Submitted:
24 April 2026
Posted:
28 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
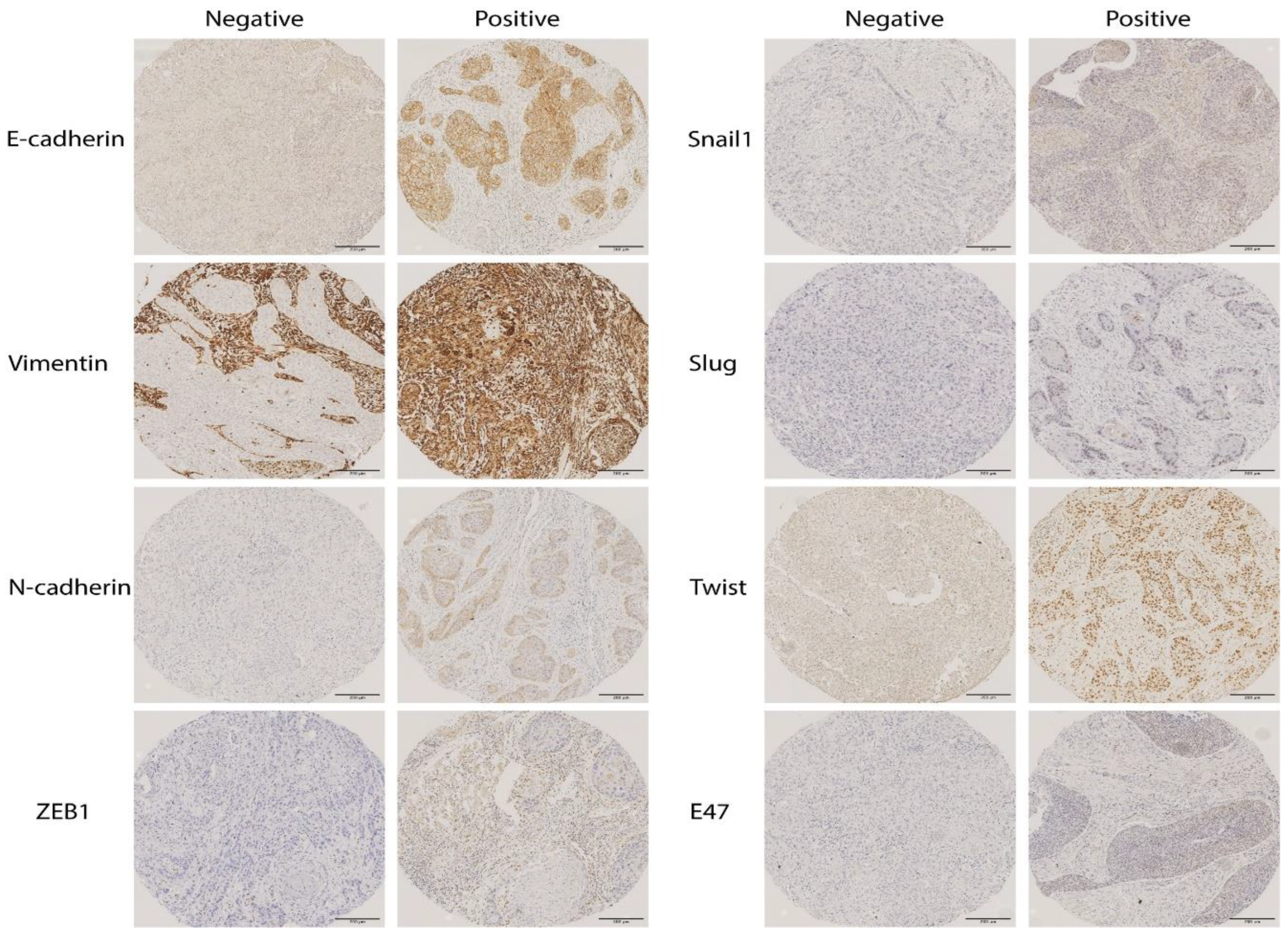
2.1. Immunohistochemical Analysis of EMT Markers in OSCC
2.2. Associations with Clinicopathological Variables and Prognosis
2.3. Survival Analysis
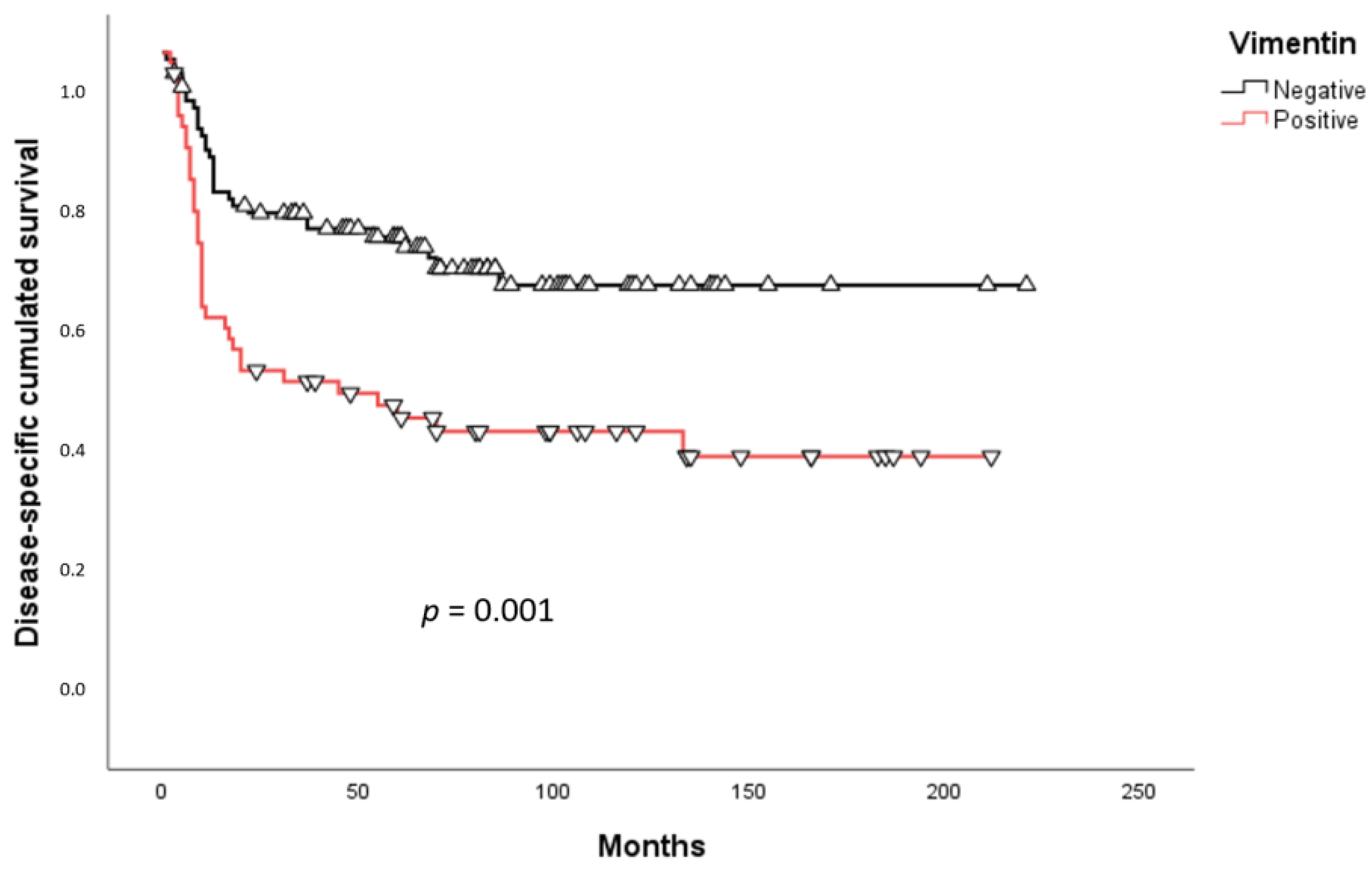
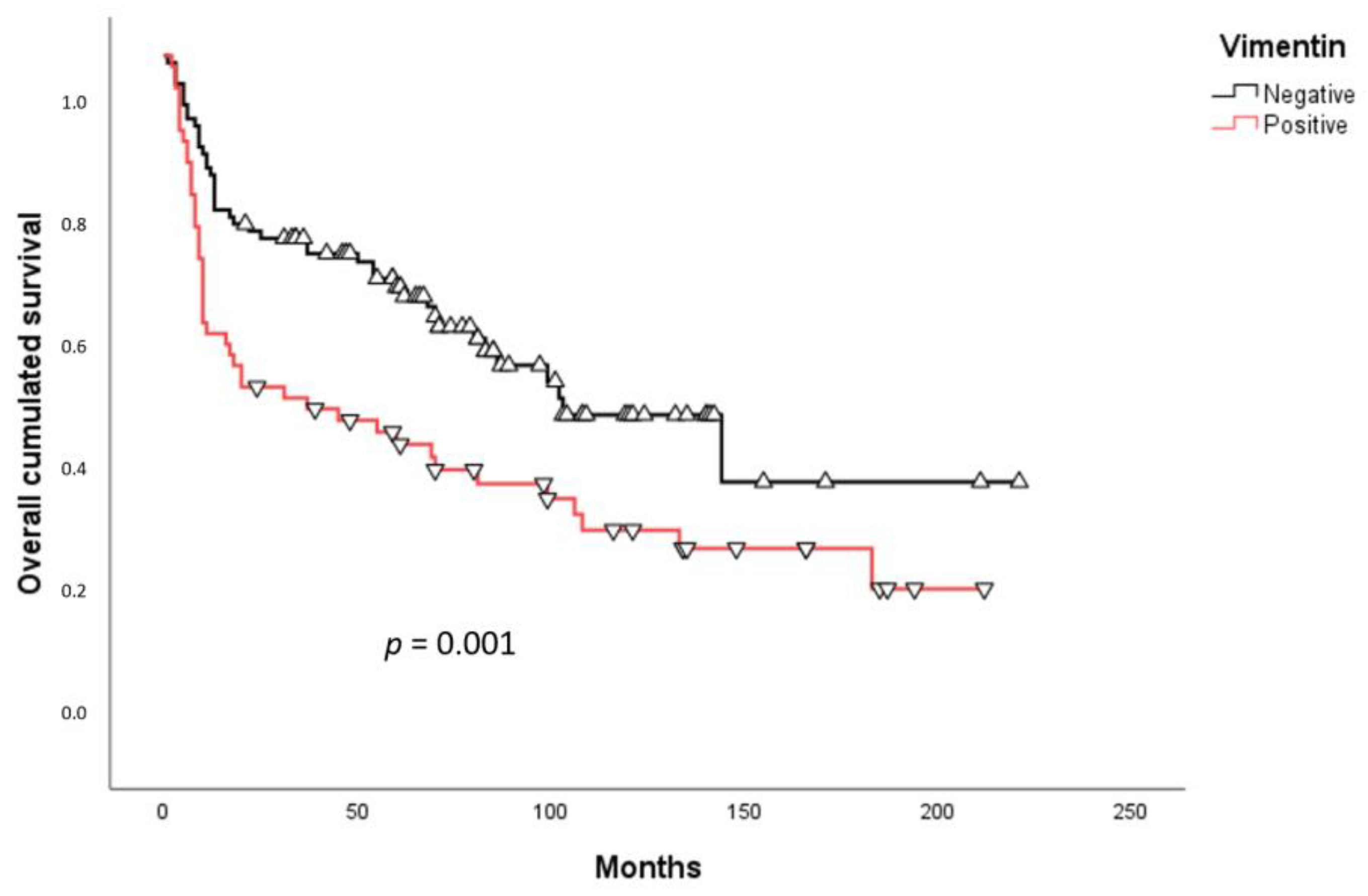
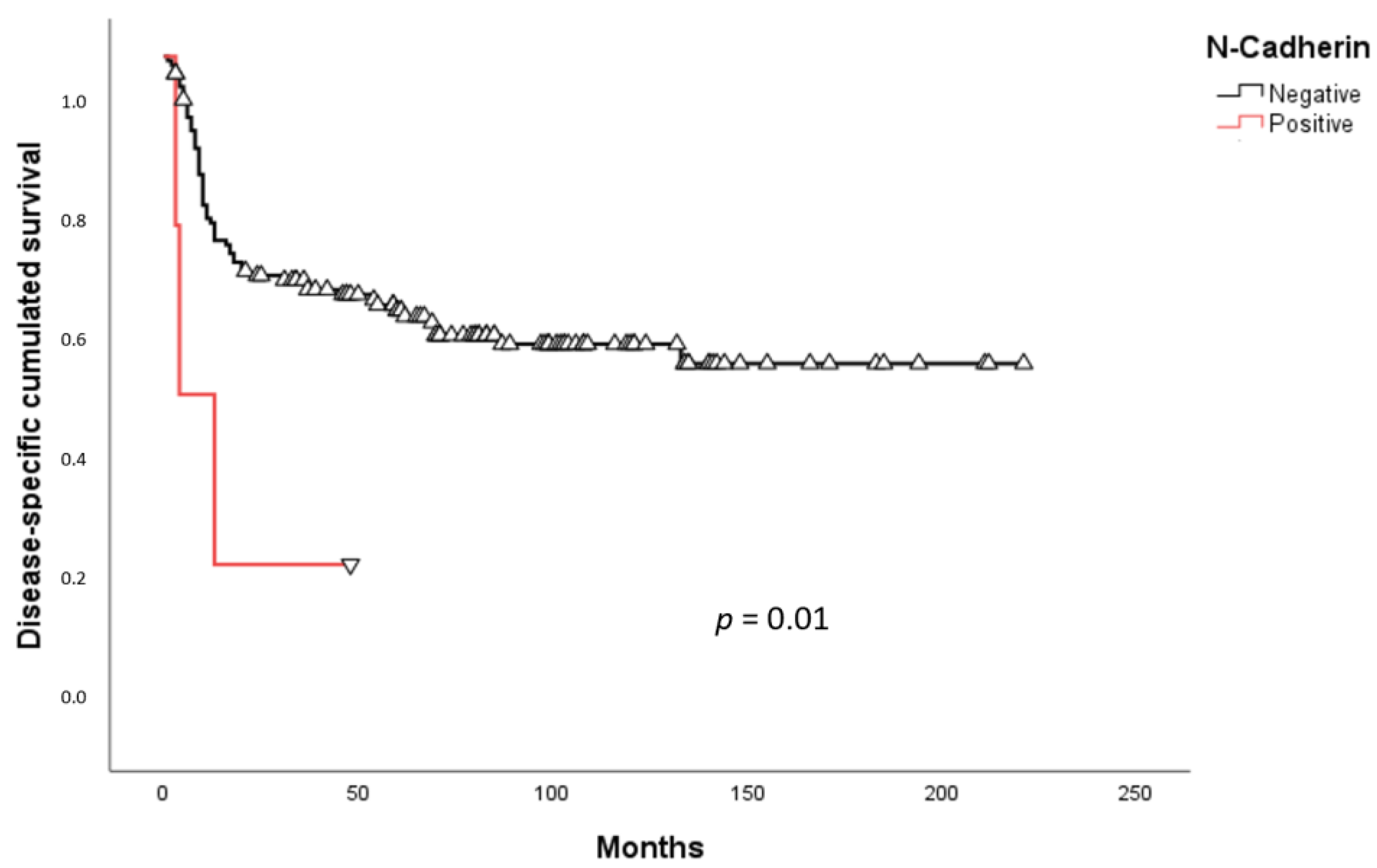
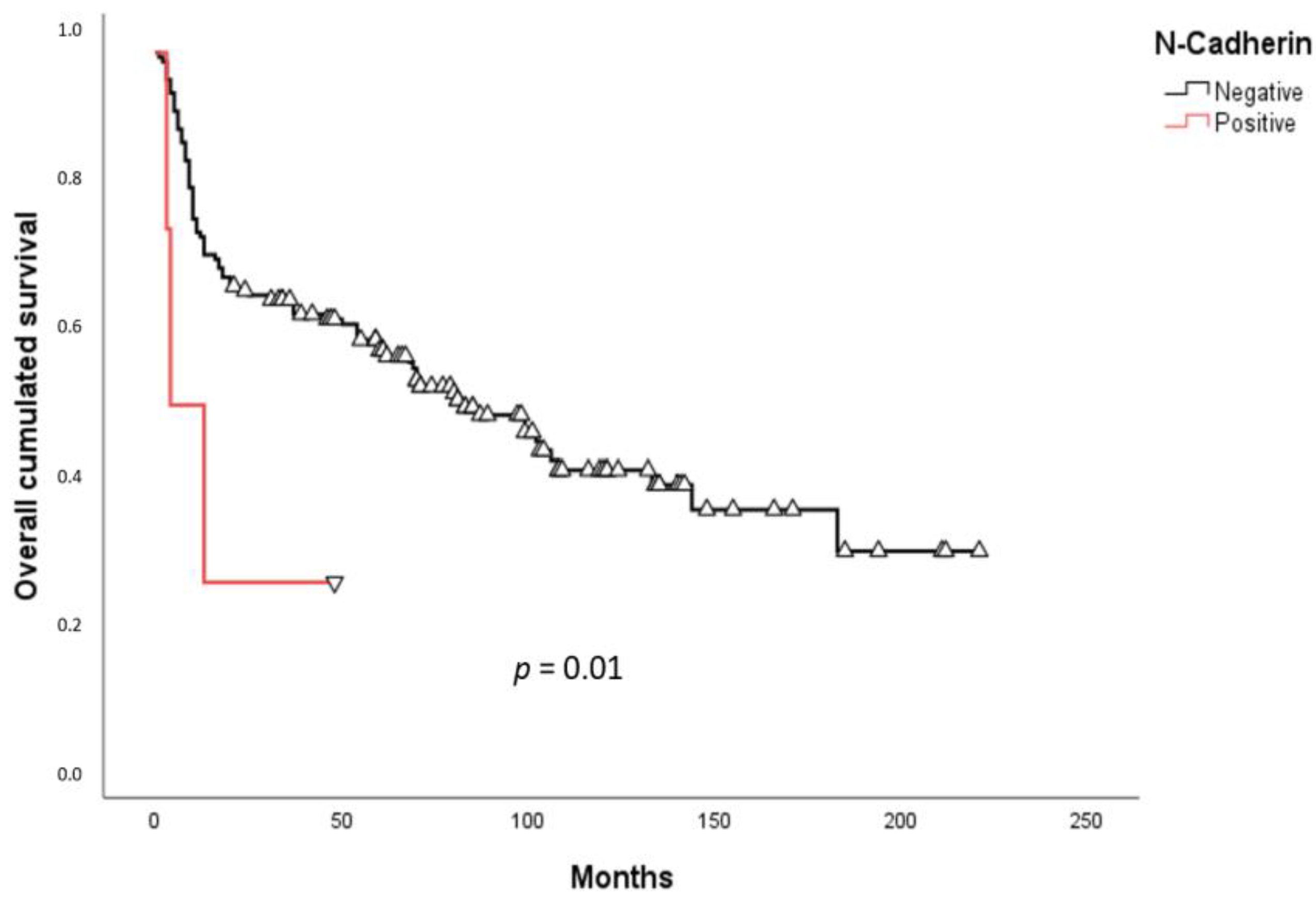
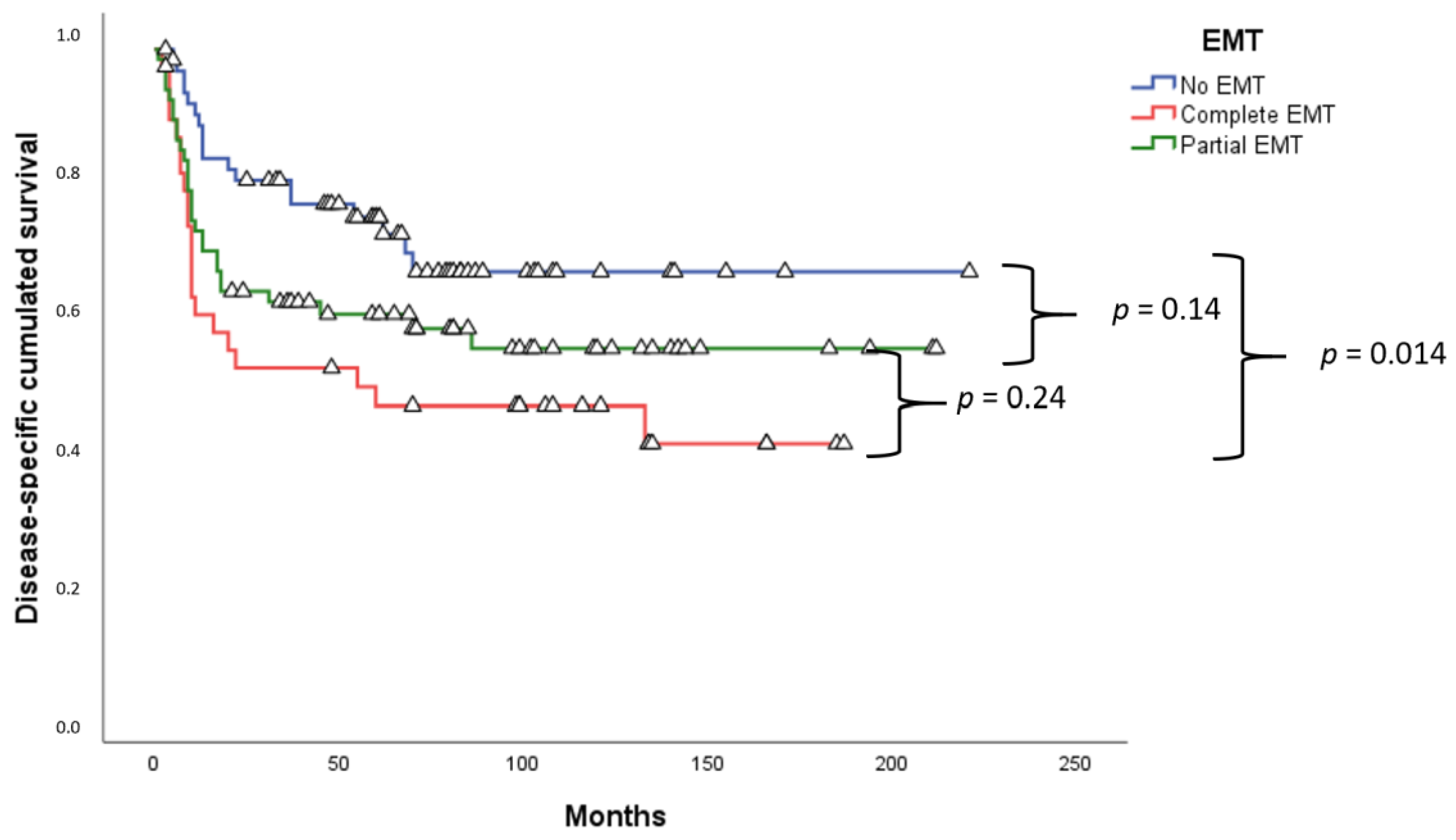
3. Discussion
4. Materials and Methods
4.1. Patients and Tissue Specimens
4.2. Immunohistochemistry (IHC)
4.3. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| tOSCC | Oral squamous cell carcinoma |
| EMT | Epithelial–mesenchymal transition |
| EMT-TFs | Transcription factors capable of inducing EMT |
| DSS | Disease-specific survival |
| OS | Overall survival |
| HR | Hazard ratio |
| CI | Confidence interval |
References
- Graillon, N.; Iocca, O.; Carey, R.M.; Benjamin, K.; Cannady, S.B.; Hartner, L.; Newman, J.G.; Rajasekaran, K.; Brant, J.A.; Shanti, R.M. What has the National Cancer Database taught us about oral cavity squamous cell carcinoma? Int. J. Oral. Maxillofac. Surg. 2022, 51, 10–17. [Google Scholar] [CrossRef]
- Zanoni, D.K.; Montero, P.H.; Migliacci, J.C.; Shah, J.P.; Wong, R.J.; Ganly, I.; Patel, S.G. Survival outcomes after treatment of cancer of the oral cavity (1985-2015). Oral. Oncol. 2019, 90, 115–121. [Google Scholar] [CrossRef] [PubMed]
- Kang, Y.; Massagué, J. Epithelial-mesenchymal transitions: twist in development and metastasis. Cell 2004, 118, 277–279. [Google Scholar] [CrossRef]
- Zidar, N.; Boštjančič, E.; Malgaj, M.; Gale, N.; Dovšak, T.; Didanovič, V. The role of epitelial-mesenchymal transition in squamous cell carcinoma of the oral cavity. Virchows Arch. 2018, 472, 237–245. [Google Scholar] [CrossRef]
- Tsai, J.H.; Donaher, J.L.; Murphy, D.A.; Chau, S.; Yang, J. Spatiotemporal regulation of epithelial-mesenchymal transition is essential for squamous cell carcinoma metastasis. Cancer Cell. 2012, 22, 725–736. [Google Scholar] [CrossRef]
- Ocaña, O.H.; Córcoles, R.; Fabra, A.; Moreno-Bueno, G.; Acloque, H.; Vega, S.; Barrallo-Gimeno, A.; Cano, A.; Nieto, M.A. Metastatic colonization requires the repression of the epithelial-mesenchymal transition inducer Prrx1. Cancer Cell. 2012, 22, 709–724. [Google Scholar] [CrossRef] [PubMed]
- Fischer, K.R.; Durrans, A.; Lee, S.; Sheng, J.; Li, F.; Wong, S.T.; Choi, H.; El Rayes, T.; Ryu, S.; Troeger, J.; Schwabe, R.F.; Vahdat, L.T.; Altorki, N.K.; Mittal, V.; Gao, D. Epithelial-to-mesenchymal transition is not required for lung metastasis but contributes to chemoresistance. Nature 2015, 527, 472–476. [Google Scholar] [CrossRef]
- Zheng, X.; Carstens, J.L.; Kim, J.; Scheible, M.; Kaye, J.; Sugimoto, H.; Wu, C.C.; LeBleu, V.S.; Kalluri, R. Epithelial-to-mesenchymal transition is dispensable for metastasis but induces chemoresistance in pancreatic cancer. Nature 2015, 527, 525–530. [Google Scholar] [CrossRef]
- Chui, M.H. Insights into cancer metastasis from a clinicopathologic perspective: Epithelial-mesenchymal transition is not a necessary step. Int. J. Cancer 2013, 132, 1487–1495. [Google Scholar] [CrossRef]
- Tarin, D.; Thompson, E.W.; Newgreen, D.F. The fallacy of epithelial mesenchymal transition in neoplasia. Cancer Res. 2005, 65, 5996-6000; discussion 6000-1. [Google Scholar] [CrossRef] [PubMed]
- Tarin, D. Inappropriate gene expression in human cancer and its far-reaching biological and clinical significance. Cancer Metastasis Rev. 2012, 31, 21–39. [Google Scholar] [CrossRef]
- Tarin, D. Role of the host stroma in cancer and its therapeutic significance. Cancer Metastasis Rev. 2013, 32, 553–566. [Google Scholar] [CrossRef]
- Scanlon, C.S.; Van Tubergen, E.A.; Inglehart, R.C.; D'Silva, N.J. Biomarkers of epithelial-mesenchymal transition in squamous cell carcinoma. J. Dent. Res. 2013, 92, 114–121. [Google Scholar] [CrossRef]
- Hakim, S.G.; Taubitz, C.; Hoppe, S.; Steller, D.; Rades, D.; Ribbat-Idel, J.; Alsharif, U.; Falougy, M. Prognostic impact of the loss of E-cadherin and de novo expression of N-cadherin at the invasive front of primary and recurrent oral squamous cell carcinoma. Front. Oncol. 2023, 13, 1151879. [Google Scholar] [CrossRef] [PubMed]
- Wangmo, C.; Charoen, N.; Jantharapattana, K.; Dechaphunkul, A.; Thongsuksai, P. Epithelial-mesenchymal transition predicts survival in oral squamous cell carcinoma. Pathol. Oncol. Res. 2020, 26, 1511–1518. [Google Scholar] [CrossRef]
- Wolf, G.T.; Winter, W.; Bellile, E.; Nguyen, A.; Donnelly, C.R.; McHugh, J.B.; Thomas, D.; Amlani, L.; Rozek, L.; Lei, Y.L. Head and Neck SPORE Program. Histologic pattern of invasion and epithelial-mesenchymal phenotype predict prognosis in squamous carcinoma of the head and neck. Oral. Oncol. 2018, 87, 29–35. [Google Scholar] [CrossRef]
- Takkunen, M.; Grenman, R.; Hukkanen, M.; Korhonen, M.; García de Herreros, A.; Virtanen, I. Snail-dependent and -independent epithelial-mesenchymal transition in oral squamous carcinoma cells. J. Histochem. Cytochem. 2006, 54, 1263–1275. [Google Scholar] [CrossRef] [PubMed]
- Vallina, C.; López-Pintor, R.M.; González-Serrano, J.; de Vicente, J.C.; Hernández, G.; Lorz, C. Genes involved in the epithelial-mesenchymal transition in oral cancer: A systematic review. Oral. Oncol. 2021, 117, 105310. [Google Scholar] [CrossRef] [PubMed]
- Yoshino, I.; Kometani, T.; Shoji, F.; Osoegawa, A.; Ohba, T.; Kouso, H.; Takenaka, T.; Yohena, T.; Maehara, Y. Induction of epithelial-mesenchymal transition-related genes by benzo[a]pyrene in lung cancer cells. Cancer 2007, 110, 369–374. [Google Scholar] [CrossRef]
- Göppel, J.; Möckelmann, N.; Münscher, A.; Sauter, G.; Schumacher, U. Expression of epithelial-mesenchymal transition regulating transcription factors in head and neck squamous cell carcinomas. Anticancer Res. 2017, 37, 5435–5440. [Google Scholar] [CrossRef]
- Riechelmann, H.; Steinbichler, T.B.; Sprung, S.; Santer, M.; Runge, A.; Ganswindt, U.; Gamerith, G.; Dudas, J. The epithelial-mesenchymal transcription factor Slug predicts survival benefit of up-front surgery in head and neck cancer. Cancers 2021, 13, 772. [Google Scholar] [CrossRef]
- Wen, B.; Xu, L.Y.; Li, E.M. LOXL2 in cancer: regulation, downstream effectors and novel roles. Biochim. Biophys. Acta Rev. Cancer 2020, 1874, 188435. [Google Scholar] [CrossRef]
- Kumar, D.; Gupta, A.; Agrahari, S.; Singh, S.; Gupta, S.; Kumar, V.; Singh, S.K. Association of epithelial to mesenchymal transition markers on prognosis and clinicopathological characteristics in oral squamous cell carcinoma: A systematic review and meta-analysis. Head. Neck. Pathol. 2025, 19, 124. [Google Scholar] [CrossRef]
- Vanini, J.V.; Koyama, L.K.S.; de Matos, L.L.; Junior, J.M.F.; Cernea, C.R.; Nagano, C.P.; Coutinho-Camillo, C.M.; Hsieh, R.; Lourenço, S.V. Epithelial-mesenchymal transition related to bone invasion in oral squamous cell carcinoma. J. Bone Oncol. 2022, 33, 100418. [Google Scholar] [CrossRef]
- Kisoda, S.; Mouri, Y.; Kitamura, N.; Yamamoto, T.; Miyoshi, K.; Kudo, Y. The role of partial-EMT in the progression of head and neck squamous cell carcinoma. J. Oral. Biosci. 2022, 64, 176–182. [Google Scholar] [CrossRef]
- Mehendiratta, M.; Solomon, M.C.; Boaz, K.; Guddattu, V.; Mohindra, A. Clinico-pathological correlation of E-cadherin expression at the invasive tumor front of Indian oral squamous cell carcinomas: An immunohistochemical study. J. Oral. Maxillofac. Pathol. 2014, 18, 217–222. [Google Scholar] [CrossRef] [PubMed]
- Lorenzo-Pouso, A.I.; Silva, F.F.E.; Pérez-Jardón, A.; Chamorro-Petronacci, C.M.; Oliveira-Alves, M.G.; Álvarez-Calderón-Iglesias, Ó.; Caponio, V.C.A.; Pinti, M.; P errotti, V.; Pérez-Sayáns, M. Overexpression of E-cadherin Is a favorable prognostic biomarker in oral squamous cell carcinoma: A systematic review and meta-analysis. Biology 2023, 12, 239. [Google Scholar] [CrossRef] [PubMed]
- de Farias Morais, H.G.; de Morais, E.F.; Carlan, L.M.; de Pontes Santos, H.B.; da Silveira, É.J.D.; de Almeida Freitas, R. Epithelial-mesenchymaltransition modulates lower lip carcinogenesis and promotes cancer progression. Arch. Oral. Biol. 2022, 136, 105387. [Google Scholar] [CrossRef] [PubMed]
- Ukpo, O.C.; Thorstad, W.L.; Zhang, Q.; Lewis, J.S., Jr. Lack of association of cadherin expression and histopathologic type, metastasis, or patient outcome in oropharyngeal squamous cell carcinoma: a tissue microarray study. Head. NeckPathol 2012, 6, 38–47. [Google Scholar] [CrossRef]
- Cheng, S.Y.; Shi, K.; Bai, X.R.; Wu, Q.W.; Lv, X.Q. Double-staining of E-cadherin and podoplaninofferhelp in thepathological diagnosis ofindecisiveearly-invasive oral squamouscellcarcinoma. Int. J. Clin.Exp. Pathol. 2018, 11, 38–47. [Google Scholar] [CrossRef]
- Al-Rawi, N.; Al Ani, M.; Quadri, A.; Hamdoon, Z.; Awwad, A.; Al Kawas, S.; Al Nuaimi, A. Prognostic significance of E-Cadherin, β-Catenin and cyclin D1 in oral squamous cell carcinoma: a tissue microarray study. Histol.Histopathol 2021, 36, 1073–1083. [Google Scholar] [CrossRef]
- Ali, A.N.; Ghoneim, S.M.; Ahmed, E.R.; El-Farouk Abdel Salam, L.O.; Anis Saleh, S.M. Cadherin switching in oral squamous cell carcinoma: A clinicopathologicalstudy. J. Oral. Biol.Craniofac. Res. 2023, 13, 486–494. [Google Scholar] [CrossRef]
- Hashimoto, T.; Soeno, Y.; Maeda, G.; Taya, Y.; Aoba, T.; Nasu, M.; Kawashiri, S.; Imai, K. Progression of oral squamous cell carcinoma accompanied with reduced E-cadherin expression but not cadherin switch. PLoSOne 2012, 7, e47899. [Google Scholar] [CrossRef]
- Nieman, M.T.; Prudoff, R.S.; Johnson, K.R.; Wheelock, M.J. N-cadherinpromotesmotility in human breastcancercellsregardless of their E-cadherin expression. J. Cell. Biol. 1999, 147, 631–44. [Google Scholar] [CrossRef]
- Balasundaram, P.; Singh, M.K.; Dinda, A.K.; Thakar, A.; Yadav, R. Studyof β-catenin, E-cadherin and vimentin in oral squamous cell carcinoma with and without lymph node metastases. Diagn.Pathol 2014, 9, 145. [Google Scholar] [CrossRef]
- Sgaramella, N.; Wilms, T.; Boldrup, L.; Loljung, L.; Gu, X.; Coates, P.J.; Hassellöf, P.; Califano, L.; Lo Muzio, L.; Fåhraeus, R.; Norberg Spaak, L.; Franco, R.; Tartaro; Colella, G.; Santagata, M.; Dell'AversanaOrabona, G.; Chirico, F.; Danielsson, K.; Troiano, G.; Ardito, F.; Nylander, K. Ethnicity based variation in expression of E-cadherin in patients with squamous cell carcinoma of the oral tongue. Oncol. Lett. 2018, 16, 6603–6607. [Google Scholar] [CrossRef]
- Nambiyar, K.; Ahuja, A.; Bhardwaj, M. A study of epithelial-mesenchymal transition immunohistochemical markers in primary oral squamous cell carcinoma. Oral. Surg. Oral. Med. Oral. Pathol. Oral. Radiol. 2021, 132, 680–686. [Google Scholar] [CrossRef] [PubMed]
- Yokoyama, K.; Kamata, N.; Hayashi, E.; Hoteiya, T.; Ueda, N.; Fujimoto, R.; Nagayama, M. Reverse correlation of E-cadherin and snail expression in oral squamouscell carcinoma cells in vitro. Oral. Oncol. 2001, 37, 65–71. [Google Scholar] [CrossRef] [PubMed]
- Baqai, N.; Amin, R.; Fatima, T.; Ahmed, Z.; Faiz, N. Expression profiling of EMT transcriptional regulators ZEB1 and ZEB2 in different histopathological grades of oral squamous cell carcinoma patients. Curr. Genom. 2024, 25, 140–151. [Google Scholar] [CrossRef]
- van der Heijden, M.; Essers, P.B.M.; Verhagen, C.V.M.; Willems, S.M.; Sanders, J.; de Roest, R.H.; Vossen, D.M.; Leemans, C.R.; Verheij, M.; Brakenhoff, R.H.; van den Brekel, M.W.M.; Vens, C. Epithelial-to-mesenchymal transition is a prognostic marker for patient outcome in advanced stage HNSCC patients treated with chemoradiotherapy. Radiother. Oncol. 2020, 147, 186–194. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Liu, H.; Borok, Z.; Davies, K.J.; Ursini, F.; Forman, H.J. Cigarettessmokeextractstimulatesepithelial-mesenchymaltransitionthroughSrcactivation. Free Radic. Biol.Med 2012, 52, 1437–1442. [Google Scholar] [CrossRef]
- Yu, M.A.; Kiang, A.; Wang-Rodriguez, J.; Rahimy, E.; Haas, M.; Yu, V.; Ellies, L.G.; Chen, J.; Fan, J.B.; Brumund, K.T.; Weisman, R.A.; Ongkeko, W.M. Nicotinepromotesacquisitionofstemcell and epithelial-to-mesenchymalproperties in head and necksquamouscell carcinoma. PLoSOne 2012, 7, e51967. [Google Scholar] [CrossRef]
- Ghantous, Y.; Mozalbat, S.; Nashef, A.; Abdol-Elraziq, M.; Sudri, S.; Araidy, S.; Tadmor, H.; Abu El-Naaj, I. EMT dynamics in lymphnodemetastasisoforal squamous cell carcinoma. Cancers 2024, 16, 1185. [Google Scholar] [CrossRef]
- Sakamoto, K.; Imanishi, Y.; Tomita, T.; Shimoda, M.; Kameyama, K.; Shibata, K.; Sakai, N.; Ozawa, H.; Shigetomi, S.; Fujii, R.; Fujii, M.; Ogawa, K. Overexpression of SIP1 and downregulation of E-cadherin predict delayed neck metastasis in stage I/II oral tongue squamouscell carcinoma after partial glossectomy. A nn.Surg. Oncol. 2012, 19, 612–619. [Google Scholar] [CrossRef] [PubMed]
- Cook, D.P.; Vanderhyden, B.C. Context specificity of the EMT transcriptional response. Nat. Commun. 2020, 11, 2142. [Google Scholar] [CrossRef] [PubMed]
- Horny, K.; Sproll, C.; Peiffer, L.; Furtmann, F.; Gerhardt, P.; Gravemeyer, J.; Stoecklein, N.H.; Spassova, I.; Becker, J.C. Mesenchymal-epithelialtransition in lymphnodemetastasesoforal squamouscell carcinoma is accompanied by ZEB1 expression. J.Transl.Med. 2023, 21, 267. [Google Scholar] [CrossRef]
- Yang, J.; Antin, P.; Berx, G.; Blanpain, C.; Brabletz, T.; Bronner, M.; Campbell, K.; Cano, A.; Casanova, J.; Christofori, G. Guidelines and definitions for research on epithelial-esenchymal transition. Nat. Rev. Mol. Cell. Biol. 2020, 21, 341–352. [Google Scholar] [CrossRef]
- Vasaikar, S.V.; Deshmukh, A.P.; den Hollander, P.; Addanki, S.; Kuburich, N.A.; Kudaravalli, S.; Joseph, R.; Chang, J.T.; Soundararajan, R.; Mani, S.A. EMTome: a resource for pan-cancer analysis of epithelial-mesenchymal transition genes and signatures. Br. J.Cancer. 2021, 124, 259–269. [Google Scholar] [CrossRef]
- Pal, A.; Barrett, T.F.; Paolini, R.; Parikh, A.; Puram, S.V. Partial EMT in head and neck cancer biology: a spectrum instead of a switch. Oncogene 2021, 40, 5049–5065. [Google Scholar] [CrossRef]
- Heguedusch, D.; Carvalho, G.L.; Tomo, S.; Aguiar, E.M.G.; Custódio, M.; Siqueira, J.M.; da unhaMercante, A.M.; Cury, P.M.; Tajara, E.H.; De Cicco, R.; Nunes, F.D. The patterns of P53, E-Cadherin, β-Catenin, CXCR4 and Podoplanin expression in oral squamous cell carcinoma suggests a hybrid invasion model: an immunohistochemical study on tissue microarrays. Head. NeckPathol 2025, 19, 6. [Google Scholar] [CrossRef] [PubMed]
| Variable | Snail1 (%) (-) (+) |
p | Slug (%) (-) (+) |
p | Zeb1 (%) (-) (+) |
p | Twist (%) (-) (+) |
p |
E47 (%) (-) (+) |
p |
|---|---|---|---|---|---|---|---|---|---|---|
|
Gender Men Women |
39 (35) 74 (65) 28 (54) 24 (46) |
0.02 | 16 (14) 97 (86) 13 (25) 39 (75) |
0.08 | 111 (98) 2 (2) 49 (94) 3 (6) |
0.18 | 6 (5) 106 (95) 3 (6) 49 (94) |
1.0 | 109 (96) 4 (4) 49 (94) 3 (6) |
0.67 |
|
Tobacco use Smoker Non-smoker |
36 (34) 71 (66) 31 (53) 27 (47) |
0.01 | 14 (13) 93 (87) 15 (26) 43 (74) |
0.04 | 105 (98) 2 (2) 55 (95) 3 (5) |
0.34 | 4 (4) 102 (96) 5 (9) 53 (91) |
0.28 | 104 (97) 3 (3) 54 (93) 4 (7) |
0.24 |
|
Alcohol use Drinker Non-drinker |
32 (36) 57 (64) 35 (46) 41 (54) |
0.18 | 10 (11) 79 (89) 19 (25) 57 (75) |
0.02 | 88 (99) 1 (1) 72 (95) 4 (5) |
0.18 | 2 (2) 86 (98) 7 (9) 69 (91) |
0.08 | 85 (95) 4 (5) 73 (96) 3 (4) |
1.0 |
|
pT pT1 + 2 pT3 + 4 |
42 (37) 72 (63) 20 (47) 23 (53) |
0.26 | 12 (11) 102 (89) 14 (33) 29 (67) |
0.001 | 112 (98) 2 (2) 40 (93) 3 (7) |
0.12 | 4 (4) 109 (96) 5 (12) 38 (88) |
0.11 |
111 (97) 3(3) 39 (91) 4 (9) |
0.09 |
|
pN pN0 pN+ |
39 (41) 56 59) 23 (37) 40 (63) |
0.56 | 13 (14) 82 (86) 12 (19) 51 (81) |
0.36 | 92 (97) 3 (3) 61 (97) 2 (3) |
0.66 | 3 (3) 91 (97) 4 (6) 59 (94) |
0.44 | 91 (96) 4 (4) 60 (95) 3 (5) |
1.0 |
|
Clinical stage I + II III + IV |
32 (36) 56 (64) 35 (46) 42 (54) |
0.23 | 8 (9) 80 (91) 21 (27) 56 (73) |
0.002 | 86 (98) 2 (2) 74 (96) 3 (4) |
0.66 | 2 (2) 85 (98) 7 (9) 70 (90) |
0.05 | 85 (97) 3 (3) 73 (95) 4 (5) |
0.70 |
|
G status Well Moderate - poor |
44 (42) 61 (58) 23 (38) 37 (62) |
0.65 | 18 (17) 87 (83) 11 (18) 49 (82) |
0.84 | 102 (97) 3 (3) 58 (97) 2 (3) |
1.0 | 5 (5) 99 (95) 4 (7) 56 (93) |
0.72 | 101 (96) 4 (4) 57 (95) 3 (5) |
0.70 |
|
Tumor recurrence No Yes |
29 (35) 54 (65) 38 (46) 44 (54) |
0.13 |
11 (13) 72 (87) 18 (22) 64 (78) |
0.14 | 81 (98) 2 (2) 79 (96) 3 (4) |
0.68 | 3 (4) 79 (96) 6 (7) 76 (93) |
0.49 |
81 (98) 2 (2) 77 (94) 5 (6) |
0.27 |
|
Clinical outcome Alive Dead of index cancer |
38 (39) 60 (61) 29 (43) 38 (57) |
0.56 | 14 (14) 84 (86) 15 (22) 52 (78) |
0.17 | 95 (97) 3 (3) 65 (97) 2 (3) |
1.0 | 4 (4) 93 (96) 5 (7) 62 (93) |
0.48 | 95 (97) 3 (3) 63 (94) 4 (6) |
0.44 |
| Variable | Number (%) | E-cadherin(%) (-)(+) |
p | N-cadherin(%) (-) (+) |
p | Vimentin (%) (-) (+) |
p |
|---|---|---|---|---|---|---|---|
|
Gender Men Women |
113 (69) 52 (31) |
48 (43) 64 (57) 27 (52) 25 (48) |
0.27 | 108 (97) 3 (3) 49 (98) 1 (2) |
0.63 | 67 (60) 45 (40) 32 (61) 20 (39) |
0.83 |
|
Tobacco use Smoker Non-smoker |
107 (65) 58 (35) |
45 (42) 61 (58) 30 (52) 28 (48) |
0.25 | 103 (98) 2 (2) 54 (96) 2 (4) |
0.61 | 67 (63) 39 (37) 32 (55) 26 (45) |
0.31 |
|
Alcohol use Drinker Non-drinker |
89 (54) 76 (46) |
35 (40) 53 (60) 40 (53) 36 (47) |
0.09 | 73 (99) 1 (1) 84 (97) 3 (3) |
0.62 | 55 (63) 33 (37) 44 (58) 32 (42) |
0.54 |
|
pT pT1 + 2 pT3 + 4 |
114 (73) 43 (27) |
48 (42) 65 (58) 20 (46) 23 (54) |
0.65 | 110 (99) 1 (1) 39 (93) 3 (7) |
0.06 | 73 (64) 41 (36) 23 (55) 19 (45) |
0.29 |
|
pN pN0 pN+ |
95 (60) 63 (40) |
39 (41) 56 (59) 31 (50) 31 (50) |
0.27 | 91 (97) 3 (3) 60 (98) 1 (2) |
0.48 | 66 (69) 29 (31) 28 (45) 34 (55) |
0.002 |
|
Clinical stage I + II III + IV |
88 (53) 77 (47) |
34 (39) 54 (61) 41 (54) 35 (46) |
0.03 | 85 (99) 1 (1) 72 (96) 3 (4) |
0.33 | 61 (69) 27 (31) 38 (50) 38 (50) |
0.009 |
|
G status Well Moderate-poor |
105 (64) 60 (36) |
46 (44) 58 (56) 29 (48) 31 (51) |
0.61 | 102 (100) 0 (0) 55 (93) 4 (7) |
0.01 | 73 (70) 32 (30) 26 (44) 33 (56) |
0.001 |
|
Tumor recurrence No Yes |
83 (50) 82 (50) |
33 (40) 49 (60) 42 (51) 40 (49) |
0.15 | 79 (99) 1 (1) 78 (96) 3 (4) |
0.62 | 61 (73) 22 (27) 38 (47) 43 (53) |
0.001 |
|
Clinical status at the end of the follow-up Alive Dead of index cancer |
98 (59) 67 (41) |
41 (42) 56 (58) 34 (51) 35 (49) |
0.28 | 94 (99) 1 (1) 63 (96) 3 (4) |
0.30 | 69 (70) 29 (30) 30 (45) 36 (55) |
0.001 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




