Submitted:
24 April 2026
Posted:
27 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
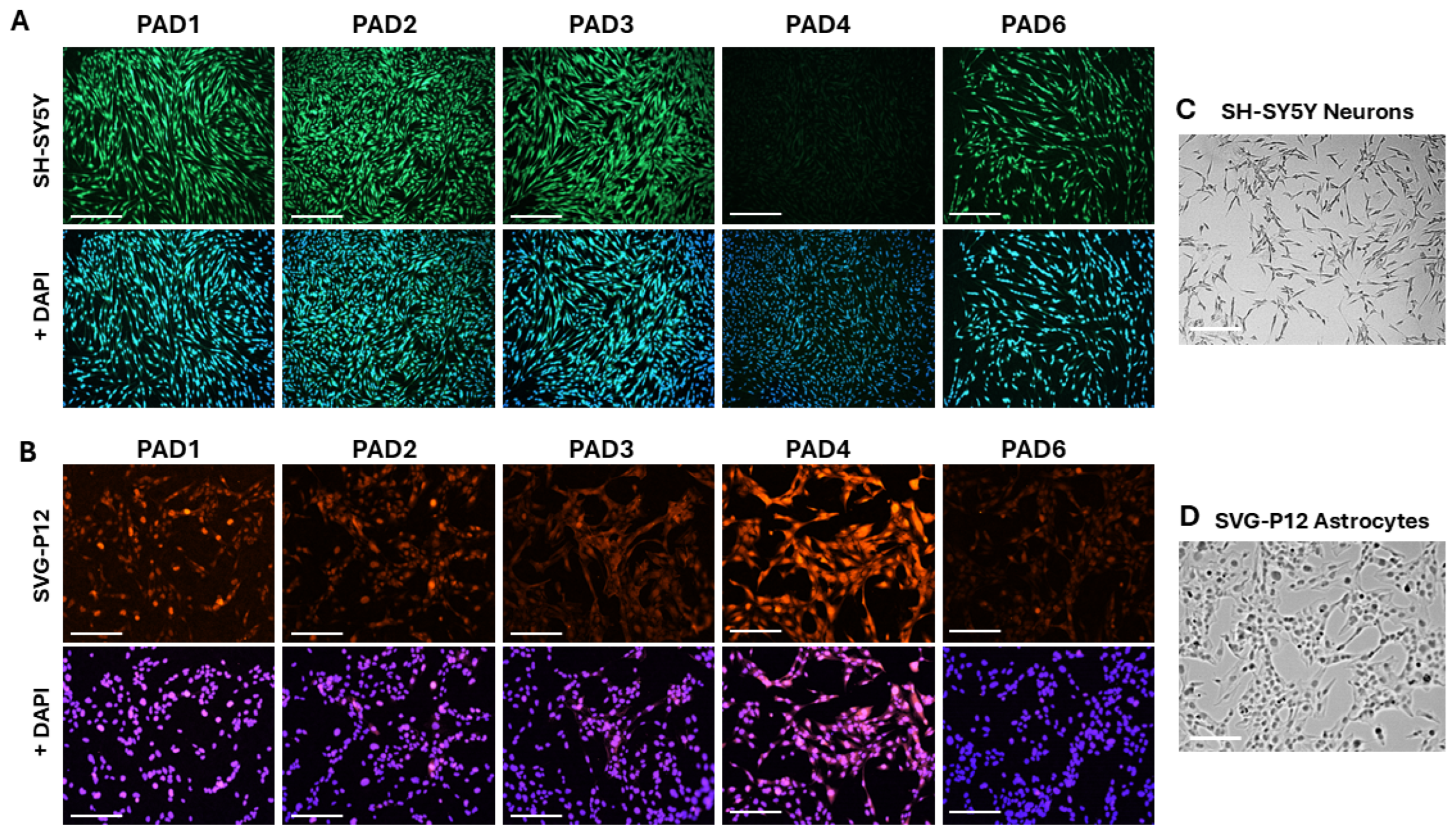
2.1. PAD Isozyme Detection Differs in Neuronal and Astrocytic Cells
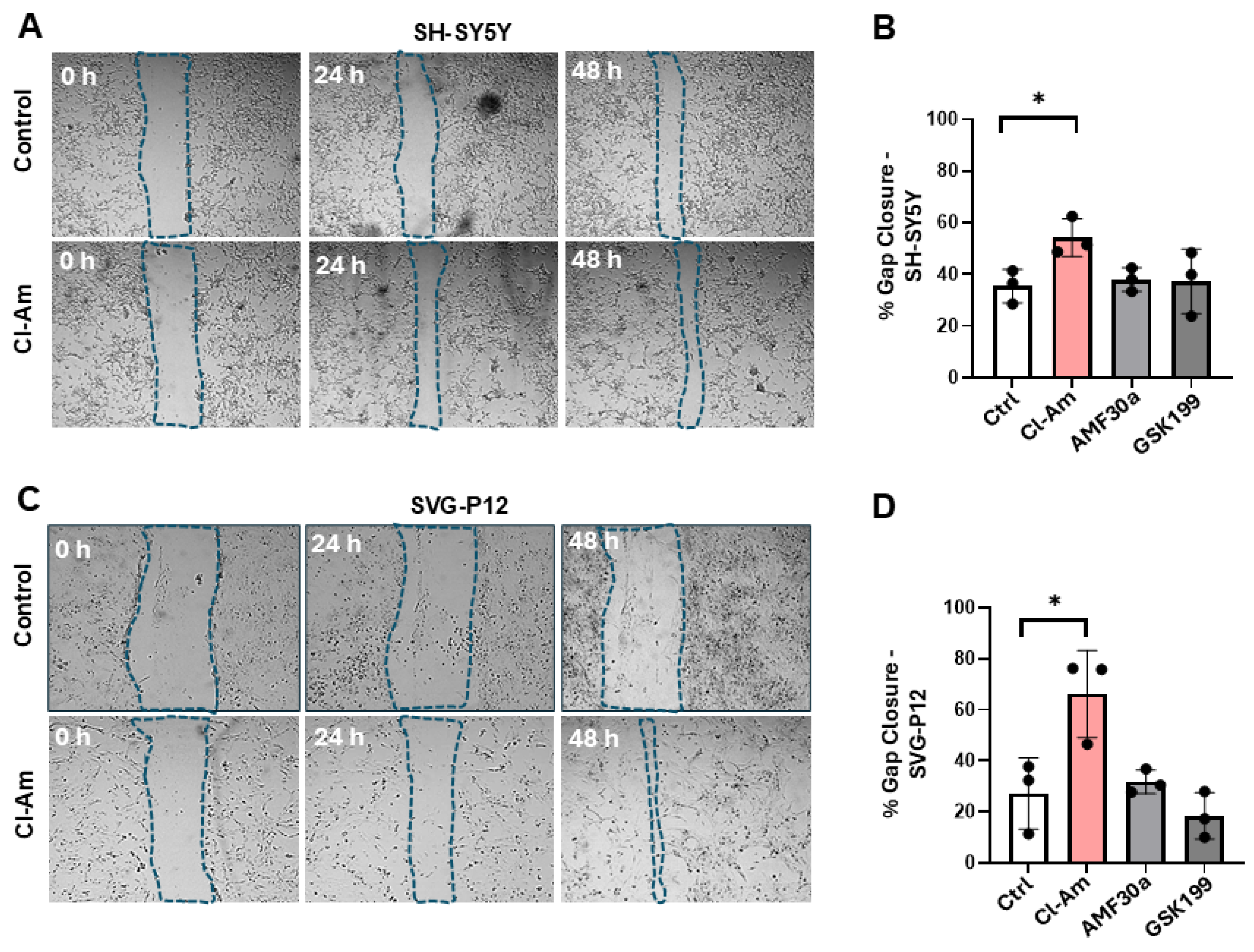
2.2. Pharmacological PAD-Inhibition Affects Scratch-Injury Closure in Neuronal and Astrocytic Cells Under Normoxic Conditions
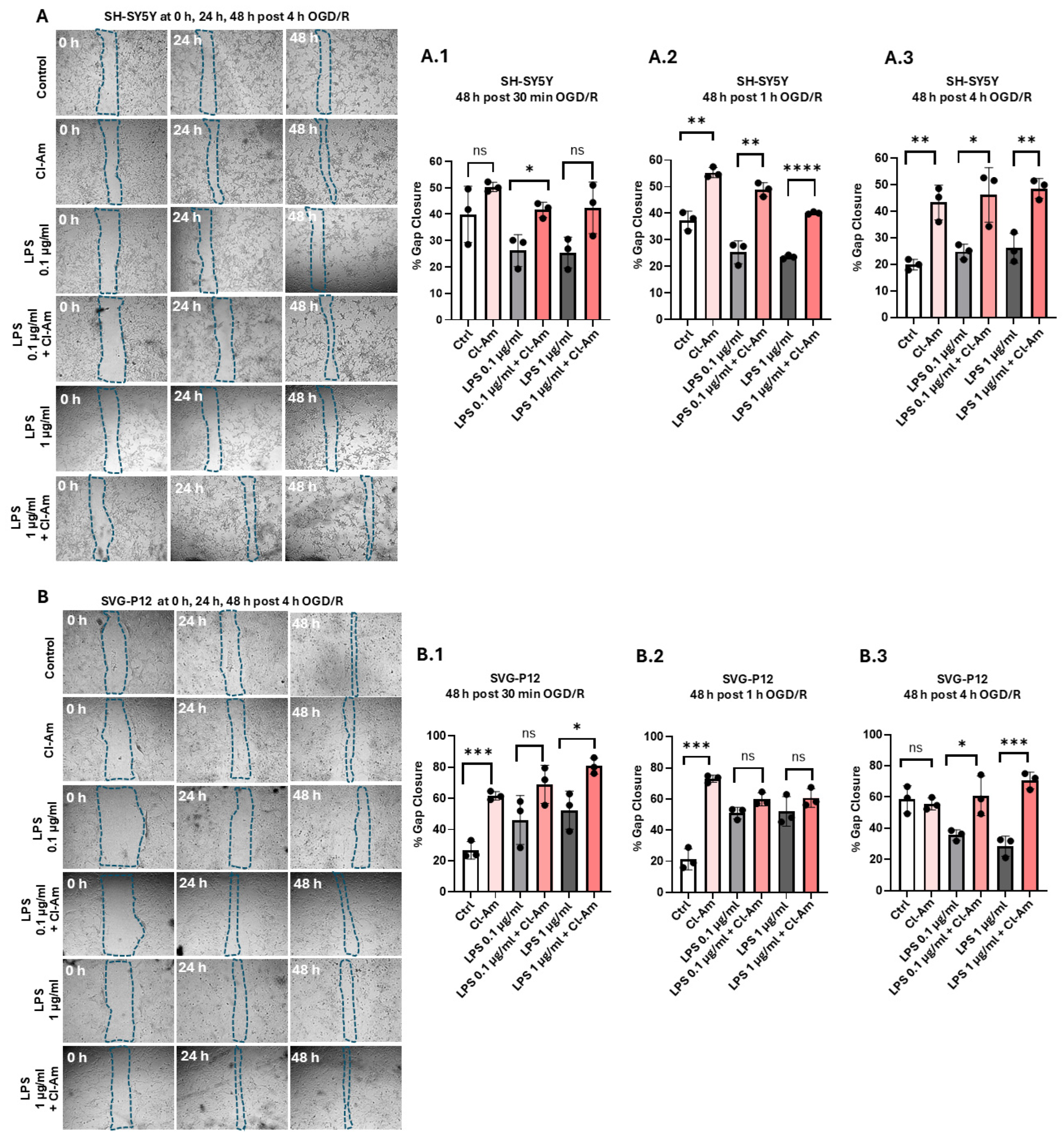
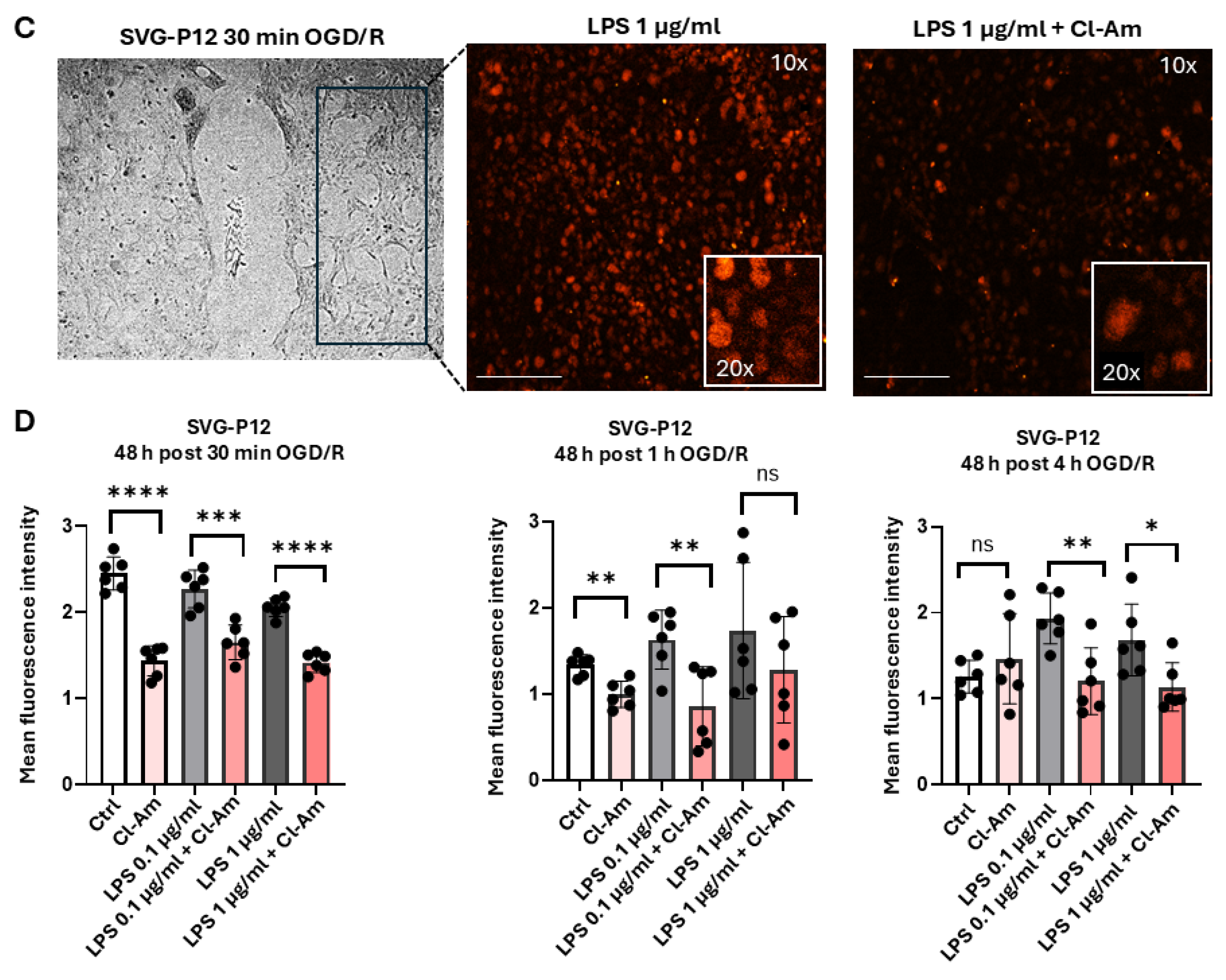
2.3. Effects of Pan-PAD-Inhibitor on Scratch-Injury of Neuronal Cells and Astrocytes Following OGD/R and LPS Stimulation
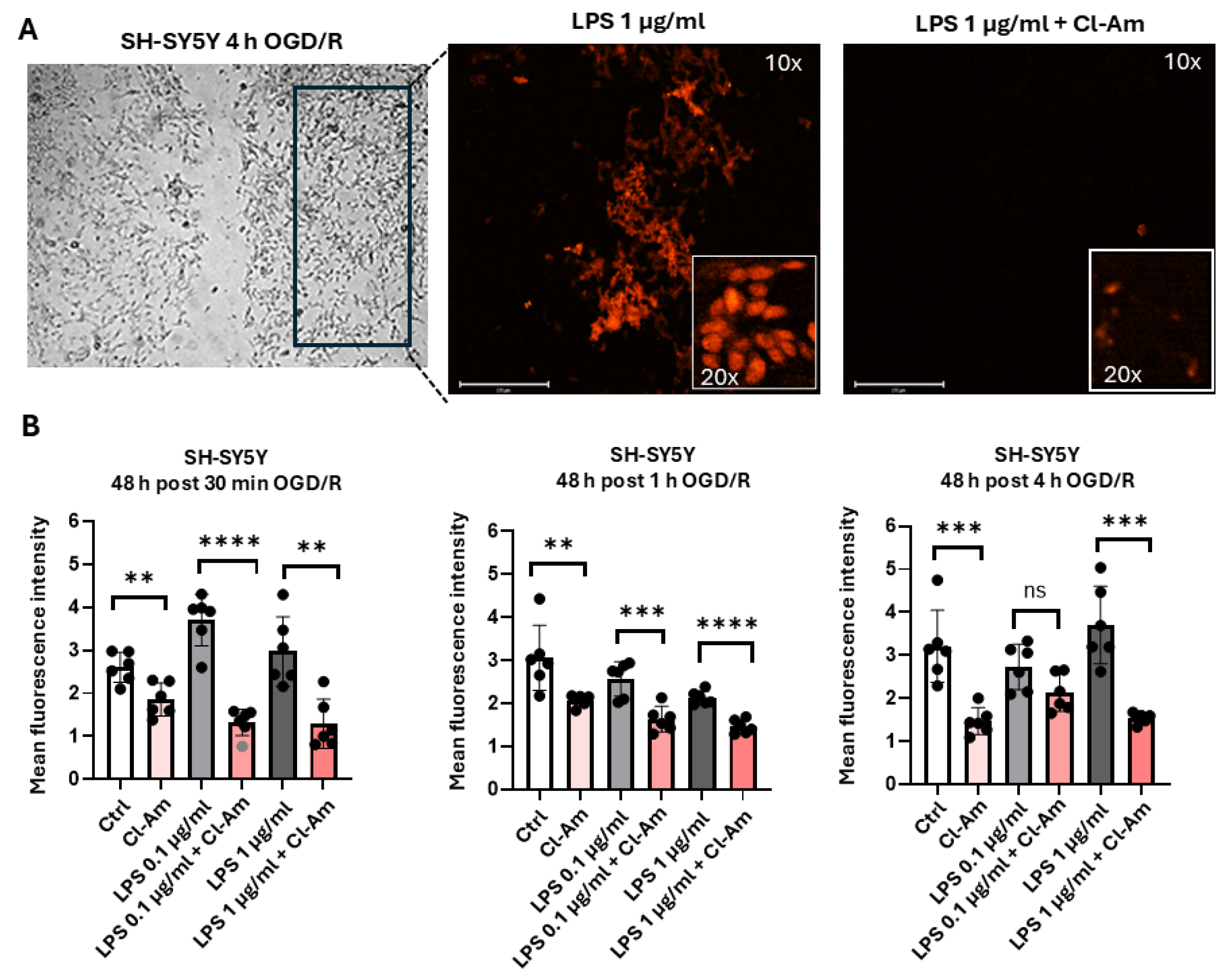
2.4. Pan-PAD Inhibitor Cl-Amidine Reduces Histone H3 Citrullination (CitH3) in Neuronal and Astrocyte Cells in OGD/R
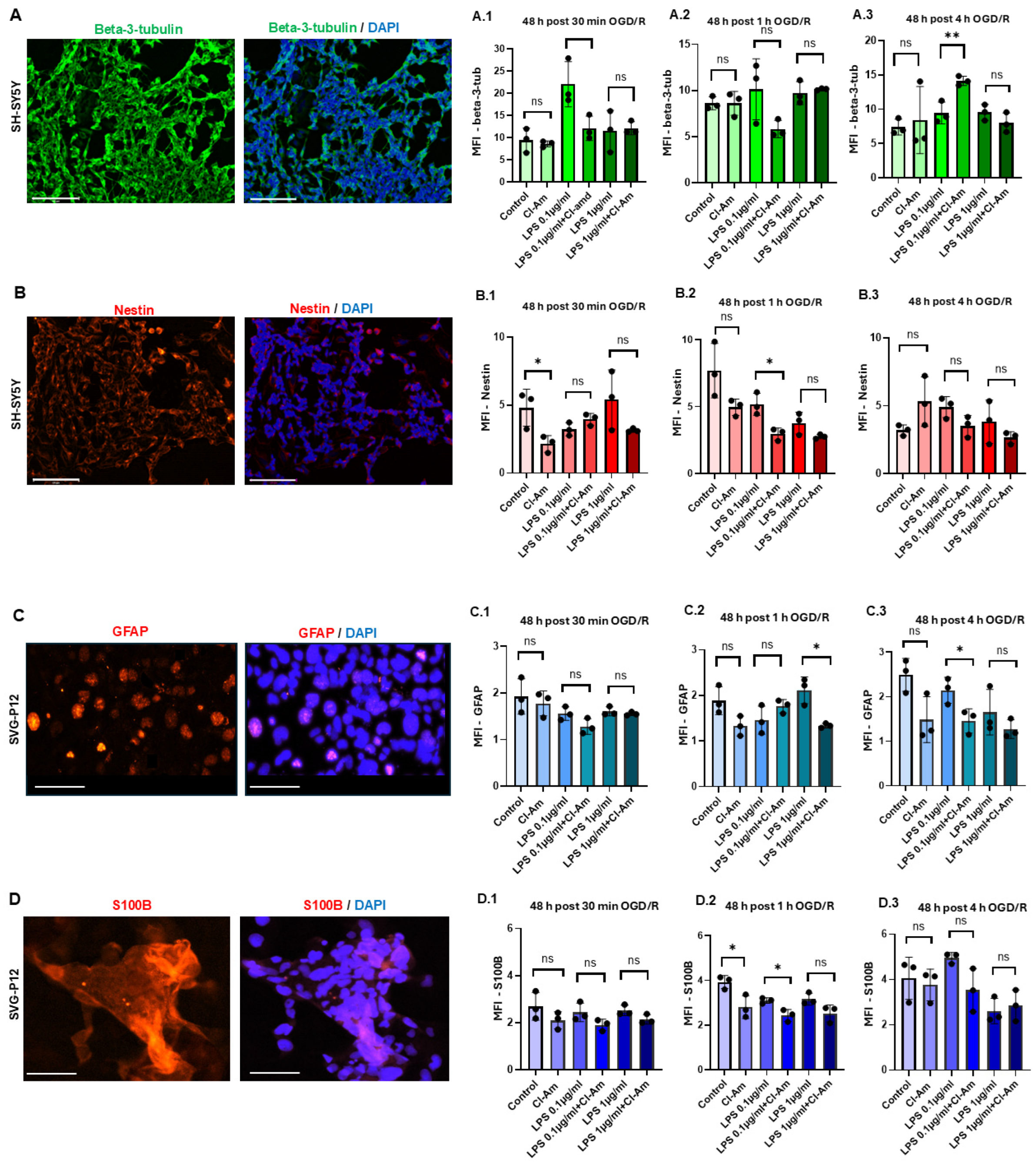
2.5. Pan-PAD Inhibitior Modifies Neuronal (β-3-Tubulin), Stemness (Nestin) and Astrocytic (GFAP and S100B) Markers in OGD/R
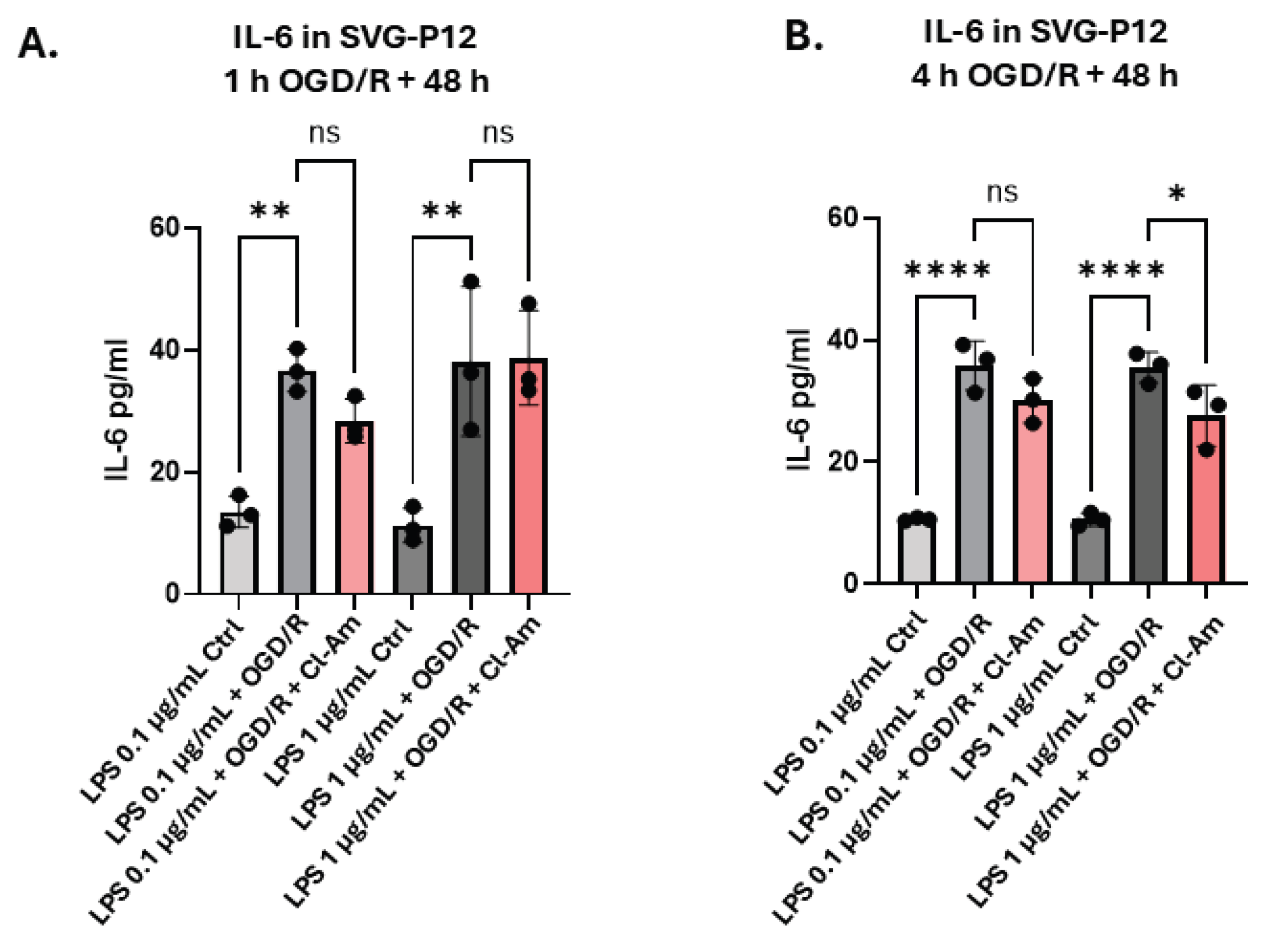
2.6. Effects of Pan-PAD Inhibitor Cl-Amidine on Pro-Inflammatory Cytokines IL-1β and IL-6 in Neurons and Astrocytes
3. Discussion
4. Materials and Methods
4.1. Cell Culture and Neuronal Cell Differentiation
4.2. Scratch (Wound Healing) Assay
4.3. OGD/R in Conjunction with Scratch Injury
4.4. Immunocytochemistry on Differentiated SH-SY5Y and SVG-P12 Cells
4.5. Assessment of PAD Inhibition on Inflammatory Cytokines IL-1β and IL-6 by ELISA
4.6. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Pilipović, K.; Harej Hrkać, A.; Kučić, N.; Mršić-Pelčić, J. Modeling Central Nervous System Injury In Vitro: Current Status and Promising Future Strategies. Biomedicines 2022, 11, 94. [Google Scholar] [CrossRef] [PubMed]
- Acun, C.; Lavu, R.; Liu, W.; Nicoletti, N.; Ramsey, J.; Aly, H. Therapeutic hypothermia in mild hypoxic ischemic encephalopathy: A clinical dilemma with uncertain long-term outcomes. Early Hum. Dev. 2026, 212, 106427. [Google Scholar] [CrossRef] [PubMed]
- Omelchenko, A.; Singh, N.K.; Firestein, B.L. Current Advances in In Vitro Models of Central Nervous System Trauma. Curr. Opin. Biomed. Eng. 2020, 14, 34–41. [Google Scholar] [CrossRef]
- Lange, S.; Gögel, S.; Leung, K.Y.; Vernay, B.; Nicholas, A.P.; Causey, C.P.; Thompson, P.R.; Greene, N.D.E.; Ferretti, P. Protein Deiminases: New Players in the Developmentally Regulated Loss of Neural Regenerative Ability. Dev. Biol. 2011, 355, 205–214. [Google Scholar] [CrossRef] [PubMed]
- Lange, S.; Rocha-Ferreira, E.; Thei, L.; Mawjee, P.; Bennett, K.; Thompson, P.R.; Subramanian, V.; Nicholas, A.P.; Peebles, D.; Hristova, M.; Raivich, G. Peptidylarginine Deiminases: Novel Drug Targets for Prevention of Neuronal Damage Following Hypoxic Ischemic Insult (HI) in Neonates. J. Neurochem. 2014, 130(4), 555–562. [Google Scholar] [CrossRef]
- Lange, S.; Gallagher, M.; Kholia, S.; Kosgodage, U.S.; Hristova, M.; Hardy, J.; Inal, J.M. Peptidylarginine Deiminases—Roles in Cancer and Neurodegeneration and Possible Avenues for Therapeutic Intervention via Modulation of Exosome and Microvesicle (EMV) Release? Int. J. Mol. Sci. 2017, 18, 1196. [Google Scholar] [CrossRef] [PubMed]
- Lange, S. Peptidylarginine Deiminases as Drug Targets in Neonatal Hypoxic-Ischemic Encephalopathy. Front. Neurol. 2016, 7, 22. [Google Scholar] [CrossRef]
- Lange, S. Peptidylarginine Deiminases and Extracellular Vesicles: Prospective Drug Targets and Biomarkers in Central Nervous System Diseases and Repair. Neural Regen. Res. 2021, 16, 934–938. [Google Scholar] [CrossRef]
- Stadler, S.C.; Vincent, C.T.; Fedorov, V.D.; Patsialou, A.; Cherrington, B.D.; Wakshlag, J.J.; Mohanan, S.; Zee, B.M.; Zhang, X.; Garcia, B.A.; et al. Dysregulation of PAD4-Mediated Citrullination of Nuclear GSK3β Activates TGF-β Signaling and Induces Epithelial-to-Mesenchymal Transition in Breast Cancer Cells. Proc. Natl. Acad. Sci. USA 2013, 110, 11851–11856. [Google Scholar] [CrossRef]
- Lazarus, R.C.; Buonora, J.E.; Jacobowitz, D.M.; Mueller, G.P. Protein Citrullination: A Proposed Mechanism for Pathology in Traumatic Brain Injury. Front. Neurol. 2015, 6, 204. [Google Scholar] [CrossRef]
- Attilio, P.J.; Flora, M.; Kamnaksh, A.; Bradshaw, D.J.; Agoston, D.V.; Mueller, G.P. The Effects of Blast Exposure on Protein Deimination in the Brain. Oxid. Med. Cell. Longev. 2017, 2017, 8398072. [Google Scholar] [CrossRef]
- Beato, M.; Sharma, P. Peptidyl Arginine Deiminase 2 (PADI2)-Mediated Arginine Citrullination Modulates Transcription in Cancer. Int. J. Mol. Sci. 2020, 21, 1351. [Google Scholar] [CrossRef]
- Boon, L.; Ugarte-Berzal, E.; Martens, E.; Fiten, P.; Vandooren, J.; Janssens, R.; Blanter, M.; Yu, K.; Boon, M.; Struyf, S.; Proost, P.; Opdenakker, G. Citrullination as a Novel Posttranslational Modification of Matrix Metalloproteinases. Matrix Biol. 2021, 95, 68–83. [Google Scholar] [CrossRef]
- Sancandi, M.; Uysal-Onganer, P.; Kraev, I.; Mercer, A.; Lange, S. Protein Deimination Signatures in Plasma and Plasma-EVs and Protein Deimination in the Brain Vasculature in a Rat Model of Pre-Motor Parkinson’s Disease. Int. J. Mol. Sci. 2020, 21, 2743. [Google Scholar] [CrossRef]
- Mercer, A.; Jaunmuktane, Z.; Hristova, M.; Lange, S. Differential, Stage Dependent Detection of Peptidylarginine Deiminases and Protein Deimination in Lewy Body Diseases—Findings from a Pilot Study. Int. J. Mol. Sci. 2022, 23, 13117. [Google Scholar] [CrossRef]
- Mercer, A.; Sancandi, M.; MacLatchy, A.; Lange, S. Brain-Region-Specific Differences in Protein Citrullination/Deimination in a Pre-Motor Parkinson’s Disease Rat Model. Int. J. Mol. Sci. 2024, 25, 11168. [Google Scholar] [CrossRef] [PubMed]
- Kosgodage, U.S.; Uysal-Onganer, P.; MacLatchy, A.; Kraev, I.; Chatterton, N.P.; Nicholas, A.P.; Inal, J.M.; Lange, S. Peptidylarginine Deiminases Post-Translationally Deiminate Prohibitin and Modulate Extracellular Vesicle Release and MicroRNAs in Glioblastoma Multiforme. Int. J. Mol. Sci. 2019, 20, 103. [Google Scholar] [CrossRef]
- Uysal-Onganer, P.; MacLatchy, A.; Mahmoud, R.; Kraev, I.; Thompson, P.R.; Inal, J.M.; Lange, S. Peptidylarginine Deiminase Isozyme-Specific PAD2, PAD3 and PAD4 Inhibitors Differentially Modulate Extracellular Vesicle Signatures and Cell Invasion in Two Glioblastoma Multiforme Cell Lines. Int. J. Mol. Sci. 2020, 21, 1495. [Google Scholar] [CrossRef] [PubMed]
- Yusuf, I.O.; Parsi, S.; Ostrow, L.W.; Brown, R.H.; Thompson, P.R.; Xu, Z. PAD2 Dysregulation and Aberrant Protein Citrullination Feature Prominently in Reactive Astrogliosis and Myelin Protein Aggregation in Sporadic ALS. Neurobiol. Dis. 2024, 192, 106414. [Google Scholar] [CrossRef]
- Liang, H.; Hunt, J.B., Jr.; Ma, C.; Kovalenko, A.; Calahatian, J.; Pedersen, C.; Liu, H.; Li, J.; Serrano, M.; Blazier, D.; Watler, M.; Rocha-Rangel, P.; Saunders, C.; Blair, L.J.; Breydo, L.; Nash, K.; Quadri, Z.; Kraemer, B.; Nelson, P.; Norris, C.; Abner, E.L.; Uversky, V.N.; Chaput, D.; Selenica, M.B.; Lee, D.C. Probing Tau Citrullination in Alzheimer’s Disease Brains and Mouse Models of Tauopathy. Acta Neuropathol. 2025, 150, 61. [Google Scholar] [CrossRef]
- Bashir, F.; Awais, H.; Waseem, A.; Shahzad, A.; Babar Khan, A.; Ali, S.A.; Shafiq, L.; Ahmed Bhatti, M. Structural and Mechanistic Insights into Peptidylarginine Deiminase (PAD2/PAD4)-Mediated Citrullination and Therapeutic Targeting: A Review. Int. J. Biol. Macromol. 2025, 332, 148586. [Google Scholar] [CrossRef]
- Dakin, L.A.; Xing, L.; Hall, J.; Ding, W.; Vajdos, F.F.; Pelker, J.W.; Ramsey, S.; Balbo, P.; Sahasrabudhe, P.V.; Banker, M.E.; Choi, W.Y.; Wright, S.W.; Chang, J.S.; Curto, J.M.; Davoren, J.E.; Drozda, S.E.; Fennell, K.F.; Futatsugi, K.; Kortum, S.; Lee, K.L.; Liu, S.; Lovering, F.; Nicki, J.A.; Trujillo, J.I.; Vincent, F.; Schnute, M.E. Inhibiting Peptidylarginine Deiminases (PAD1-4) by Targeting a Ca2+-Dependent Allosteric Binding Site. Nat. Commun. 2025, 16, 4579. [Google Scholar] [CrossRef]
- Kijak-Boćkowska, M.; Czerwińska, J.; Owczarczyk-Saczonek, A. Peptidylarginine Deiminases: An Overview of Recent Advances in Citrullination Research. Int. J. Mol. Sci. 2025, 26(24), 12060. [Google Scholar] [CrossRef]
- Arun, P.; Abu-Taleb, R.; Valiyaveettil, M.; Wang, Y.; Long, J.B.; Nambiar, M.P. Studies on Blast Traumatic Brain Injury Using In Vitro Model with Shock Tube. NeuroReport 2011, 22, 379–384. [Google Scholar] [CrossRef]
- Hatic, H.; Kane, M.J.; Saykally, J.N.; Citron, B.A. Modulation of Transcription Factor Nrf2 in an In Vitro Model of Traumatic Brain Injury. J. Neurotrauma 2012, 29, 1188–1196. [Google Scholar] [CrossRef]
- Bae, Y.-H.; Joo, H.; Bae, J.; Kim, Y.; Lee, H.; Kim, H.; Kim, D.; Kim, K.-S.; Lee, J.; Kim, S. Brain Injury Induces HIF-1α-Dependent Transcriptional Activation of LRRK2 That Exacerbates Brain Damage. Cell. Death Dis. 2018, 9, 1125. [Google Scholar] [CrossRef]
- Chen, W.C.; Chang, L.H.; Huang, S.S.; Huang, Y.J.; Chih, C.L.; Kuo, H.C.; Lee, Y.H.; Lee, I.H. Aryl Hydrocarbon Receptor Modulates Stroke-Induced Astrogliosis and Neurogenesis in the Adult Mouse Brain. J. Neuroinflammation 2019, 16(1), 187. [Google Scholar] [CrossRef]
- Jing, Y.; Yang, D.; Fu, Y.; Wang, W.; Yang, G.; Yuan, F.; Chen, H.; Ding, J.; Chen, S.; Tian, H. Neuroprotective Effects of Serpina3k in Traumatic Brain Injury. Front. Neurol. 2019, 10, 1215. [Google Scholar] [CrossRef]
- Meyer, L.J.; Lotze, F.P.; Riess, M.L. Simulated Traumatic Brain Injury in In Vitro Mouse Neuronal and Brain Endothelial Cell Culture Models. J. Pharmacol. Toxicol. Methods 2022, 114, 107159. [Google Scholar] [CrossRef]
- Gallart-Palau, X.; Lee, B.S.; Adav, S.S.; Qian, J.; Serra, A.; Park, J.E.; Lai, M.K.; Chen, C.P.; Kalaria, R.N.; Sze, S.K. Gender Differences in White Matter Pathology and Mitochondrial Dysfunction in Alzheimer’s Disease with Cerebrovascular Disease. Mol. Brain 2016, 9, 27. [Google Scholar] [CrossRef]
- Shi, G.; Liu, L.; Cao, Y.; Ma, G.; Zhu, Y.; Xu, J.; Zhang, X.; Li, T.; Mi, L.; Jia, H.; Zhang, Y.; Liu, X.; Zhou, Y.; Li, S.; Yang, G.; Liu, X.; Chen, F.; Wang, B.; Deng, Q.; Zhang, S.; Zhang, J. Inhibition of Neutrophil Extracellular Trap Formation Ameliorates Neuroinflammation and Neuronal Apoptosis via STING-Dependent IRE1α/ASK1/JNK Signaling Pathway in Mice with Traumatic Brain Injury. J. Neuroinflammation 2023, 20, 222. [Google Scholar] [CrossRef]
- Wu, Y.H.; Rosset, S.; Lee, T.R.; Dragunow, M.; Park, T.; Shim, V. In Vitro Models of Traumatic Brain Injury: A Systematic Review. J. Neurotrauma 2021, 38, 2336–2372. [Google Scholar] [CrossRef]
- Chen, K.Z.; Liu, S.X.; Li, Y.W.; He, T.; Zhao, J.; Wang, T.; Qiu, X.X.; Wu, H.F. Vimentin as a Potential Target for Diverse Nervous System Diseases. Neural Regen. Res. 2023, 18(5), 969–975. [Google Scholar] [CrossRef]
- Liu, Q.; Jin, Z.; Xu, Z.; Yang, H.; Li, L.; Li, G.; et al. Antioxidant Effects of Ginkgolides and Bilobalide against Cerebral Ischemia Injury by Activating the Akt/Nrf2 Pathway In Vitro and In Vivo. Cell. Stress Chaperones 2019, 24, 441–452. [Google Scholar] [CrossRef]
- Skrzypczak-Wiercioch, A.; Sałat, K. Lipopolysaccharide-Induced Model of Neuroinflammation: Mechanisms of Action, Research Application and Future Directions for Its Use. Molecules 2022, 27, 5481. [Google Scholar] [CrossRef]
- Luo, Y.; Arita, K.; Bhatia, M.; Knuckley, B.; Lee, Y.H.; Stallcup, M.R.; Sato, M.; Thompson, P.R. Inhibitors and Inactivators of Protein Arginine Deiminase 4: Functional and Structural Characterization. Biochemistry 2006, 45, 11727–11736. [Google Scholar] [CrossRef]
- Wang, Y.; Lyu, Y.; Tu, K.; Xu, Q.; Yang, Y.; Salman, S.; Le, N.; Lu, H.; Chen, C.; Zhu, Y.; Wang, R.; Liu, Q.; Semenza, G.L. Histone Citrullination by PADI4 Is Required for HIF-Dependent Transcriptional Responses to Hypoxia and Tumor Vascularization. Sci. Adv. 2021, 7, eabe3771. [Google Scholar] [CrossRef]
- Ahmed, D.; Puthussery, H.; Basnett, P.; Knowles, J.C.; Lange, S.; Roy, I. Controlled Delivery of Pan-PAD-Inhibitor Cl-Amidine Using Poly(3-Hydroxybutyrate) Microspheres. Int. J. Mol. Sci. 2021, 22, 12852. [Google Scholar] [CrossRef]
- Muth, A.; Subramanian, V.; Beaumont, E.; Nagar, M.; Kerry, P.; McEwan, P.; Srinath, H.; Clancy, K.; Parelkar, S.; Thompson, P.R. Development of a Selective Inhibitor of Protein Arginine Deiminase 2. J. Med. Chem. 2017, 60, 3198–3211. [Google Scholar] [CrossRef]
- Willis, V.C.; Banda, N.K.; Cordova, K.N.; Chandra, P.E.; Robinson, W.H.; Cooper, D.C.; Lugo, D.; Mehta, G.; Taylor, S.; Tak, P.P.; Lewis, H.D.; Holers, V.M. Protein Arginine Deiminase 4 Inhibition Is Sufficient for the Amelioration of Collagen-Induced Arthritis. Clin. Exp. Immunol. 2017, 188, 263–274. [Google Scholar] [CrossRef]
- Byun, D.J.; Lee, J.; Yu, J.W.; Hyun, Y.M. NLRP3 Exacerbates NETosis-Associated Neuroinflammation in an LPS-Induced Inflamed Brain. Immune Netw. 2023, 23(3), e27. [Google Scholar] [CrossRef]
- Frizzo, J.K.; Tramontina, F.; Bortoli, E.; Gottfried, C.; Leal, R.B.; Lengyel, I.; Donato, R.; Dunkley, P.R.; Gonçalves, C.A. S100B-Mediated Inhibition of the Phosphorylation of GFAP Is Prevented by TRTK-12. Neurochem. Res. 2004, 29, 735–740. [Google Scholar] [CrossRef]
- Guerra, M.C.; Tortorelli, L.S.; Galland, F.; Da Ré, C.; Negri, E.; Engelke, D.S.; Rodrigues, L.; Leite, M.C.; Gonçalves, C.A. Lipopolysaccharide Modulates Astrocytic S100B Secretion: A Study in Cerebrospinal Fluid and Astrocyte Cultures from Rats. J. Neuroinflammation 2011, 8, 128. [Google Scholar] [CrossRef]
- Yang, Z.; Wang, K.K. Glial Fibrillary Acidic Protein: From Intermediate Filament Assembly and Gliosis to Neurobiomarker. Trends Neurosci. 2015, 38, 364–374. [Google Scholar] [CrossRef] [PubMed]
- Janigro, D.; Mondello, S.; Posti, J.P.; Unden, J. GFAP and S100B: What You Always Wanted to Know and Never Dared to Ask. Front. Neurol. 13 2022, 835597. [Google Scholar] [CrossRef] [PubMed]
- Richardson, L.S.; Emezienna, N.; Burd, I.; Taylor, B.D.; Peltier, M.R.; Han, A.; Menon, R. Adapting an Organ-on-Chip Device to Study the Effect of Fetal Sex and Maternal Race/Ethnicity on Preterm Birth Related Intraamniotic Inflammation Leading to Fetal Neuroinflammation. Am. J. Reprod. Immunol. 2022, 88, e13638. [Google Scholar] [CrossRef] [PubMed]
- Cuende, J.; Moreno, S.; Bolaños, J.P.; Almeida, A. Retinoic Acid Downregulates Rae1 Leading to APC(Cdh1) Activation and Neuroblastoma SH-SY5Y Differentiation. Oncogene 2008, 27, 3339–3344. [Google Scholar] [CrossRef]
- Cheung, Y.T.; Lau, W.K.W.; Yu, M.S.; Lai, C.S.W.; Yeung, S.C.; So, K.F.; Chang, R.C.C. Effects of All-Trans-Retinoic Acid on Human SH-SY5Y Neuroblastoma as In Vitro Model in Neurotoxicity Research. Neurotoxicology 2009, 30, 127–135. [Google Scholar] [CrossRef]
- Xie, H.; Hu, L.; Li, G. SH-SY5Y Human Neuroblastoma Cell Line: In Vitro Cell Model of Dopaminergic Neurons in Parkinson’s Disease. Chin. Med. J. 2010, 123, 1086–1092. [Google Scholar]
- Skotak, M.; Wang, F.; Chandra, N. An In Vitro Injury Model for SH-SY5Y Neuroblastoma Cells: Effect of Strain and Strain Rate. J. Neurosci. Methods 2012, 205, 159–168. [Google Scholar] [CrossRef]
- Filograna, R.; Civiero, L.; Ferrari, V.; Codolo, G.; Greggio, E.; Bubacco, L.; Beltramini, M.; Bisaglia, M. Analysis of the Catecholaminergic Phenotype in Human SH-SY5Y and BE(2)-M17 Neuroblastoma Cell Lines upon Differentiation. PLoS ONE 2015, 10, e0136769. [Google Scholar] [CrossRef] [PubMed]
- Elnagar, M.R.; Walls, A.B.; Helal, G.K.; Hamada, F.M.; Thomsen, M.S.; Jensen, A.A. Functional Characterization of α7 Nicotinic Acetylcholine and NMDA Receptor Signaling in SH-SY5Y Neuroblastoma Cells in an ERK Phosphorylation Assay. Eur. J. Pharmacol. 2018, 826, 106–113. [Google Scholar] [CrossRef]
- Juntunen, M.; Hagman, S.; Moisan, A.; Narkilahti, S.; Miettinen, S. In Vitro Oxygen-Glucose Deprivation-Induced Stroke Models with Human Neuroblastoma Cell- and Induced Pluripotent Stem Cell-Derived Neurons. Stem Cells Int. 2020, 2020, 8841026. [Google Scholar] [CrossRef] [PubMed]
- Wan, F.; Jin, L.; Qin, Y.; Zeng, Y. Modulation of Muscarinic Receptors by Anisodine Hydrobromide in Cerebral Ischemia. Cell. Mol. Biol. 2023, 69, 17–24. [Google Scholar] [CrossRef]
- Thakor, F.K.; Wan, K.W.; Welsby, P.J.; Welsby, G. Pharmacological Effects of Asiatic Acid in Glioblastoma Cells under Hypoxia. Mol. Cell. Biochem. 2017, 430, 179–190. [Google Scholar] [CrossRef]
- Demircan, T.; Yavuz, M.; Kaya, E.; Akgül, S.; Altuntaş, E. Cellular and Molecular Comparison of Glioblastoma Multiform Cell Lines. Cureus 2021, 13, e16043. [Google Scholar] [CrossRef]
- Cao, Y.; Shi, M.; Liu, L.; Zuo, Y.; Jia, H.; Min, X.; Liu, X.; Chen, Z.; Zhou, Y.; Li, S.; Yang, G.; Liu, X.; Deng, Q.; Chen, F.; Chen, X.; Zhang, S.; Zhang, J. Inhibition of Neutrophil Extracellular Trap Formation Attenuates NLRP1-Dependent Neuronal Pyroptosis via STING/IRE1α Pathway after Traumatic Brain Injury in Mice. Front. Immunol. 2023, 14, 1125759. [Google Scholar] [CrossRef]
- Zhang, T.; Liu, P.; Yao, G.; Zhang, X.; Cao, C. A complex heterozygous mutation in PADI6 causes early embryo arrest: A case report. Front Genet. 2023, 13, 1104085. [Google Scholar] [CrossRef]
- Williams, J.P.C.; Mouilleron, S.; Trapero, R.H.; Bertran, M.T.; Marsh, J.A.; Walport, L.J. Structural Insight into the Function of Human Peptidyl Arginine Deiminase Comput. Struct. Biotechnol. J. 2024, 23, 3258–3269. [Google Scholar] [CrossRef]
- Zhu, Y.; Xu, J.; Chai, Y.; Li, P.; Liu, L.; Zhang, S.; Zhang, J.; Chen, X. Neutrophil Extracellular Traps Aggravate Blood-Brain Barrier Disruption via ZBP1/FSP1-Mediated Ferroptosis after Traumatic Brain Injury. Fluids Barriers CNS 2025, 23, 2. [Google Scholar] [CrossRef]
- Lai, N.S.; Yu, H.C.; Tung, C.H.; Huang, K.Y.; Huang, H.B.; Lu, M.C. Increased Peptidylarginine Deiminases Expression during the Macrophage Differentiation and Participated Inflammatory Responses. Arthritis Res. Ther. 2019, 21, 108. [Google Scholar] [CrossRef]
- Chen, Y.; Zhang, H.; Hu, X.; Cai, W.; Ni, W.; Zhou, K. Role of NETosis in Central Nervous System Injury. Oxid. Med. Cell. Longev. 2022, 2022, 3235524. [Google Scholar] [CrossRef]
- Savi, M.; Su, F.; Sterchele, E.D.; Bogossian, E.G.; Demailly, Z.; Baggiani, M.; Casu, G.S.; Taccone, F.S. Targeting NETosis in Acute Brain Injury: A Systematic Review of Preclinical and Clinical Evidence. Cells 2024, 13, 1553. [Google Scholar] [CrossRef]
- Qiao, S.; Yuan, J.; Zhang, S.C.; Lu, Y.Y.; Zhou, P.; Xin, T. Neutrophil Extracellular Traps in Central Nervous System Disorders: Mechanisms, Implications, and Emerging Perspective. Front. Immunol. 2025, 16, 1602336. [Google Scholar] [CrossRef]
- Seol, S.I.; Oh, S.A.; Davaanyam, D.; Lee, J.K. Blocking Peptidyl Arginine Deiminase 4 Confers Neuroprotective Effect in the Post-Ischemic Brain through Both NETosis-Dependent and -Independent Mechanisms. Acta Neuropathol. Commun. 2025, 13, 33. [Google Scholar] [CrossRef]
- Zhou, R.; Zhang, T.; Sun, J.; Tan, M.; Li, T.; Yang, T.; Dai, S.S.; Liu, Y.W. Neutrophil Extracellular Traps Aggravate Neutrophil Reverse Transendothelial Migration during Traumatic Brain Injury. Biochem. Biophys. Res. Commun. 2025, 778, 152387. [Google Scholar] [CrossRef] [PubMed]
- Li, A.; Pei, T.W.; Qi, H.; Song, L.B.; Fang, J.; Ding, Z.S.; Chen, T. Study on the Function and Mechanism of Neutrophil Extracellular Traps in Regulating Necroptosis Following Traumatic Brain Injury. Brain Behav. 2026, 16, e71275. [Google Scholar] [CrossRef]
- Wood, L.M.; Moore, J.K. β3 Accelerates Microtubule Plus End Maturation through a Divergent Lateral Interface. Mol. Biol. Cell. 2025, 36(4), ar36. [Google Scholar] [CrossRef]
- Poirier, K.; Saillour, Y.; Bahi-Buisson, N.; Jaglin, X.H.; Fallet-Bianco, C.; Nabbout, R.; Castelnau-Ptakhine, L.; Roubertie, A.; Attié-Bitach, T.; Desguerre, I.; et al. Mutations in the Neuronal β-Tubulin Subunit TUBB3 Result in Malformation of Cortical Development and Neuronal Migration Defects. Hum. Mol. Genet. 2010, 19, 4462–4473. [Google Scholar] [CrossRef] [PubMed]
- Kaverina, I.; Straube, A. Regulation of Cell Migration by Dynamic Microtubules. Semin. Cell. Dev. Biol. 2011, 22, 968–974. [Google Scholar] [CrossRef] [PubMed]
- Quigley, E.B.; DeVore, S.B.; Khan, S.A.; Geisterfer, Z.M.; Rothfuss, H.M.; Sequoia, A.O.; Thompson, P.R.; Gatlin, J.C.; Cherrington, B.D.; Navratil, A.M. GnRH Induces Citrullination of the Cytoskeleton in Murine Gonadotrope Cells. Int. J. Mol. Sci. 2024, 25, 3181. [Google Scholar] [CrossRef]
- Moskowitz, P.F.; Oblinger, M.M. Sensory Neurons Selectively Upregulate Synthesis and Transport of the Beta III-Tubulin Protein during Axonal Regeneration. J. Neurosci. 1995, 15, 1545–1555. [Google Scholar] [CrossRef]
- Latremoliere, A.; Cheng, L.; DeLisle, M.; Wu, C.; Chew, S.; Hutchinson, E.B.; Sheridan, A.; Alexandre, C.; Latremoliere, F.; Sheu, S.H.; et al. Neuronal-Specific TUBB3 Is Not Required for Normal Neuronal Function but Is Essential for Timely Axon Regeneration. Cell. Rep. 2018, 24, 1865–1879.e9. [Google Scholar] [CrossRef]
- Puri, D.; Barry, B.J.; Engle, E.C. TUBB3 and KIF21A in Neurodevelopment and Disease. Front. Neurosci. 2023, 17, 1226181. [Google Scholar] [CrossRef]
- Guo, Y.; Wang, Y.Y.; Sun, T.T.; Xu, J.J.; Yang, P.; Ma, C.Y.; Guan, W.J.; Wang, C.J.; Liu, G.F.; Liu, C.Q. Neural Progenitor Cells Derived from Fibroblasts Induced by Small Molecule Compounds under Hypoxia for Treatment of Parkinson’s Disease in Rats. Neural Regen. Res. 2023, 18, 1090–1098. [Google Scholar] [CrossRef]
- Hartmann, J.; Henschel, N.; Bartmann, K.; Dönmez, A.; Brockerhoff, G.; Koch, K.; Fritsche, E. Molecular and Functional Characterization of Different BrainSphere Models for Use in Neurotoxicity Testing on Microelectrode Arrays. Cells 2023, 12, 1270. [Google Scholar] [CrossRef]
- Kim, J.T.; Cho, S.M.; Youn, D.H.; Hong, E.P.; Park, C.H.; Lee, Y.; Jung, H.; Jeon, J.P. Therapeutic Effect of a Hydrogel-Based Neural Stem Cell Delivery Sheet for Mild Traumatic Brain Injury. Acta Biomater. 2023, 167, 335–347. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; Wang, W.; Zhang, Y.; Gou, X.; Zhang, Q.; Huang, Y.; Zhang, K.; Zhang, H.; Yang, J.; Li, Y. Ethanol Changes Nestin-Promoter Induced Neural Stem Cells to Disturb Newborn Dendritic Spine Remodeling in the Hippocampus of Mice. Neural Regen. Res. 2024, 19, 416–424. [Google Scholar] [CrossRef] [PubMed]
- Wilhelmsson, U.; Lebkuechner, I.; Leke, R.; Marasek, P.; Yang, X.; Antfolk, D.; Chen, M.; Mohseni, P.; Lasič, E.; Bobnar, S.T.; Stenovec, M.; Zorec, R.; Nagy, A.; Sahlgren, C.; Pekna, M.; Pekny, M. Nestin Regulates Neurogenesis in Mice through Notch Signaling from Astrocytes to Neural Stem Cells. Cereb. Cortex 2019, 29, 4050–4066. [Google Scholar] [CrossRef] [PubMed]
- Gilyarov, A.V. Nestin in Central Nervous System Cells. Neurosci. Behav. Physiol. 2008, 38, 165–169. [Google Scholar] [CrossRef]
- Xiao, Q.X.; Xue, L.L.; Tan, Y.X.; Huangfu, L.R.; Chen, L.; Zhai, C.Y.; Ma, R.F.; Al-Hawwas, M.; Zhou, H.S.; Wang, T.H.; Zhou, X.F.
- Wong, A.; Ghassemi, E.; Yellowley, C.E. Nestin Expression in Mesenchymal Stromal Cells: Regulation by Hypoxia and Osteogenesis. BMC Vet. Res. 2014, 10, 173. [Google Scholar] [CrossRef] [PubMed]
- Briot, J.; Simon, M.; Méchin, M.-C. Deimination, Intermediate Filaments and Associated Proteins. Int. J. Mol. Sci. 2020, 21, 8746. [Google Scholar] [CrossRef]
- Mafuika, N.S.; Naicker, T.; Harrichandparsad, R.; Lazarus, L. The Potential of Serum S100 Calcium-Binding Protein B and Glial Fibrillary Acidic Protein as Biomarkers for Traumatic Brain Injury. Transl. Res. Anat. 2022, 29, 100228. [Google Scholar] [CrossRef]
- Vos, P.E.; Jacobs, B.; Andriessen, T.M.; Lamers, K.J.; Borm, G.F.; Beems, T.; Edwards, M.; Rosmalen, C.F.; Vissers, J.L. GFAP and S100B Are Biomarkers of Traumatic Brain Injury: An Observational Cohort Study. Neurology 2010, 75, 1786–1793. [Google Scholar] [CrossRef]
- Agoston, D.V.; Shutes-David, A.; Peskind, E.R. Biofluid Biomarkers of Traumatic Brain Injury. Brain Inj. 2017, 31, 1195–1203. [Google Scholar] [CrossRef]
- Czeiter, E.; Amrein, K.; Gravesteijn, B.Y.; Lecky, F.; Menon, D.K.; Mondello, S.; Newcombe, V.F.J.; Richter, S.; Steyerberg, E.W.; Vyvere, T.V.; Verheyden, J.; Xu, H.; Yang, Z.; Maas, A.I.R.; Wang, K.K.W.; Büki, A. CENTER-TBI Participants and Investigators. Blood Biomarkers on Admission in Acute Traumatic Brain Injury: Relations to Severity, CT Findings and Care Path in the CENTER-TBI Study. EBioMedicine 2020, 56, 102785. [Google Scholar] [CrossRef]
- Gao, K.; Wang, C.R.; Jiang, F.; Wong, A.Y.K.; Su, N.; Jiang, J.H.; Chai, R.C.; Vatcher, G.; Teng, J.; Chen, J.; Jiang, Y.-W.; Yu, A.C.H. Traumatic Scratch Injury in Astrocytes Triggers Calcium Influx to Activate the JNK/c-Jun/AP-1 Pathway and Switch on GFAP Expression. Glia 2013, 61, 2063–2077. [Google Scholar] [CrossRef]
- Kesherwani, V.; Tarang, S.; Barnes, R.; Agrawal, S.K. Fasudil Reduces GFAP Expression after Hypoxic Injury. Neurosci. Lett. 2014, 576, 45–50. [Google Scholar] [CrossRef]
- Filous, A.R.; Silver, J. Targeting Astrocytes in CNS Injury and Disease: A Translational Research Approach. Prog. Neurobiol. 2016, 144, 173–187. [Google Scholar] [CrossRef] [PubMed]
- Yousaf, M.A.; Scartezzini, A.; Colombo, C.; Bachetti, T.; Sarto, E.; Bella, D.D.; Lorenzi, P.; Tinazzi, M.; Fabrizi, G.M.; Vattemi, G.; Savoia, A. A Novel De Novo GFAP Variant Causes a Juvenile-Onset Alexander Disease with Bilateral Vocal Cord Paralysis. Gene 2025, 951, 149388. [Google Scholar] [CrossRef]
- Rodnight, R.; Gonçalves, C.A.; Wofchuk, S.T.; Leal, R. Control of the Phosphorylation of the Astrocyte Marker Glial Fibrillary Acidic Protein (GFAP) in the Immature Rat Hippocampus by Glutamate and Calcium Ions: Possible Key Factor in Astrocytic Plasticity. Braz. J. Med. Biol. Res. 1997, 30, 325–338. [Google Scholar] [CrossRef]
- Herskowitz, J.H.; Seyfried, N.T.; Duong, D.M.; Xia, Q.; Rees, H.D.; Gearing, M.; Peng, J.; Lah, J.J.; Levey, A.I. Phosphoproteomic Analysis Reveals Site-Specific Changes in GFAP and NDRG2 Phosphorylation in Frontotemporal Lobar Degeneration. J. Proteome Res. 2010, 9, 6368–6379. [Google Scholar] [CrossRef]
- Sullivan, S.M.; Sullivan, R.K.; Miller, S.M.; Ireland, Z.; Björkman, S.T.; Pow, D.V.; Colditz, P.B. Phosphorylation of GFAP Is Associated with Injury in the Neonatal Pig Hypoxic-Ischemic Brain. Neurochem. Res. 2012, 37, 2364–2378. [Google Scholar] [CrossRef]
- Snider, N.T.; Omary, M.B. Post-Translational Modifications of Intermediate Filament Proteins: Mechanisms and Functions. Nat. Rev. Mol. Cell. Biol. 2014, 15, 163–177. [Google Scholar] [CrossRef] [PubMed]
- Battaglia, R.A.; Beltran, A.S.; Delic, S.; Dumitru, R.; Robinson, J.A.; Kabiraj, P.; Herring, L.E.; Madden, V.J.; Ravinder, N.; Willems, E.; Goldman, J.E.; Quinlan, R.A.; Snider, N.T. Site-Specific Phosphorylation and Caspase Cleavage of GFAP Are New Markers of Alexander Disease Severity. eLife 2019, 8, e47789. [Google Scholar] [CrossRef] [PubMed]
- Kanuri, S.H.; Sirrkay, P.J. Deciphering the Structural Biology of GFAP: Connotations of Its Potency in Presaging the Diagnosis for Traumatic Brain Injury and AD. Neurol. Int. 2025, 17, 134. [Google Scholar] [CrossRef] [PubMed]
- Klein, M.A.; Möller, J.C.; Jones, L.L.; Bluethmann, H.; Kreutzberg, G.W.; Raivich, G. Impaired Neuroglial Activation in Interleukin-6 Deficient Mice. Glia 1997, 19, 227–233. [Google Scholar] [CrossRef]
- Brunello, A.G.; Weissenberger, J.; Kappeler, A.; Vallan, C.; Peters, M.; Rose-John, S.; Weis, J. Astrocytic Alterations in Interleukin-6/Soluble Interleukin-6 Receptor Alpha Double-Transgenic Mice. Am. J. Pathol. 2000, 157, 1485–1493. [Google Scholar] [CrossRef]
- Levison, S.W.; Jiang, F.J.; Stoltzfus, O.K.; Ducceschi, M.H. IL-6-Type Cytokines Enhance Epidermal Growth Factor-Stimulated Astrocyte Proliferation. Glia 2000, 32, 328–337. [Google Scholar] [CrossRef]
- Okada, S.; Nakamura, M.; Katoh, H.; Miyao, T.; Shimazaki, T.; Ishii, K.; Yamane, J.; Yoshimura, A.; Iwamoto, Y.; Toyama, Y.; Okano, H. Conditional Ablation of Stat3 or Socs3 Discloses a Dual Role for Reactive Astrocytes after Spinal Cord Injury. Nat. Med. 2006, 12, 829–834. [Google Scholar] [CrossRef] [PubMed]
- Krasovska, V.; Doering, L.C. Regulation of IL-6 Secretion by Astrocytes via TLR4 in the Fragile X Mouse Model. Front. Mol. Neurosci. 2018, 11, 272. [Google Scholar] [CrossRef] [PubMed]
- Pons-Espinal, M.; Blasco-Agell, L.; Fernandez-Carasa, I.; Andrés-Benito, P.; di Domenico, A.; Richaud-Patin, Y.; Baruffi, V.; Marruecos, L.; Espinosa, L.; Garrido, A.; Tolosa, E.; Edel, M.J.; Otero, M.J.; Mosquera, J.L.; Ferrer, I.; Raya, A.; Consiglio, A. Blocking IL-6 Signaling Prevents Astrocyte-Induced Neurodegeneration in an iPSC-Based Model of Parkinson’s Disease. JCI Insight 2024, 9, e163359. [Google Scholar] [CrossRef] [PubMed]
- Ishigami, A.; Ohsawa, T.; Hiratsuka, M.; Taguchi, H.; Kobayashi, S.; Saito, Y.; Murayama, S.; Asaga, H.; Toda, T.; Kimura, N.; et al. Abnormal Accumulation of Citrullinated Proteins Catalyzed by Peptidylarginine Deiminase in Hippocampal Extracts from Patients with Alzheimer’s Disease. J. Neurosci. Res. 2005, 80, 120–128. [Google Scholar] [CrossRef]
- Jang, B.; Jin, J.K.; Jeon, Y.C.; Cho, H.J.; Ishigami, A.; Choi, K.C.; Carp, R.I.; Maruyama, N.; Kim, Y.S.; Choi, E.K. Involvement of Peptidylarginine Deiminase-Mediated Post-Translational Citrullination in Pathogenesis of Sporadic Creutzfeldt-Jakob Disease. Acta Neuropathol. 2010, 119, 199–210. [Google Scholar] [CrossRef]
- Yusuf, I.O.; Qiao, T.; Parsi, S.; Tilvawala, R.; Thompson, P.R.; Xu, Z. Protein Citrullination Marks Myelin Protein Aggregation and Disease Progression in Mouse ALS Models. Acta Neuropathol. Commun. 2022, 10, 135. [Google Scholar] [CrossRef]
- McNee, G.; Eales, K.L.; Wei, W.; Williams, D.S.; Barkhuizen, A.; Bartlett, D.B.; Essex, S.; Anandram, S.; Filer, A.; Moss, P.A.; Davies, C.C.; Tennant, D.A. Citrullination of Histone H3 Drives IL-6 Production by Bone Marrow Mesenchymal Stem Cells in MGUS and Multiple Myeloma. Leukemia 2017, 31, 373–381. [Google Scholar] [CrossRef]
- De Conto, V.; Cheung, V.; Maubon, G.; Souguir, Z.; Maubon, N.; Vandenhaute, E.; Bérézowski, V. In Vitro Differentiation Modifies the Neurotoxic Response of SH-SY5Y Cells. Toxicol. In Vitr. 2021, 77, 105235. [Google Scholar] [CrossRef]
- U, K.P.; Subramanian, V.; Nicholas, A.P.; Thompson, P.R.; Ferretti, P. Modulation of Calcium-Induced Cell Death in Human Neural Stem Cells by the Novel Peptidylarginine Deiminase-AIF Pathway. Biochim. Biophys. Acta Mol. Cell. Res. 2014, 1843, 1162–1171. [Google Scholar] [CrossRef]
- Kaneva, M.K.; Kerrigan, M.J.; Grieco, P.; Curley, G.P.; Locke, I.C.; Getting, S.J. Chondroprotective and Anti-Inflammatory Role of Melanocortin Peptides in TNF-α Activated Human C-20/A4 Chondrocytes. Br. J. Pharmacol. 2012, 167, 67–79. [Google Scholar] [CrossRef]
- Can, V.C.; Locke, I.C.; Kaneva, M.K.; Kerrigan, M.J.P.; Merlino, F.; De Pascale, C.; Grieco, P.; Getting, S.J. Novel Anti-Inflammatory and Chondroprotective Effects of the Human Melanocortin MC1 Receptor Agonist BMS-470539 Dihydrochloride and Human Melanocortin MC3 Receptor Agonist PG-990 on Lipopolysaccharide Activated Chondrocytes. Eur. J. Pharmacol. 2020, 872, 172971. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



