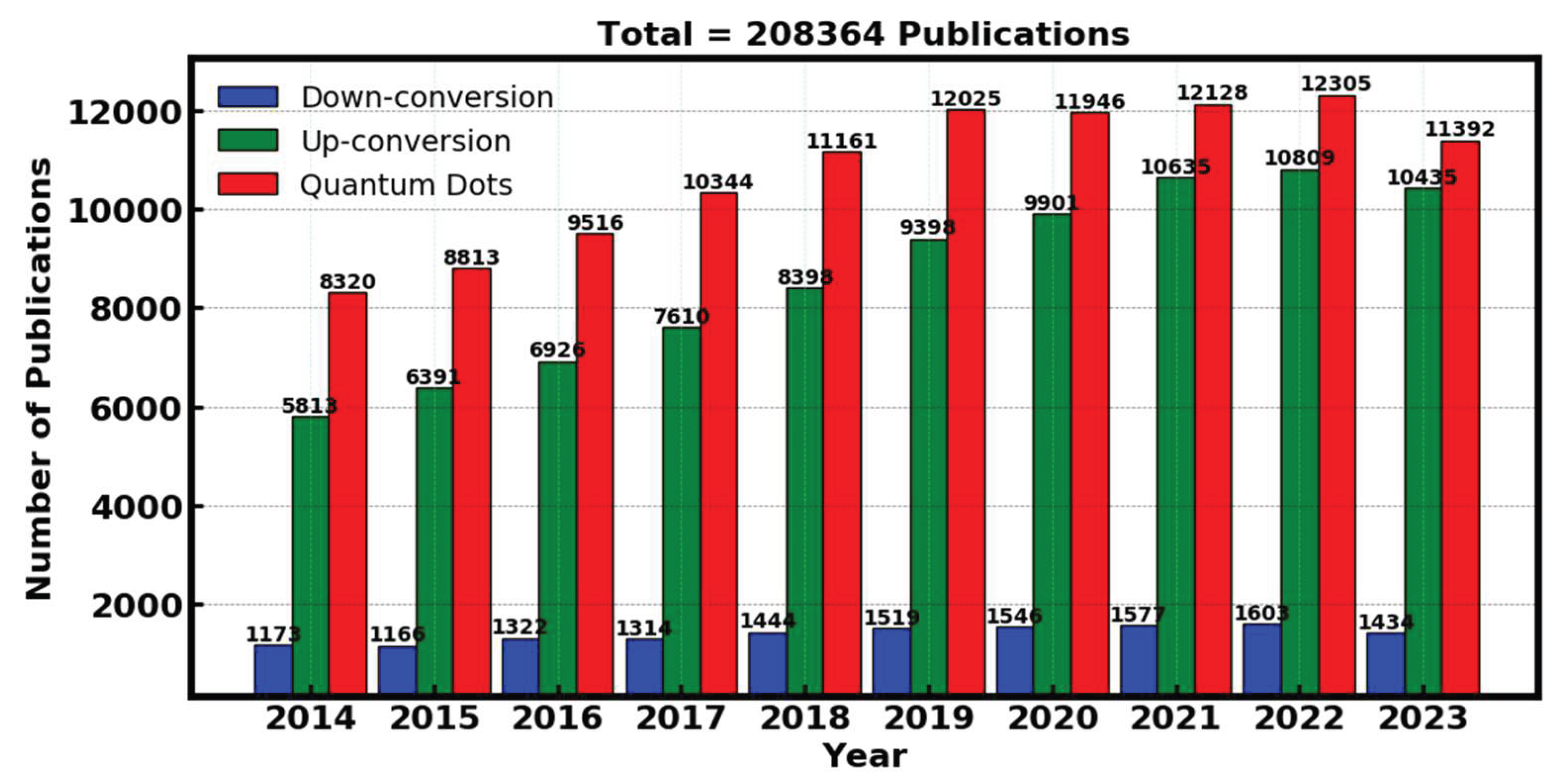
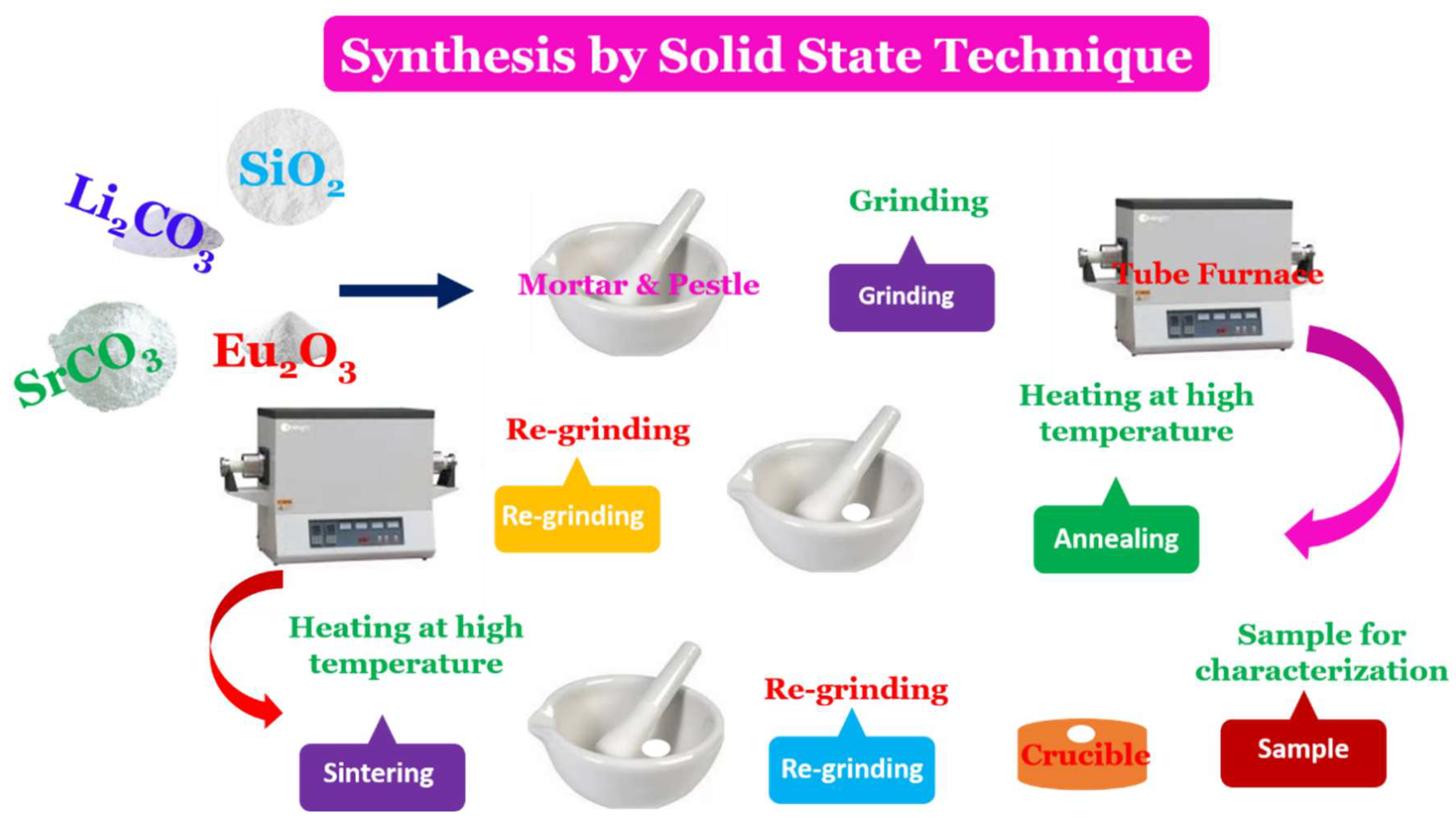
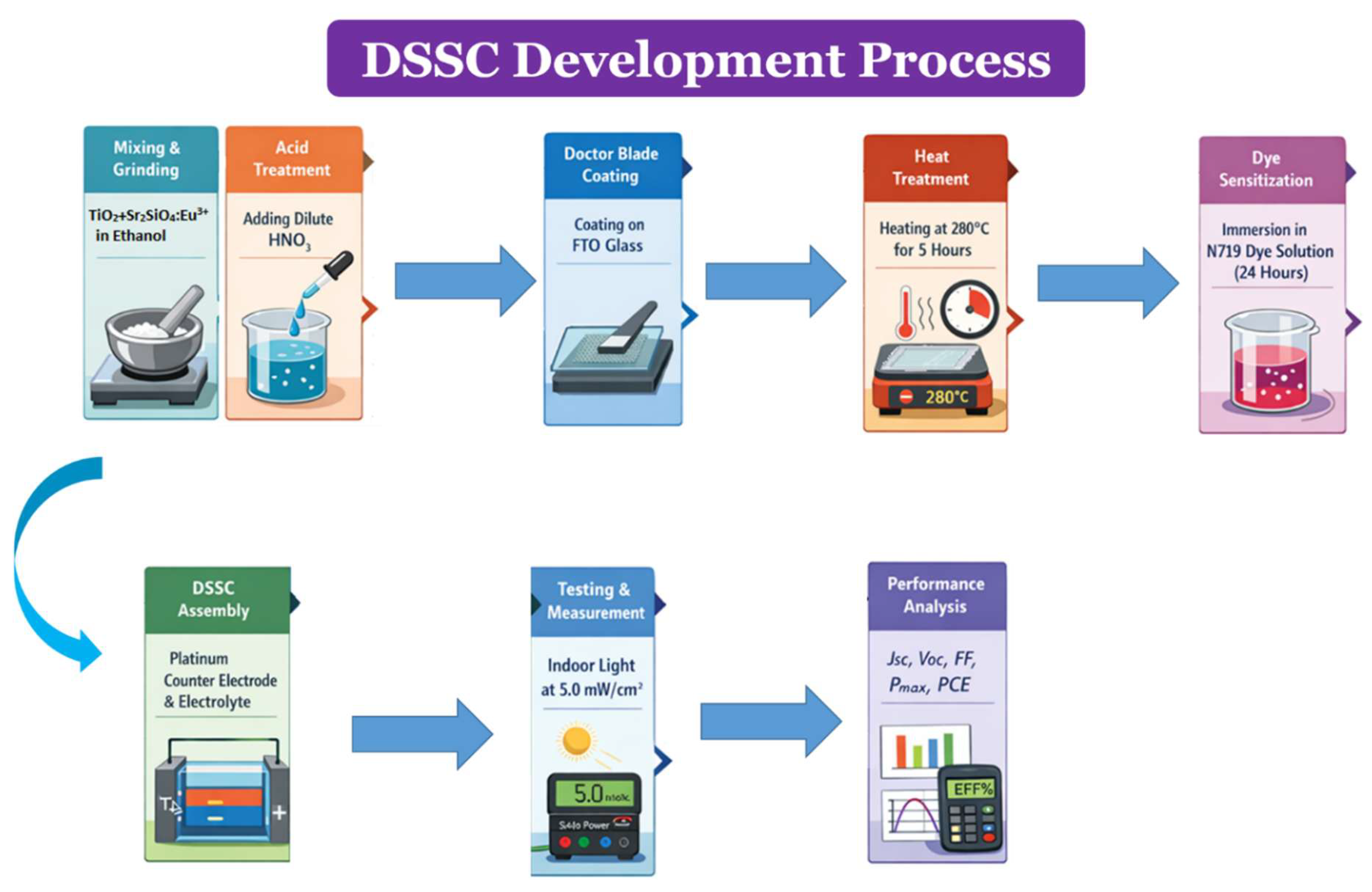
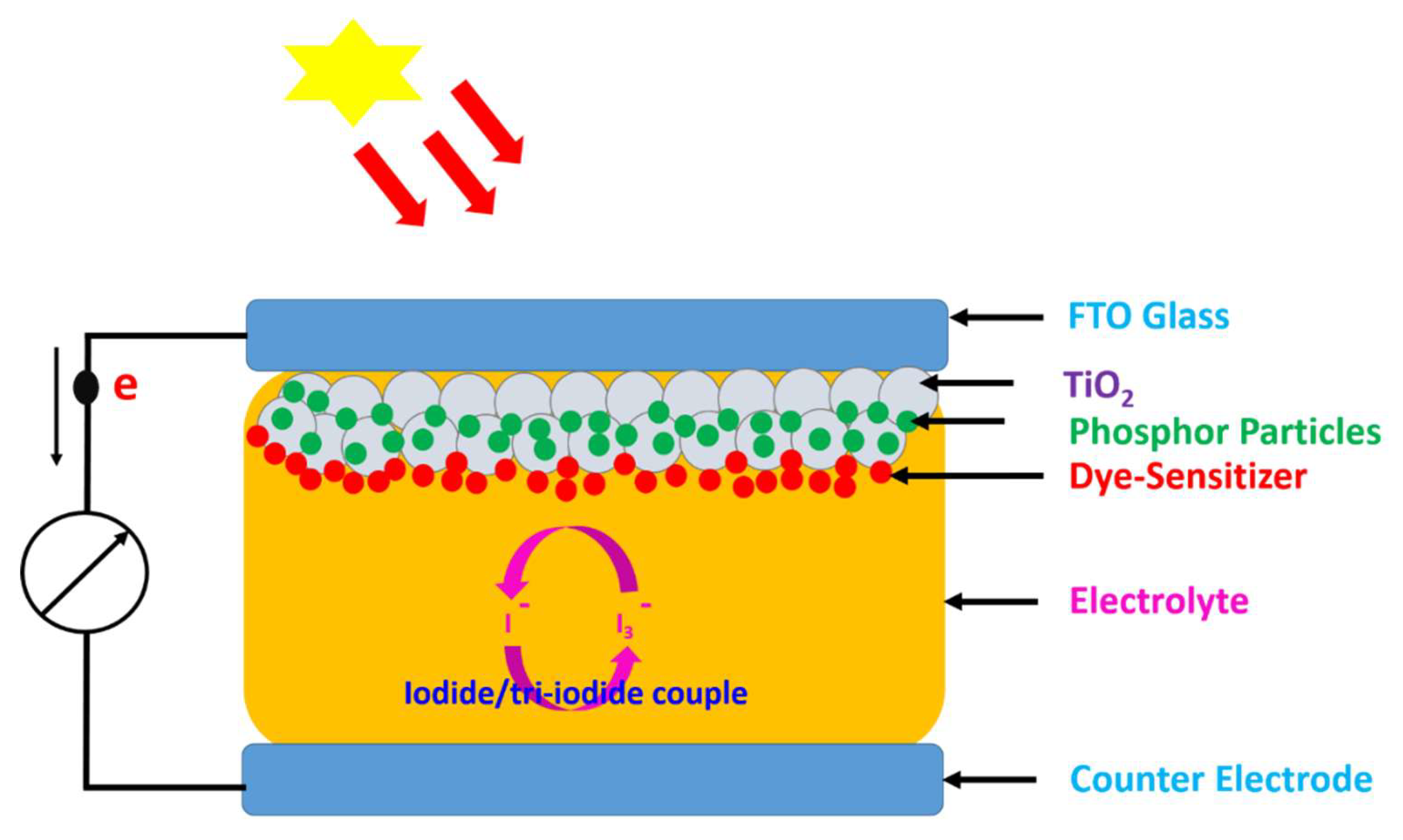
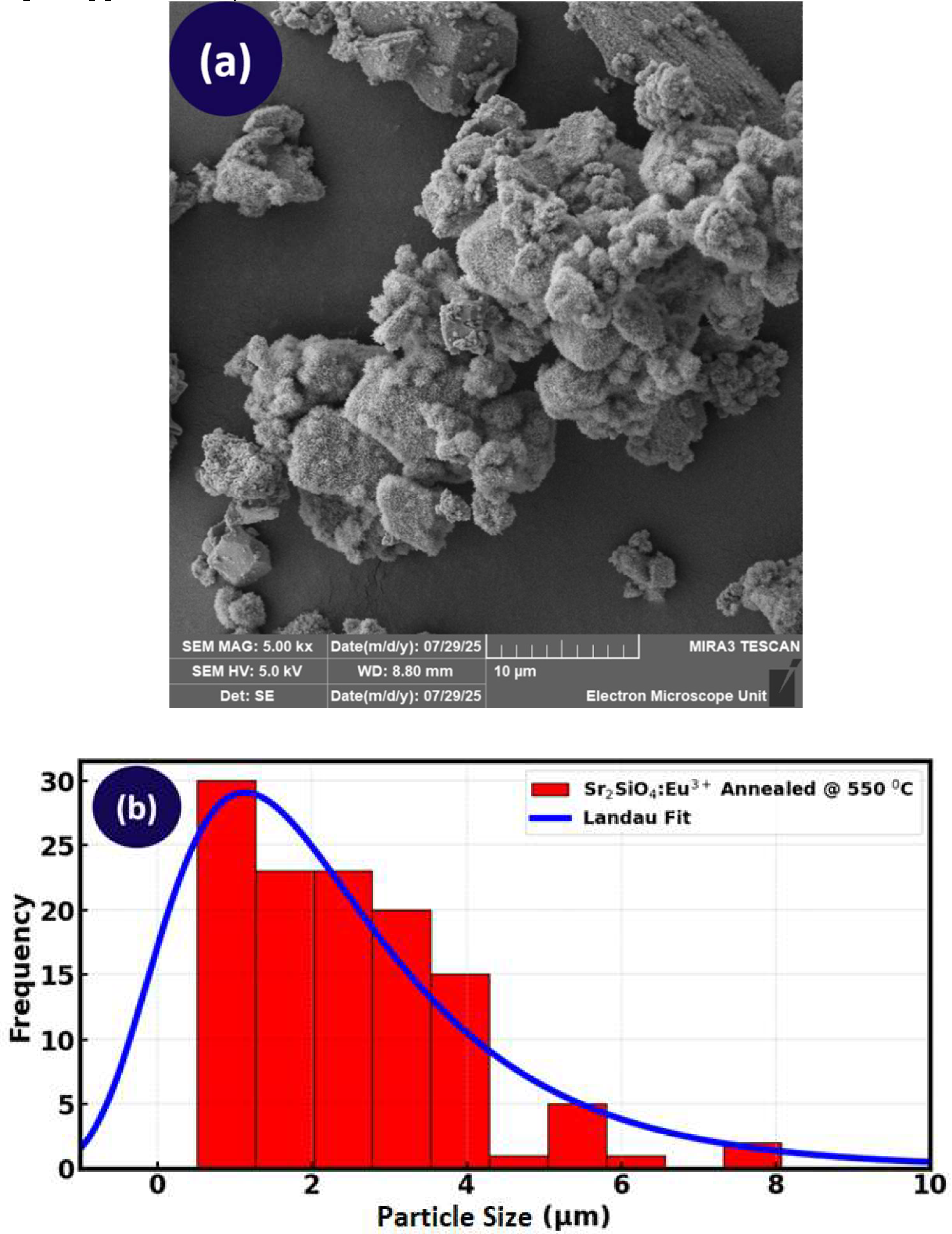
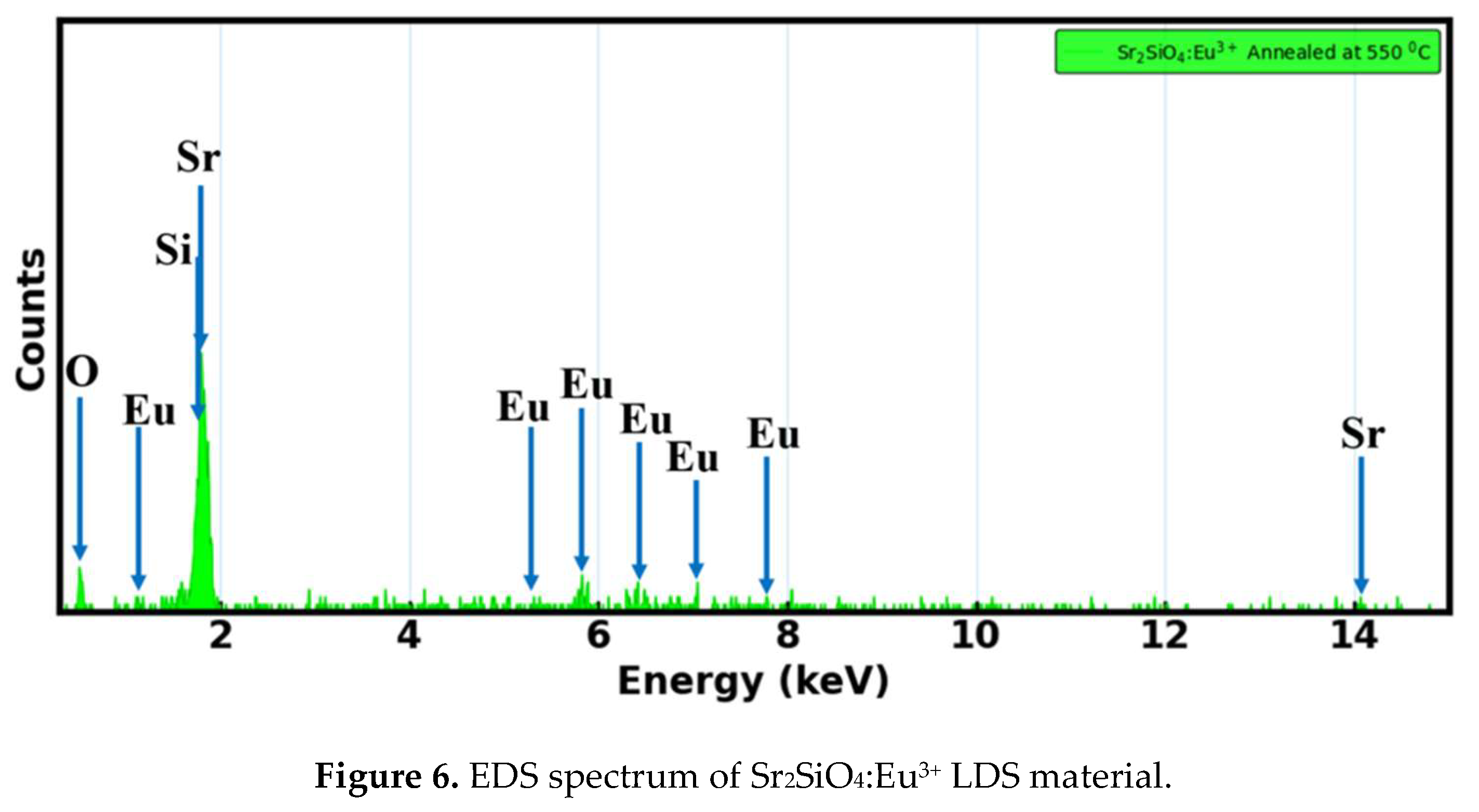
3.3. XRD Analysis
Powder X-ray diffraction (XRD) measurement of the LDS material was performed on a Panalytical Powder diffractometer in the 2theta range from 10 to 80
0 with monochromatized Cu-Kα radiation (λ = 0.15406 nm).
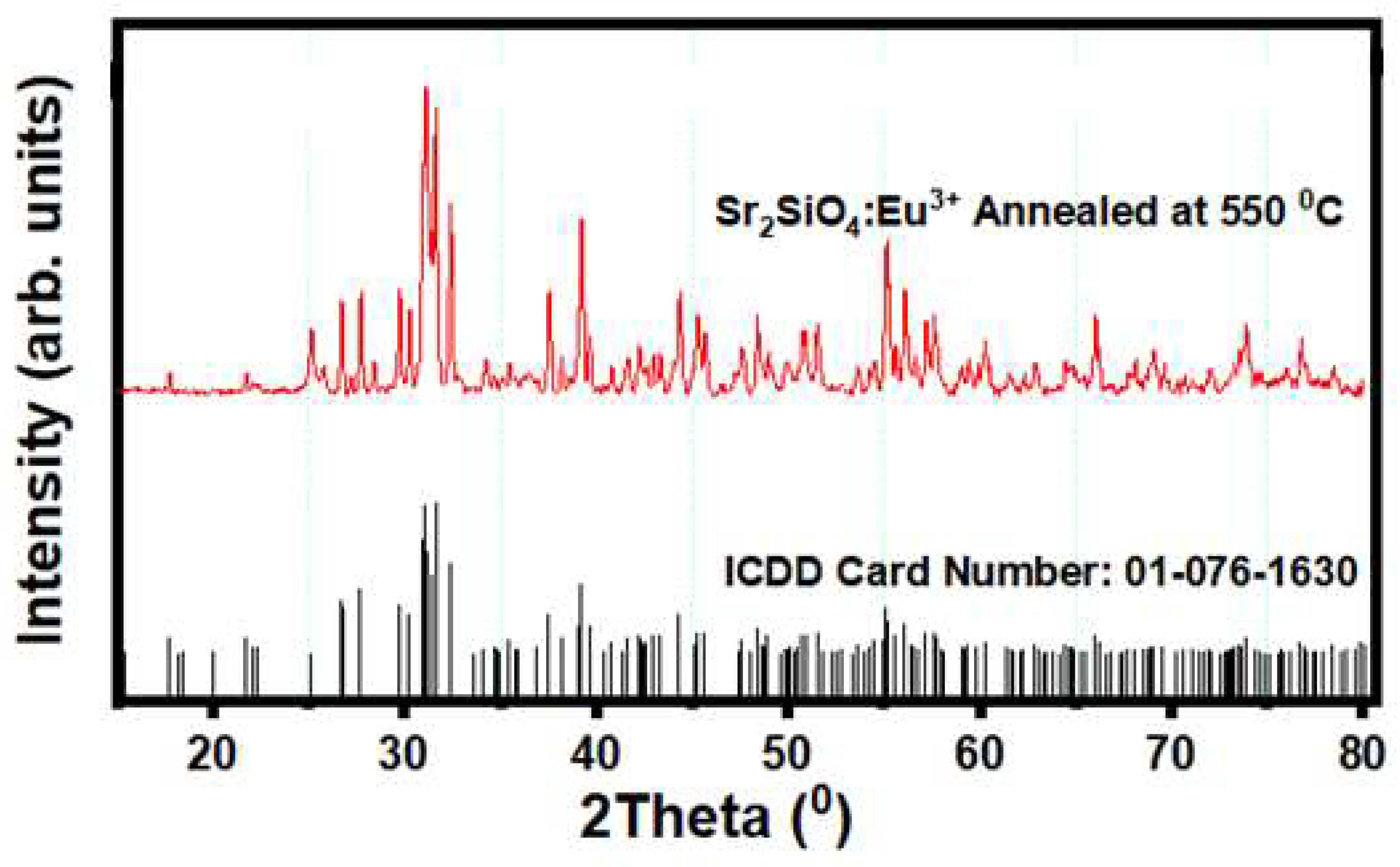
Figure 7 represents the XRD patterns of the Sr
2SiO
4:Eu
3+, with the corresponding international Centre for Diffraction Data (ICDD) card number 01 – 076 – 1630. The diffraction peaks corresponded with one another and are in agreement with the ICDD card number profile, which indicates that the solid state technique applied ensured effective synthesis of the strontium silicate material. Introduction of the activator Eu
3+ ions did not significantly alter the crystal structure of the Sr
2SiO
4, indicating that Eu
3+ ions was effectively incorporated into the host lattice. The Sr
2SiO
4 exhibits a monoclinic structure with space group: P21/n [
56,
57]. The lattice parameters were determined to be a = 5.663 Å, b = 7.084 Å, c = 9.767 Å, α = β = γ = 90
0, V = 391.39 Å
3.
“The monoclinic structure, space group and lattice parameters of Sr2SiO4 were determined by the ICDD pattern using the experimental data. It is pertinent to state that what majorly determines the crystal system (monoclinic or orthorhombic structure) is the space group, not the lattice parameters which can change depending on the synthesis technique. The intensity of the peaks suggests that the material has undergone a complete phase transformation and that the crystal grains have become more ordered. This enhanced crystallinity is advantageous for applications requiring efficient charge transport and stable structural properties, as it minimizes defects and grain boundaries that can act as charge traps or recombination centers. The crystal structural property is expected to translate into better optical and electronic properties, making the material suitable for advanced applications such as light-emitting devices and, potentially, dye-sensitized solar cells (DSSCs).
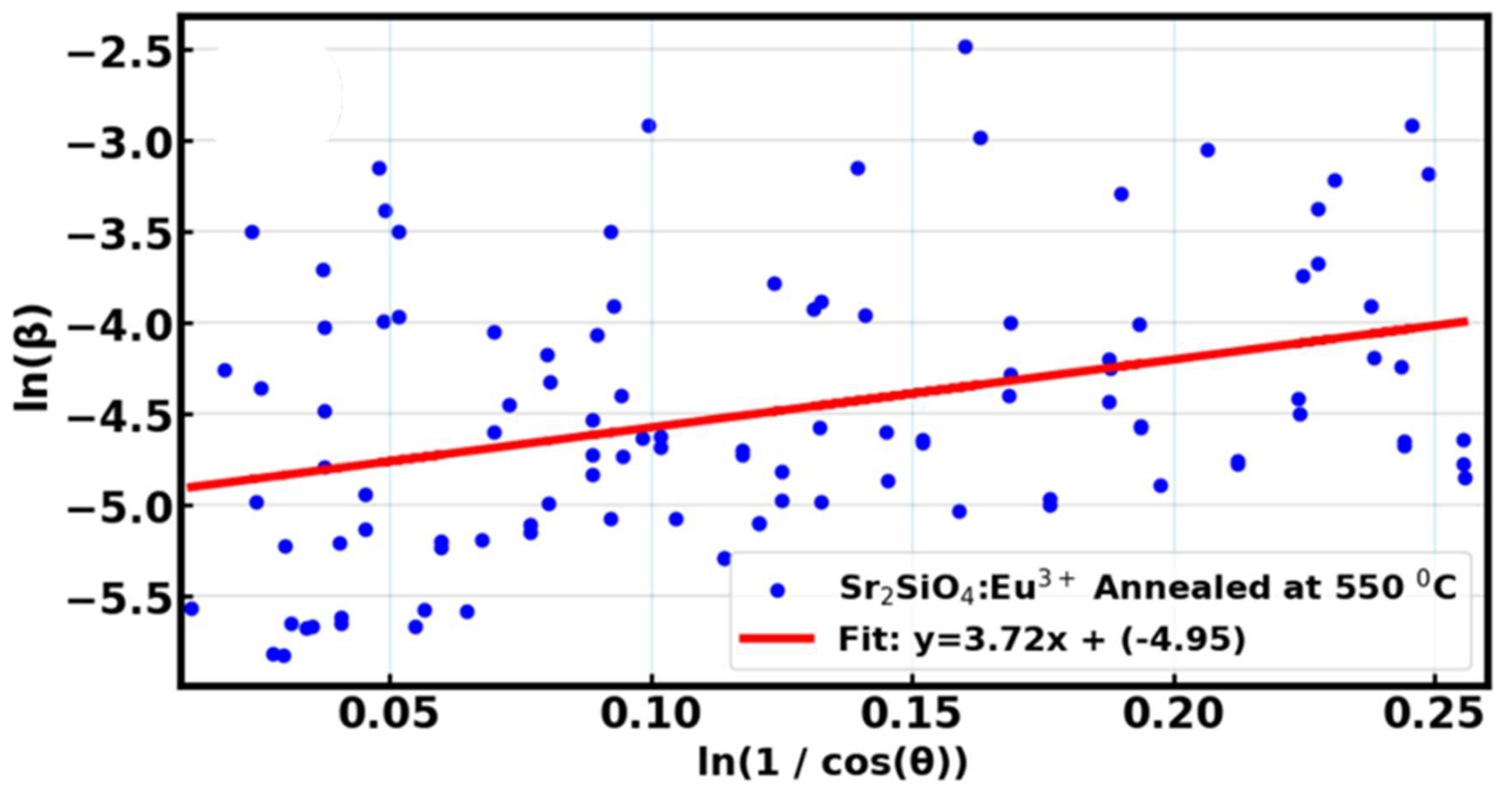
In order to quantitatively determine the crystallite size of the sample, the modified Scherrer equation remains advantageous at decreasing the sum of absolute values of errors,
and developing a line through the data points to extrapolate the intercept [
58,
59], and can be of great use in determining whether samples are of the same crystallite size.
Figure 8 provides the linear regression plots of
verses
for determining the crystallite size of the material.
Table 3 provides the quantitative estimated crystallite size value with the respective standard error sample. The crystallite size was estimated to be 19.5 nm for the LDS material, showing a relatively small crystallite size, however the sample annealed at 650
0C exhibits smaller crystallite size of 14.86 nm with a similar standard error (0.91 nm), suggesting that temperature treatment in this case leads to inhibited crystal growth or enhanced recrystallization processes that produce fine crystallite. Moreover, small crystallite size increases the surface area to volume ratio of the phosphor material, which can enhance the material’s light absorption capacity and equally improve the material’s luminescent efficiency, thereby resulting in better light emission [
60]. The behavior of excitons (electron-hole pairs) is also affected by crystallite size. In small crystallites, the increase in the movement and interaction of excitons, potentially leads to more efficient energy transfer and absorption of light, which is crucial in promoting the light absorption capacity and the performance of DSSCs.
3.5. Diffused Reflectance
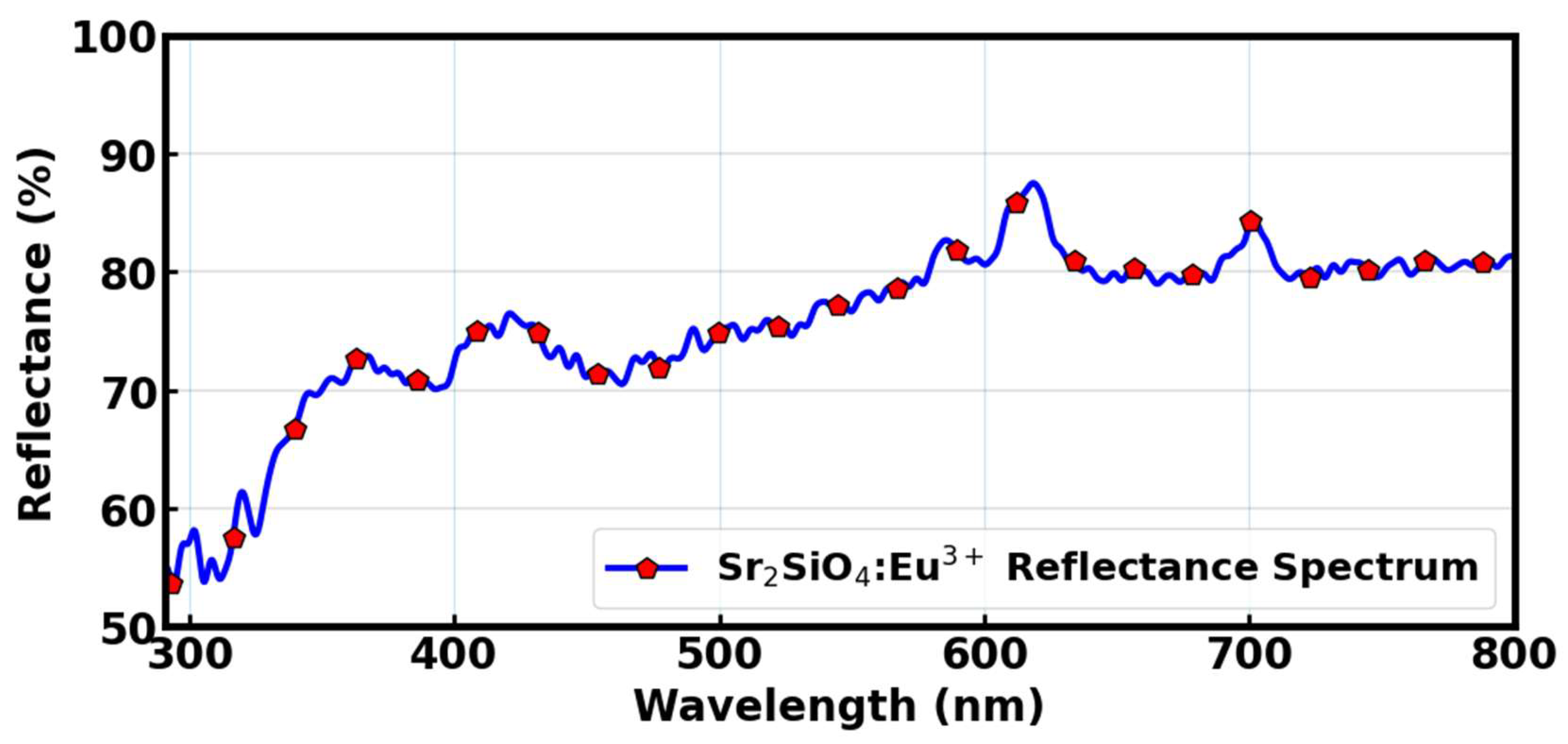
Diffused reflectance spectrum of the samples are shown in
Figure 10. The material exhibits strong reflectance between 410 and 800 nm, corresponding to the visible and near-infrared regions, while a marked decline in reflectance occurs below 400 nm, within the ultraviolet (UV) range. This decline indicates efficient absorption of UV photons, a behaviour linked to the material’s luminescent downshifting ability – specifically, its capacity to absorb high-energy UV light and re-emit it as visible light. This process is facilitated by electronic transitions from the f – f energy levels of Eu
3+ ions. Given its strong UV absorption, Sr
2SiO
4:Eu
3+ has potential as a UV-harvesting material in optoelectronic systems, such as dye-sensitized solar cells (DSSCs), where it can convert unused UV radiation into usable visible light, thereby improving power conversion efficiency.
The sample’s reflectivity started at lower (~55%) and increases more modestly to ~78–82% in the visible region. In the visible range of the solar spectrum, this material reflected substantial amount of light – unlike conventional sensitizing dyes such as N719 and various organic dyes, which are designed to absorb visible light efficiently. As a result, Sr2SiO4:Eu3+ is not suitable for use as a surface coating, since its high reflectivity in the visible spectrum would interfere with light absorption. Instead, it is more effective when integrated within the device architecture, such as in an internal or interfacial layer, where it can selectively absorb UV light without disrupting visible light transmission. While it is not meant to replace traditional dye molecules, it can serve as a complementary sensitizer, extending the device’s absorption capacity into the UV region and aiding in the generation and transport of photo-induced charge carriers.
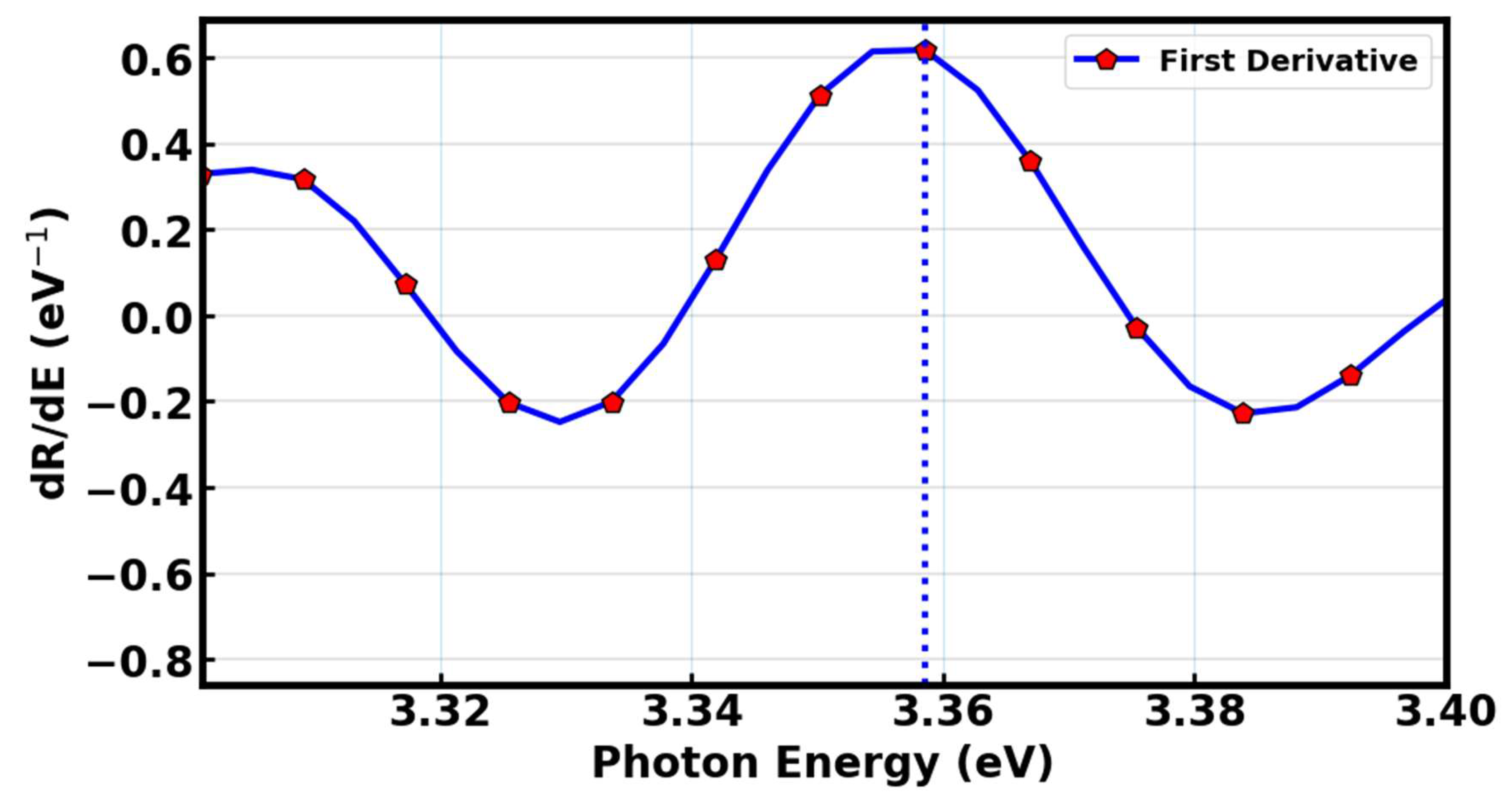
To further study the optical properties of the annealed Sr
1.97SiO
4:0.03Eu
3+ phosphor samples, we have estimated the optical band gap from the first derivative of the diffuse reflectance spectrums as shown in
Figure 11. We have located the absorption edge with respect to photon energy
. The prominent peak observed corresponds to the energy at which photon absorption initiates electronic transitions from the valence band to the conduction band. The x-axis value (photon energy) where this peak intersects indicates the band gap energy. For Sr
2SiO
4:Eu
3+ LDS material, this intersection occurred near 3.36 eV; suggesting direct band gaps that fall within the ultraviolet (UV) portion of the electromagnetic spectrum [
63]
.
In the context of dye-sensitized solar cells (DSSCs), this band gap implies that Sr
2SiO
4:Eu
3+ is not suitable as the main light-absorbing material or as a photo-anode, since it does not significantly absorb visible light—the most intense part of the solar spectrum. However, its strong absorption in the UV range makes it highly suitable as a luminescent downshifting material. As previously discussed alongside the material’s reflectance behaviour, Sr
2SiO
4:Eu
3+ can efficiently capture UV photons and re-emit them in the visible range through the characteristic sharp emission peaks, corresponding to the transition from the excited state
5D
0 to the ground states
7F
j (j =0, 1, 2, 3 and 4) in the 4f
6 configuration of the Eu
3+ ions. This re-emitted visible light can subsequently be absorbed by common sensitizer dyes used in DSSCs, such as N719 (ruthenium-based) and various organic dyes, which operate effectively in the 400–700 nm range [
64,
65,
66]. Thus, these band gaps confirm Sr
2SiO
4:Eu
3+’s potential as a spectral conversion material. While not contributing directly to visible light absorption, it enhances solar cell efficiency indirectly by transforming non-utilized UV light into a form that dye molecules can absorb. Additionally, it offers the added benefit of shielding the device from UV-induced damage, contributing to the longevity and overall performance of the DSSC.
3.6. Photoluminescence Analysis
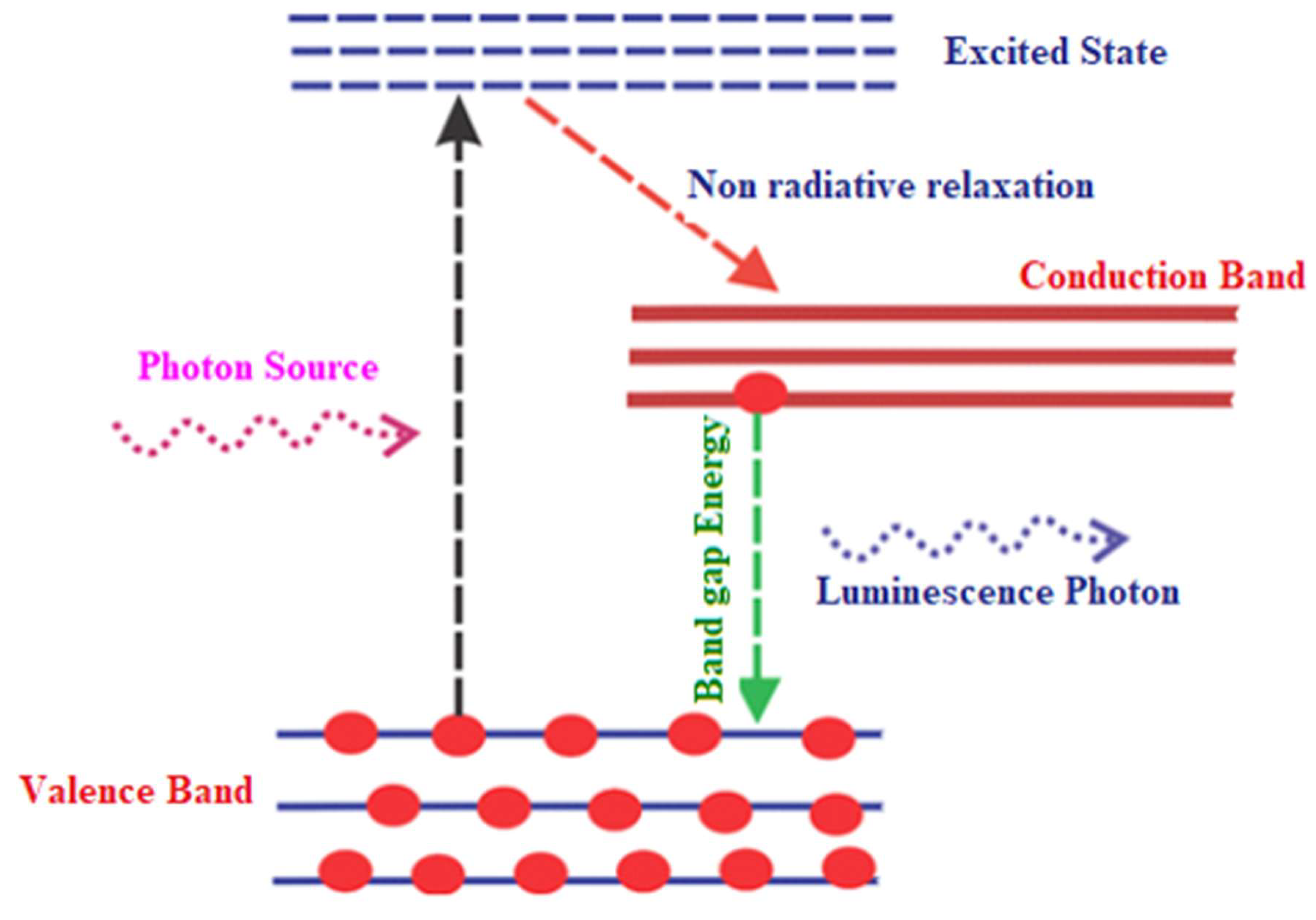
In typical photo-luminescent process, light of suitable energy is directed onto a sample material, where it is absorbed, exciting electrons from valence band to higher energy excited state, a process known as
photo-excitation. When the excited electrons subsequently relax back to ground energy states, they emit energy in a process known as PL photoluminescence [
67]. Therefore, PL is essentially the spontaneous emission of light from a material under optical excitation [
68]. The light emitted can be collected and analyzed spectroscopically to obtain valuable information about the material’s electronic structure, bandgap energy, defect states, and recombination mechanisms. Moreover, the intensity and wavelength distribution of the PL signal provide insights into the purity, crystallinity, and defect density of the material. A material under photo-excitation will have its electrons moved from the valence band to the allowed excited states, creating electron hole pairs called excitons. As these electrons and holes recombine, the system may release energy, which may include emission of light (luminescence/radiative process) or may not (non-radiative process, where the energy is dissipated as heat through phonon interactions), as demonstrated in
Figure 12. The energy of the emitted light i.e., PL relates to the band gap energy between the excited and the equilibrium states. In semiconductor devices, the most common radiative transition occurs between the conduction and the valence band of the material, and their energy difference is known as the band gap. During PL spectroscopy experiment, excitation is provided by a light source with the energy that is larger than the semiconductor material’s band gap. The photo-excited carriers are the electrons and holes, which relax within their respective band edges and recombines by emitting light known as a luminescence photon.
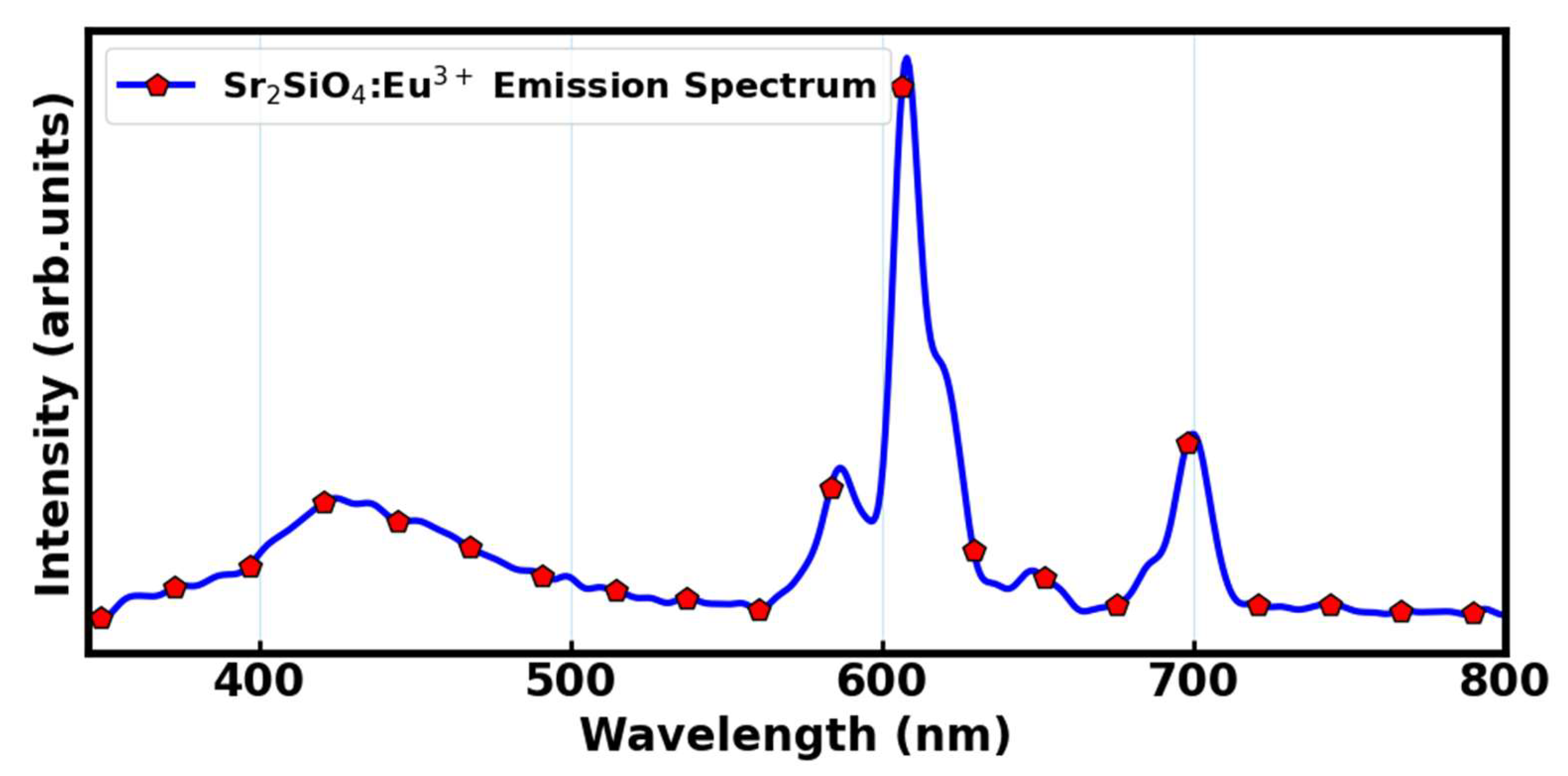
The Sr
2SiO
4:Eu
3+ is a promising material due to its sharp and intense luminescence characteristics 4f – 4f transition. Among rare-earth-doped phosphors, Eu
3+ is particularly attractive for applications that demand tunable emission and thermal stability [
26]. These features make Sr
2SiO
4:Eu
3+ not only suitable for solid-state lighting and display technologies, but also relevant in the context of energy conversion devices, such as dye-sensitized solar cells. From
Figure 13, the PL emission spectrum of the sample spanned in the wavelength range of approximately 350 – 800 nm which illustrates the characteristic luminescence response of the Eu
3+ and are dominated by sharp and intense luminescence, originating from characteristic 4f – 4f transitions, which are parity-forbidden but become allowed due to the asymmetric ligand field around Eu
3+ ions. Several distinct emission bands were observed, predominantly in the UV and near infra-red spectral region. These bands are attributed to the well-known
5D
0 →
7F
J (J = 0–4) transitions of Eu
3+ ions [
69]. Among them, the most intense emission appears in the infrared region around 612–620 nm, which is associated with the electric dipole transition
5D
0 →
7F
2 emanating from the local symmetry of the Eu
3+ sites. This transition is highly sensitive to the local crystal field and is a hallmark of Eu
3+ occupying sites lacking inversion symmetry within the host lattice, with higher intensity indicating a more distorted or non-centrosymmetric environment.
In conventional DSSCs, the performance is limited by the spectral response of the dye molecules, which often absorb inefficiently in the UV region. Phosphors such as Sr
2SiO
4:Eu
3+ can be strategically applied as spectral converters or luminescent down-shifting layers. When integrated into the photo-anode architecture or applied as a top layer, these materials can absorb high-energy UV or blue photons and re-emit them in the visible and near infra-red spectral range, which matches better with the absorption profile of typical dyes such as N719 or organic D-π-A molecules. This re-emission can then be absorbed by the dye, effectively increasing the number of photons available for charge generation and improving the overall photocurrent [
70]
.
Among the various dyes developed for DSSCs, the N719 dye has become a standard due to its broad absorption in the visible range, good chemical stability, and favorable alignment of energy levels for electron injection and regeneration [
71]. One major limitation in DSSC performance arises from the mismatch between the solar spectrum and the dye’s absorption characteristics. In particular, UV photons below 350 nm are poorly utilized by many DSSC dyes, including N719, either due to low absorption or inefficient electron injection. Moreover, prolonged UV exposure can cause photo-degradation of the dye or the electrolyte, limiting the long-term stability of the device. A promising strategy to address these challenges involves the use of luminescent materials such as phosphors that can absorb high-energy UV photons and re-emit them in the visible range where the dye has strong absorption [
17]. This approach, referred to as spectral down-conversion or luminescent down-shifting, has potential not only to increase the number of useful photons incident on the dye but also to shield sensitive components from harmful UV radiation.
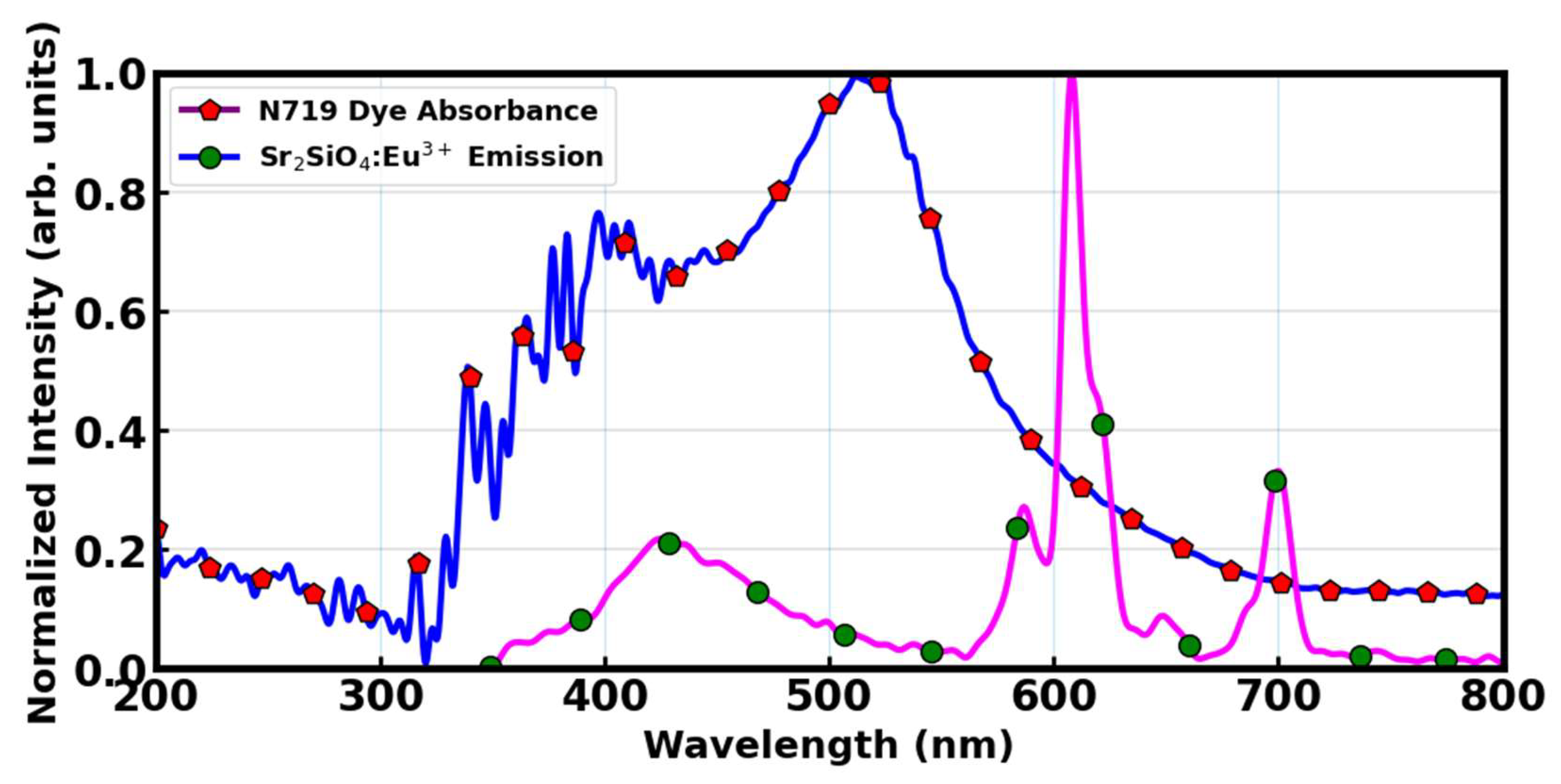
Figure 14 shows that comparing N719 dye absorption and the Sr
2SiO
4:Eu
3+ emission revealed that the N719 dye showed strong absorbance between 350 – 600 nm. While the absorbance significantly decreased beyond 600 nm.
This is particularly relevant because Sr
2SiO
4:Eu
3+ emits strongly at approximately 612 nm, meaning that photons emitted by the phosphor at this wavelength can still be absorbed by the N719 dye. This overlap, although not ideal, is sufficient to propose the use of Sr
2SiO
4:Eu
3+ as a spectral converter in DSSCs utilizing N719 [
72]. When UV photons (e.g., < 350 nm) that are otherwise under-utilized are absorbed by the phosphor, the resultant visible emission can contribute to enhanced light harvesting if those emitted photons fall within the dye’s absorptive window. The degree of spectral overlap between the phosphor’s emission and the dye’s absorption is a key factor in determining the effectiveness of the down-shifting process [
72,
73]. FRET spectral integral between the emission spectrum of Sr
2SiO
4:Eu
3+ and the absorption spectrum of the N719 dye molecules showed that strong overlap was achieved between the phosphor material and the dye molecules, and an estimated spectral overlap integral J(
λ) value of 5.91
x 10
10 M
-1cm
-1nm
4 was realized from the material. This value is sufficient enough, indicating an efficient energy transfer process could occur [
48,
74]. The overlap strength realized is appropriate as it depicted proper correspondence between the absorption spectrum of the N719 dye acceptor and the emission spectrum of the Sr
2SiO
4:Eu
3+ donor.
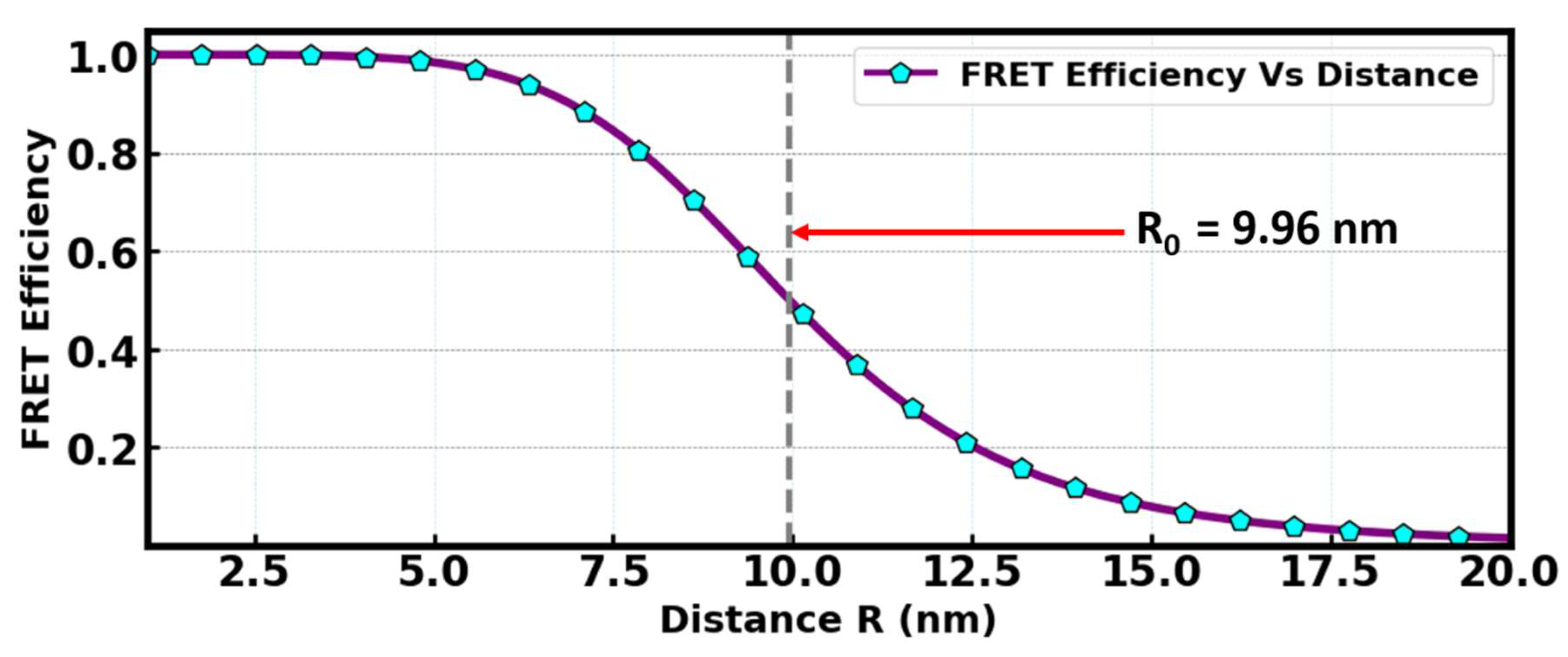
FRET efficiency as a function of distance between the donor and the acceptor molecules is depicted as shown in
Figure 15 below. It can be deduced that the FRET efficiency decreases as the distance between the donor and the acceptor increases, which demonstrates that FRET is always effective at a typically close distance. R
0 = 9.96 nm indicated the participation of FRET mechanisms, which fall within the range 1–10 nm. Therefore, at such distances, the energy transfer between the donor, Sr
2SiO
4:Eu
3+, and the acceptor, the N719 dye, would be 50% efficient. Estimation of R
0 was computed from the relationship governing the Förster distance, the quantum yield of the donor in the absence of the acceptor, the spectra overlap between the donor and acceptor molecules, the refractive index of the medium containing the donor and the acceptor molecules, and the relative orientation of the transition dipoles of the donor and acceptor.
Generally, the development of an efficient energy transfer system is strongly dependent on the Förster distance between the donor and acceptor molecules as well as the strength of the spectra overlap integral between the donor and acceptor. A system with a Förster distance between 1 – 10 nm and a strong overlap between the donor and the acceptor molecules would produce efficient energy transfer [
14,
75]. This mechanism in DSSC would facilitate generation of charge carriers that would promote the power output and the overall performance of the device.
3.7. Energy Level Diagram
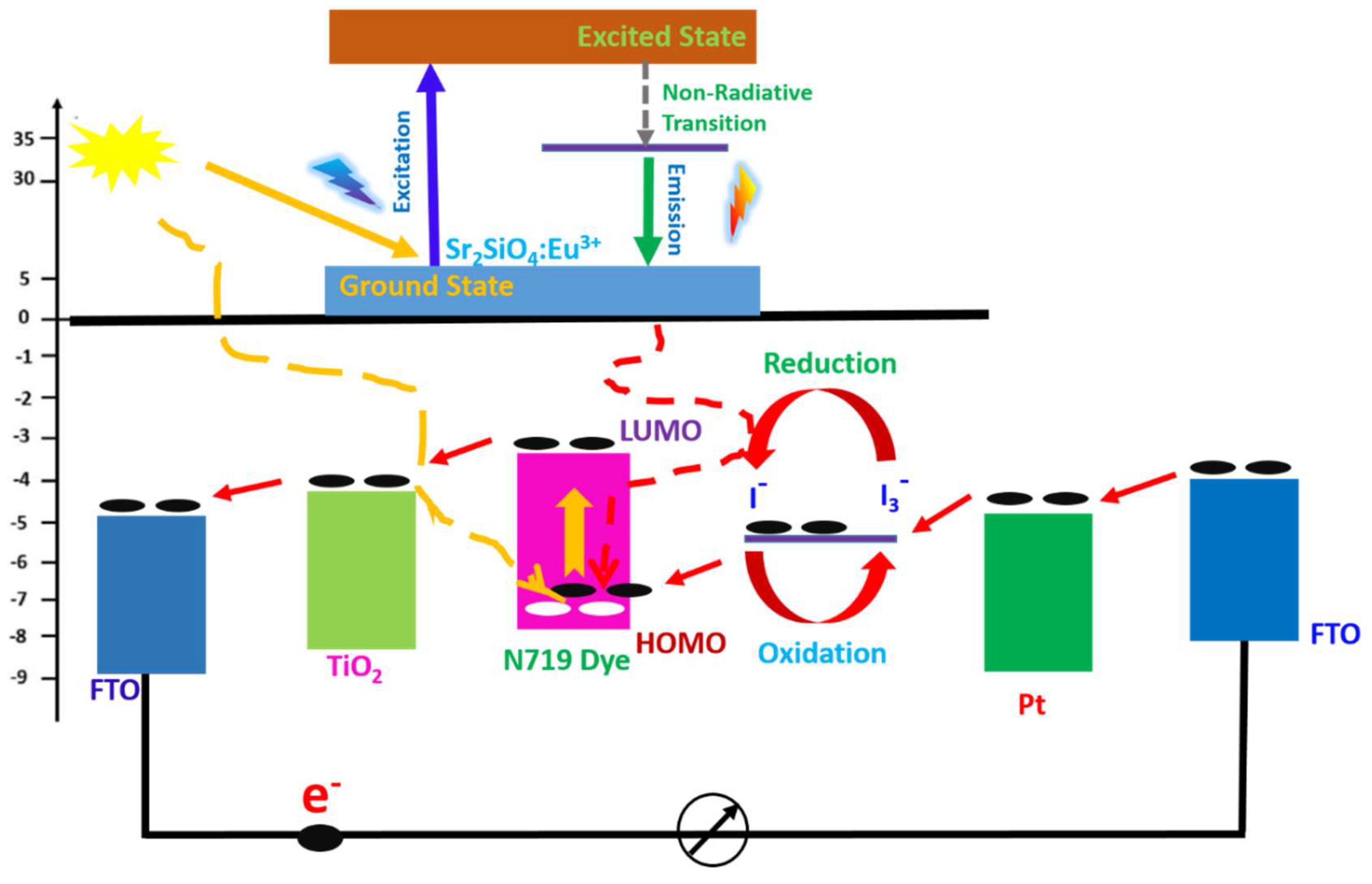
The diagram in
Figure 16 illustrates how Sr
2SiO
4:Eu
3+ is integrated into a dye-sensitized solar cell (DSSC) to enhance light harvesting and charge transfer. At the top, the Eu
3+ luminescent centers show a typical photo-physical process: incident high-energy photons excite electrons from the ground state to an excited state (blue upward arrow). After excitation, part of the energy is lost through a non-radiative transition, followed by radiative emission (green downward arrow). This emission corresponds to visible light, meaning the phosphor acts as a down-shifting layer, converting higher-energy UV photons into wavelengths better matched to the dye absorption. The emitted light from Sr
2SiO
4:Eu
3+ is then reabsorbed by the sensitizer, here N719 dye. In the diagram, the dye’s HOMO–LUMO levels are clearly positioned between the conduction band of TiO
2 and the redox electrolyte. Upon photon absorption (either direct sunlight or re-emitted light from Eu
3+), electrons in the dye are excited from the HOMO to the LUMO. Because the LUMO lies above the TiO
2 conduction band edge, the excited electrons are efficiently injected into TiO
2, which is essential for photocurrent generation [
76]. Once injected, electrons percolate through the TiO
2 network and are collected by the transparent conducting electrode, typically FTO. The energy alignment shown indicates that TiO
2 has a slightly higher (less negative) conduction band than FTO, allowing downhill electron transport. The diagram’s arrows emphasize this directional flow toward the external circuit, minimizing recombination losses when the interfaces are well optimized. Meanwhile, the oxidized dye is regenerated by the electrolyte, commonly the, I
−/ I
3− redox couple. The redox potential is positioned above the dye HOMO, enabling electron donation to the dye. This regeneration step is critical to sustain continuous operation. The cyclic red arrows in the diagram represent the oxidation of iodide (I
− → I
3−) at the dye interface and its subsequent reduction at the counter electrode. At the counter electrode, typically coated with Platinum, the tri-iodide (I
3−) is reduced back to iodide by electrons returning from the external circuit. This closes the electrochemical loop. The energy level of Pt facilitates fast charge transfer kinetics, reducing over-potential and improving fill factor. Overall, the inclusion of Sr
2SiO
4:Eu
3+ introduces a spectral conversion mechanism that broadens the usable portion of the solar spectrum. By converting UV photons (which are poorly absorbed by N719 dye) into visible emission, it enhances photon utilization. The diagram also highlights that efficient DSSC operation depends on precise energy level alignment: dye LUMO above TiO
2 conduction band for injection, dye HOMO below electrolyte redox potential for regeneration, and seamless electron transport through FTO and Pt.
3.8. Photovoltaic Performance Analysis
Measurement of the current density (J) – voltage (V) characteristics of solar cells is vital to determine the overall performance of the cells. The power – voltage characteristics can also be determined, as power is proportional to the product of current and the voltage. From the J – V and P – V characteristics, a comprehensive set of important photovoltaic parameters can be determined, providing critical insight into the electrical behaviour and overall energy-conversion capability of the device [
77,
78,
79]. These curves enable the determination of key performance indicators, including the short-circuit current density (Jsc), open-circuit voltage (Voc), maximum power output (Pmax), fill factor (FF), and power conversion efficiency (PCE), which collectively reflect the charge-generation efficiency, recombination dynamics, and operational stability of the solar cell.
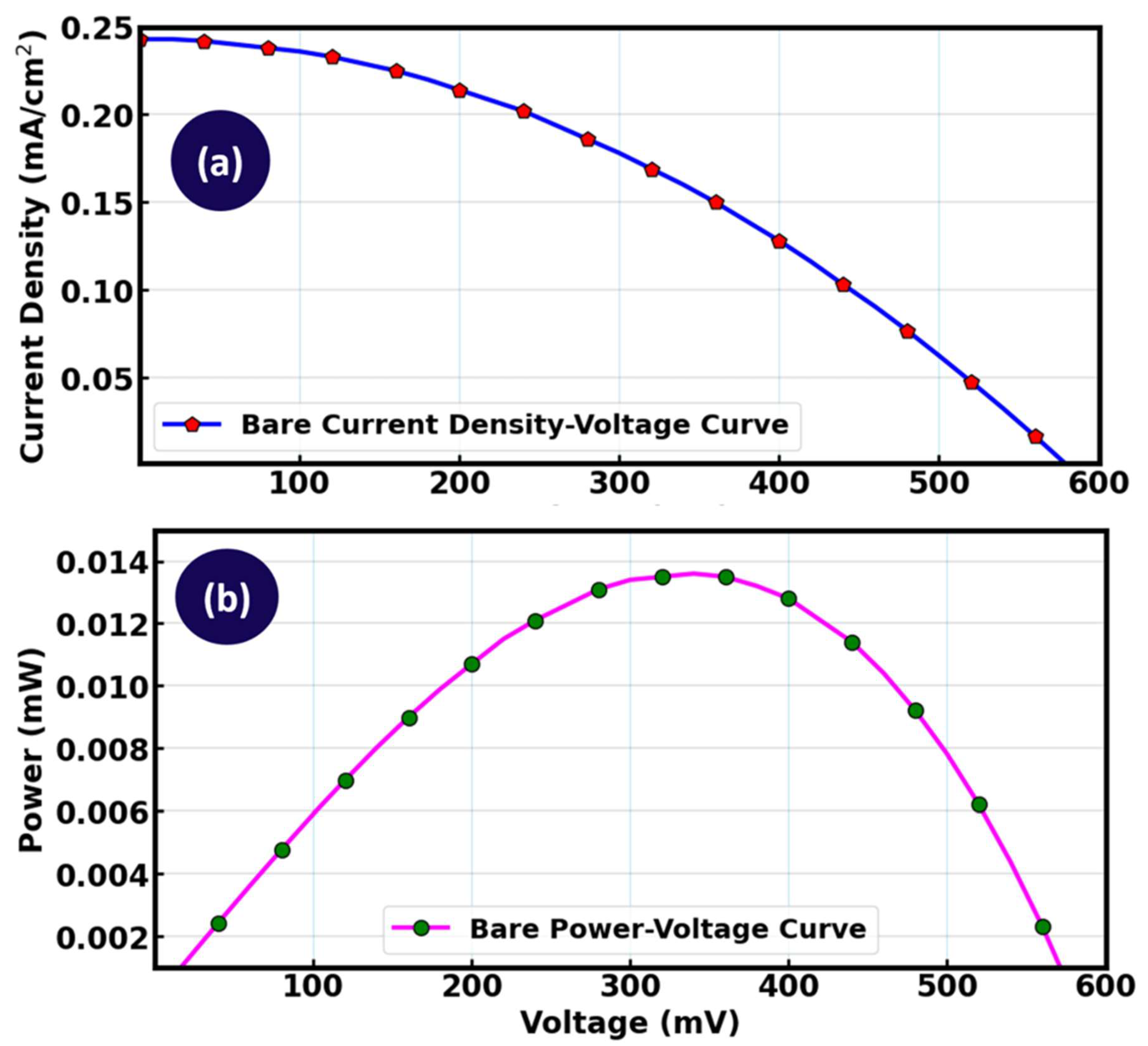
Figure 17(a) illustrates the current density – voltage (J – V), characteristics of a bare dye-sensitized solar cell (DSSC) without the incorporation of any luminescent material, while
Figure 17(b) shows the corresponding power–voltage (P–V) characteristics. As the applied voltage increased from 0 to approximately 600 mV, the current density decreased steadily from approximately 0.25 mA/cm
2 to nearly zero, demonstrating the expected non-linear relationship between current and voltage in a photovoltaic device [
80]. The high current density at low voltage corresponds to the short-circuit current-density region, whereas the decreased current density at higher voltage reflected the approach toward the open-circuit voltage (Voc). On the other hand, the power output initially increased with voltage, reached a maximum around 300-350 mV, and then declined sharply as the device approached Voc. This bell-shaped curve is characteristic of DSSC performance, with the peak representing the maximum power point (MPP), the operating condition under which the device produces its highest electrical power output.
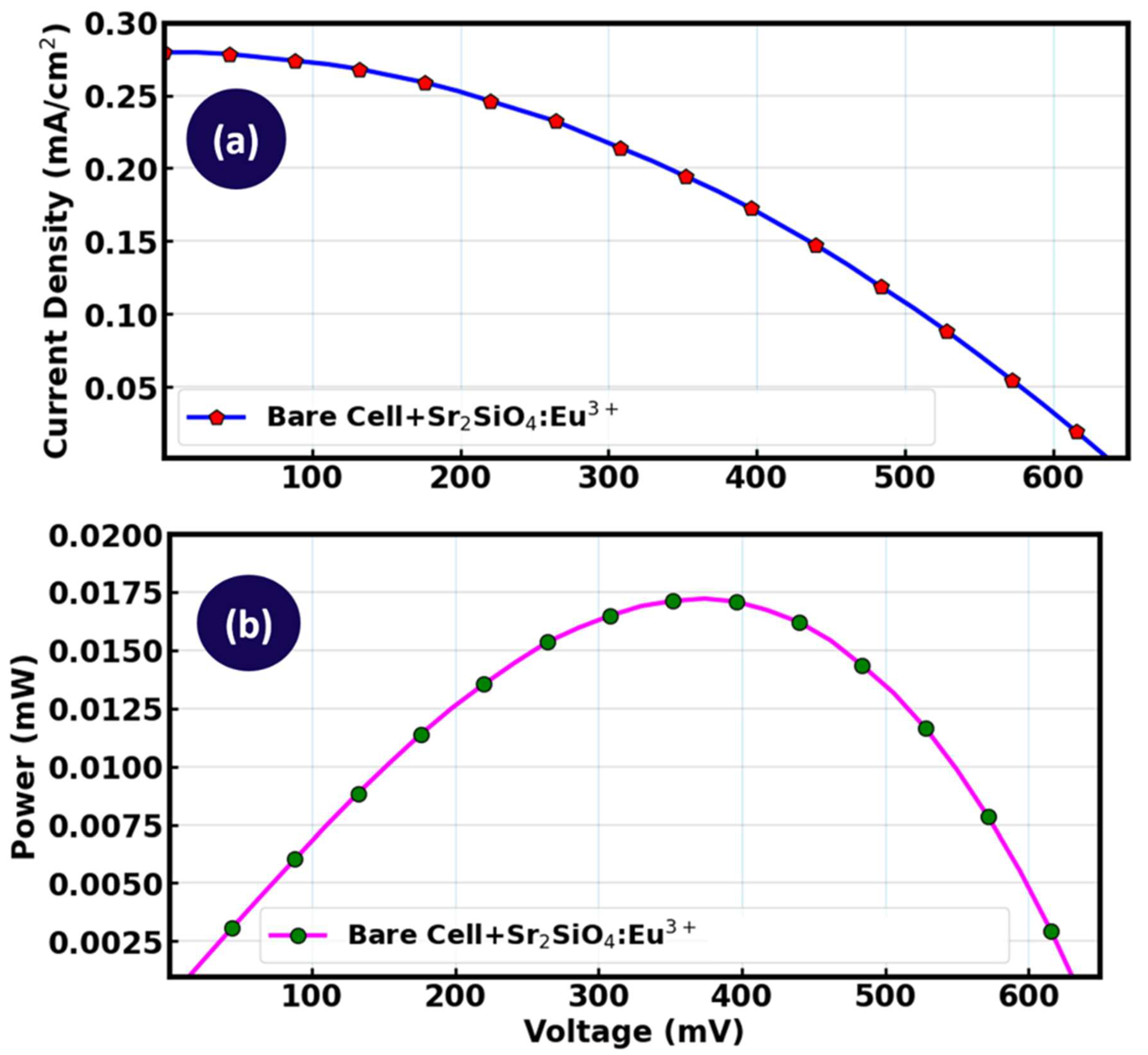
The integration of Sr
2SiO
4:Eu
3+ phosphor, annealed at 550 °C into the DSSC structure has led to a clear enhancement in device performance when compared to the bare cell. The current density–voltage (J – V) and power–voltage (P – V) characteristics are shown in
Figure 18 (a & b). Similarly to the bare, the J-V curve exhibits a progressively decreasing current density as the applied voltage approaches the open-circuit region, which is still expected of dye-sensitized solar cells [
81]. Compared with the bare cell, the short-circuit current density (Jsc) increased from 0.243 mA/cm
2 in the bare DSSC to 0.279 mA/cm
2 in the phosphor-modified Sr
2SiO
4:Eu
3+ device, denoting an increase of around 14.8%. This improvement indicates that the phosphor material enhances photon harvesting and light-conversion efficiency, most likely through its down-conversion or spectral-conversion capability. By converting high-energy UV photons into visible wavelengths that align with the N719 dye’s absorption range, more electrons are injected into the TiO
2 conduction band, resulting in a higher photocurrent. The curve shape remains smooth and consistent, suggesting that the phosphor layer does not introduce significant recombination pathways or series resistance losses within the cell.
Figure 18 (b) shows the corresponding power–voltage (P–V) characteristics for the LDS incorporated device. The power output increases with voltage until reaching a maximum at the device’s maximum power point (MPP), located around 350–400 mV with a peak power of approximately 0.017–0.018 mW. This peak value is higher than that observed for the bare DSSC, further confirming that the incorporation of Sr
2SiO
4:Eu
3+ LDS material contributes positively to cell performance. The improved MPP signifies more efficient harvesting and utilization of the incident solar irradiance. The nearly symmetrical rise and fall of the P–V curve reflected stable and predictable device behaviour, indicating that the phosphor material integrates well with the photo-anode without creating interfacial instability or optical loss. Collectively, these results demonstrated that adding Sr
2SiO
4:Eu
3+ LDS material enhanced both photocurrent generation and power output, confirming its effectiveness as a luminescent down-shifting material for boosting DSSC efficiency.”
The summary of the photovoltaic performance of the bare and the phosphor enhanced devices are depicted in
Table 4. The bare DSSC has the following photovoltaic performance parameters: J
sc = 0.243 mA/cm
2, V
oc = 580 mV, P
max = 0.0136 mW, FF = 38.60% and PCE = 1.09%. The DSSC with Sr
2SiO
4:Eu
3+ LDS material has the following performance parameters: J
sc = 0.279 mA/cm
2, V
oc = 638 mV, P
max = 0.0172 mW, FF = 38.65%, and PCE = 1.38%. Compared to the bare DSSC, the cell with Sr
2SiO
4:Eu
3+ LDS material had an enhancement of 14.8% in the short circuit current density (J
sc), from 0.243 mA/cm
2 to 0.279 mA/cm
2. The open circuit voltage (V
oc) yielded an improvement of 10% from 580 mV to 638 mV. Maximum power output (P
max) produced a boost of 26.5% from 0.0136 mW to 0.0172 mW. Fill factor yielded an improvement of 0.1% from 38.60% to 38.65%. The efficiency improvement was at 26.6% from 1.09% to 1.38%.
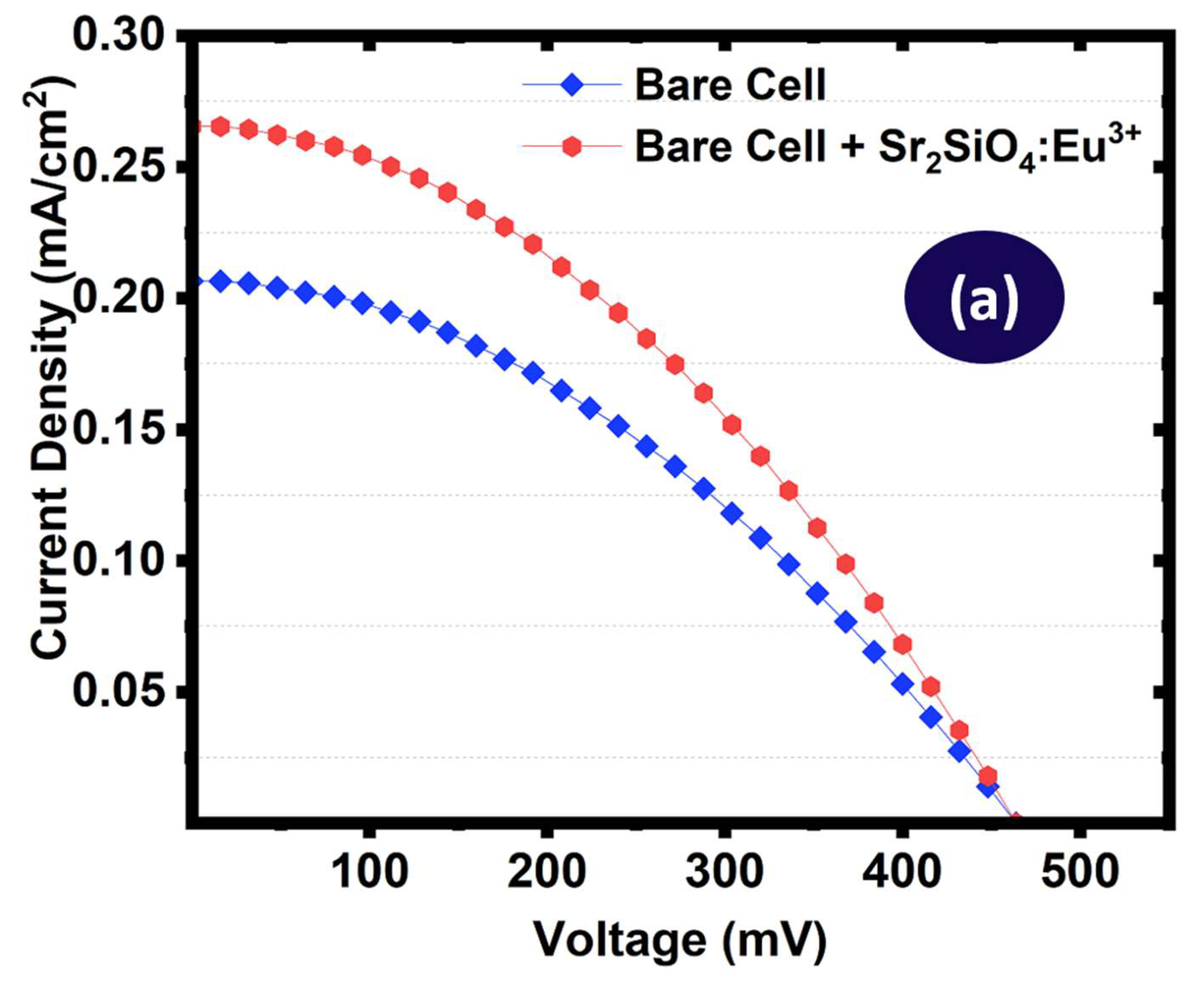
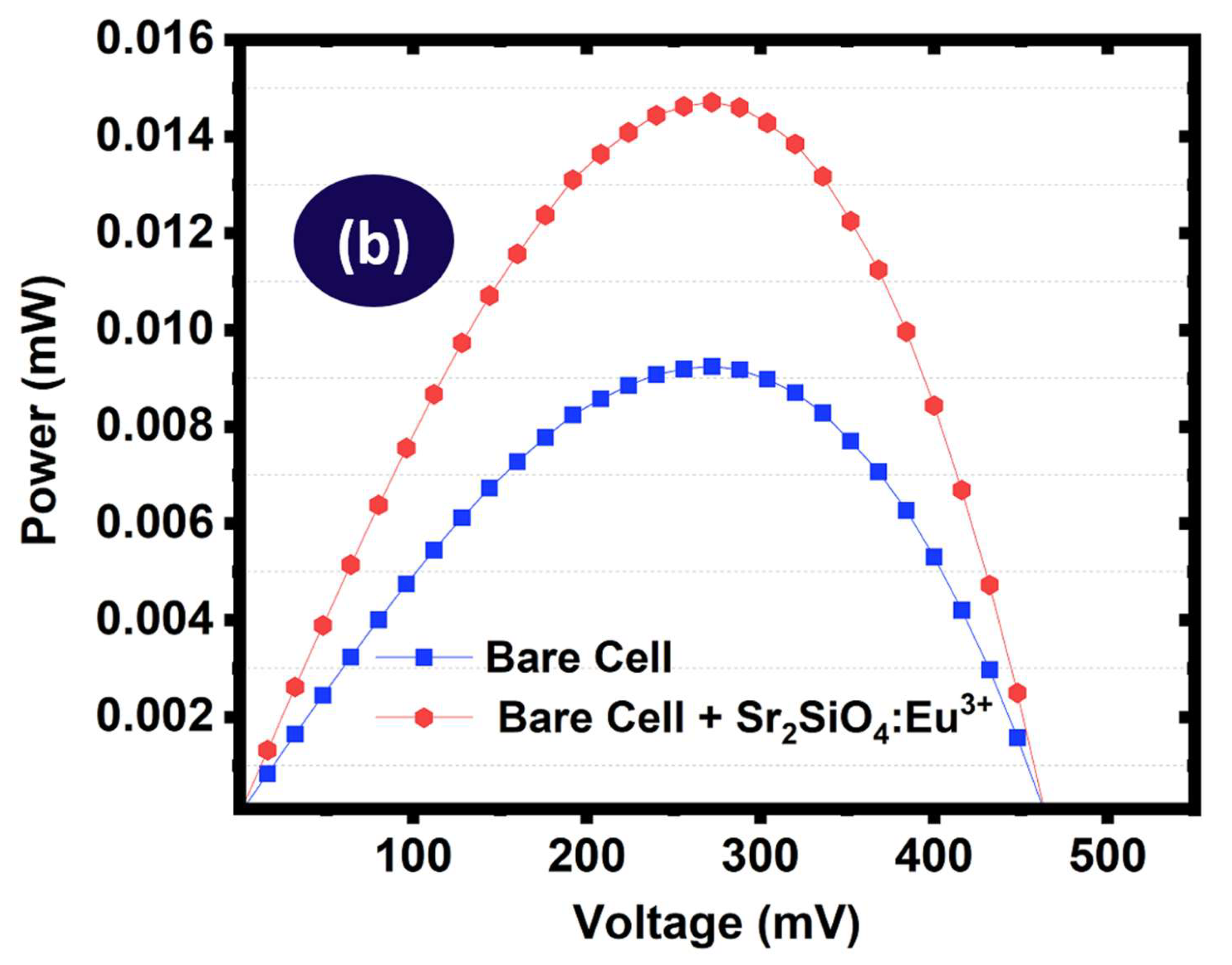
To enable direct comparison, the current density – voltage curves and the power – voltage curves of the bare and phosphor modified cells were plotted on the same axis as shown in
Figure 19(a & b). The baseline device exhibited relatively low photovoltaic performance, with a short-circuit current density of 0.243 mA cm
−2 and maximum power output of 0.0136 mW, leading to a power conversion efficiency of 1.09%. The relatively low values likely reflected inherent limitations in light harvesting and spectral mismatch between the incident solar spectrum and the absorption range of the dye sensitizers. Upon incorporation of Sr
2SiO
4:Eu
3+ phosphor annealed at 550 °C, improvements were observed across all photovoltaic parameters. The short-circuit current density increased from 0.243 mA cm
−2 to 0.279 mA cm
−2. The maximum power output improved from 0.0136 mW to 0.0172 mW (26.5% increase), resulting in a corresponding increase in power conversion efficiency from 1.09% to 1.38%, representing a 26.6% improvement relative to the bare device.”
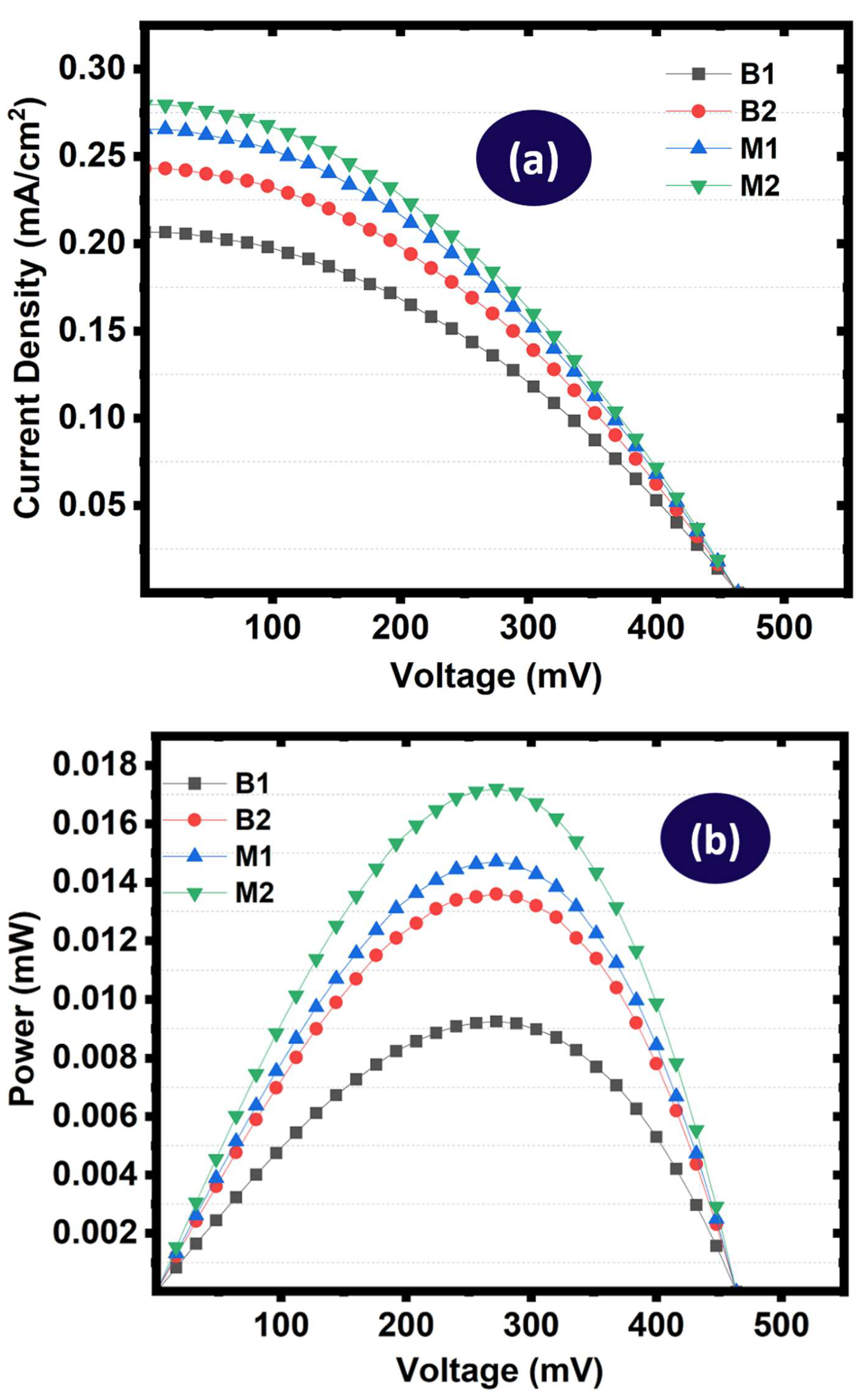
To evaluate the reliability and reproducibility of the fabricated dye-sensitized solar cells (DSSCs), a statistical analysis was conducted using the photovoltaic parameters obtained from reproduced measurements of the J–V and P–V curves. The key parameters considered in this analysis were the short-circuit current density (J
sc), open-circuit voltage (V
oc), maximum output power (P
max), and power conversion efficiency (PCE). These parameters were extracted from the J-V and P-V characteristics measurements for two bare DSSCs and two modified DSSCs incorporating Sr
2SiO
4:Eu
3+ (SSO: Eu
3+) phosphor, which are depicted in
Figure 20 (a & b). The purpose of this analysis was to assess the reproducibility of device performance and to determine how the incorporation of the luminescent material influences the overall photovoltaic behavior.
Table 5.
Summary of reproduced photovoltaic parameters.
Table 5.
Summary of reproduced photovoltaic parameters.
| Device Type |
Device ID |
Jsc (mA/cm2) |
Voc (V) |
Pmax (mW) |
PCE (%) |
| Bare Cell - 1 |
B1 |
0.207 |
0.464 |
0.0120 |
0.96 |
| Bare Cell – 2 |
B2 |
0.243 |
0.580 |
0.0136 |
1.09 |
| Modified (SSO: Eu3+) - 1 |
M1 |
0.266 |
0.574 |
0.0153 |
1.22 |
| Modified (SSO: Eu3+) - 2 |
M2 |
0.279 |
0.638 |
0.0172 |
1.38 |
To quantitatively evaluate the reproducibility of the photovoltaic parameters, the mean values and standard deviations (SD) [
82,
83] were calculated for each device category and depicted in
Table 6. The bare DSSCs exhibited an average J
sc of 0.225 ± 0.026 mA cm
−2 and an average V
oc of 0.522 ± 0.082 V. The mean P
max and PCE values were found to be 0.013 ± 0.001 mW and 1.02 ± 0.09%, respectively. The relatively larger uncertainty observed in V
oc for the bare devices suggests that slight variations in fabrication or interface quality may influence the open-circuit voltage. For the DSSCs modified with SSO:Eu
3+, the mean J
sc increased to 0.272 ± 0.009 mA cm
−2, indicating improved and more consistent photocurrent generation. The average V
oc increased to 0.606 ± 0.045 V, while the mean P
max rose to 0.016 ± 0.001 mW. The corresponding average PCE reached 1.30 ± 0.11%. The smaller uncertainty observed for J
sc compared to the bare cells suggested improved uniformity in photocurrent generation among the modified devices.”
“In addition to the mean and standard deviation, the coefficient of variation (CV) [
84,
85] was calculated to assess the relative variability in device efficiency as shown in
Table 7. The CV is an important statistical indicator that expresses the ratio of standard deviation to the mean, most often as a percentage, providing insight into the reproducibility of the measurements. The bare DSSCs exhibited a CV of 8.8% for PCE, indicating moderate variability between repeated devices. After modification with SSO:Eu
3+, the CV decreased slightly to 8.5%, suggesting improved reproducibility. The reduction in variability can be attributed to improved optical and structural properties introduced by the SSO:Eu
3+ phosphor. The luminescent material may act as a light-scattering medium and spectral converter, increasing the effective absorption of incident photons by the dye molecules. Additionally, improved film morphology and reduced defect density may contribute to more efficient charge transport and reduced recombination losses. These factors collectively enhance both the efficiency and the reproducibility of the DSSCs.”