Submitted:
24 April 2026
Posted:
28 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
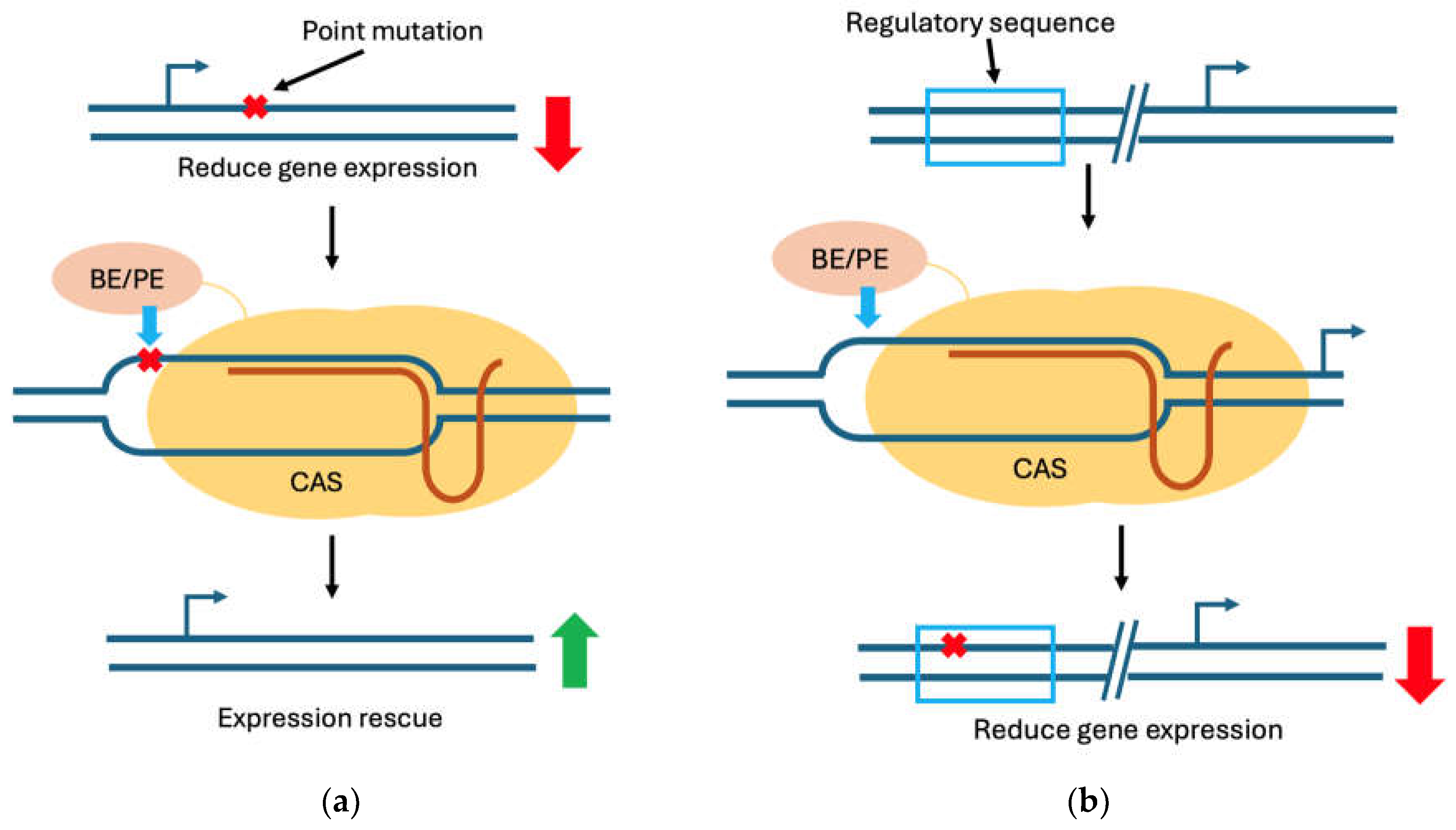
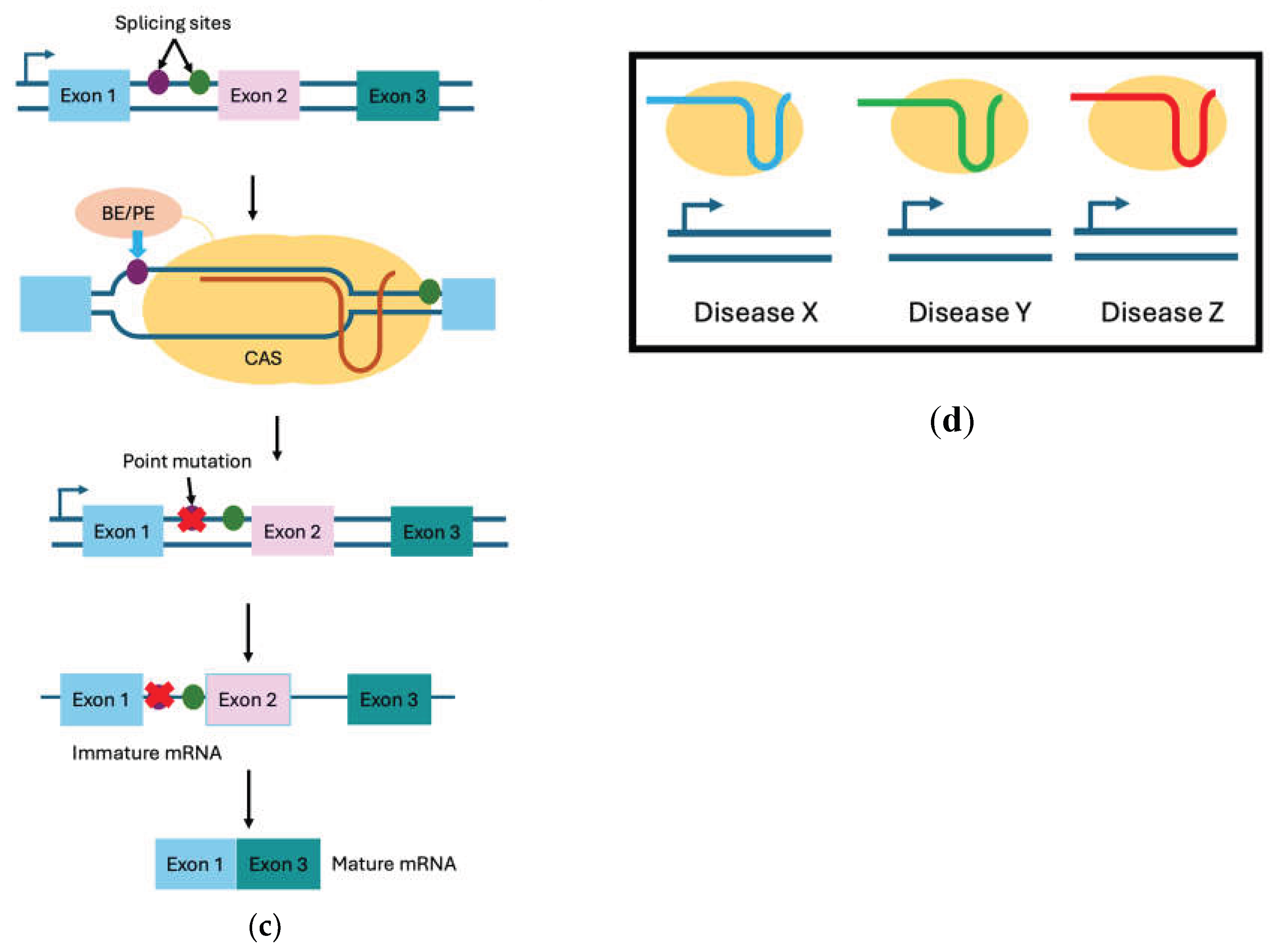
2. CRISPR Mechanisms of Action and Toolkits
2.1. Double-Strand Break–Dependent CRISPR Systems
2.2. Base Editing Systems
2.3. Prime Editing Systems
2.4. Delivery Systems for CRISPR Machinery
3. Personalized Medicine Applications
4. CRISPR-Based Gene Editing Therapies Case Studies
4.1. Base Editing for the Treatment of CSP1 Deficiency
4.2. Base Editing for the Treatment of Duchenne Muscular Dystrophy
4.3. Prime Editing for the Treatment of p47phox-Deficient Chronic Granulomatous Disease
4.4. Base-Editing for the Treatment of Familial Hypercholesterolemia
5. Challenges in Platform Development of CRISPR-Based Gene Editing Therapies
5.1. Delivery Barriers
5.1.1. Viral Vectors
5.1.1. Lipid Nanoparticles
5.2. Off-Target Effects
5.3. Ethical Concerns
5.4. Scalability and Regulatory Legislation
6. Interventional Genetics
7. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wu, D.W.; Bernstein, J.A.; Bejerano, G. Discovering monogenic patients with a confirmed molecular diagnosis in millions of clinical notes with MonoMiner. Genet. Med. 2022, 24(10), 2091–2102. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Wu, Z.-Y. Gene therapy for monogenic disorders: challenges, strategies, and perspectives. J. Genet. Genom. 2024, 51(2), 133–143. [Google Scholar] [CrossRef]
- Wang, Y.; et al. Gene therapy strategies for rare monogenic disorders with nuclear or mitochondrial gene mutations. Biomaterials 2021, 277, 121108. [Google Scholar] [CrossRef] [PubMed]
- Yang, G.; et al. The national economic burden of rare disease in the United States in 2019. Orphanet J. Rare Dis. 2022, 17(1), 163. [Google Scholar] [CrossRef]
- Pavel-Dinu, M.; Borna, S.; Bacchetta, R. Rare immune diseases paving the road for genome editing-based precision medicine. In Frontiers in Genome Editing; 2023; pp. 5–2023. [Google Scholar]
- Pradhan, A.; Kalin, T.V.; Kalinichenko, V.V. Genome Editing for Rare Diseases. Curr. Stem Cell. Rep. 2020, 6(3), 41–51. [Google Scholar] [CrossRef]
- Anzalone, A.V.; Koblan, L.W.; Liu, D.R. Genome editing with CRISPR–Cas nucleases, base editors, transposases and prime editors. Nat. Biotechnol. 2020, 38(7), 824–844. [Google Scholar] [CrossRef]
- Yang, X.; et al. Exploring the Potential and Challenges of CRISPR Delivery and Therapeutics for Genetic Disease Treatment. Adv. Funct. Mater. 2024, 34(38), 2402630. [Google Scholar] [CrossRef]
- Newby, G.A.; Liu, D.R. Mol. Ther. 2021; 29, 11, pp. 3107–3124.
- Ahrens-Nicklas, R.C.; Musunuru, K. How to create personalized gene editing platforms: Next steps toward interventional genetics. Am. J. Hum. Genet. 2025, 112(12), 2826–2829. [Google Scholar] [CrossRef]
- Amitai, G.; Sorek, R. CRISPR–Cas adaptation: insights into the mechanism of action. Nat. Rev. Microbiol. 2016, 14(2), 67–76. [Google Scholar] [CrossRef]
- Nussenzweig, P.M.; Marraffini, L.A. Molecular Mechanisms of CRISPR-Cas Immunity in Bacteria. Annu. Rev. Genet. 2020, 2020. 54((Volume 54), 93–120. [Google Scholar] [CrossRef] [PubMed]
- Hille, F.; et al. The Biology of CRISPR-Cas: Backward and Forward. Cell 2018, 172(6), 1239–1259. [Google Scholar] [CrossRef]
- Jiang, F.; Doudna, J.A. CRISPR–Cas9 Structures and Mechanisms. Annu. Rev. Biophys. 2017, 46((Volume 46), 505–529. [Google Scholar] [CrossRef]
- Staals, R.H.J.; et al. Interference-driven spacer acquisition is dominant over naive and primed adaptation in a native CRISPR–Cas system. Nat. Commun. 2016, 7(1), 12853. [Google Scholar] [CrossRef]
- Li, T.; et al. CRISPR/Cas9 therapeutics: progress and prospects. Signal Transduct. Target. Ther. 2023, 8(1), 36. [Google Scholar] [CrossRef]
- Saber Sichani, A.; et al. A Review on Advanced CRISPR-Based Genome-Editing Tools: Base Editing and Prime Editing. Mol. Biotechnol. 2023, 65(6), 849–860. [Google Scholar] [PubMed]
- Liao, H.; et al. CRISPR-Cas9-mediated homology-directed repair for precise gene editing. Mol. Ther. Nucleic Acids 2024, 35(4). [Google Scholar]
- Fichter, K.M.; Setayesh, T.; Malik, P. Strategies for precise gene edits in mammalian cells. Mol. Ther. Nucleic Acids 2023, 32, 536–552. [Google Scholar] [CrossRef] [PubMed]
- Liao, Q.; et al. Global prevalence of myotonic dystrophy: an updated systematic review and meta-analysis. Neuroepidemiology 2022, 56(3), 163–173. [Google Scholar] [CrossRef]
- Jin, Y.-Y.; et al. Enhancing homology-directed repair efficiency with HDR-boosting modular ssDNA donor. Nat. Commun. 2024, 15(1), 6843. [Google Scholar] [CrossRef]
- Mengistu, G.; et al. CRISPR–Cas Systems in Human Disease Therapy: Advances, Clinical Applications, Limitations, and Future Directions. J. Gene Med. 2026, 28(4), e70091. [Google Scholar] [CrossRef]
- Lee, J.; Kweon, J.; Kim, Y. Emerging trends in prime editing for precision genome editing. Exp. Mol. Med. 2025, 57(7), 1381–1391. [Google Scholar] [CrossRef]
- Zhao, Z.; et al. Prime editing: advances and therapeutic applications. Trends Biotechnol. 2023, 41(8), 1000–1012. [Google Scholar] [CrossRef]
- Rees, H.A.; Liu, D.R. Base editing: precision chemistry on the genome and transcriptome of living cells. Nat. Rev. Genet. 2018, 19(12), 770–788. [Google Scholar] [CrossRef]
- Mawson, S.J.; et al. Precision genome editing with DNA base editors. Nat. Rev. Methods Prim. 2026, 6(1), 23. [Google Scholar] [CrossRef]
- Lee, J.; et al. Prime editing with genuine Cas9 nickases minimizes unwanted indels. Nat. Commun. 2023, 14(1), 1786. [Google Scholar] [CrossRef] [PubMed]
- Gao, H.; et al. Research progress of base editing and prime editing tools based on the CRISPR/Cas system. Mol. Ther. Nucleic Acids 2025, 36(4). [Google Scholar] [CrossRef] [PubMed]
- Porto, E.M.; et al. Base editing: advances and therapeutic opportunities. Nat. Rev. Drug. Discov. 2020, 19(12), 839–859. [Google Scholar] [CrossRef]
- Perrotta, R.M.; et al. Engineered base editors with reduced bystander editing through directed evolution. Nat. Biotechnol. 2025. [Google Scholar] [CrossRef] [PubMed]
- Valdez, I.; et al. A streamlined base editor engineering strategy to reduce bystander editing. Nat. Commun. 2025, 16(1), 8115. [Google Scholar] [CrossRef]
- Tan, J.; et al. Engineering of high-precision base editors for site-specific single nucleotide replacement. Nat. Commun. 2019, 10(1), 439. [Google Scholar] [CrossRef]
- Murray, J.B.; Harrison, P.T.; Scholefield, J. Prime editing: therapeutic advances and mechanistic insights. Gene Ther. 2025, 32(2), 83–92. [Google Scholar] [CrossRef]
- Brooks, I.R.; et al. Prime editing in mammals: From promise to practicalities. Mol. Ther. Nucleic Acids 2025, 36(4). [Google Scholar] [CrossRef]
- Zhang, G.; et al. Enhancement of prime editing via xrRNA motif-joined pegRNA. Nat. Commun. 2022, 13(1), 1856. [Google Scholar] [CrossRef]
- Cavazza, A.; et al. Advanced delivery systems for gene editing: A comprehensive review from the GenE-HumDi COST Action Working Group. Mol. Ther. Nucleic Acids 2025, 36(1), 102457. [Google Scholar] [CrossRef] [PubMed]
- Geng, G.; et al. Viral and non-viral vectors in gene therapy: current state and clinical perspectives. eBioMedicine 2025, 118. [Google Scholar] [CrossRef] [PubMed]
- Bulcha, J.T.; et al. Viral vector platforms within the gene therapy landscape. Signal Transduct. Target. Ther. 2021, 6(1), 53. [Google Scholar] [CrossRef]
- Mohsen, M.O.; Bachmann, M.F. Virus-like particle vaccinology, from bench to bedside. Cell. Mol. Immunol. 2022, 19(9), 993–1011. [Google Scholar] [CrossRef] [PubMed]
- Hou, X.; et al. Lipid nanoparticles for mRNA delivery. Nat. Rev. Mater. 2021, 6(12), 1078–1094. [Google Scholar] [CrossRef]
- The Lancet Global, H. The landscape for rare diseases in 2024. Lancet Glob. Health 2024, 12(3), e341. [Google Scholar] [CrossRef]
- Fermaglich, L.J.; Miller, K.L. A comprehensive study of the rare diseases and conditions targeted by orphan drug designations and approvals over the forty years of the Orphan Drug Act. Orphanet J. Rare Dis. 2023, 18(1), 163. [Google Scholar] [CrossRef]
- Musunuru, K.; et al. Patient-Specific In Vivo Gene Editing to Treat a Rare Genetic Disease. N. Engl. J. Med. 2025, 392(22), 2235–2243. [Google Scholar] [CrossRef]
- Kluesner, M.G.; et al. CRISPR-Cas9 cytidine and adenosine base editing of splice-sites mediates highly-efficient disruption of proteins in primary and immortalized cells. Nat. Commun. 2021, 12(1), 2437. [Google Scholar] [CrossRef]
- Fontana, L.; et al. Multiplex base editing of BCL11A regulatory elements to treat sickle cell disease. Cell. Rep. Med. 2025. 6, 10. [Google Scholar] [CrossRef] [PubMed]
- Levesque, S.; Bauer, D.E. CRISPR-based therapeutic genome editing for inherited blood disorders. Nat. Rev. Drug. Discov. 2025, 24(12), 907–925. [Google Scholar] [CrossRef] [PubMed]
- Zebanaz, A.; et al. Personalized Medicine in Treating Rare Genetic Disorders: A Review. J. Pharm. Bioallied Sci. 2025. 17, (Suppl 1), S59–S62. [Google Scholar] [CrossRef] [PubMed]
- Cabré-Romans, J.-J.; Cuella-Martin, R. CRISPR-dependent base editing as a therapeutic strategy for rare monogenic disorders. In Frontiers in Genome Editing; 2025; pp. 7–2025. [Google Scholar]
- Wu, T.; Hu, Y.; Tang, L.V. Gene therapy for polygenic or complex diseases. Biomark. Res. 2024, 12(1), 99. [Google Scholar] [CrossRef]
- Khan, A.; et al. Genomic medicine and personalized treatment: a narrative review. Ann. Med. Surg. 2025, 87(3), 1406–1414. [Google Scholar] [CrossRef]
- Abbas, A.; et al. Orphan drug policies in different countries. J. Pharm. Health Serv. Res. 2019, 10(3), 295–302. [Google Scholar] [CrossRef]
- Kim-McManus, O.; et al. A framework for N-of-1 trials of individualized gene-targeted therapies for genetic diseases. Nat. Commun. 2024, 15(1), 9802. [Google Scholar] [CrossRef]
- Dong, H.; et al. Clinical features and CPS1 variants in Chinese patients with carbamoyl phosphate synthetase 1 deficiency. BMC Pediatr. 2024, 24(1), 539. [Google Scholar] [CrossRef]
- Zhang, L.; et al. Unraveling the therapeutic potential of carbamoyl phosphate synthetase 1 (CPS1) in human diseases. Bioorganic Chem. 2023, 130, 106253. [Google Scholar] [CrossRef]
- Wang, S.; et al. Clinical and genetic analysis of a case of late onset carbamoyl phosphate synthase I deficiency caused by CPS1 mutation and literature review. BMC Med. Genom. 2023, 16(1), 145. [Google Scholar] [CrossRef]
- Duan, D.; et al. Duchenne muscular dystrophy. Nat. Rev. Dis. Prim. 2021, 7(1), 13. [Google Scholar] [CrossRef]
- Haque, U.S.; Yokota, T. Gene editing for duchenne muscular dystrophy: from experimental models to emerging therapies. Degener. Neurol. Neuromuscul. Dis. 2025, 17–40. [Google Scholar] [CrossRef]
- Echevarría, L.; Aupy, P.; Goyenvalle, A. Exon-skipping advances for Duchenne muscular dystrophy. Hum. Mol. Genet. 2018, 27(R2), R163–R172. [Google Scholar] [CrossRef] [PubMed]
- HuidaGene Therapeutics Co., L. NCT06594094; An Open-label, Multidose Dose-escalation Study to Understand the Safety of CRISPR Gene-editing Therapy and Its Long-Lasting Effects in DMD Patients (MUSCLE). cited. 2026.
- Shimizu-Motohashi, Y. Gene therapy for Duchenne muscular dystrophy. Brain Dev. 2025, 47(5), 104424. [Google Scholar] [CrossRef]
- Aartsma-Rus, A.; et al. The Dilemma of Choice for Duchenne Patients Eligible for Exon 51 Skipping The European Experience. J. Neuromuscul. Dis. 2023, 10(3), 315–325. [Google Scholar] [CrossRef]
- A Single-arm, Open-label, Single-center Study to Evaluate the Safety and Tolerability of Intravenous GEN6050X Gene Therapy in Ambulatory Boys With Duchenne Muscular Dystrophy (DMD). 2024.
- Jin, M.; et al. Correction of human nonsense mutation via adenine base editing for Duchenne muscular dystrophy treatment in mouse. Mol. Ther. Nucleic Acids 2024, 35(2). [Google Scholar] [CrossRef]
- Yu, H.-H.; Yang, Y.-H.; Chiang, B.-L. Chronic Granulomatous Disease: a Comprehensive Review. Clin. Rev. Allergy Immunol. 2021, 61(2), 101–113. [Google Scholar] [CrossRef] [PubMed]
- Chiriaco, M.; et al. Chronic granulomatous disease: clinical, molecular, and therapeutic aspects. Pediatr. Allergy Immunol. 2016, 27(3), 242–253. [Google Scholar] [CrossRef] [PubMed]
- Gori, J.L.; et al. Prime Editing for p47phox-Deficient Chronic Granulomatous Disease. N. Engl. J. Med. 2026, 394(12), 1195–1203. [Google Scholar] [CrossRef]
- A Phase 1/2 Study Evaluating Gene Therapy by Transplantation of Autologous CD34+ Stem Cells Modified Ex Vivo Using Prime Editing (PM359) in Participants With Autosomal Recessive Chronic Granulomatous Disease Due to Mutations in the NCF1 Gene. 2024.
- Varghese, M.J. Familial hypercholesterolemia: A review. Ann. Pediatr. Cardiol. 2014, 7(2), 107–117. [Google Scholar] [CrossRef]
- Abifadel, M.; Boileau, C. Genetic and molecular architecture of familial hypercholesterolemia. J. Intern. Med. 2023, 293(2), 144–165. [Google Scholar] [CrossRef]
- Futema, M.; et al. Comparison of the mutation spectrum and association with pre and post treatment lipid measures of children with heterozygous familial hypercholesterolaemia (FH) from eight European countries. Atherosclerosis 2021, 319, 108–117. [Google Scholar] [CrossRef]
- Bao, X.; et al. Targeting proprotein convertase subtilisin/kexin type 9 (PCSK9): from bench to bedside. Signal Transduct. Target. Ther. 2024, 9(1), 13. [Google Scholar] [CrossRef] [PubMed]
- Flight, P.; et al. † VERVE-102, a clinical stage in vivo base editing medicine, leads to potent and precise inactivation of PCSK9 in preclinical studies. J. Clin. Lipidol. 2025. 19, (3 Supplement, e68. [Google Scholar] [CrossRef]
- Horie, T.; Ono, K. VERVE-101: a promising CRISPR-based gene editing therapy that reduces LDL-C and PCSK9 levels in HeFH patients. Eur. Heart J.-Cardiovasc. Pharmacother. 2024, 10(2), 89–90. [Google Scholar] [CrossRef] [PubMed]
- Open-label, Phase 1b, Single-ascending Dose and Optional re Dosing Study to Evaluate the Safety of VERVE-101 Administered to Patients With Heterozygous Familial Hypercholesterolemia, Atherosclerotic Cardiovascular Disease, and Uncontrolled Hypercholesterolemia. 2022.
- Hooper, A.J.; Tang, X.L.; Burnett, J.R. VERVE-101, a CRISPR base-editing therapy designed to permanently inactivate hepatic PCSK9 and reduce LDL-cholesterol. Expert Opin. Investig. Drugs 2024, 33(8), 753–756. [Google Scholar] [CrossRef]
- Inc., V.T. Verve 102. 2026. Available online: https://www.vervetx.com/our-programs/verve-102.
- Wan, P.; et al. In vivo base editing gene therapy for heterozygous familial hypercholesterolemia: a phase 1 trial. Nat. Med. 2026, 32(3), 1045–1051. [Google Scholar] [CrossRef] [PubMed]
- Mollé, L.M.; et al. Nanoparticles for vaccine and gene therapy: Overcoming the barriers to nucleic acid delivery. WIREs Nanomed. Nanobiotechnology 2022, 14(6), e1809. [Google Scholar] [CrossRef]
- Raguram, A.; Banskota, S.; Liu, D.R. Cell 2022, 185(15), 2806–2827.
- Wang, J.-H.; et al. Adeno-associated virus as a delivery vector for gene therapy of human diseases. Signal Transduct. Target. Ther. 2024, 9(1), 78. [Google Scholar] [CrossRef]
- Zhao, Z.; Anselmo, A.C.; Mitragotri, S. Viral vector-based gene therapies in the clinic. Bioeng. Transl. Med. 2022, 7(1), e10258. [Google Scholar] [CrossRef]
- Batty, P.; Lillicrap, D. Adeno-associated viral vector integration: implications for long-term efficacy and safety. J. Thromb. Haemost. 2024, 22(11), 2945–2960. [Google Scholar] [CrossRef]
- Glenn, J.D.; et al. The presence of CpGs in AAV gene therapy vectors induces a plasmacytoid dendritic cell-like population very early after administration. Cell. Immunol. 2024, 399-400, 104823. [Google Scholar] [CrossRef]
- Mehta, N.; et al. Optimization of adeno-associated viral (AAV) gene therapies vectors for balancing efficacy, longevity and safety for clinical application. Gene Ther. 2025, 32(3), 197–210. [Google Scholar] [CrossRef] [PubMed]
- Bushman, F.D. Retroviral Insertional Mutagenesis in Humans: Evidence for Four Genetic Mechanisms Promoting Expansion of Cell Clones. Mol. Ther. 2020, 28(2), 352–356. [Google Scholar] [CrossRef] [PubMed]
- Parman, T.; et al. Mutagenesis and Carcinogenesis Risk Evaluation for AAV and Lentiviral Gene Therapies. Int. J. Toxicol. 2025, 44(3), 183–195. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; et al. Lipid Nanoparticles for Drug Delivery. Adv. NanoBiomed Res. 2022, 2(2), 2100109. [Google Scholar] [CrossRef]
- Zhang, H.; et al. Novel Lipid Nanoparticle (LNP) Delivery Systems Enabling the Advancement of RNA Therapeutics. Adv. Healthc. Mater. n/a(n/a), e05340.
- Jozić, A.; et al. In vivo endosomal escape assay identifies mechanisms for efficient hepatic LNP delivery. Nat. Biotechnol. 2026. [Google Scholar] [CrossRef] [PubMed]
- Hu, M.; et al. Physiological barriers and strategies of lipid-based nanoparticles for nucleic acid drug delivery. Adv. Mater. 2024, 36(22), 2303266. [Google Scholar] [CrossRef] [PubMed]
- Li, T.; et al. Engineered internal architecture of core-shell lipid nanoparticles promotes efficient mRNA endosomal release. Nat. Commun. 2026, 17(1), 2187. [Google Scholar] [CrossRef]
- Lee, Y.; et al. Immunogenicity of lipid nanoparticles and its impact on the efficacy of mRNA vaccines and therapeutics. Exp. Mol. Med. 2023, 55(10), 2085–2096. [Google Scholar] [CrossRef] [PubMed]
- Skorzynski, M.; Krol, M.; Braniewska, A. Hepatotoxicity of nanoparticle-based anti-cancer drugs: insights into toxicity and mitigation strategies. Int. J. Nanomed. 2025, 11697–11715. [Google Scholar] [CrossRef]
- Zhao, Y.; et al. Replacing cholesterol and PEGylated lipids with zwitterionic ionizable lipids in LNPs for spleen-specific mRNA translation. Sci. Adv. 2025, 11(41), eady6460. [Google Scholar] [CrossRef]
- Kalter, N.; et al. Off-target effects in CRISPR-Cas genome editing for human therapeutics: Progress and challenges. Mol. Ther. Nucleic Acids 2025, 36(3), 102636. [Google Scholar] [CrossRef]
- Zhang, C.; et al. Prediction of base editor off-targets by deep learning. Nat. Commun. 2023, 14(1), 5358. [Google Scholar] [CrossRef]
- Donohoue, P.D.; et al. Conformational control of Cas9 by CRISPR hybrid RNA-DNA guides mitigates off-target activity in T cells. Mol. Cell. 2021, 81(17), 3637–3649.e5. [Google Scholar] [CrossRef]
- Rueda, J.; de Miguel Beriain, Í.; Montoliu, L. Affordable Pricing of CRISPR Treatments is a Pressing Ethical Imperative. Cris. J. 2024, 7(5), 220–226. [Google Scholar]
- Witkowsky, L.; et al. Towards affordable CRISPR genomic therapies: a task force convened by the Innovative Genomics Institute. Gene Ther. 2023, 30(10), 747–752. [Google Scholar] [CrossRef]
- Lee, T.-L.; Sawai, T. Navigating equity in global access to genome therapy expanding access to potentially transformative therapies and benefiting those in need requires global policy changes. In Frontiers in Genetics; 2024; pp. 15–2024. [Google Scholar]
- Gaviglio, A.M.; et al. Gene-targeted therapies: Towards equitable development, diagnosis, and access. In American Journal of Medical Genetics Part C: Seminars in Medical Genetics; Wiley Online Library, 2023. [Google Scholar]
- Cornetta, K.; et al. Equitable access to gene therapy: a call to action for the American Society of gene and cell therapy. Mol. Ther. 2018, 26(12), 2715–2716. [Google Scholar] [CrossRef]
- Coquerelle, S.; et al. Innovative Curative Treatment of Beta Thalassemia: Cost-Efficacy Analysis of Gene Therapy Versus Allogenic Hematopoietic Stem-Cell Transplantation. Hum. Gene Ther. 2019, 30(6), 753–761. [Google Scholar] [CrossRef]
- Nicosia, L. Balancing personalized medicine and scalability in base editing for phenylketonuria. Mol. Ther. Nucleic Acids 2026, 37(1), 102824. [Google Scholar] [CrossRef]
- Quigley, A.; et al. Base editing strategies for in vivo correction of two highly recurrent phenylketonuria variants. Mol. Ther. Nucleic Acids 2025, 36(4). [Google Scholar] [CrossRef] [PubMed]
- Tavridou, A.; et al. Genome-editing medicinal products: the EMA perspective. Nat. Rev. Drug. Discov. 2024. [Google Scholar] [CrossRef]
- Human Gene Therapy Products Incorporating Human Genome Editing; 2024.
- Kliegman, M.; et al. A roadmap for affordable genetic medicines. Nature 2024, 634(8033), 307–314. [Google Scholar] [CrossRef] [PubMed]
- Considerations for the use of the Plausible Mechanism Framework to Develop Individualized Therapies that Target Specific Genetic Conditions with Known Biological Cause. 2026.
- Schambach, A.; et al. A new age of precision gene therapy. The Lancet 2024, 403(10426), 568–582. [Google Scholar] [CrossRef] [PubMed]
- Lekstrom-Himes, J.; et al. Moving away from one disease at a time: Screening, trial design, and regulatory implications of novel platform technologies. Am. J. Med. Genet. Part C. Semin. Med. Genet. 2023, 193(1), 30–43. [Google Scholar] [CrossRef]
- Giannikopoulos, P.; et al. Interventional Genomics: Bridging Germline Diagnosis and Therapeutic Action; Elsevier, 2026; p. 102532. [Google Scholar]
- Wenger, T.L.; Adam, M.P. Commentary: Interventional Genetics. Am. J. Med. Genet. Part C. Semin. Med. Genet. 2025, 199(3), 151–153. [Google Scholar] [CrossRef]
| Trial ID | Phase | Study design | Number of participants | Status | Target disease and gene (mutation if applicable) | Delivery system | Brief description |
|---|---|---|---|---|---|---|---|
| NCT06860672 | Phase 1 | Open label, single group assignment | 1 | Recruiting | Snijders Blok-Campeau syndrome Heterozygous mutation of c.3073C>T, p. (Arg1025Trp) in the CHD3 gene |
Dual Vector AAV | In vivo dual AAV delivery base editing therapy. Aim to rescue the mutant A-T base pair via a TadA-embedded adenine base editor (TeABE). |
| NCT06559176 | Phase 1/2 | Open-label, single-arm, multicenter | 12 | Recruiting by invitation | Autosomal recessive Chronic Granulomatous Disease Deletion mutation (c.75_76delGT) in the NCF1 gene |
Electroporation | Prime Editing autologous CD34+ stem cells ex vivo therapy. Aim to rescue exon 2 del GT mutation in the NCF1 gene, encoding p47phox protein. |
| NCT06959771 | Phase 1/2 | Open label, single group assignment | 1 | Recruiting | X-linked hyper-IgM (HIGM) syndrome Point mutation (c.658C>T; p.Q220X) in CD40L gene |
Electroporation | Base editing autologous hematopoietic stem/progenitor cells (HSPC) and T cells (BE T) ex vivo therapy. Aim to restore CD40L expression by rescuing c.658C>T point mutation |
| NCT07176923 & NCT07371767 |
Phase 1 | Open-label, single-arm, dose-escalation | 15 | Recruiting | Familial chylomicronemia syndrome (FCS) & Hyperchylomicronemia APOC3 gene. |
Lipid nanoparticles | In vivo base editing therapy delivered by lipid nanoparticles targeting APOC3 gene. Aim to introduce mutations that can reduce ApoC3 expression to reduce triglyceride serum levels and lowering pancreatitis risk. |
| NCT06325709 | Phase 1/2 | Open label, single group assignment | 10 | Recruiting | Chronic Granulomatous Disease Missense mutation (c.676C>T) in CYBB gene |
Electroporation | Base editing autologous hematopoietic stem and progenitor cells (HSPCs) ex vivo therapy. Aim to restore mutations in CYBB gene in HSPCs and later engraft into patients. Modify HSPCs can differentiate into functional phagocytes with restored NADPH oxidase activity. |
| NCT06851767 | Phase 1/2 | Non-randomized, open label, single group assignment | 18 | Enrolling by invitation | X-linked severe combined immunodeficiency IL2RG gene |
Electroporation | Base editing autologous hematopoietic stem and progenitor cells (HSPCs) ex vivo therapy. Aim to rescue mutations in the IL2RG gene in different patients. |
| NCT06065189, NCT07000318, NCT06565026, NCT06024876, & NCT06479616 | Phase 1 | Open label, single-arm | 5 | Active or recruiting | Major β – thalassemia and Severe sickle cell disease BCL11A binding site in Hemoglobin Subunit Gamma (HBG) promoter |
Electroporation | Base editing autologous hematopoietic stem cell transplantation (HSPCs) ex vivo therapy. Aim to modify HBG promoter in BCL11A binding site to prevent BCL11A inhibitory effect in γ-globin chain synthesis. This to increase the concentration of fetal hemoglobin in the blood. |
| NCT06025032 | Phase 1 | Open-label, multiple-cohort, dose-finding | 0 | Withdrawn due to lack of patients in China | Auditory neuropathy (hearing loss) Nonsense mutation c.2485C>T (p. Q829X) in OTOF gene |
AAV9 vector | In vivo AAV9 delivered CRISPR/Cas13 RNA base-editing therapy. Aim to restore otoferlin expression in the sensory inner hair cells. |
| NCT07489196 | Phase 2 | Open label, single-arm | 20 | Not yet recruiting | Major β – thalassemia BCL11A binding site in Hemoglobin Subunit Gamma (HBG) promoter |
Electroporation | Base editing autologous hematopoietic stem cell transplantation (HSPCs) ex vivo therapy. Aim to modify HBG promoter in BCL11A binding site to prevent BCL11A inhibitory effect in γ-globin chain synthesis. This to increase the concentration of fetal hemoglobin in the blood. |
| NCT06594094 |
Phase 1 | Open-label, multidose, dose-escalation | 4 | Completed | Duchenne muscular dystrophy Exon 51 splice donor site of DMD gene |
AAV vector |
In vivo AAV delivered CRISPR/hfCas12Max base-editing therapy. Aim to modify DMD exon 51 splice donor site to induce skipping of exon 51. Exon 51 skipping can restore the reading frame and recover dystrophin production in patients carrying different single and multi-exon deletions. |
| NCT05398029 | Phase 1 | Open-label, single-ascending dose | 13 | Completed | Heterozygous familial hypercholesterolemia PCSK9 gene (Splicing site) |
Lipid nanoparticles | In vivo LNP delivered CRISPR/ABE base-editing therapy. Aim to induce point mutation in PCSK9 splicing site to impair PSCK9 expression levels and reduce LDL-C levels in blood. |
| NCT06735755 | Phase 1 & 2 | Open-label, single ascending-dose, multicenter | 36 | Recruiting | Glycogen storage disease type-Ia (von Gierke disease) Missense mutation c.247C > T (p.R83C) in the G6PC1 gene |
Lipid nanoparticles | In vivo LNP delivered CRISPR/ABE base-editing therapy. Aim to correct c.247C > T missense mutation on G6PC1 gene to rescue glucose-6-phosphatase-α production. |
| NCT05456880 | Phase 1 & 2 | Open-label, single-arm, multicenter | 15 | Recruiting | Sickle Cell Disease and Severe Vaso-Occlusive Crises HBG1/2 gene promoters |
Electroporation | Base editing autologous CD34+ hematopoietic stem cells ex vivo therapy. Aim to disrupt BCL11A binding sites in HBG1/2 gene promoters |
| NCT06389877 | Phase 1 & 2 | Open-label, multicenter, dose-exploration and dose expansion | 106 | Recruiting | Alpha-1 antitrypsin deficiency (AATD) Missense mutation (E342K) in the SERPINA1 gene |
Lipid nanoparticles | In vivo LNP delivered CRISPR/ABE base-editing therapy. Aim to rescue α1-antitrypsin expression by amending E342K point mutation. |
| NCT06164730 | Phase 1 | Open-label, single-ascending Dose | 85 | Recruiting | Familial Hypercholesterolemia or Premature Coronary Artery Disease PCSK9 gene (Splicing site) |
Lipid nanoparticles conjugated with GalNAc | In vivo LNP delivered CRISPR/ABE base-editing therapy. Aim to induce point mutation in PCSK9 splicing site to impair PSCK9 expression levels and reduce LDL-C levels in blood. |
| NCT06461702, & NCT06458010 | Phase 1 | Open-label, single arm, single dose escalation | 13 & 20 | Recruiting | Familial Hypercholesterolemia. Exon 1 splice donor site of PCSK9. |
Lipid nanoparticles conjugated with GalNAc | In vivo LNP delivered CRISPR/ hpABE5 base-editing therapy. Aim to induce point mutation in PCSK9 splicing site to impair PSCK9 expression levels and reduce LDL-C levels in blood. |
| NCT06451770 | Phase 1 | Open-label, single-ascending dose | 36 | Recruiting | Familial Hypercholesterolemia and Refractory Hyperlipidemia ANGPTL3 gene |
Lipid nanoparticles conjugated with GalNAc | In vivo LNP delivered CRISPR/ ABE8.8 base-editing therapy. Aim to inactivate the expression of the ANGPTL3 gene and reduce LDL-C and triglycerides levels in blood. |
| NCT06392724 | Phase 1 | Open-label, single-arm, single-center | 3 | Active, not recruiting | Duchenne muscular dystrophy 5’ splicing site of exon 50 of the DMD gene |
Dual ss.AAV9 vector | In vivo AAV delivered CRISPR/CBE base-editing therapy. Aim to modify DMD exon 50 5’ splice site to induce skipping of exon 50. Exon 50 skipping can restore the reading frame and recover dystrophin production in patients carrying different single and multi-exon deletions. |
| N/A | N/A | Single-patient expanded-access Investigational New Drug application, | 1 | N/A | Carbamoyl-phosphate synthetase 1 (CPS1) deficiency Missense mutation c.1003C→T (p.Gln335Ter) in CPS1 gene |
Lipid nanoparticles | In vivo personalized lipid nanoparticle delivered CRISPR/ NGC-ABE8eV106W base-editing therapy. Aim to rescue carbamoyl-phosphate synthetase 1 production by restoring c.1003C→T point mutation. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




