Submitted:
25 April 2026
Posted:
27 April 2026
You are already at the latest version
Abstract
Keywords:
Introduction
Materials and Methods
Study Design
Setting
Participants
Variables
Data Sources/Measurement
Study Size
Quantitative Variables
Statistical Methods
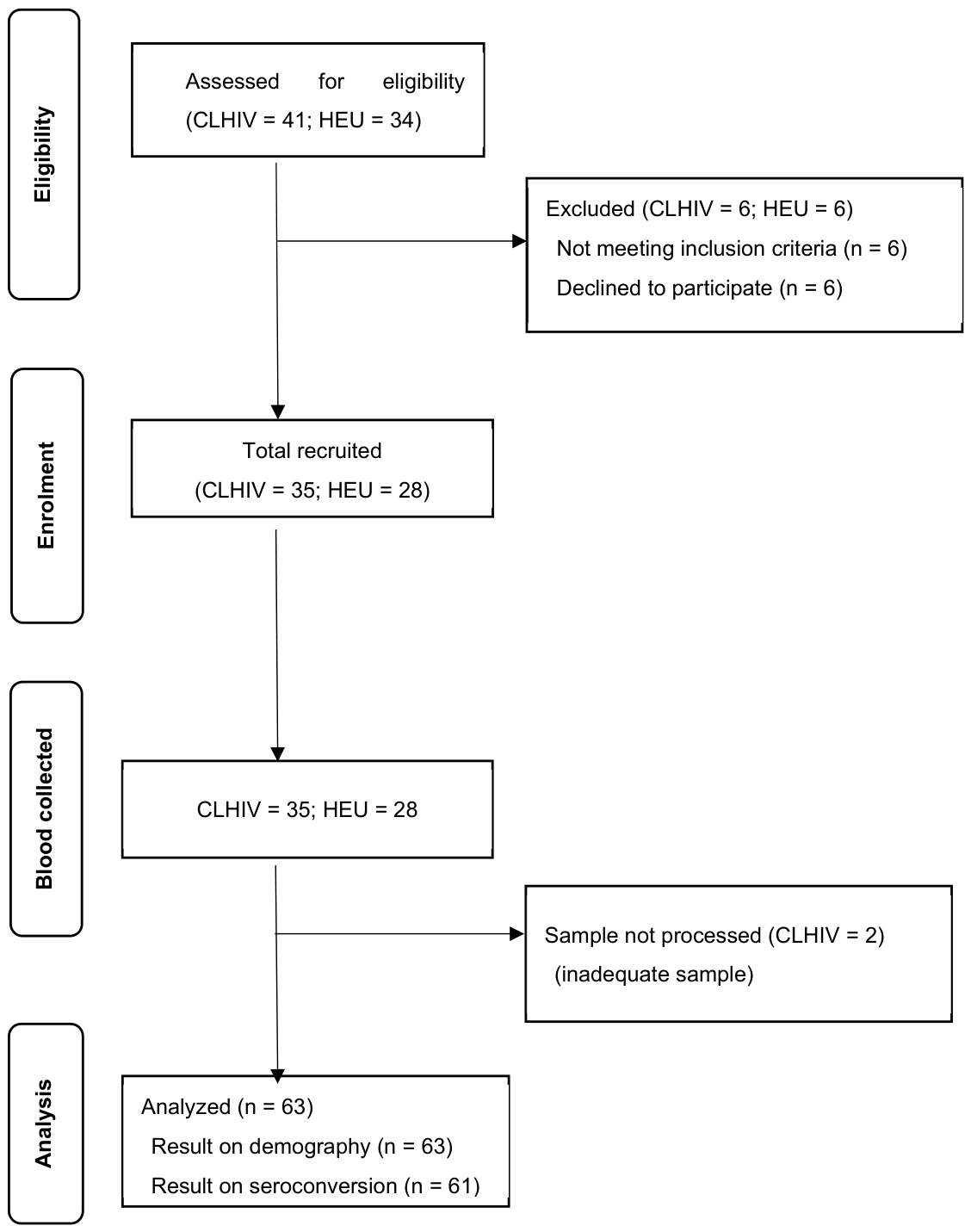
Results
Participants
Descriptive Data
Outcome Data
Main Results
Discussion
Limitations
Conclusions
Ethical Statement
References
- World Health Organization (WHO); Regional Office for South-East Asia. Midterm review of the “Strategic plan for measles elimination and rubella and congenital rubella syndrome control in the South-East Asia Region: 2014–2020” [Internet]. World Health Organization. Regional Office for South-East Asia, 2018. Available online: https://apps.who.int/iris/handle/10665/279993.
- Feemster, K.A.; Szipszky, C. Resurgence of measles in the United States: how did we get here? Curr. Opin. Pediatr. 2020, 32, 139–144. [Google Scholar] [CrossRef]
- Mehtani, N.J.; Rosman, L.; Moss, W.J. Immunogenicity and Safety of the Measles Vaccine in HIV-Infected Children: An Updated Systematic Review. Am. J. Epidemiol. 2019, 188, 2240–51. [Google Scholar] [CrossRef] [PubMed]
- Simani, O.E.; Adrian, P.V.; Violari, A.; et al. Effect of in-utero HIV exposure and antiretroviral treatment strategies on measles susceptibility and immunogenicity of measles vaccine. AIDS 2013, 27, 1583–1591. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization (WHO). Measles and rubella strategic framework 2021–2030; Licence: CC BY-NC-SA 3.0 IGO; World Health Organization: Geneva, 2020. [Google Scholar]
- Siberry, G.K.; Patel, K.; Bellini, W.J.; Karalius, B.; Purswani, M.U.; Burchett, S.K.; Meyer, W.A., 3rd; Sowers, S.B.; Ellis, A.; Van Dyke, R.B. Pediatric HIV AIDS Cohort Study (PHACS); Pediatric HIV AIDS Cohort Study PHACS. Immunity to Measles, Mumps, and Rubella in US Children With Perinatal HIV Infection or Perinatal HIV Exposure Without Infection. Clin. Infect. Dis. 2015, 61, 988–95. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Chen, R.T.; Markowitz, L.E.; Albrecht, P.; Stewart, J.A.; Mofenson, L.M.; Preblud, S.R.; Orenstein, W.A. Measles antibody: reevaluation of protective titers. J. Infect. Dis. 1990, 162, 1036–42. [Google Scholar] [CrossRef] [PubMed]
- World Health Organisation (WHO). ISSN 0049-8114 [Internet]. In Weekly Epidemiological Record; World Health Organization: Geneva, 2017; vol. 92, p. 17. Available online: https://www.who.int/publications/i/item/who-wer9217-205-227.
- Mutsaerts, E.A.; Nunes, M.C.; Van Rijswijk, M.N.; Klipstein-Grobusch, K.; Otwombe, K.; Cotton, M.F.; Violari, A.; Madhi, S.A. Measles immunity at 4.5 years of age following vaccination at 9 and 15–18 months of age among human immunodeficiency virus (HIV)–infected, HIV-exposed–uninfected, and HIV-unexposed children. Clin. Infect. Dis. 2019, 69, 687–96. [Google Scholar] [CrossRef] [PubMed]
- Mutsaerts, E.A.; Nunes, M.C.; van Rijswijk, M.N.; Klipstein-Grobusch, K.; Grobbee, D.E.; Madhi, S.A. Safety and immunogenicity of measles vaccination in HIV-infected and HIV-exposed uninfected children: a systematic review and meta-analysis. E Clin. Med. 2018, 1, 28–42. [Google Scholar] [CrossRef] [PubMed]
- Tejiokem, M.C.; Desselas, E.; Noumsi, T.J.; Ateba Ndongo, F.; Tetang Ndiang, S.; Fossi, M.A.; Guemkam, G.; Zangue Kenfack Tekougang, B.; Tagnouokam-Ngoupo, P.A.; Penda, I.C.; Faye, A. Pre-and Post-Vaccination Measles Antibody and Persistence Up to 5 Years of Age Among Early ART-Treated HIV-Infected, HIV-Exposed Uninfected and HIV-Unexposed Children in Cameroon. Vaccines 2025, 13, 584. [Google Scholar] [CrossRef] [PubMed]
- Jain, S.; Seth, A.; Khare, S.; Chandra, J. Seroprevalence of transplacentally acquired measles antibodies in HIV-exposed versus HIV-unexposed infants at six months of age. Indian J. Med. Res. 2017, 145, 536–42. [Google Scholar] [CrossRef] [PubMed]
- National AIDS and STD Control Programme Phase-V (2021-26). Strategy booklet. National AIDS Control Organisation (NACO), Ministry of Health & Family Welfare, Government of India: New Delhi, 2021. Available online: https://naco.gov.in/sites/default/files/NACP_V_Strategy_Booklet.pdf.
- NACO. Early infant diagnosis. National AIDS Control Organisation: New Delhi, 26 Sep 2023. Available online: https://naco.gov.in/sites/default/files/DBS_Sample_Collection_for_EID_Module_Read_Only.pdf.
- OpenEpi. Sample Size for Unmatched Case-Control Studies [Internet]. OpenEpi. 2 Feb 2022. Available online: http://www.openepi.com/SampleSize/unmatchedcc.htm.
- International Business Machines Corp. IBM SPSS Statistics for Windows, Version 23.0; IBM Corp.: Armonk (NY), 2015; Available online: https://www.ibm.com/products/spss-statistics.
- Python Software Foundation. Python language reference, version 3.11; Python Software Foundation: Wilmington (DE), 2023; Available online: http://www.python.org.
- Haban, H.; Benchekroun, S.; Sadeq, M.; Tajounte, L.; Ahmed, H.J.; Benjouad, A.; Amzazi, S.; Oumzil, H.; Elharti, E. Seroprevalence of measles vaccine antibody response in vertically HIV-infected children, in Morocco. BMC Infect. Dis. 2018, 18, 680. [Google Scholar] [CrossRef] [PubMed]
- Murhekar, M.V.; Gupta, N.; Hasan, A.Z.; Kumar, M.S.; Kumar, V.S.; Prosperi, C.; Sapkal, G.N.; Thangaraj, J.W.; Kaduskar, O.; Bhatt, V.; Deshpande, G.R. Evaluating the effect of measles and rubella mass vaccination campaigns on seroprevalence in India: A before-and-after cross-sectional household serosurvey in four districts, 2018–2020. Lancet Glob. Health 2022, 10, e1655-64. [Google Scholar] [CrossRef] [PubMed]
- Kumar, P.; Dhalaria, P.; Sapru, M.; Brahma, D.; Datta, E.; Priyadarshini, P.; Singh, A.K.; Taneja, G.; Tripathi, B. Trend Analysis of Routine Immunization Coverage in India: A Composite Measure Using Cross-Sectional Data of National Family Health Surveys-4 & 5. VeriXiv 2025, 2, 190. [Google Scholar]
- Bolotin, S.; Osman, S.; Hughes, S.L.; Ariyarajah, A.; Tricco, A.C.; Khan, S.; Li, L.; Johnson, C.; Friedman, L.; Gul, N.; Jardine, R. In elimination settings, measles antibodies wane after vaccination but not after infection: a systematic review and meta-analysis. J. Infect. Dis. 2022, 226, 1127–39. [Google Scholar] [CrossRef] [PubMed]
- Available online: https://www.cdc.gov/global-measles-vaccination/data-research/global-measles-outbreaks/index.html.
- Ministry of Health and Family Welfare; Government of India. Union Health Minister Shri J P Nadda launches National Zero Measles-Rubella Elimination Campaign on the occasion of World Immunization Week [press release]. PIB. 24 Apr 2025. Available online: https://www.pib.gov.in/PressReleasePage.aspx?PRID=2124032.
- Kang, H.J.; Han, Y.W.; Kim, S.J.; Kim, Y.J.; Kim, A.R.; Kim, J.A.; Jung, H.D.; Eom, H.E.; Park, O.; Kim, S.S. An increasing, potentially measles-susceptible population over time after vaccination in Korea. Vaccine 2017, 35, 4126–32. [Google Scholar] [CrossRef] [PubMed]
- Kontio, M.; Jokinen, S.; Paunio, M.; Peltola, H.; Davidkin, I. Waning antibody levels and avidity: implications for MMR vaccine-induced protection. J. Infect. Dis. 2012, 206, 1542–8. [Google Scholar] [CrossRef] [PubMed]
- Scott, P.; Moss, W.J.; Gilani, Z.; Low, N. Measles vaccination in HIV-infected children: systematic review and meta-analysis of safety and immunogenicity. J. Infect. Dis. 2011, 204, S164–78. [Google Scholar] [CrossRef] [PubMed]
- Mossong, J.; Muller, C.P. Modelling measles re-emergence as a result of waning of immunity in vaccinated populations. Vaccine 2003, 21, 4597–603. [Google Scholar] [CrossRef] [PubMed]
- Lefebvre, M.; Secher, S.; Bouchez, S.; Vandamme, Y.M.; Fialaire, P.; Leautez, S.; Blanchi, S.; Michau, C.; Coste-Burel, M.; Brunet-Cartier, C.; Reliquet, V. Measles seroprevalence in human immunodeficiency virus-infected adults born in the era of measles vaccination. AIDS 2022, 36, 1273–8. [Google Scholar] [CrossRef] [PubMed]
- Cohen, B.J.; Parry, R.P.; Doblas, D.; Samuel, D.; Warrener, L.; Andrews, N.; Brown, D. Measles immunity testing: comparison of two measles IgG ELISAs with plaque reduction neutralisation assay. J. Virol. Methods 2006, 131, 209–12. [Google Scholar] [CrossRef] [PubMed]
| HIV exposed (n=28) Number (%) |
HIV infected (n=35) Number (%) |
Difference between groups |
|
|---|---|---|---|
| Age (mean ± SD) in months | 52.1 (19.6) | 62.1 (18.3) | t=2.07; p =0.04 * |
| Birth weight (mean ± SD) in Kg | 2.7 (0.7) | 2.9 (0.4) | t=1.23; p=0.22 |
| Months from MR2 (mean ± SD) | 36.3 (23.8) | 40.6 (23.9) | t=0.61; p=0.55 |
| Sex | |||
| Male | 13 (46.4) | 16 (45.7) | X2 = 0.003 (DF 1; p=0.96) |
| Female | 15 (53.6) | 19 (54.3) | |
| Weight for Agea) | |||
| Normal | 17 (60.7) | 21 (61.8) | X2 = 1.7 (DF 2; p=0.42) |
| Underweight | 3 (10.7) | 7 (20.6) | |
| Severely underweight | 8 (28.6) | 6 (17.6) | |
| Characteristic | Number | % |
|---|---|---|
| Age of starting ART | ||
| Mean (SD) in months | 29.13 (17.24) | |
| Median (Q1-Q3 range) in months | 24 (18 – 41) | |
| Within 24 months | 19 | 54.3 |
| After 24 months | 12 | 34.3 |
| Missing record on ART initiation | 4 | 11.4 |
| Viral Load Suppression | ||
| Undetectable/Suppressed | 32 | 91.4 |
| Unsuppressed | 03 | 08.6 |
| CD4% or CD4 count at ART initiation | ||
| Immunosuppressed [ < 25% or <350] | 4 | 11.4 |
| Immunocompetent [≥ 25% or ≥ 350] | 27 | 77.1 |
| Missing record on ART initiation | 4 | ----- |
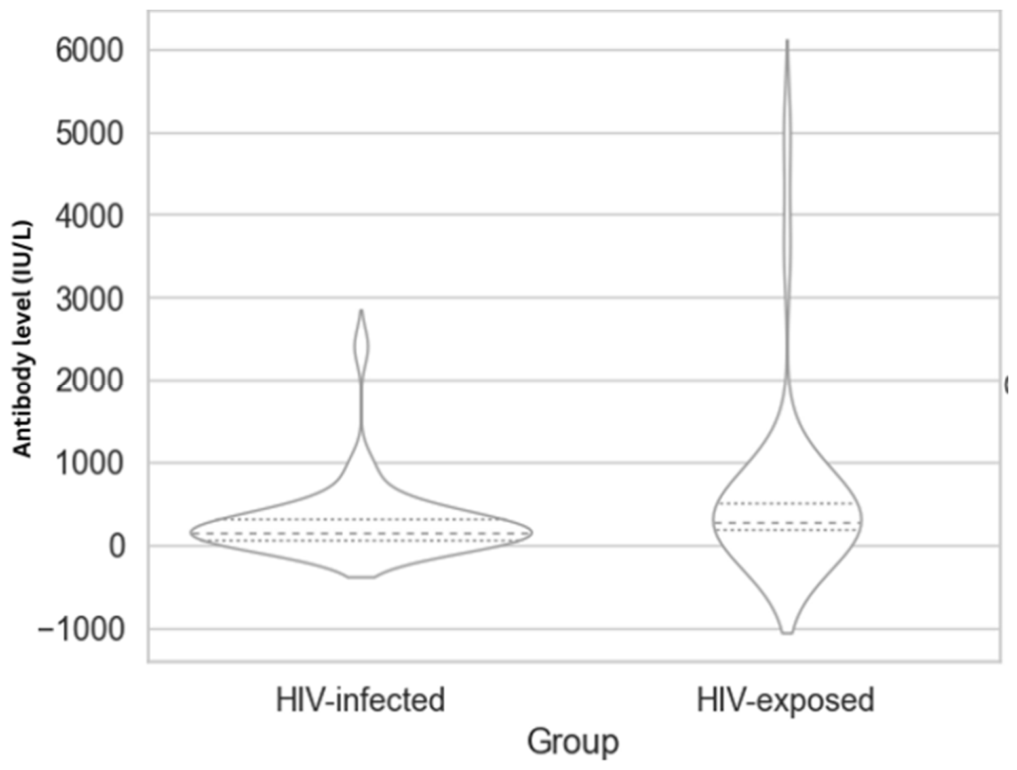
| Mean (SD) | Median (IQR) | Skewness | Kurtosis | |
|---|---|---|---|---|
| HIV infectedb) (n=33) |
290.62 (440.09) | 147.79 (2362.96) | 3.86 | 17.43 |
| HIV exposed (n=28) |
609.42 (1083.63) | 272.88 (4950.00) | 3.44 | 11.79 |
| Immunity against measles | HIV exposed (n=28) Number (%) |
HIV infectedb) (n=33) Number (%) |
Total (n=61) Number (%) |
Chi-square test for independence | Odds Ratio (95% CI) |
|---|---|---|---|---|---|
| Seroconversion | 14 (50) | 11 (33.7) | 25 (41) | χ2 = 1.73 (DF 1; p=0.18) |
0.5 (0.18-1.41) |
| Non-conversion | 14 (50) | 22 (66.7) | 36 (59) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




