Kaygorodov, K. L., Skiba, E. A., Tarabanko, N., Chelbina, Y. V., Baybakova, O. V., ... Djakovitch, L. Processing pine wood into vanillin and glucose by sequential catalytic oxidation and enzymatic hydrolysis. J. Wood Chem. Technol. 2017, 37, 43–51. [CrossRef]
| Oxidation conditions | With the catalyst | Without catalyst | ||||||
| Mean value and arithmetic mean error | Mean value and arithmetic mean error | |||||||
| Oxidation duration (min) | 15 | 12 | 12 | 12 | 12 | 12 | ||
| Vanillin (wt.% of the initial lignin) | 48.6 | 50.4 | 48.3 | 49.1±0.87 | 41.2 | 40.5 | 44.6 | 42.1±1.7 |
| Acetovanillone (wt.% of the initial lignin) | 4.02 | 2.99 | 2.27 | 3.09±0.61 | 3.26 | 5.71 | 3.11 | 4.03±1.12 |
| Vanillic acid (wt.% of the initial lignin) | 3.11 | 5.15 | 5.56 | 5.25±0.85 | 4.73 | 4.54 | 4.91 | 4.73±0.73 |
| Sum of the monophenols (wt.% of the initial lignin) | 55.76 | 58.58 | 56.13 | 56.8±1.2 | 50.23 | 50.75 | 52.65 | 51.2±0.96 |
| Sum of the monophenols (mol.% of the initial lignin) | 64.84 | 68.01 | 65.27 | 66.04±1,83 | 57.05 | 58.7 | 61.08 | 58.94±2.01 |
| References | Oxidant | Vanillin yield (wt.% of the initial lignin) | Vanillin yield (mol.% of the initial lignin) | Yield of the monophenols (wt.% of the initial lignin) | Yield of the mono-phenols (mol.% of the initial lignin) | Vanillin concentration (g/L) | Oxidation duration (min) |
|---|---|---|---|---|---|---|---|
| [7] | Nitrobenzene | 27 | 32 | - | - | - | 180 |
| [8] | 36 | 42 | - | - | 9.6 | 180 | |
| [18] | 35-40 | 41-47 | 39-43 | 45-50 | - | 90-120 | |
| [4] | - | - | 30.2 | - | - | 120 | |
| [13] | Molecular oxygen | 23 | 27 | - | - | 4.5 | 20 |
| [4] | 32 | 38 | 33.8 | - | 0.97 | 60 | |
| [9] | 25-28 | 29-33 | - | - | - | - | |
| [21] | 32-37 | 38-44 | 38-42 | 44-49 | 5 | 9-15 | |
| This paper | 42-49 | 49-58 | 51-57 | 59-66 | 5-6.5 | 9-15 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




