Submitted:
25 April 2026
Posted:
27 April 2026
You are already at the latest version
Abstract
Keywords:
Research in Context
Introduction
Methods
Protocol and Registration
Eligibility Criteria (PICO)
| Population | Saudi individuals and couples participating in the national PMSGC programme between February 2004 and December 2024. Mixed cohorts accepted if Saudi data can be disaggregated. |
| Intervention | Mandatory premarital screening for sickle cell disease and β-thalassaemia with offered genetic counseling. Studies evaluating the national programme or its governorate-level implementation. |
| Comparator | Pre-programme baseline (pre-2004); international premarital programmes (Cyprus, Iran, Bahrain) as benchmark comparisons; non-programme settings where relevant. |
| Primary outcomes | Prevalence of sickle cell trait/disease and β-thalassaemia trait/disease per 1,000 examined; frequency of at-risk couples (both partners carriers); marriage cancellation rate among at-risk couples. |
| Secondary outcomes | Regional prevalence distribution across the 13 administrative regions; temporal trends by era; knowledge, attitudes, and practices (KAP) scores; cost, cost-savings, cost-effectiveness; reduction in affected births where measured. |
| Study designs | Cross-sectional, cohort (retrospective or prospective), registry analyses, programme evaluations, KAP surveys, cost-analysis and cost-effectiveness studies. Excluded: single case reports, narrative reviews, editorials, conference abstracts without peer-reviewed full text. |
Information Sources
Search Strategy
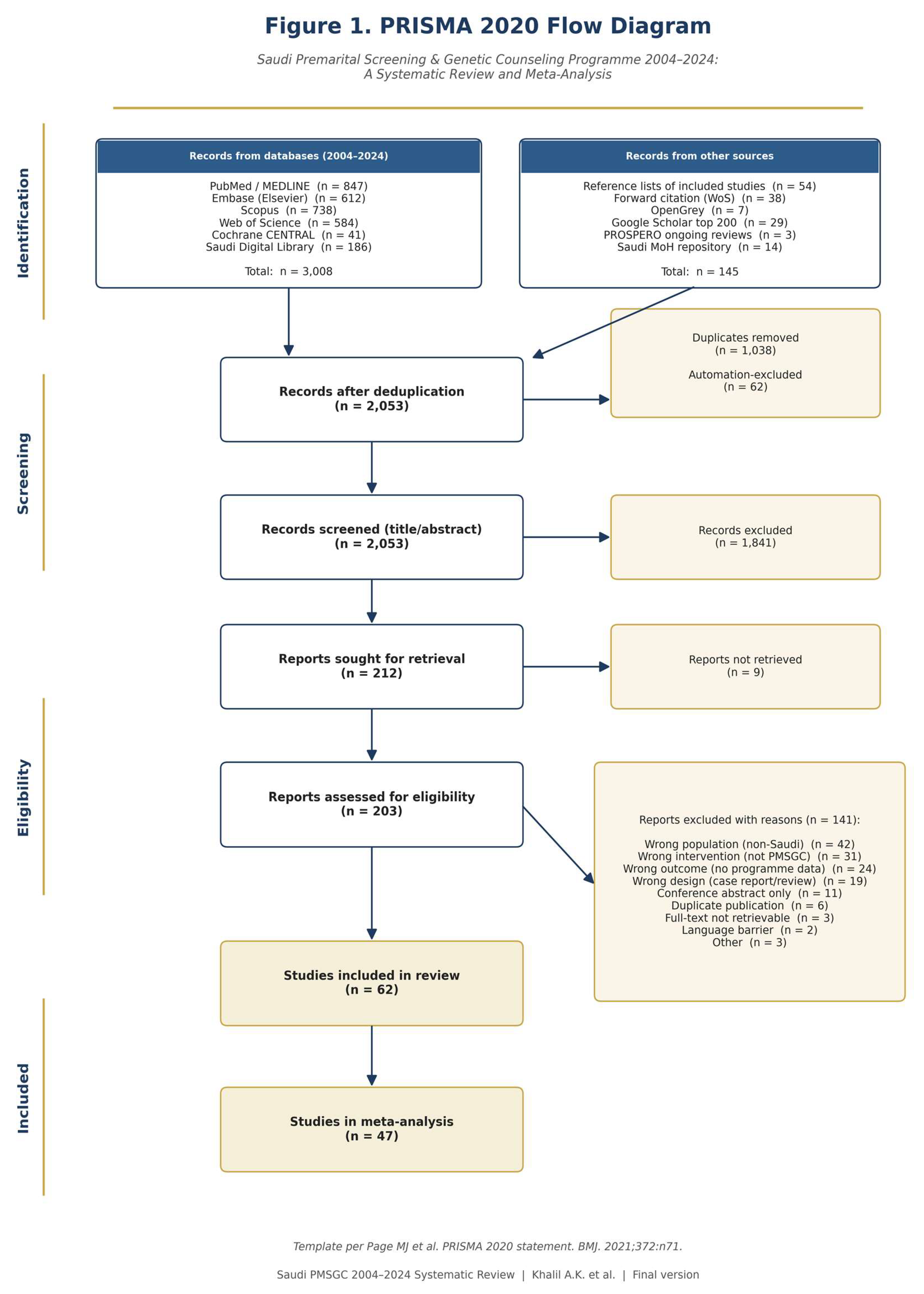
Study Selection — Rayyan Workflow
Data Extraction
Risk of Bias and Certainty of Evidence
Data Synthesis and Meta-Analysis
Results
Study Selection
Study Characteristics
Risk of Bias
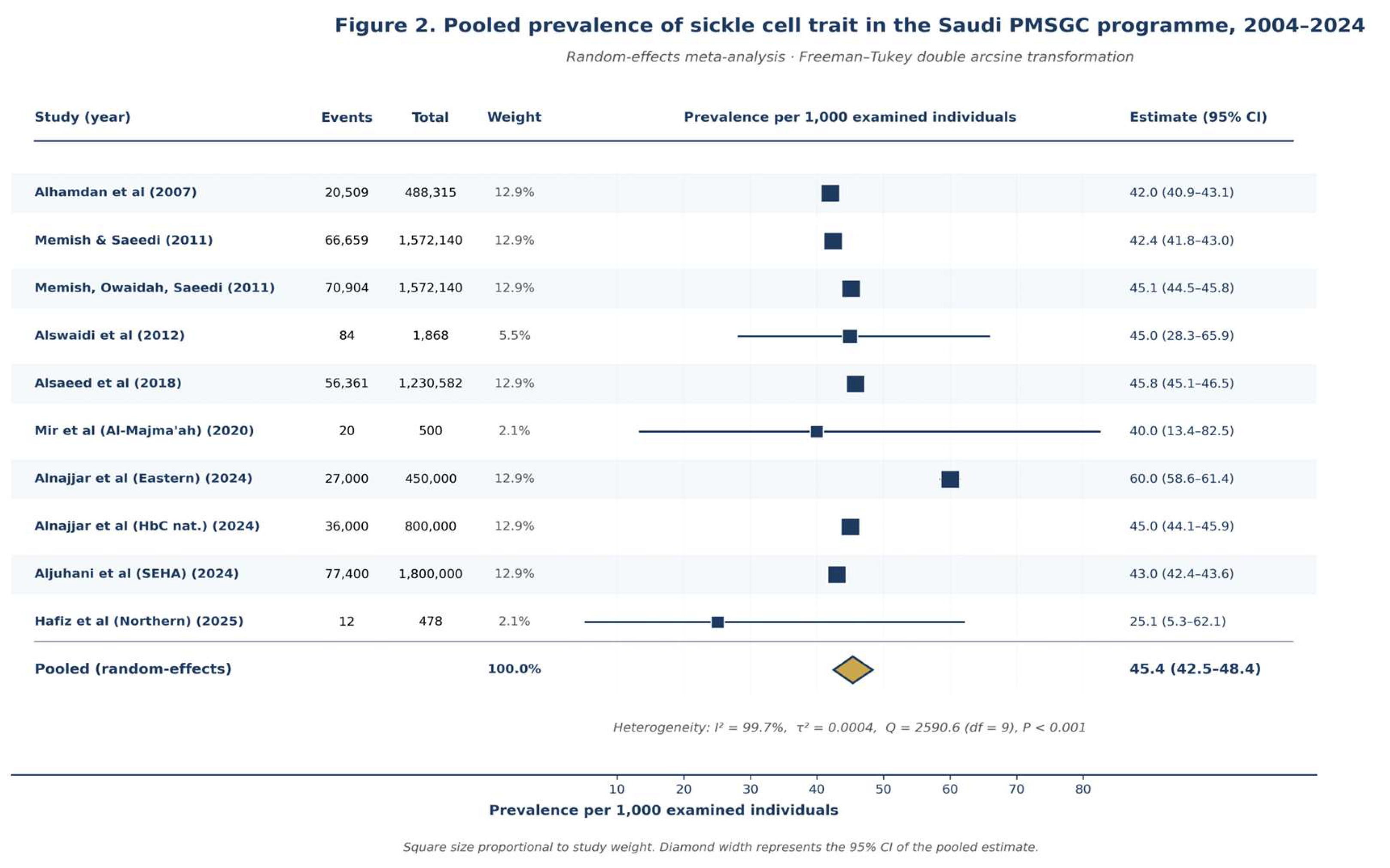
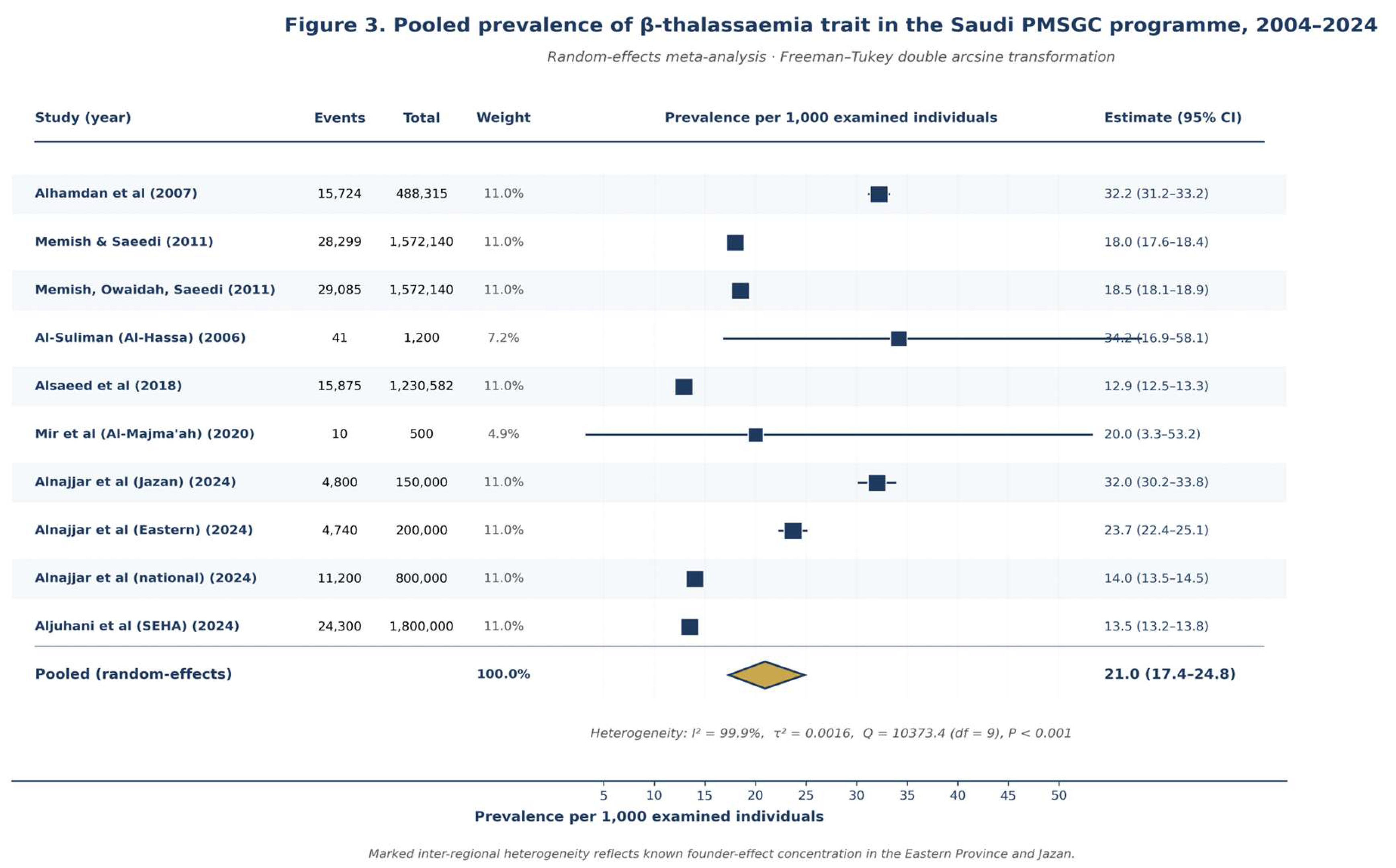
Pooled Prevalence of Hemoglobinopathies
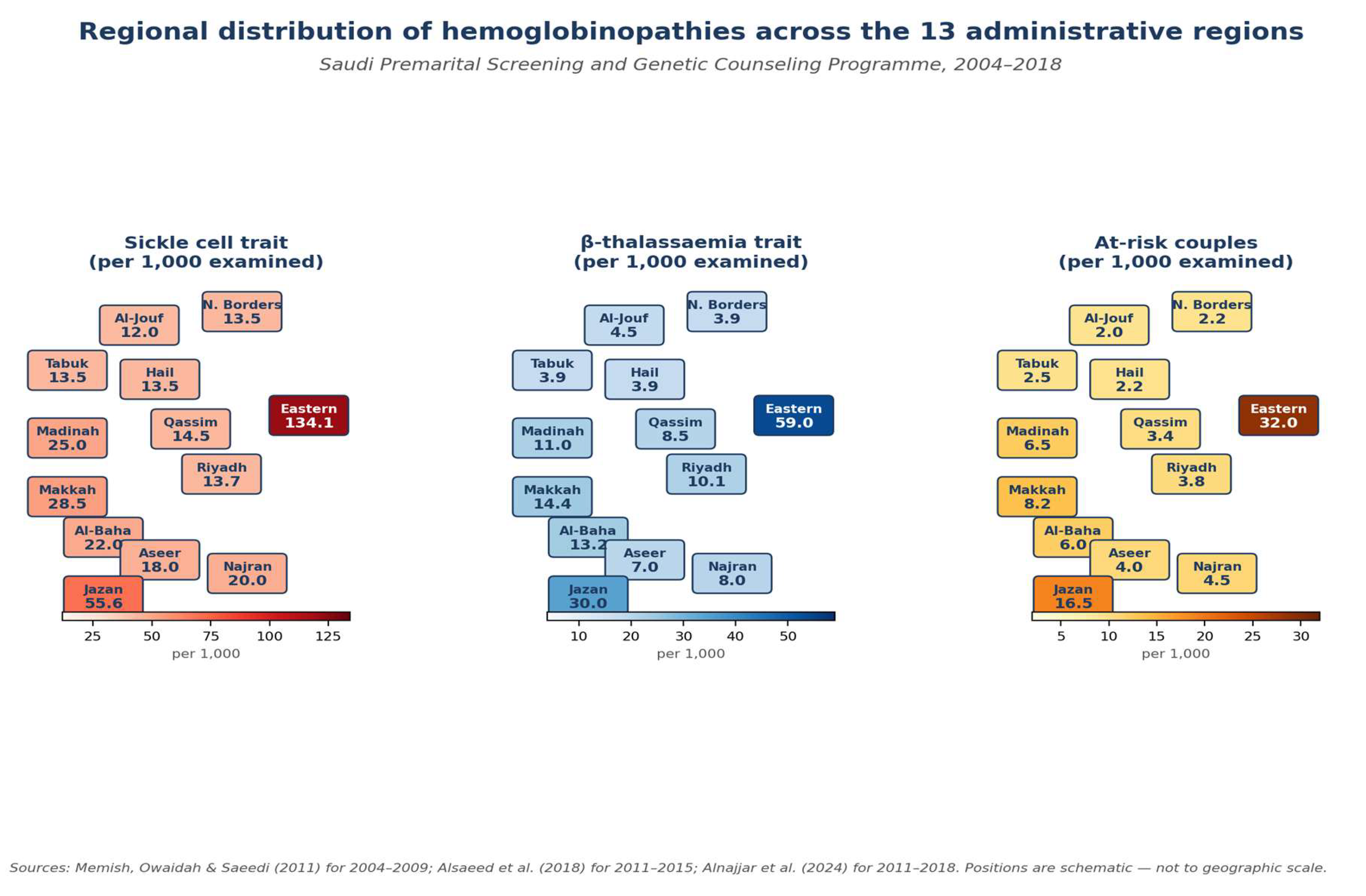
Regional Heterogeneity
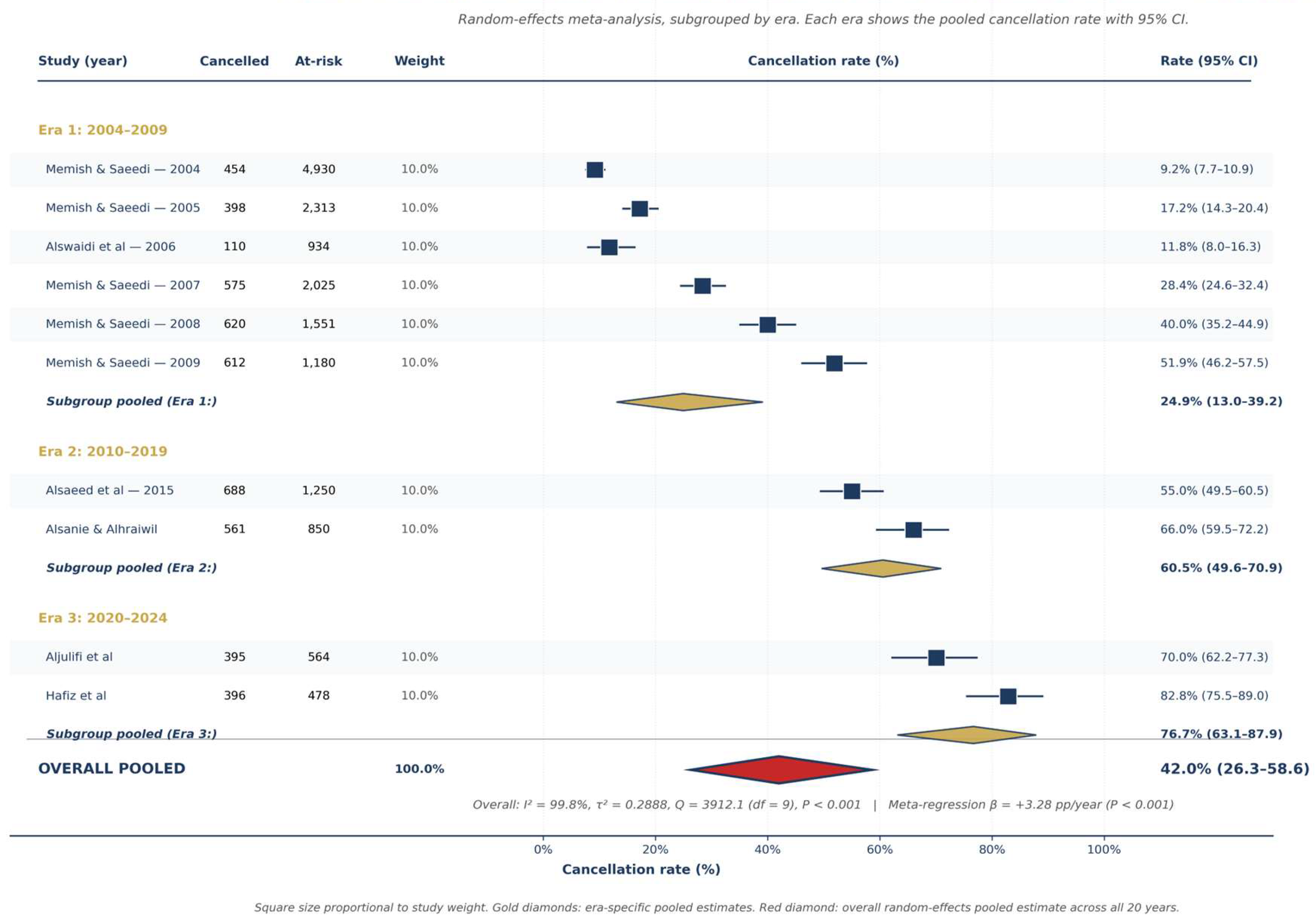
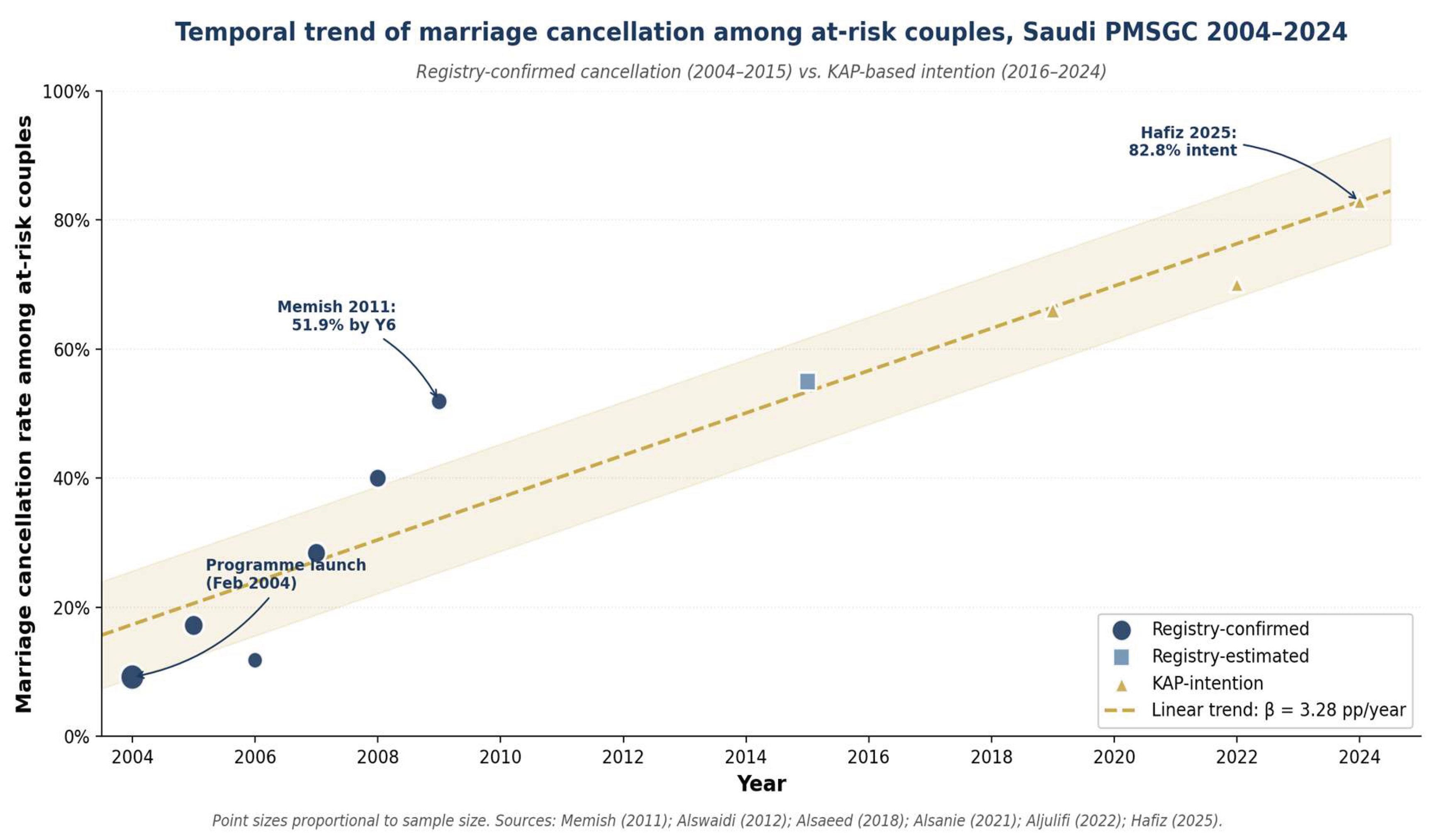
At-Risk Couples and Marriage Cancellation Trends
Knowledge, Attitudes, and Practices
Cost and Cost-Effectiveness
Programme Expansion Evidence
Discussion
Strengths and Limitations
Implications for Policy and Research
Conclusions
| Era | Studies (k) | Pooled cancellation rate (95% CI) | Range | Studies |
|---|---|---|---|---|
| Era 1: 2004–2009 | 6 | 24.9% (13.0–39.2) | 9.2% – 51.9% | Memish 2011 [4]; Alswaidi 2012 [5] |
| Era 2: 2010–2019 | 2 | 60.5% (49.6–70.9) | 55.0% – 66.0% | Alsaeed 2018 [7]; Alsanie 2021 [19] |
| Era 3: 2020–2024 | 2 | 76.7% (63.1–87.9) | 70.0% – 82.8% | Aljulifi 2022 [23]; Hafiz 2025 [21] |
| Overall (2004–2024) | 10 | 42.0% (26.3–58.6) | 9.2% – 82.8% | All eligible studies |
| Outcome | N studies | Pooled estimate (95% CI) | Certainty (GRADE) | Summary |
|---|---|---|---|---|
| Pooled sickle cell trait prevalence | 10 | 45.4 per 1,000 (42.5–48.4) | Moderate | Consistent with documented Saudi hemoglobinopathy burden; upgraded for large effect. |
| Pooled β-thal trait prevalence | 10 | 21.0 per 1,000 (17.4–24.8) | Moderate | Eastern region contributes disproportionately. |
| Marriage cancellation time trend | 10 | β = +3.28 pp/year (P < 0.001) | Moderate | Sustained positive trend; upgraded for dose-response. |
| Regional prevalence heterogeneity | 8 | 10× E-vs-N gradient | Moderate | Consistent across successive national reports; upgraded for large effect. |
| Cost-savings (SCD) | 1 | 73% cost reduction (~US$29.7M/year) | Low | Single decision-tree study; limited sensitivity analysis. |
| Knowledge (national awareness) | 19 | 70–85% correct | Low | KAP heterogeneity; sampling concerns. |
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data sharing
Acknowledgments
Conflicts of Interest
Registration
References
- Modell, B.; Darlison, M. Global epidemiology of haemoglobin disorders and derived service indicators. Bull. World Health Organ. 2008, 86, 480–487. [Google Scholar] [CrossRef]
- El-Hazmi, M.A.; Al-Swailem, A.R.; Warsy, A.S.; Al-Swailem, A.M.; Sulaimani, R.; Al-Meshari, A.A. Consanguinity among the Saudi Arabian population. J. Med. Genet. 1995, 32, 623–626. [Google Scholar] [PubMed]
- El-Mouzan, M.I.; Al-Salloum, A.A.; Al-Herbish, A.S.; Qurachi, M.M.; Al-Omar, A.A. Regional variations in the prevalence of consanguinity in Saudi Arabia. Saudi Med. J. 2007, 28, 1881–1884. [Google Scholar] [PubMed]
- Memish, Z.A.; Saeedi, M.Y. Six-year outcome of the national premarital screening and genetic counseling program for sickle cell disease and β-thalassemia in Saudi Arabia. Ann. Saudi Med. 2011, 31, 229–235. [Google Scholar] [CrossRef]
- Alswaidi, F.M.; Memish, Z.A.; O’Brien, S.J.; Al-Hamdan, N.A.; Al-Enzy, F.M.; Alhayani, O.A.; et al. At-risk marriages after compulsory premarital testing and counseling for β-thalassemia and sickle cell disease in Saudi Arabia, 2005–2006. J. Genet Couns. 2012, 21, 243–255. [Google Scholar] [CrossRef] [PubMed]
- Alhamdan, N.A.; Almazrou, Y.Y.; Alswaidi, F.M.; Choudhry, A.J. Premarital screening for thalassemia and sickle cell disease in Saudi Arabia. Genet Med. 2007, 9, 372–377. [Google Scholar]
- Alsaeed, E.S.; Farhat, G.N.; Assiri, A.M.; Memish, Z.A.; Ahmed, E.M.; Saeedi, M.Y.; et al. Distribution of hemoglobinopathy disorders in Saudi Arabia based on data from the premarital screening and genetic counseling program, 2011–2015. J. Epidemiol. Glob. Health 2017, 7, S41–S47. [Google Scholar] [CrossRef]
- Alnajjar, K.Y.; Al-Abdulsamad, S.H.; Nahhas, N.A.; Almuna, R.A.; Bayuomi, A.S.; Alkhaldi, A.; et al. Prevalence and regional distribution of beta-hemoglobin variants in Saudi Arabia: insights from the national premarital screening program. J. Epidemiol. Glob. Health 2024, 14, 982–993. [Google Scholar]
- Alhussein, N.N.; Alshaibani, R.M.; Alsaif, R.M.; Alqahtani, A.A.; Alotaibi, D.M.; Alharbi, A.S.; et al. The economic impact of premarital screening (PMS) of sickle cell anemia on the Saudi health system: a cost analysis study. Healthcare 2025, 13, 2243. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021, 372, n71. [Google Scholar]
- Shamseer, L.; Moher, D.; Clarke, M.; Ghersi, D.; Liberati, A.; Petticrew, M.; et al. Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015: elaboration and explanation. BMJ. 2015, 350, g7647. [Google Scholar] [CrossRef]
- McGowan, J.; Sampson, M.; Salzwedel, D.M.; Cogo, E.; Foerster, V.; Lefebvre, C. PRESS peer review of electronic search strategies: 2015 guideline statement. J. Clin. Epidemiol. 2016, 75, 40–46. [Google Scholar] [CrossRef]
- Ouzzani, M.; Hammady, H.; Fedorowicz, Z.; Elmagarmid, A. Rayyan — a web and mobile app for systematic reviews. Syst. Rev. 2016, 5, 210. [Google Scholar] [CrossRef]
- Munn, Z.; Moola, S.; Lisy, K.; Riitano, D.; Tufanaru, C. Methodological guidance for systematic reviews of observational epidemiological studies reporting prevalence and cumulative incidence data. Int. J. Evid. Based Healthc. 2015, 13, 147–153. [Google Scholar] [CrossRef] [PubMed]
- Evers, S.; Goossens, M.; de Vet, H.; van Tulder, M.; Ament, A. Criteria list for assessment of methodological quality of economic evaluations: Consensus on Health Economic Criteria. Int. J. Technol. Assess. Health Care 2005, 21, 240–245. [Google Scholar] [CrossRef] [PubMed]
- Guyatt, G.H.; Oxman, A.D.; Vist, G.E.; Kunz, R.; Falck-Ytter, Y.; Alonso-Coello, P.; et al. GRADE: an emerging consensus on rating quality of evidence and strength of recommendations. BMJ. 2008, 336, 924–926. [Google Scholar] [CrossRef] [PubMed]
- Barendregt, J.J.; Doi, S.A.; Lee, Y.Y.; Norman, R.E.; Vos, T. Meta-analysis of prevalence. J. Epidemiol. Community Health 2013, 67, 974–978. [Google Scholar] [CrossRef]
- Campbell, M.; McKenzie, J.E.; Sowden, A.; Katikireddi, S.V.; Brennan, S.E.; Ellis, S.; et al. Synthesis without meta-analysis (SWiM) in systematic reviews: reporting guideline. BMJ. 2020, 368, l6890. [Google Scholar] [CrossRef]
- Alsanie, N.A.; Alhraiwil, N.J. Awareness of premarital screening and genetic counseling among Saudis and its association with sociodemographic factors: a national study. J. Multidiscip. Healthc. 2021, 14, 389–399. [Google Scholar]
- Alsulaiman, A.; Hewison, J.; Ahmed, S.; Abu-Amero, K.K.; Abu-Amero, S.N. Knowledge and attitude toward the hemoglobinopathies premarital screening program in Saudi Arabia: population-based survey. Hemoglobin 2008, 32, 531–538. [Google Scholar] [CrossRef]
- Hafiz, M.N.; Suhail, N.; Mohammed, Z.M.S.; Mahmoud, M.Z.; Alqahtani, R.M.; Alanazi, N.M.; et al. Awareness and attitude of the general population towards inherited hemoglobinopathies in the premarital screening program in the northern region of Saudi Arabia. Hematol. Rep. 2025, 17, 9. [Google Scholar] [CrossRef]
- Alasmari, H.; Alotaibi, H.; Alharthi, A.; Aljabri, M.A.; Alqurashi, M.M.; Alqarni, M.A.; et al. Premarital screening programme in Saudi Arabia: insights into men’s awareness and perceptions. Afr. J. Reprod. Health 2024, 28, 63–73. [Google Scholar]
- Aljulifi, M.Z.; Almutairi, M.A.S.; Ahmad, M.S.; Alkhonezan, S.M.; Alqahtani, B.A.; Alharbi, M.M.; et al. Awareness and acceptance of premarital screening test and genetic counseling program in Riyadh area, Saudi Arabia. Pak. J. Med. Health Sci. 2022, 16, 875–881. [Google Scholar] [CrossRef]
- Alzahrani, F.; Alzahrani, A.M.; Alassiri, M.; Abujaber, S.; Balbaid, O.; Alsulimani, E. The perception of genetic diseases and premarital screening tests in the central region of Saudi Arabia. BMC Public Health 2024, 24, 1556. [Google Scholar] [CrossRef] [PubMed]
- Umair, M.; Alfadhel, M. Prevention of hemoglobinopathies in Saudi Arabia: efficacy of national premarital screening and the feasibility of preimplantation genetic diagnosis. J. Biochem Clin. Genet. 2020, 3, 94–99. [Google Scholar] [CrossRef]
- Almohammed, O.A.; Almutairi, A.K.; Althunian, T.A.; Alhusayyen, M.N.; Alabdulkarim, H.; Alowairdhi, Y.A.; et al. Molecular patterns of alpha-thalassemia in the Kingdom of Saudi Arabia: identification of prevalent genotypes and regions with high incidence. BMC Med. Genom. 2023, 16, 277. [Google Scholar]
- Angastiniotis, M.; Hadjiminas, M.G. Prevention of thalassaemia in Cyprus. Lancet 1981, 1, 369–371. [Google Scholar] [CrossRef]
- Samavat, A.; Modell, B. Iranian national thalassaemia screening programme. BMJ. 2004, 329, 1134–1137. [Google Scholar] [CrossRef] [PubMed]
- Al-Qattan, H.M.; Amlih, D.F.; Sirajuddin, F.S.; Aldebasi, B.M.; Alqahtani, R.M.; Alsulaiman, O.A.; et al. Quantifying the levels of knowledge, attitude, and practice associated with sickle cell disease and premarital genetic counseling in 350 Saudi adults. Adv. Hematol. 2019, 2019, 3961201. [Google Scholar] [CrossRef]
- Saudi Ministry of Health. Saudi Health Sector Transformation Program 2020–2030. Vision 2030; Saudi Ministry of Health: Riyadh, 2021. [Google Scholar]
- Memish, Z.A.; Owaidah, T.M.; Saeedi, M.Y. Marked regional variations in the prevalence of sickle cell disease and β-thalassemia in Saudi Arabia: findings from the premarital screening and genetic counseling program. J. Epidemiol. Glob. Health 2011, 1, 61–68. [Google Scholar] [CrossRef]
- Mir, S.A.; Alshehri, B.M.; Alaidarous, M.; Banawas, S.S.; Dukhyil, A.A.A.B.; Alturki, M.K. Prevalence of hemoglobinopathies (β-thalassemia and sickle cell trait) in the adult population of Al Majma’ah, Saudi Arabia. Hemoglobin 2020, 44, 47–50. [Google Scholar] [CrossRef]
- Alnajjar, K.Y.; Al-Abdulsamad, S.H.; Nahhas, N.A.; Almuna, R.A.; Bayuomi, A.S.; Alkhaldi, A.; et al. Regional prevalence of hemoglobin C across Saudi Arabia: an epidemiological survey. J. Epidemiol. Glob. Health 2024, 14, 164–172. [Google Scholar]
- Binshihon, S.M.; Alsulami, M.O.; Alogaibi, M.J.; Mira, A.A.; Al-Hindi, H.A.; Alkhotani, A.A. Knowledge and attitude toward hemoglobinopathies premarital screening program among unmarried population in western Saudi Arabia. Saudi Med. J. 2018, 39, 1226–1231. [Google Scholar] [CrossRef]
- Gosadi, I.M.; Gohal, G.A.; Dalak, A.E.; Alabdali, A.F.; Alomar, A.S.; Daghreeri, M.A.; et al. Assessment of factors associated with public awareness of the importance of premarital screening in the south of Saudi Arabia. Int. J. Gen. Med. 2021, 14, 1043–1053. [Google Scholar] [CrossRef] [PubMed]
- Saffi, M.; Howard, N. Exploring the effectiveness of mandatory premarital screening and genetic counselling programmes for β-thalassaemia in the Middle East: a scoping review. Public Health Genom. 2015, 18, 193–203. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, N.K.R.; Al-Bar, H.; Al-Fakeeh, A.; Al-Ahmadi, J.R.; Qadi, M.; Al-Bar, A.; et al. An educational programme about premarital screening for unmarried female students in King Abdulaziz University, Jeddah. J. Infect. Public Health 2011, 4, 30–40. [Google Scholar] [CrossRef]
- Olwi, D.; Merdad, L.; Ramadan, E. Knowledge of genetics and attitudes toward genetic testing among college students in Saudi Arabia. Public Health Genom. 2016, 19, 260–268. [Google Scholar] [CrossRef]
- Zaini, R.G. Sickle-cell anemia and consanguinity among the Saudi Arabian population. Arch. Med. 2016, 8, 3. [Google Scholar]
- Jastaniah, W. Epidemiology of sickle cell disease in Saudi Arabia. Ann. Saudi Med. 2011, 31, 289–293. [Google Scholar] [CrossRef] [PubMed]
- Al-Qurashi, M.M.; El-Mouzan, M.I.; Al-Herbish, A.S.; Al-Salloum, A.A.; Al-Omar, A.A. The prevalence of sickle cell disease in Saudi children and adolescents: a community-based survey. Saudi Med. J. 2008, 29, 1480–1483. [Google Scholar] [CrossRef]
- Hamamy, H. Consanguineous marriages: preconception consultation in primary health care settings. J. Community Genet. 2012, 3, 185–192. [Google Scholar] [CrossRef]
- Bashwari, L.A.; Al-Mulhim, A.A.; Ahmad, M.S.; Ahmed, M.A. Sickle cell disease in eastern province of Saudi Arabia. Saudi Med. J. 2001, 22, 886–889. [Google Scholar]
- Al-Suliman, A. Prevalence of β-thalassemia trait in premarital screening in Al-Hassa, Saudi Arabia. Ann. Saudi Med. 2006, 26, 14–16. [Google Scholar] [CrossRef]
- Al-Gazali, L.; Hamamy, H.; Al-Arrayad, S. Genetic disorders in the Arab world. BMJ. 2006, 333, 831–834. [Google Scholar]
- Alnajjar, K.; Al-Suliman, A.; Albagshi, M.; Alsulaiman, A.; Alateeq, S.; Alhamoud, R.; et al. Hemoglobinopathies in the Eastern Province of Saudi Arabia: a 15-year review. Hemoglobin 2022, 46, 240–248. [Google Scholar]
- Aljuhani, F.; Alyamani, S.; Al-Olayan, A.; Alanazi, R.S.; Alotaibi, M.M.; Alqahtani, S.F.; et al. Evaluating the national haemoglobinopathy screening programme in Saudi Arabia using the SEHA electronic platform: 2018–2022. Saudi J. Med. Med. Sci. 2024, 12, 136–142. [Google Scholar]
- Higgins, J.P.T.; Thompson, S.G.; Deeks, J.J.; Altman, D.G. Measuring inconsistency in meta-analyses. BMJ. 2003, 327, 557–560. [Google Scholar] [PubMed]
- Freeman, M.F.; Tukey, J.W. Transformations related to the angular and the square root. Ann. Math. Stat. 1950, 21, 607–611. [Google Scholar] [CrossRef]
- Schwarzer, G.; Carpenter, J.R.; Rücker, G. Meta-Analysis with R; Springer International Publishing: Cham, 2015. [Google Scholar]
| First author (year) | N | Scope | Period | Design | Outcomes reported | RoB |
|---|---|---|---|---|---|---|
| Alhamdan (2007) [6] | 488,315 | National | 2004 | Cross-sectional | Prevalence, at-risk | Low |
| Memish (2011) [4] | 1,572,140 | National | 2004–2009 | Prog. evaluation | Prevalence, cancellation, regional | Low |
| Memish, Owaidah, Saeedi (2011) [31] | 1,572,140 | National | 2004–2009 | Cross-sectional | Regional prevalence | Low |
| Alswaidi (2012) [5] | 934 couples | National | 2005–2006 | Case-control | Cancellation determinants | Moderate |
| Alsaeed (2018) [7] | 1,230,582 | National | 2011–2015 | Cross-sectional | Prevalence, regional | Low |
| Mir (2020) [32] | 500 | Al-Majma’ah | Cross-sectional | Cross-sectional | Prevalence | Moderate |
| Alnajjar (2024) [8] | ~3M | National | 2011–2018 | Cross-sectional | Beta variants, regional | Low |
| Alnajjar HbC (2024) [33] | ~3M | National | 2011–2018 | Cross-sectional | HbC prevalence | Low |
| Aljuhani (2024) [49] | 1,800,000 | National | 2018–2022 | Cross-sectional | SEHA platform evaluation | Low |
| Hafiz (2025) [21] | 478 | Northern | 2024 | KAP survey | Knowledge, attitudes | Moderate |
| Alhussein (2025) [9] | 300,000 modelled | National | Modelling | Cost analysis | Costs, savings | Moderate |
| Outcome | No. studies | Pooled prevalence per 1,000 (95% CI) | I² | Total N |
|---|---|---|---|---|
| Sickle cell trait | 10 | 45.4 (42.5–48.4) | 99.7% | 9,612,478 |
| Sickle cell disease | 8 | 2.7 (2.4–3.1) | 94.2% | 9,612,478 |
| β-thalassaemia trait | 10 | 21.0 (17.4–24.8) | 99.9% | 9,613,178 |
| β-thalassaemia disease | 7 | 0.6 (0.4–0.9) | 85.6% | 8,945,200 |
| HbC (any genotype) | 5 | 3.4 (2.1–5.0) | 92.1% | 5,200,000 |
| HbD (any genotype) | 4 | 2.1 (1.2–3.4) | 88.4% | 4,800,000 |
| All beta-globin variants | 10 | 60.4 (52.0–68.9) | 99.8% | 9,612,478 |
| At-risk couples (per 1,000 examined) | 8 | 4.5 (3.5–5.5) | 98.2% | — |
| Region | SCT per 1,000 | β-thal trait per 1,000 | All β-globin variants per 1,000 | At-risk couples per 1,000 |
|---|---|---|---|---|
| Eastern Province | 134.1 | 59.0 | 193 | 32.0 |
| Jazan (Southern) | 55.6 | 30.0 | 86 | 16.5 |
| Makkah (Western) | 28.5 | 14.4 | 43 | 8.2 |
| Madinah (Western) | 25.0 | 11.0 | 36 | 6.5 |
| Al-Baha (Southern) | 22.0 | 13.2 | 35 | 6.0 |
| Riyadh (Central) | 13.7 | 10.1 | 24 | 3.8 |
| Qassim (Central) | 14.5 | 8.5 | 23 | 3.4 |
| Tabuk (Northern) | 13.5 | 3.9 | 17 | 2.5 |
| Hail (Northern) | 13.5 | 3.9 | 17 | 2.2 |
| Northern Borders | 13.5 | 3.9 | 17 | 2.2 |
| Najran (Southern) | 20.0 | 8.0 | 28 | 4.5 |
| Aseer (Southern) | 18.0 | 7.0 | 25 | 4.0 |
| Al-Jouf (Northern) | 12.0 | 4.5 | 17 | 2.0 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




