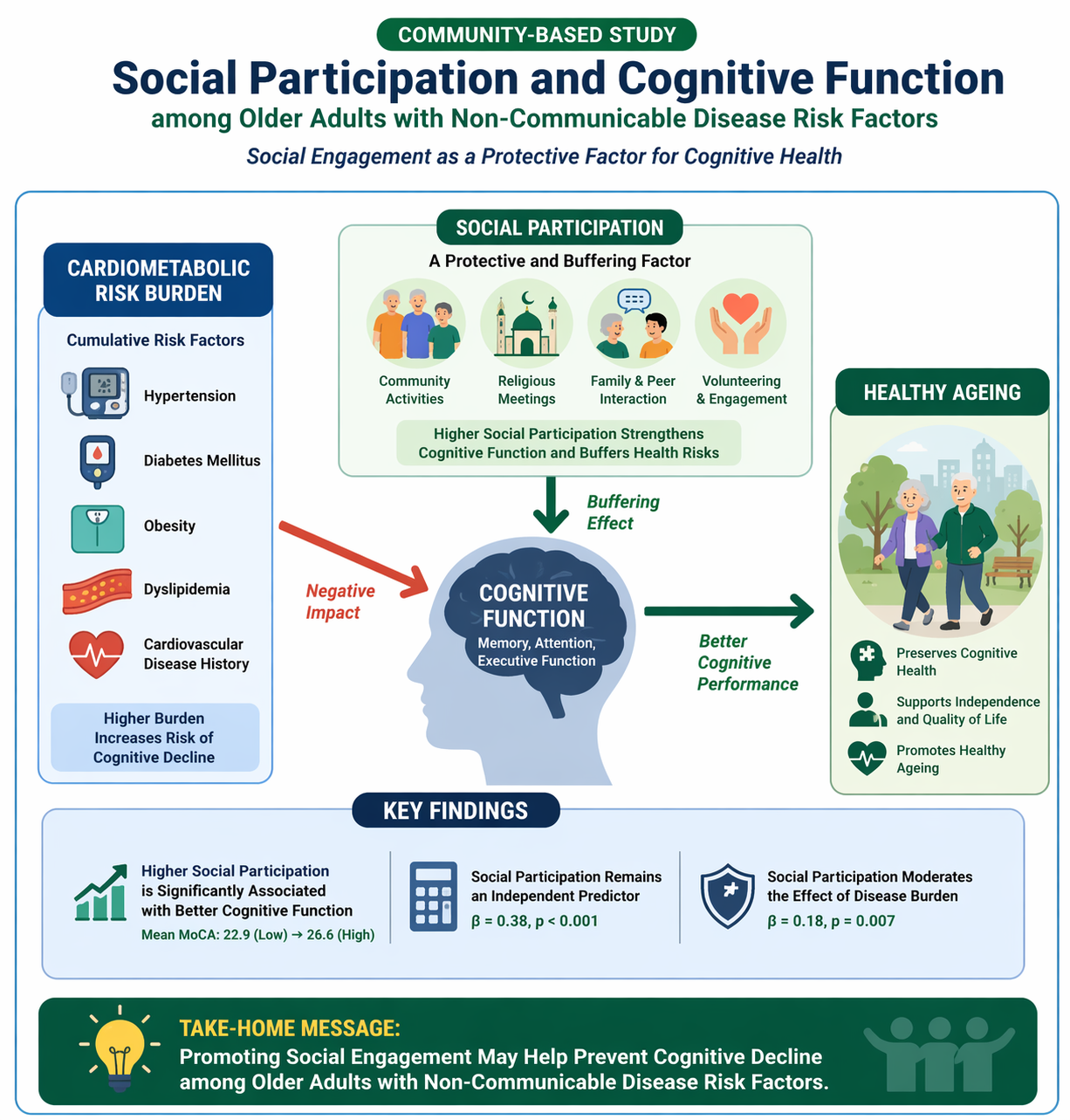
Principal Findings
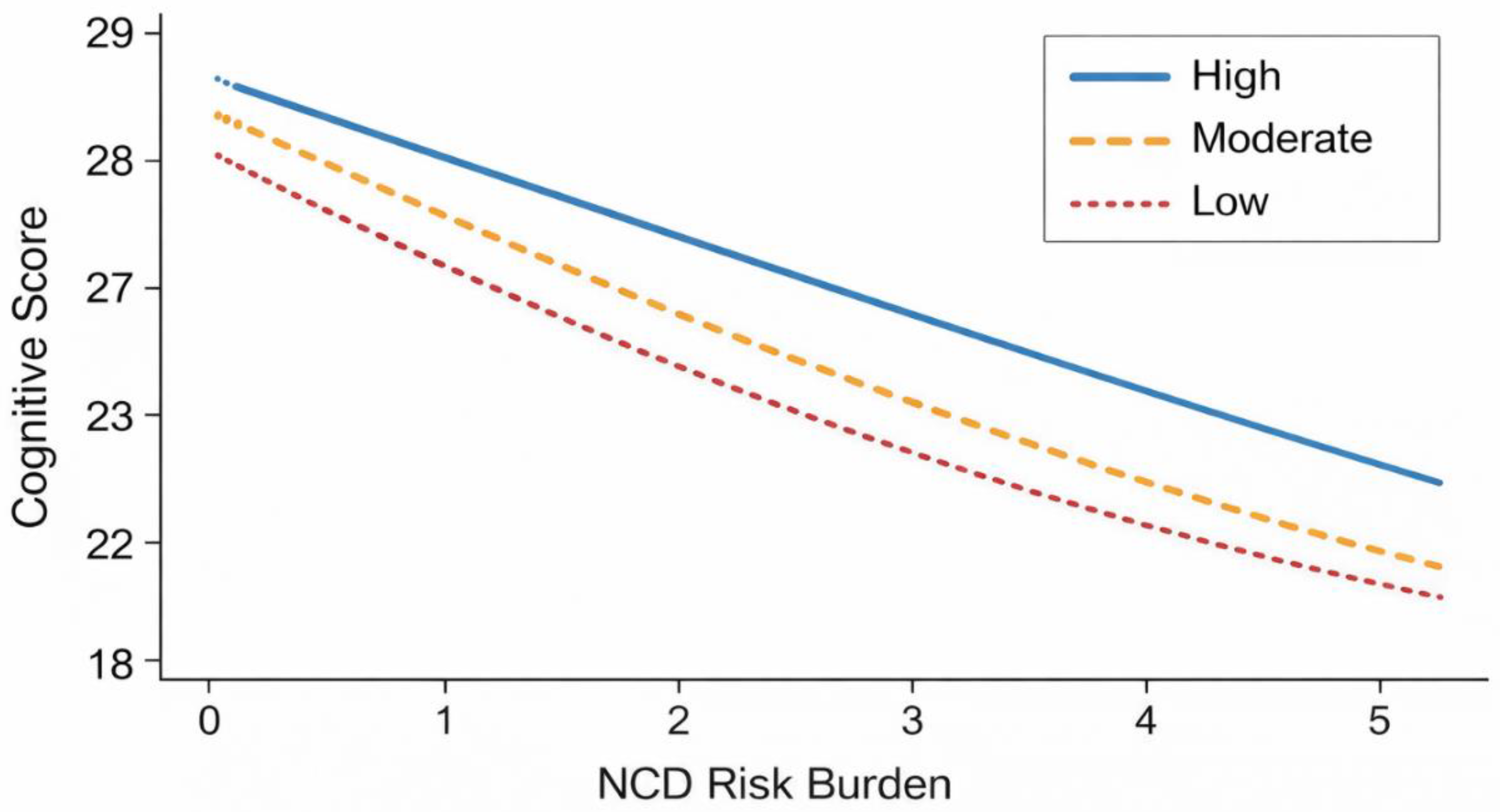
This study examined the association between social participation and cognitive function among 214 community-dwelling older adults with non-communicable disease risk factors. A clear gradient was observed across levels of social participation. Participants with low social participation demonstrated a mean cognitive score of 22.9 ± 3.4, compared with 24.7 ± 2.8 among those with moderate participation and 26.6 ± 2.5 among those with high participation. This difference of 3.7 points between low and high engagement groups suggests clinically meaningful variation in cognitive performance. Multivariate regression analysis confirmed that social participation remained independently associated with cognitive function after adjusting for demographic and behavioral covariates (β = 0.38, p < 0.001). In contrast, non-communicable disease risk burden was negatively associated with cognition (β = −0.41, p = 0.001). Furthermore, moderation analysis demonstrated a significant interaction effect (β = 0.18, p = 0.007), indicating that higher social participation attenuated the negative association between cardiometabolic risk and cognitive function. These findings support the hypothesis that social engagement may buffer biological vulnerability in ageing populations.
Comparison with Previous Studies
The present findings are consistent with longitudinal epidemiological evidence demonstrating that social engagement is associated with slower cognitive decline trajectories [
1,
9]. Previous multidomain lifestyle studies have shown that behavioral factors, including social participation, physical activity, and cognitive stimulation, contribute to preserved intrinsic capacity in older adults [
4,
5]. Additionally, research examining cardiometabolic multimorbidity has reported accelerated cognitive decline among individuals with multiple non-communicable disease risk factors [
6]. The current study extends this evidence by demonstrating a moderation effect, indicating that social participation not only independently predicts cognitive function but also modifies the impact of cardiometabolic burden. This interaction supports integrative models suggesting that biological and social determinants jointly influence cognitive ageing [
10].
Mechanisms Linking Social Participation and Cognitive Function
Several mechanisms may explain the observed association. First, social participation may enhance cognitive reserve through sustained cognitive stimulation, including communication, problem solving, and decision making. Second, social engagement may reduce depressive symptoms, which were associated with lower cognitive scores in the present study. Participants with depressive symptoms showed a mean cognitive score of 23.1 compared with 25.3 among those without depression. Third, socially active individuals may engage in healthier behaviors, including increased physical activity and better disease management. Fourth, social participation may reduce chronic stress and inflammation, mechanisms implicated in vascular cognitive impairment. These multidimensional pathways align with previous research highlighting psychosocial determinants of cognitive ageing [
7,
8].
Public Health Implications
From a public health perspective, the findings identify social participation as a modifiable determinant of cognitive health. The observed difference of 3.7 points in cognitive scores between low and high participation groups suggests meaningful functional implications. Identifying older adults with both cardiometabolic risk and low social participation may help prioritize high-risk populations. Integrating social participation screening into primary care services and community-based programmes may therefore support multidomain approaches to cognitive decline prevention. These implications align with global healthy ageing frameworks emphasizing social determinants of functional ability and intrinsic capacity [
1,
10].
Strengths
This study has several strengths. First, it focused on a biologically vulnerable population of older adults with non-communicable disease risk factors. Second, the inclusion of moderation analysis allowed evaluation of interaction effects between social and biological determinants. Third, multiple covariates, including depressive symptoms, education, and physical activity, were controlled. Fourth, the regression model explained 42% of variance in cognitive function, indicating substantial explanatory power. Finally, community-based recruitment enhances ecological validity.
Limitations
Several limitations should be considered. First, the cross-sectional design precludes causal inference, and reverse causality cannot be excluded. Second, consecutive sampling may introduce selection bias, as participants attending community health activities may be more socially active than the general older population. Third, generalizability may be limited because participants were recruited from specific community-based settings, which may not represent institutionalized or rural populations. Fourth, non-communicable disease risk factors were partly based on self-reported medical history, which may introduce misclassification bias. Fifth, several variables, including social participation and depressive symptoms, were obtained through interviews, raising the possibility of common-method bias. Sixth, residual confounding from unmeasured variables such as nutrition or socioeconomic status cannot be excluded. Finally, the absence of longitudinal follow-up limits evaluation of cognitive trajectories over time. Future longitudinal studies and intervention trials are required to confirm causal relationships and assess long-term effects of structured social participation programs [
9].
Generalizability
Despite these limitations, the findings may be generalizable to community-dwelling older adults with cardiometabolic risk profiles, particularly in similar primary care and community health settings. The consistency of observed associations with previous international studies suggests that the relationship between social participation and cognitive function may extend across diverse populations, although contextual and cultural differences should be considered.
Policy and Research Implications
The findings suggest that promoting social participation may enhance cognitive resilience among older adults with cardiometabolic risk. Community health centers and local organizations could implement structured group activities, peer support programmes, and social engagement initiatives. Screening for social isolation during routine health assessments may help identify individuals at risk. At the policy level, age-friendly environments that promote social interaction may support healthy ageing strategies. These recommendations align with global public health approaches emphasizing social determinants of cognitive health [
10,
11].