Submitted:
24 April 2026
Posted:
28 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
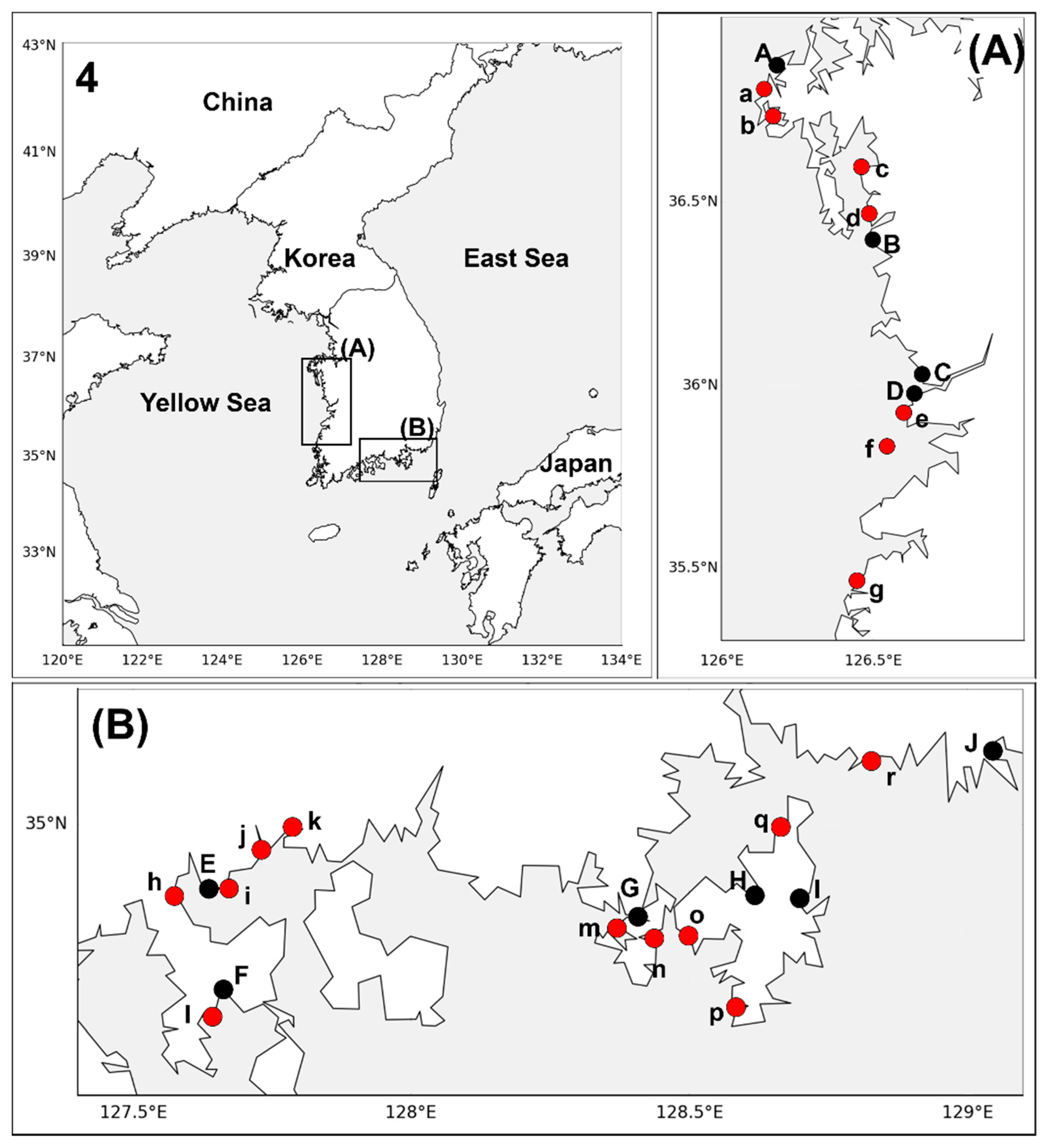
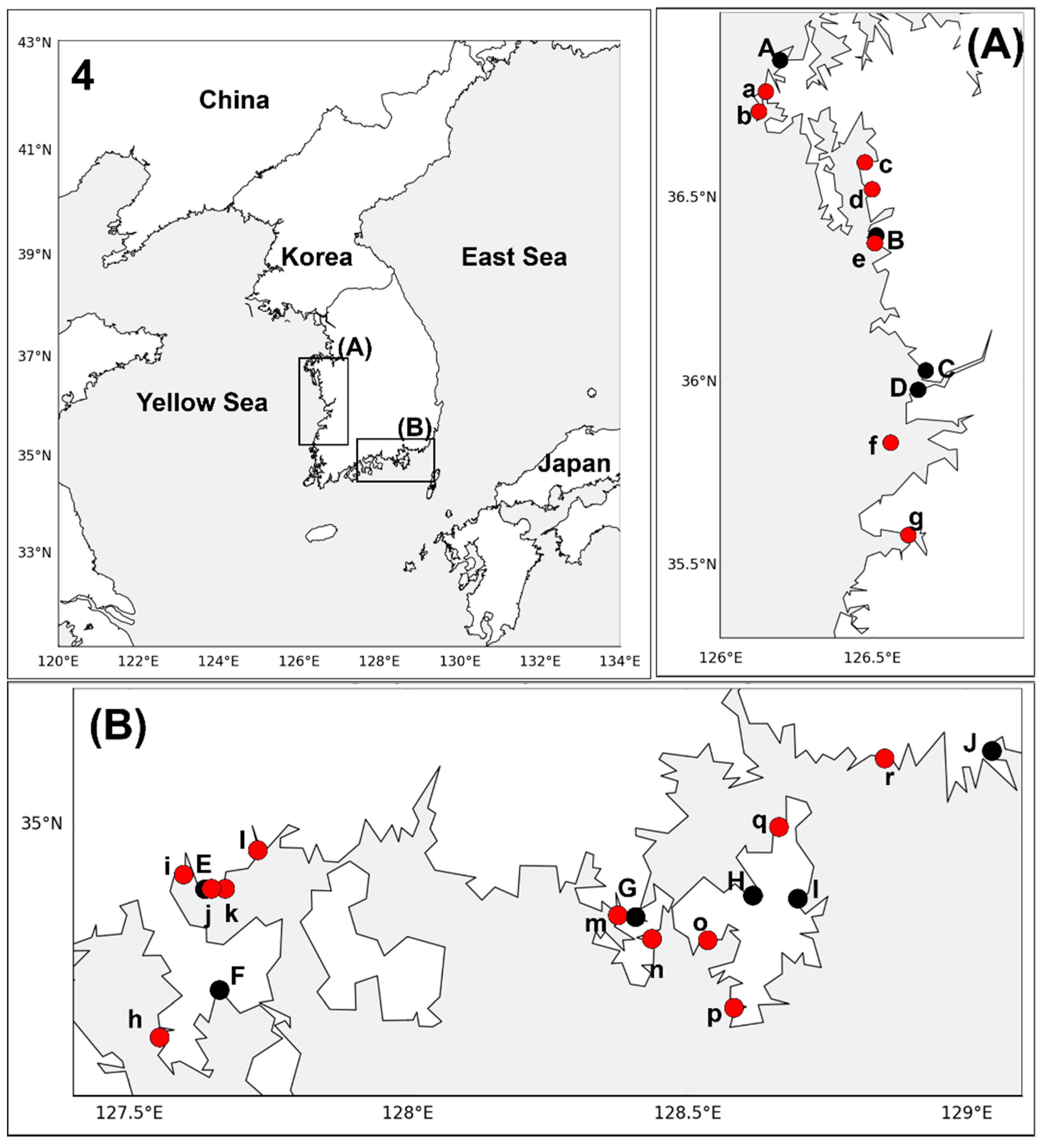
2.1. Sample Collection
2.2. PCR Assay
3. Results
3.1. PCR Screening Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Oidtmann, B.; Johnston, C.; Klotins, K.; Mylrea, G.; Van, P.T.; Cabot, S.; Martin, P.R.; Ababouch, L.; Berthe, F. Assessment of the Safety of Aquatic Animal Commodities for International Trade: The OIE Aquatic Animal Health Code. Transbound. Emerg. Dis. 2013, 60, 27–38. [Google Scholar] [CrossRef]
- Rodgers, C.J.; Mohan, C.V.; Peeler, E.J. The Spread of Pathogens through Trade in Aquatic Animals and Their Products. Rev. Sci. Tech. OIE 2011, 30, 241–256. [Google Scholar] [CrossRef] [PubMed]
- Fattorini, D. Bioaccumulation of Trace Elements in Mussels as Sentinels of Environmental Pollution in the Mediterranean Sea: A Review. EER 2025, 2, 8078. [Google Scholar] [CrossRef]
- Pagenkopp Lohan, K.M.; Hill-Spanik, K.M.; Torchin, M.E.; Fleischer, R.C.; Carnegie, R.B.; Reece, K.S.; Ruiz, G.M. Phylogeography and Connectivity of Molluscan Parasites: Perkinsus Spp. in Panama and Beyond. Int. J. Parasitol. 2018, 48, 135–144. [Google Scholar] [CrossRef] [PubMed]
- Itoh, N.; Komatsu, Y.; Maeda, K.; Hirase, S.; Yoshinaga, T. First Discovery of Perkinsus beihaiensis in Mediterranean Mussels (Mytilus galloprovincialis) in Tokyo Bay, Japan. J. Invertebr. Pathol. 2019, 166, 107226. [Google Scholar] [CrossRef]
- Yisrael, L.; DiMaria, R.A.; Santos-Ciminera, P.; Aguirre-Macedo, M.L.; Vidal-Martínez, V.M.; Ruiz, G.M.; Torchin, M.E.; Hill-Spanik, K.M.; Gnanadesikan, A.; Pagenkopp Lohan, K.M. Expanding the Phylogeography and Connectivity of Perkinsus Species across North and Central America. Divers. Distrib. 2024, 30, e13807. [Google Scholar] [CrossRef]
- Oyarzún, P.A.; Toro, J.E.; Nuñez, J.J.; Ruiz-Tagle, G.; Gardner, J.P.A. The Mediterranean Mussel Mytilus galloprovincialis (Mollusca: Bivalvia) in Chile: Distribution and Genetic Structure of a Recently Introduced Invasive Marine Species. Animals 2024, 14, 823. [Google Scholar] [CrossRef]
- Le Roux, F.; Lorenzo, G.; Peyret, P.; Audemard, C.; Figueras, A.; Vivarès, C.; Gouy, M.; Berthe, F. Molecular Evidence for the Existence of Two Species of Marteilia in Europe. J. Eukaryot. Microbiol. 2001, 48, 449–454. [Google Scholar] [CrossRef]
- Novoa, B.; Posada, D.; Figueras, A. Polymorphisms in the Sequences of Marteilia Internal Transcribed Spacer Region of the Ribosomal RNA Genes (ITS-1) in Spain: Genetic Types Are Not Related with Bivalve Hosts. J. Fish. Dis. 2005, 28, 331–338. [Google Scholar] [CrossRef]
- Lattos, A.; Papadopoulos, D.K.; Feidantsis, K.; Karagiannis, D.; Giantsis, I.A.; Michaelidis, B. Are Marine Heatwaves Responsible for Mortalities of Farmed Mytilus galloprovincialis? A Pathophysiological Analysis of Marteilia Infected Mussels from Thermaikos Gulf, Greece. Animals 2022, 12, 2805. [Google Scholar] [CrossRef]
- Carella, F.; Fernandez Tejedor, M.; Villari, G.; Andree, K.B.; De Vico, G. The Endoparasite Perkinsus Olseni Affecting the Mediterranean Mussels (Mytilus galloprovincialis) in the Italian and Spanish Waters: A New Possible Threat for Mussel Aquaculture and Wild Animal Population. Front. Mar. Sci. 2023, 10, 1116837. [Google Scholar] [CrossRef]
- Kim, S.-H.; Bathige, S.D.N.K.; Jeon, H.-B.; Lee, D.; Choi, K.-S.; Kim, H.J.; Park, K.-I. First Report of Perkinsus Marinus Occurrence Associated with Wild Pacific Oysters Crassostrea gigas from the West Coast of Korea. J. Invertebr. Pathol. 2024, 204, 108119. [Google Scholar] [CrossRef]
- Lynch, S.A.; Abollo, E.; Ramilo, A.; Cao, A.; Culloty, S.C.; Villalba, A. Observations Raise the Question If the Pacific Oyster, Crassostrea gigas, Can Act as Either a Carrier or a Reservoir for Bonamia ostreae or Bonamia Exitiosa. Parasitol. 2010, 137, 1515–1526. [Google Scholar] [CrossRef]
- Yang, M.; Wang, Q.; Chen, J.; Wu, H. The Occurrence of Potential Pathogenic Bacteria on International Ships’ Ballast Water at Yangshan Port, Shanghai, China. Mar. Pollut. Bull. 2022, 184, 114190. [Google Scholar] [CrossRef]
- Kang, H.-S.; Yang, H.-S.; Reece, K.S.; Cho, Y.-G.; Lee, H.-M.; Kim, C.-W.; Choi, K.-S. Survey on Perkinsus Species in Manila Clam Ruditapes philippinarum in Korean Waters Using Species-Specific PCR. Fish. Pathol. 2017, 52, 202–205. [Google Scholar] [CrossRef]
- Shin, J.-S.; Kim, H.J.; Hwang, J.Y.; Kwon, M.G.; Cho, Y.-G.; Choi, K.-S. Monitoring of Oie-Listed Parasitic Pathogens in the Pacific Oyster (Crassostrea gigas) on the South Coast of Korea Using Species-Specific PCR. J. Shellfish Res. 2022, 41. [Google Scholar] [CrossRef]
- Kim, S.-H.; Long, J.; Song, T.-S.; Lee, D.; Kim, H.J.; Jeon, H.-B.; Chen, Y.; Udayantha, H.M.V.; Lee, H.-M.; Arzul, I.; et al. First Report of Bonamia ostreae in Wild Flat Oysters (Ostrea denselamellosa) from the West Coast of Korea: Molecular and Histopathological Confirmation. J. Invertebr. Pathol. 2026, 214, 108489. [Google Scholar] [CrossRef] [PubMed]
- Cochennec, N.; Le Roux, F.; Berthe, F.; Gerard, A. Detection of Bonamia ostreae Based on Small Subunit Ribosomal Probe. J. Invertebr. Pathol. 2000, 76, 26–32. [Google Scholar] [CrossRef]
- Atencio, W.; Ziegler, S.; Greiman, S.; Carroll, J. Drivers of Perkinsus marinus and Haplosporidium nelsoni Prevalence and Intensity in Oyster Reefs around Sapelo Island, Georgia. Parasitol. 2025, 1–12. [Google Scholar] [CrossRef]
- Audemard, C.; Reece, K.S.; Burreson, E.M. Real-Time PCR for Detection and Quantification of the Protistan Parasite Perkinsus marinus in Environmental Waters. Appl. Environ. Microbiol. 2004, 70, 6611–6618. [Google Scholar] [CrossRef] [PubMed]
- Moss, J.A.; Xiao, J.; Dungan, C.F.; Reece, K.S. Description of Perkinsus beihaiensis n. Sp., a New Perkinsus Sp. Parasite in Oysters of Southern China. J. Eukaryot. Microbiol. 2008, 55, 117–130. [Google Scholar] [CrossRef] [PubMed]

| Species | Primers | Primers sequence (5’-3’) | Size (bp) | Target gene | Reference |
|---|---|---|---|---|---|
| Perkinsus marinus | PmarITS-70F | F: CTT TTG YTW GAG WGT TGC GAG ATG | 509 | Inter Transcribed Spacer of rDNA gene | [20] |
| PmarITS-600R | R: CGA GTT TGC GAG TAC CTC KAG AG | ||||
| P. olseni | HS12 | F: CGA AAC TAG CGG TCT TGC TTC GGC | 490 | Inter Transcribed Spacer of rDNA gene | Kang et al. (2017) |
| HS13 | R: AGG CGC GGT CCT CCT CKC G | ||||
| P. beihaiensis | PerkITS-85 | F: CCG CTT TGT TTG GAT CCC | 460 | Inter Transcribed Spacer of rDNA gene | [21] |
| PerkITS-430R | R: TCT GAG GGG CTA CAA TCA T | ||||
|
Bonamia ostreae, B. exitiosa |
BO | F: CAT TTA ATT GGT CGG GCC GC | 300 (B. ostreae) 304 (B. exitiosa) |
Small Sub Unit of rDNA gene | Cochennec et al. (2000) |
| BOAS | R: CTG ATC GTC TTC GAT CCC CC | ||||
| Marteilia refringens | Pr4 | F: CCG CAC ACG TTC TTC ACT CC | 412 (M type) 413 (O type) |
Inter Transcribed Spacer of rDNA gene | Le Roux et al. (2001) |
| Pr5 | R: CTC GCG AGT TTC GAC AGA CG |
| Site | N | SL (mm) mean±SD | TWT (g) mean±SD | Infection prevalence (%) | ||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Perkinsus | Bonamia | Marteilia | ||||||||||||||||||||
| marinus | olseni | beihaiensis | ostreae | exitiosa | refringens | |||||||||||||||||
| May | Sep. | May | Sep. | May | Sep. | May | Sep. | May | Sep. | May | Sep. | May | Sep. | May | Sep. | |||||||
| Western Coast | Taean Port | Eoeundol Port | 60 | 59.9±7.6 | 27.5±3.9 | 4.6±2.2 | 0.6±0.2 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |||
| Tonggae Port | 60 | 35.5±7.9 | 60.7±7.1 | 1.6±1.2 | 5.6±1.9 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |||||
| Boryeong Port | Gungri | 60 | 57.9±3.8 | 62.4±7.1 | 6.1±1.4 | 7.0±2.1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | ||||
| Namdang Port | 60 | 49.3±5.2 | 59.0±6.3 | 4.2±1.4 | 6.2±2.2 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |||||
| Janghang Port | Bieung Port | 60 | 67.2±6.8 | 31.0±2.9 | 7.3±2.1 | 0.8±0.3 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | ||||
| Gunsan Port | Jangja-do | 60 | 55.1±9.6 | 36.5±6.9 | 5.2±2.3 | 1.3±0.8 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | ||||
| Gusipo Port | 60 | 33.7±2.4 | 63.6±6.0 | 1.4±0.3 | 6.7±2.0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |||||
| South Coast | Gwangyang Port | Gwangyang Police Station | 60 | 59.9±9.8 | 70.6±7.1 | 5.9±3.2 | 9.3±3.4 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |||
| Dochon Ferry Terminal |
60 | 50.5±2.2 | 74.9±8.3 | 5.4±1.2 | 9.1±2.6 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |||||
| Johwa-ri | 60 | 42.2±4.9 | 35.3±9.4 | 2.9±1.1 | 1.3±1.1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |||||
| Mangdeok Port | 60 | 46.6±5.8 | 57.8±6.7 | 4.2±1.7 | 3.7±1.6 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |||||
| Yeosu Port | Hodu-ri | 60 | 60.7±4.0 | 58.5±6.7 | 5.3±1.0 | 4.3±1.5 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | ||||
| Tongyeong Port | Pyeongrim Port | 60 | 49.1±7.5 | 63.5±6.7 | 5.3±2.4 | 4.2±1.4 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | ||||
| Hwasam-ri | 60 | 50.0±4.2 | 74.0±6.2 | 5.1±1.5 | 9.6±2.9 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |||||
| Gohyeon Port | Eogu-ri | 60 | 46.5±7.8 | 30.3±2.7 | 3.8±2.0 | 0.7±0.2 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | ||||
| Myeongsa-Po | 60 | 67.5±5.7 | 55.9±8.1 | 5.9±1.5 | 3.8±1.7 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |||||
| Okpo Port | Jangmok-ri | 60 | 65.6±5.6 | 39.2±3.6 | 6.5±2.3 | 1.6±0.5 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | ||||
| Busan Port | Angolpo | 60 | 46.7±8.3 | 60.8±8.3 | 4.0±2.6 | 5.5±3.1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | ||||
| Site | N | SL (mm) mean±SD | TWT (g) mean±SD | Infection prevalence (%) | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Perkinsus | Bonamia | Marteilia | |||||||||||||||||||||
| marinus | olseni | beihaiensis | ostreae | exitiosa | refringens | ||||||||||||||||||
| May | Sep. | May | Sep. | May | Sep. | May | Sep. | May | Sep. | May | Sep. | May | Sep. | May | Sep. | ||||||||
| Western Coast | Taean Port | Mohang Port | 60 | 56.8±8.5 | 43.4±5.9 | 2.5±1.0 | 2.1±1.1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | ||||
| Pado-ri | 60 | 79.1±11.5 | 61.5±6.3 | 6.2±2.3 | 3.5±1.5 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | ||||||
| Boryeong Port | Gungri | 60 | 67.8±7.0 | 63.5±12.1 | 3.6±0.8 | 3.7±1.6 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |||||
| Suryong Port | 60 | 49.3±6.7 | 59.9±14.8 | 2.0±0.5 | 3.3±1.8 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | ||||||
| Janghang Port | Janghang Port | 60 | 21.3±4.7 | 45.3±7.9 | 3.1±1.5 | 1.0±0.3 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |||||
| Gunsan Port | Seonyu Port | 60 | 53.1±7.4 | 52.7±7.1 | 5.4±1.4 | 3.1±0.9 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |||||
| Gomso Port | 60 | 50.0±8.3 | 55.6±9.6 | 3.9±1.6 | 3.4±1.2 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | ||||||
| South Coast | Gwangyang Port | Gwangyang Port | 60 | 58.7±11.0 | 55.1±8.0 | 6.5±2.0 | 2.6±1.5 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | ||||
| Gwangyang Police Station | 60 | 77.2±11.2 | 40.9±6.6 | 6.8±2.7 | 0.9±0.7 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | ||||||
| Chonam Bridge | 60 | 80.0±14.2 | 54.6±7.1 | 6.0±3.5 | 2.9±1.0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | ||||||
| Taein Bridge | 60 | 73.0±10.7 | 45.4±6.5 | 5.4±1.8 | 1.4±0.5 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | ||||||
| Yeosu Port | Imok-ri | 60 | 42.0±5.0 | 43.6±6.9 | 1.4±0.4 | 1.7±0.5 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |||||
| Tongyeong Port | Pyeongrim Port | 60 | 66.0±10.0 | 75.4±21.3 | 5.8±2.6 | 8.4±7.2 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |||||
| Dongam Port | 60 | 63.8±13.5 | 57.2±7.7 | 4.4±2.2 | 2.3±0.6 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | ||||||
| Gohyeon Port | Daepo Port | 60 | 43.7±5.5 | 47.0±4.8 | 1.5±0.6 | 2.3±0.8 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |||||
| Seondaldo Ferry Terminal |
60 | 56.4±8.8 | 54.4±20.1 | 4.1±1.6 | 3.5±2.9 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | ||||||
| Okpo Port | Jangmok-ri | 60 | 75.0±17.9 | 53.1±13.1 | 13.3±7.3 | 3.1±2.2 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |||||
| Busan Port | Busan New Port | 60 | 54.4±6.1 | 58.7±7.8 | 2.9±0.8 | 4.0±1.3 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




