2.2. Model Structure
The model aimed to estimate the health benefits of treatment with sparsentan compared to irbesartan, the active comparator in the PROTECT trial. It is a cohort-level state transition model which utilises clinically established definitions of stages of CKD in Kidney Disease: Improving Global Outcomes (KDIGO) guidelines [
22]. These stages are defined based on eGFR, a measure of how well a patient’s kidneys can filter waste, and are displayed in
Table 2.
Previous economic models in IgAN, and CKD more generally, have typically modelled treatment effects based on patient progression through CKD stages defined by eGFR alone, or based on proteinuria alone [
14,
15,
16,
17,
18,
19,
20,
21]. However, both eGFR and proteinuria are independent predictors of patient prognosis. While eGFR provides a direct quantification of a patient’s kidney function, proteinuria is highly predictive of the rate of decline of kidney function [
23]. Proteinuria also has the benefit of being more sensitive to changes in treatment and consequently is typically used as the primary endpoint of clinical trials in IgAN [
24].
Therefore, the developed model considers both CKD stage (defined by eGFR), but also a patient’s level of proteinuria. Data on CKD stage transitions from both the PROTECT study and the world’s largest registry of rare renal disease, UK RaDaR, was used to estimate transitions between CKD (as measured by eGFR) stages within UP/C states [
6,
11]. The model therefore considers patient outcomes, including quality of life, mortality, and healthcare resource utilisation (HCRU), conditional on both CKD stage and proteinuria level, rather than just the former. As shown in
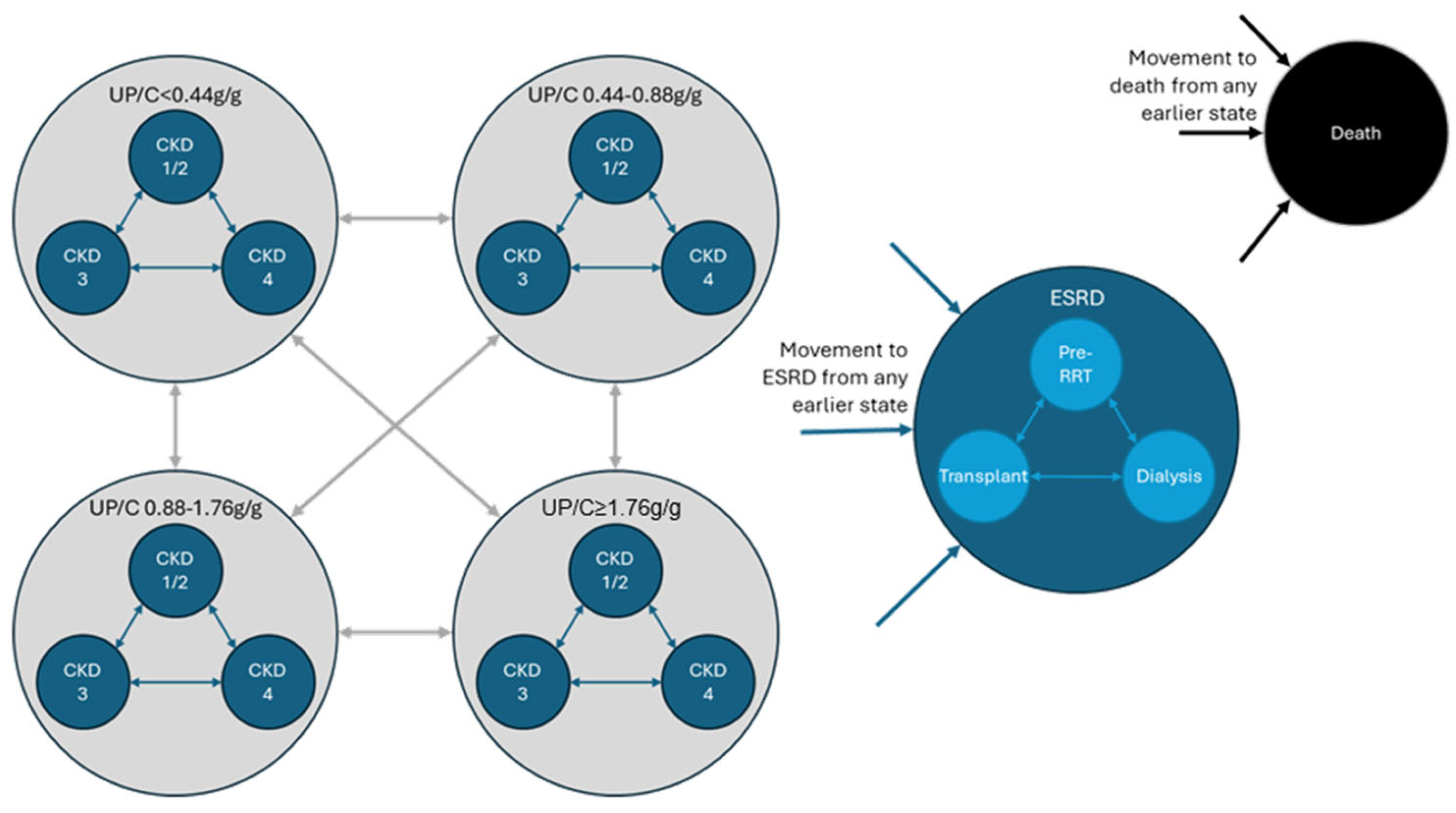
Figure 1, the model utilises 15 states (excluding death): three CKD states (CKD1/2, CKD3, and CKD4) for each of the four UP/C states (<0.44 g/g, 0.44-0.88 g/g, 0.88-1.76 g/g, and ≥1.76 g/g), equating to a total of 12 states, with an additional three health states within ESRD (CKD5) for pre-renal replacement therapy (RRT), dialysis, and transplant.
UP/C states are split into four bandings. Because these transitions are different for sparsentan and irbesartan in the PROTECT trial, they are the main driver of the differences in efficacy between the two drugs. Within the UP/C states, patients are assigned to sub-health states according to CKD stage. The first two stages of CKD, CKD1 and CKD2, are considered together because of their similar HCRU and QoL profiles. Patients in CKD5 are considered to have end-stage renal disease (ESRD) and are grouped together irrespective of their UP/C, with stratifications based on RRT modality.
Figure 1 is a schematic of this model structure.
Whilst not expected to be clinically plausible, backwards transitions and transitions that skipped stages (i.e., going from CKD1/2 to CKD4, or vice versa) were not strictly disallowed in the study’s model. This was to account for natural fluctuations in eGFR readings at the time of observation (for example a patient at the boundary of two CKD states transitioning back and forth between observations) to avoid additional manipulation or added constraints to the data. Abbreviations: CKD, chronic kidney disease; ESRD, end-stage renal disease; RRT, renal replacement therapy; UP/C, urine protein/creatinine ratio.
The model had a lifetime horizon to reflect the lifelong, incurable nature of IgAN. The cycle length was 12 weeks, reflecting the frequency of data collection in the PROTECT trial. The treatment effect of sparsentan was extrapolated beyond the duration of the trial.
2.3. Health State Transitions
Transitions between the health states defined in the model were based on three separate sources: PROTECT data for transitions between the UP/C health states and health states defined by CKD1 to CKD4 (within each UP/C state), RaDaR data for transitions from CKD 4 and to CKD5, and UK Renal Registry (UKRR) data for transitions within ESRD [
6,
11,
25]. Patients in RaDaR were matched in terms of clinical and demographic characteristics and inclusion criteria to the PROTECT clinical trial. This approach used weights obtained from a logistic regression model (for full details on this matching, see
Supplementary Materials).
The transition matrix for the first 12-week cycle is treatment-specific and is derived from a combination of the first 12 weeks of PROTECT (for treatment-specific UP/C transitions treatment-agnostic early CKD stage transitions) and the RaDaR dataset (for treatment-agnostic late CKD stage transitions) [
6,
11]. This is because the change in UP/C observed in the first 12 weeks of PROTECT was substantial: if it were used and extrapolated over the entire stage of the model, it would result in a significant overestimation of the sparsentan treatment effect over the lifetime model horizon. From Week 12 to Week 108 (the end of the PROTECT trial period), another treatment-specific transition matrix is derived from PROTECT and RaDaR data. A uniform transition matrix is employed from this point forward, made from extrapolated UP/C and CKD stage transitions based on the results of PROTECT and RaDaR.
Transitions between UP/C health states were based on patient-level data from PROTECT. Patients were sorted into four groups based on their UP/C at baseline, with movements between these groups at 12-week observation intervals recorded in both treatment arms and entered into the model. From Week 106 onwards, transitions were based on the uniform transition matrix described above.
In the model, patients who progress to ESRD are grouped together regardless of their UP/C, and distributed across three states: those who are not receiving RRT, those receiving dialysis, and those who have received a kidney transplant. A constant transition matrix was applied for these three states, with transition probabilities taken from the NICE technology appraisal guidance for targeted-related budesonide in treating IgAN (NICE TA775) [
14,
26,
27].
2.4. Adverse Events, Treatment Discontinuations, and Mortality
Data on adverse events and treatment discontinuations in the model was sourced from the PROTECT trial. All treatment-emergent adverse events (TEAE) which occurred in ≥5% of patients in either treatment arm were modelled using one-off costs and one-off utility decrements in the first cycle, sourced from Sullivan et al. 2011 [
28]. The disutilities applied for adverse events are outlined in
Table 3. All disutilities were set to last for 7 days.
Discontinuation of sparsentan was applied as a constant per-cycle rate based on rates of treatment discontinuation in PROTECT, resulting in an annual discontinuation probability of 7.08% and a per-cycle discontinuation probability of 1.68%. Additional discontinuations were included by assuming treatment would be stopped if sparsentan did not result in sufficient treatment effect.
Mortality rates could not be modelled using PROTECT data because the rate of mortality in the trial was low. A systematic literature review found no published data on IgAN-specific mortality rates [
13]. Therefore, data from the 2024 KDIGO draft guidelines were used to apply mortality hazard ratios (HRs) for CKD1/2, CKD3, and CKD4 patients, as well as ESRD patients who had not undergone RRT, versus all-cause mortality in the population of England adjusted for age and sex [
29]. For ESRD patients who were undergoing dialysis or who had received a kidney transplant, HRs calculated by Neovius et al. 2014 were multiplied by the CKD4 HR [
30]. These HRs, as applied in the model, are shown in
Table 4.