Submitted:
24 April 2026
Posted:
27 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
3. Results
3.1. AL
3.1.1. Exercise
3.1.2. HF-Focussed Interventions
3.1.3. Nutrition
3.1.4. ASCT-Related Interventions
3.1.5. HF-Unrelated Fatigue
3.1.6. Renal Involvement
3.1.7. Neuropathy
3.1.8. Sarcopenia
3.2. MM
3.2.1. Exercise
3.2.2. Bone Disease Interventions
3.2.3. Nutrition
3.2.4. ASCT-Related Interventions
3.2.5. Fatigue
3.2.6. Renal Impairment
3.2.7. Neuropathy
3.2.8. Sarcopenia
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- World Physiotherapy Policy statement: Description of physiotherapy. World Physiotherapy: London, UK, 2023; Available online: https://world.physio/policy/ps-descriptionPT.
- Kersten, P.; McPherson, K.; Lattimer, V.; George, S.; Breton, A.; Ellis, B. Physiotherapy extended scope of practice – who is doing what and why? Physiotherapy 2007, 93, 235–242. [Google Scholar] [CrossRef]
- Morris, J.; Grimmer, K.; Gilmore, L.; Perera, C.; Waddington, G.; Kyle, G.; Ashman, B.; Murphy, K. Principles to guide sustainable implementation of extended-scope-of-practice physiotherapy workforce redesign initiatives in Australia: stakeholder perspectives, barriers, supports, and incentives. J. Multidiscip. Healthc. 2014, 7, 249–258. [Google Scholar] [CrossRef]
- McBeath, B.; Chapdelaine, S.; Brabant, L.; Desmeules, F.; Cherrier, S.; Lévesque, M.C. Advancing the practice of cultural safety in physiotherapy: a scoping review. Disability and Rehabilitation 2025. Online ahead of print. [Google Scholar] [CrossRef]
- Demont, A.; Daudier, O.; Tourjansky, Y. Exploring goals, roles, and scopes of advanced practice physiotherapy in French primary and secondary specialized care settings: a qualitative multi-step study. Physiother. Theory Pract. 2025. Online ahead of print. [Google Scholar] [CrossRef]
- Agger, N.; Nørgaard Madsen, M.; Rud Budtz, C.; Desmeules, F.; Høyrup Christiansen, D. Mapping the integration of advanced practice physiotherapists in Danish hospitals. Physiotherapy 2026, 130, 101839. [Google Scholar] [CrossRef]
- Sturm, A.; Burillo Lafuente, M.; Silva Guerrero, A.V.; Roth Psy, R. The diversity and contextuality of ethical situations encountered by physiotherapists in the South America region of World physiotherapy, Spain and Portugal – a cross-cultural, qualitative analysis. Physiother. Theory Pract. 2025. Online ahead of print. [Google Scholar] [CrossRef]
- Hayes, C.; Conneely, M.; McNamara, R.; Quinn, R.; French, H.; Desmeules, F.; Kilcawley, R.; McCreesh, K.; Galvin, R. What is the role of advanced physiotherapy practice for adults in urgent care and emergency department settings? A scoping review protocol. HRB Open. Res. 2025, 8, 116. [Google Scholar] [CrossRef]
- Hamazaki, N.; Ogura, K. Implementation and Challenges of Early-Phase Physiotherapy During Cardiovascular Intensive Care. Circ. Rep. 2026, 8, 21–26. [Google Scholar] [CrossRef]
- Forslund, L.; Arntzen, C.; Nikolaisen, M.; Gramstad, A.; Eliassen, M. Contextualised physiotherapy: connecting professional practice to the achievement of the person-centred goals of rehabilitation. Eur. J. Physiother. 2026, 28, 5–18. [Google Scholar] [CrossRef]
- Roos, R. Current state of evidence-based practice in clinical physiotherapy. S. Afr. J. Physiother. 2025, 81, a2139. [Google Scholar] [CrossRef]
- Souto-Miranda, S.; Brazete Cruz, E.; Pires, D.; Ribeiro, F.; Cordeiro, N.; acome, C. Priorities in physical therapy research: A scoping review. Braz. J. Phys. Ther. 2024, 28, 101135. [Google Scholar] [CrossRef]
- Michou, I.; Fouras, A.; Chrysanthakopoulou, D.; Theodoritsi, M.; Mariettou, S.; Stellatou, S.; Koutsojannis, C. AI-Powered Physiotherapy: Evaluating LLMs Against Students in Clinical Rehabilitation Scenarios. Appl. Sci. 2026, 16, 1165. [Google Scholar] [CrossRef]
- Melnick, S. Physiotherapy and clinical approaches- an overview. Eur. J. Phys. Educ. Sport Sci. 2015, 1, 27–37. [Google Scholar]
- Ríos-Tamayo, R. Epidemiology of systemic light-chain (AL) amyloidosis. Lymphatics 2025, 3, 25. [Google Scholar] [CrossRef]
- Alaggio, R.; Amador, C.; Anagnostopoulos, I.; Attygalle, A.D.; Araujo, I.B.d.O.; Berti, E.; Bhagat, G.; Borges, A.M.; Boyer, D.; 335 Calaminici, M.; et al. The 5th edition of the World Health Organization Classification of Haematolymphoid Tumours: Lymphoid 336 Neoplasms. Leukemia 2022, 36, 1720–1748. [Google Scholar] [CrossRef] [PubMed]
- Ríos-Tamayo, R.; Krsnik, I.; Gómez-Bueno, M.; Garcia-Pavia, P.; Segovia-Cubero, J.; Huerta, A.; Salas, C.; Silvestre, R.; Sánchez, A.; Manso, M.; et al. AL Amyloidosis and Multiple Myeloma: A Complex Scenario in Which Cardiac Involvement Remains the Key Prognostic Factor. Life 2023, 13, 1518. [Google Scholar] [CrossRef]
- Ríos-Tamayo, R.; Sánchez Rodríguez, D.; Chang-Chan, D.-Y.-L.; Sánchez Pérez, M.J. Epidemiology of Multiple Myeloma. In Update on Multiple Myeloma; Al-Anazi, Khalid Ahmed, Ed.; Intech: London, UK, 2019; pp. 13–33. [Google Scholar] [CrossRef]
- Chang-Chan, D.Y.; Ríos-Tamayo, R.; Rodríguez-Barranco, M.; Redondo-Sánchez, D.; González, Y.; Marcos-Gragera, R.; Sánchez, M.J. Trends of incidence, mortality and survival of multiple myeloma in Spain. A twenty-three-year population-based study. Clin. Trans. Oncol. 2021, 23, 1429–1439. [Google Scholar] [CrossRef]
- Mateos, M.V.; Trelles, R.; Escalante Barrigón, F.; de la Rubia, F.; Marin Huarte, N.; Garcia Garcia-Porrero, A.; Cáceres Nuñez, A.; Rios-Tamayo, R.; Sampol Mayol, A.; Giménez, E.; et al. Prevalence, incidence, and outcomes of multiple myeloma patients using electronic health records and natural language processing. Blood 2025, 146, 4566–4567. [Google Scholar] [CrossRef]
- Encinas, C.; Blanchard, M.J.; Alonso Fernández, R.; Hernández-Rivas, J.-A.; Ríos-Tamayo, R.; Machado-Vílchez, M.; Martínez-López, J.; Mateos, M.V. Factors Increasing the Risk of Infection in Patients With Multiple Myeloma: From Biology to Prevention. Clin. Lymphoma Myeloma Leuk. 2025, 25, e844–e856. [Google Scholar] [CrossRef]
- Xue, Q.-L. The Frailty Syndrome: Definition and Natural History. Clin. Geriatr. Med. 2011, 27, 1–15. [Google Scholar] [CrossRef]
- Mian, H.; Wildes, T.M.; Vij, R.; Pianko, M.J.; Major, A.; Fiala, M.A. Dynamic frailty risk assessment among older adults with multiple myeloma: A population-based cohort study. Blood Cancer J. 2023, 13, 76. [Google Scholar] [CrossRef]
- Bart, N.K.; Bianchi, G.; Cuddy, S.A.M.; Goyal, P.; Griffin, M.; Hummel, S.L.; Macdonald, P.; Maurer, M.; Montgomery, E.; Nanne, M.G.; et al. Cardiac Amyloidosis in Older Adults With a Focus on Frailty. JACC Adv. 2025, 4, 101784. [Google Scholar] [CrossRef]
- Ríos-Tamayo, R.; Lecumberri, R.; Cibeira, M.T.; González-Calle, V.; Alonso, R.; Domingo-González, A.; Landete, E.; Encinas, C.; 352 Iñigo, B.; Blanchard, M.J.; et al. A Simple Frailty Score Predicts Survival and Early Mortality in Systemic AL Amyloidosis. Cancers 2024, 16, 1689. [Google Scholar] [CrossRef]
- Ríos-Tamayo, R.; Sainz, J.; Martínez-López, J.; Puerta, J.M.; Chang, D.Y.L.; Rodríguez, T.; Lahuerta, J.J. Early mortality in mul-349 tiple myeloma: The time-dependent impact of comorbidity. A population-based study in 621 real-life patients. Am. J. Hematol. 350 2016, 91, 700–705. [Google Scholar] [CrossRef]
- Ríos-Tamayo, R. The evolving landscape of anti-clonal therapy in newly diagnosed systemic light-chain (AL) amyloidosis: evidence- and time-based comparison with multiple myeloma. Life 2026, 16, 363. [Google Scholar] [CrossRef] [PubMed]
- Knips, L.; Bergenthal, N.; Streckmann, F.; Monsef, I.; Elter, T.; Skoetz, N. Aerobic physical exercise for adult patients with haematological malignancies. Cochrane Database Syst. Rev. 2019, Issue 1. Art. No., CD009075. [Google Scholar] [CrossRef] [PubMed]
- Fowles, J.R.; Sasso, J.P.; Thomas, S.G.; Shivgulam, M.E.; O’Brien, M.W.; Nagpal, T.S. Exercise in medicine: challenges and opportunities integrating qualified exercise professionals (QEPs) into Canadian healthcare. Appl. Physiol. Nutr. Metab. 2026, 51, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Wernhart, S.; Rassaf, T. Exercise, cancer, and the cardiovascular system: clinical effects and mechanistic insights. Basic Res. Cardiol. 2025, 120, 35–55. [Google Scholar] [CrossRef]
- Crevenna, R.; Hasenoehrl, T.; Wiltschke, C.; Kainberger, F.; Keilani, M. Prescribing Exercise to Cancer Patients Suffering from Increased Bone Fracture Risk Due to Metastatic Bone Disease or Multiple Myeloma in Austria—An Inter- and Multidisciplinary Evaluation Measure. Cancers 2023, 15, 1245. [Google Scholar] [CrossRef]
- Angulo, J.; El Assar, M.; Álvarez-Bustos, A.; Rodríguez-Mañas, L. Physical activity and exercise: Strategies to manage frailty. Redox Biol. 2020, 101513. [Google Scholar] [CrossRef]
- Raza Rizvi, M.; Sharma, A.; Kashoo, F.Z.; Rahim Shaik, A.; Seyam, M.K.; Hamdy Elrefaey, B.; Ali, Z.A.; Abdullah Asiri, A.; Ahmad, I.; Ahmad, F. Reversing frailty: The transformative effects of structured physical-activity-based physiotherapy on physical, cognitive and emotional health in older adults—An evidence-based systematic review. Eur. J. Clin. Invest. 2025, 56, e70158. [Google Scholar] [CrossRef] [PubMed]
- Clegg, A.; Young, J.; Olde Rikkert, M.; Rockwood, K. Frailty in elderly people. Lancet 2013, 381, 752–762. [Google Scholar] [CrossRef]
- Safarti, D.; Koczwara, B.; Jackson, C. The Impact of Comorbidity on Cancer and Its Treatment. CA Cancer J. Clin. 2016, 66, 337–350. [Google Scholar] [CrossRef]
- Cruz-Jentoft, A.J.; Volkert, D. Malnutrition in Older Adults. N. Engl. J. Med. 2025, 392, 2244–2255. [Google Scholar] [CrossRef]
- Sayer, A.A.; Cooper, R.; Arai, H.; Cawthon, P.M.; Ntsama Essomba, M.-J.; Fielding, R.A.; Grounds, M.D.; Witham, M.D.; Cruz-Jentoft, A.J. Sarcopenia. Nat. Rev. Dis. Prim. 2024, 10, 68. [Google Scholar] [CrossRef] [PubMed]
- Jost, Z.; Kujach, S. Understanding Cognitive Decline in Aging: Mechanisms and Mitigation Strategies – A Narrative Review. Clin. Interv. Aging 2025, 20 459–469. [Google Scholar] [CrossRef]
- Patrick, R.E.; Dickinson, R.A.; Gregg, A.; Kaufman, J.R.; Maciarz, J.; Merrill, J.G.; Williams, L.A.; Weisenbach, S.L. Review of Emotion Regulation in Late Life Mood Disorders. J. Psychiatry Brain Sci. 2024, 9, e240008. [Google Scholar] [CrossRef]
- Holt-Lunstad, J.; Perissinotto, C. Social Isolation and Loneliness as Medical Issues. N. Engl. J. Med. 2023, 388, 193–195. [Google Scholar] [CrossRef] [PubMed]
- Nhlanzeko Ngcobo, N. Polypharmacy and deprescribing among geriatric patients. Aging Health Res. 2025, 5, 100256. [Google Scholar] [CrossRef]
- Akseer, S.; Torkom Yeretzian, S.; Abrahamyan, L.; Puts, M.; Mohamed, M.; Kheng Soo, W.; Alibhai, S.M.H.; Sahakyan, Y. Cost-Utility of Geriatric Assessment and Management in Older Adults With Cancer: Model-Based Economic Evaluation. J. Clin. Oncol. 2025, 44, 92–102. [Google Scholar] [CrossRef]
- Redder, E.; Zhao, Q.; Bumma, N.; Kahwash, R.; Vallakati, A.; Campbell, C.; Parikh, S.; Almaani, S.; Freimer, M.; Efebera, Y.; et al. Functional Impairments of Amyloidosis Patients: Physical Therapy Assessment. Hemato 2022, 3, 414–421. [Google Scholar] [CrossRef]
- Muchtar, E.; Grogan, M.; Aus dem Siepen, F.; Waddington-Cruz, M.; Misumi, Y.; Carroll, A.S.; Clarke, J.O.; Sanchorawala, V.; Milani, P.; Caccialanza, R.; et al. Supportive care for systemic amyloidosis: International Society of Amyloidosis (ISA) expert panel guidelines. Amyloid 2025, 32, 93–116. [Google Scholar] [CrossRef]
- Ski, C.F.; Cartledge, S.; Foldager, D.; Thompson, D.R.; Fredericks, S.; Ekman, I.; Hendriks, J.M. Integrated care in cardiovascular disease: a statement of the Association of Cardiovascular Nursing and Allied Professions of the European Society of Cardiology. Eur. J. Cardiovasc. Nurs. 2023, 22, e39–e46. [Google Scholar] [CrossRef] [PubMed]
- Kittleson, M.M. Guidelines for treating heart failure. Trends Cardiovasc. Med. 2025, 35, 141–150. [Google Scholar] [CrossRef]
- Denfeld, Q.E.; Jha, S.R.; Fung, E.; Jaarsma, T.; Maurer, M.S.; Reeves, G.R.; Afilalo, J.; Beerli, N.; Bellumkonda, L.; De Geest, S.; et al. Assessing and managing frailty in advanced heart failure: An International Society for Heart and Lung Transplantation consensus statement. J. Heart Lung. Transplant. 2024, 43, 1–27. [Google Scholar] [CrossRef]
- Kittleson, M.M.; Ruberg, F.L.; Ambardekar, A.V.; Brannagan, T.H.; Cheng, R.K.; Clarke, J.O.; Dember, L.M.; Grazzini Frantz, J.; Hershberger, R.E.; Maurer, M.S.; et al. 2023 ACC Expert Consensus Decision Pathway on Comprehensive Multidisciplinary Care for the Patient With Cardiac Amyloidosis: A Report of the American College of Cardiology Solution Set Oversight Committee. J. Am. Coll. Cardiol. 2023, 81, 1076–1126. [Google Scholar] [CrossRef]
- Pugliatti, P.; Trimarchi, G.; Barocelli, F.; Pizzino, F.; Di Spigno, F.; Tedeschi, A.; Cusmà Piccione, M.; Irrera, P.; Aschieri, D.; Niccoli, G.; et al. Advancing Cardiac Amyloidosis Care Through Insights from Cardiopulmonary Exercise Testing. J. Clin. Med. 2024, 13, 7285. [Google Scholar] [CrossRef]
- Marra, A.; Palmiero, G.; Losi, M.; Mallardo, M.; Galati, A.; Bossone, E.; Esposito, G.; Calabrò, P.; Cittadini, A.; Limongelli, G. Exercise Training and Rehabilitation in Cardiac Amyloidosis (ERICA) Study: Rationale and Design. Heart Fail. Clin. 2024, 20, e55–e62. [Google Scholar] [CrossRef] [PubMed]
- Cederholm, T.; Bosaeus, I. Manutrition in adults. N. Engl. J. Med. 2024, 391, 155–165. [Google Scholar] [CrossRef]
- Cruz-Jentoft, A.J.; Volkert, D. Malnutrition in Older Adults. N. Engl. J. Med. 2025, 392, 2244–2255. [Google Scholar] [CrossRef] [PubMed]
- Kyle, R.A.; Gertz, M.A. Primary systemic amyloidosis: clinical and laboratory features in 474 cases. Semin. Hematol. 1995, 32, 45–59. [Google Scholar]
- Caccialanza, R.; Palladini, G.; Klersy, C.; Cena, H.; Vagia, C.; Cameletti, B.; Russo, P.; Lavatelli, F.; Merlini, G. Nutritional status of outpatients with systemic immunoglobulin light-chain amyloidosis. Am. J. Clin. Nutr. 2006, 83, 350–354. [Google Scholar] [CrossRef]
- Caccialanza, R.; Palladini, G.; Klersy, C.; Cereda, E.; Bonardi, C.; Cameletti, B.; Quarleri, L.; Montagna, E.; Foli, A.; Milani, P.; et al. Malnutrition at Diagnosis Predicts Mortality in Patients With Systemic Immunoglobulin Light-Chain Amyloidosis Independently of Cardiac Stage and Response to Treatment. JPEN J. Parenter. Enter. Nutr. 2014, 38, 891–894. [Google Scholar] [CrossRef] [PubMed]
- Caccialanza, R.; Palladini, G.; Cereda, E.; Bonardi, C.; Milani, P.; Cameletti, B.; Quarleri, L.; Capello, S.; Foli, A.; Lavatelli, F.; et al. Nutritional counseling improves quality of life and preserves body weight in systemic immunoglobulin light-chain (AL) amyloidosis. Nutrition 2015, 31, 1228–1234. [Google Scholar] [CrossRef] [PubMed]
- Caccialanza, R.; Goldwasser, F.; Marschal, O.; Ottery, F.; Schiefke, S.; Tilleul, P.; Zalcman, G.; Pedrazzoli, P. Unmet needs in clinical nutrition in oncology: a multinational analysis of real-world evidence. Ther. Adv. Med. Oncol. 2020, 12, 1–10. [Google Scholar] [CrossRef]
- Sanchorawala, V.; Boccadoro, M.; Gertz, M.; Hegenbart, U.; Kastritis, E.; Landau, H.; Mollee, P.; Wechalekar, A.; Palladini, G. Guidelines for high dose chemotherapy and stem cell transplantation for systemic AL amyloidosis: EHA-ISA working group guidelines. Amyloid 2022, 29, 1–7. [Google Scholar] [CrossRef]
- Casirati, A.; Salcedo, I.; Cereda, E.; Chabannon, C.; Ruggeri, A.; Kuball, J.; Clout, J.R.; Mooyaart, J.E.; Kenyon, M.; Caccialanza, R.; et al. The European Society for Blood and Marrow Transplantation (EBMT) roadmap and perspectives to improve nutritional care in patients undergoing hematopoietic stem cell transplantation on behalf of the Cellular Therapy and Immunobiology Working Party (CTIWP) and the Nurses Group (NG) of the EBMT. Bone Marrow Transplant. 2023, 58, 965–972. [Google Scholar] [CrossRef] [PubMed]
- Falcone, L.; Mancin, S.; Azzolini, E.; Colotta, F.; Ferrante, S.; Pastore, M.; Morales Palomares, S.; Lopane, D.; Sguanci, M.; Cosmai, S.; et al. Nutritional Prehabilitation Intervention in Hematological Patients Undergoing Bone Marrow Transplant: A Systematic Review of the Literature. Nutrients 2024, 16, 4387. [Google Scholar] [CrossRef]
- König, F.; Davidoff, C.; Ibanez, K.; Hwang, S.; Goldstein, I.; Feldman, D.R.; Gyurkocza, B.; Giralt, S.A.; Politikos, I.; Ponce, D.M.; et al. Post-Transplant Pain and Paralysis: Neurologic Amyotrophy as an Atypical Cause of Shoulder Dysfunction Following Hematopoietic Stem Cell Transplant. Cancers 2025, 17, 1816. [Google Scholar] [CrossRef]
- Sharpe, M.; Wilks, D. Fatigue. BMJ 2002, 325, 480. [Google Scholar] [CrossRef]
- Tachibana, S.; Bourgeois Yoshioka, C.K. Take Fatigue or Fatigues into Account in Physiotherapy Interventions? A Rapid Scoping Review. Phys. Ther. Res. 2025, 28, 157–173. [Google Scholar] [CrossRef]
- Gray, P.J. Management of Patients with Chronic Renal Failure: Role of Physical Therapy. Phys. Ther. 1982, 62, 173–176. [Google Scholar] [CrossRef]
- Sawant, A.; House, A.:A.; Overend, T.J. Anabolic Effect of Exercise Training in People with End-Stage Renal Disease on Hemodialysis: A Systematic Review with Meta-analysis. Physiother.Can 2014, 66, 44–53. [Google Scholar] [CrossRef]
- Baker, L.A.; March, D.S.; Wilkinson, T.J.; Billany, R.E.; Bishop, N.C.; Castle, E.M.; Chilcot, J.; Davies, M.D.; Graham-Brown, M.P.M.; Greenwood, S.A.; et al. Clinical practice guideline exercise and lifestyle in chronic kidney disease. BMC Nephrol. 2022, 23, 75. [Google Scholar] [CrossRef]
- Kidney Disease: Improving Global Outcomes (KDIGO) CKD Work Group. KDIGO 2024 Clinical Practice Guideline for the Evaluation and Management of Chronic Kidney Disease. Kidney Int. 2024, 105, S117–S314. [CrossRef] [PubMed]
- Villanego, F.; Arroyo, D.; Martínez-Majonero, V.; Hernández-Sánchez, S.; Esteve-Simó, V. Importance of physical exercise prescription in patients with chronic kidney disease: results of the survey of the Grupo Español Multidisciplinar de Ejercicio Físico en el Enfermo Renal [Spanish Multidisciplinary Group of Physical Exercise in Kidney Patients] (GEMEFER). Nefrologia 2023, 43, 126–132. [Google Scholar] [CrossRef]
- Battaglia, Y.; Baciga, F.; Bulighin, F.; Amicone, M.; Mosconi, G.; Storari, A.; Brugnano, R.; Pozzato, M.; Motta, D.; D’alessandro, C.; et al. Physical activity and exercise in chronic kidney disease: consensus statements from the Physical Exercise Working Group of the Italian Society of Nephrology. J. Nephrol. 2024, 37, 1735–1765. [Google Scholar] [CrossRef]
- Hirano, Y.; Kono, K.; Takahashi, R.; Tamura, Y.; Takahashi, M.; Imaoka, S.; Nomura, T.; Igaki, M. Participation of physical therapists in medical fee-based dialysis-prevention interventions: a nation wide survey in Japan. Clin. Exp. Nephrol. 2026, 30, 96–108. [Google Scholar] [CrossRef] [PubMed]
- Fudeyasu, K.; Nakashima, Y.; Iwaki, D.; Fukuhara, K.; Nagao, A.; Chishaki, R.; Mikami, Y. Long-Term Rehabilitation Therapy Is Effective for Physical Function in a Patient With Amyloid Light Chain Amyloidosis Complicated by Nephrotic Syndrome: A Case Report and Literature Review. Cureus 2024, 16, e64830. [Google Scholar] [CrossRef] [PubMed]
- Wechalekar, A.D.; Fontana, M.; Quarta, C.; Liedtke, M. AL Amyloidosis for Cardiologists: Awareness, Diagnosis, and Future Prospects: JACC: CardioOncology State-of-the-Art Review. J. Am. Coll. Cardiol. CardioOnc. 2022, 4, 427–441. [Google Scholar] [CrossRef]
- Thimm, A.; Alexander Carpinteiro, A.; Oubari, S.; Papathanasiou, M.; Luedike, P.; Kessler, L.; Rischpler, C.; Blau, T.; Reinhardt, H.C.; Rassaf, T.; et al. Prevalence and predictors of neurological manifestations in systemic AL amyloidosis. J. Neurol. Sci. 2022, 440, 120341. [Google Scholar] [CrossRef]
- Chompoopong, P.; Mauermann, M.L.; Siddiqi, H.; Peltier, A. Amyloid Neuropathy: From Pathophysiology to Treatment in Light-Chain Amyloidosis and Hereditary Transthyretin Amyloidosis. Ann. Neurol. 2024, 96, 423–440. [Google Scholar] [CrossRef]
- Kaku, M.; Berk, J.L. Neuropathy Associated with Systemic Amyloidosis. Semin. Neurol. 2019, 39, 578–588. [Google Scholar] [CrossRef] [PubMed]
- Qian, M.; Qin, L.; Shen, K.; Guan, H.; Ren, H.; Zhao, Y.; Guan, Y.; Zhou, D.; Peng, B.; Li, J.; et al. Light-Chain Amyloidosis With Peripheral Neuropathy as an Initial Presentation. Front. Neurol. 2021, 12, 707134. [Google Scholar] [CrossRef]
- Ballegaard, M.; Nelson, L.M.; Gimsing, P. Comparing neuropathy in Multiple Myeloma and AL Amyloidosis. J.Peripher. Nerv. Syst. 2021, 26, 75–82. [Google Scholar] [CrossRef]
- Jesson, T.; Runge, N.; Schmid, A.B. Physiotherapy for people with painful peripheral neuropathies: a narrative review of its efficacy and safety. Pain. Rep. 2020, 5, e834. [Google Scholar] [CrossRef]
- Chimenti, R.L.; Frey-Law, L.A.; Sluka, K.A. A Mechanism-Based Approach to Physical Therapist Management of Pain. Phys. Ther. 2018, 98, 302–314. [Google Scholar] [CrossRef]
- Dahlin, L.B.; Zimmerman, M.; Calcagni, M.; Hundepool, C.A.; van Alfen, N.; Chung, K.C. Carpal tunnel syndrome. Nat. Rev. Dis. Prim. 2024, 10, 37. [Google Scholar] [CrossRef]
- Zegri-Reiriz, I.; de Haro-del Moral, F. J.; Dominguez, F.; Salas, C.; de la Cuadra, P.; Plaza, A.; Krsnik, I.; Gonzalez-Lopez, E.; Garcia-Pavia, P. Prevalence of Cardiac Amyloidosis in Patients with Carpal Tunnel Syndrome. J. Cardiovasc. Trans. Res. 2019, 12, 507–513. [Google Scholar] [CrossRef] [PubMed]
- Muchtar, E.; Derudas, D.; Mauermann, M.; Liewluck, T.; Dispenzieri, A.; Kumar, S.K.; Dingli, D.; Lacy, M.Q.; Buadi, F.K.; Hayman, S.R.; et al. Systemic Immunoglobulin Light Chain AmyloidosiseAssociated Myopathy: Presentation, Diagnostic Pitfalls, and Outcome. Mayo Clin. Proc. 2016, 91, 1354–1361. [Google Scholar] [CrossRef] [PubMed]
- Namiranian, D.; Geisler, S. Neuromuscular Complications of Systemic Amyloidosis. Am. J. Med. 2022, 135, S13–S19. [Google Scholar] [CrossRef] [PubMed]
- Curtis, A.R.; Prado, C.M.; Orellana, L.; Daly, J.R.M.; Bauer, J.; Denehy, L.; Edbrooke, L.; Baguley, B.J.; Alston, L.; Hardcastle, N.; et al. Exploring potential predictors of low muscle mass and muscle loss in adults with cancer: A scoping review. Clin. Nutr. 2026, 56, 106531. [Google Scholar] [CrossRef]
- Kyriazopoulou Korovesi, A.A.; Pateli, A.; Kastritis, E.; Poulia, K.A.; Stamatelopoulos, K.; Karatzi, K. Diet-related poor nutritional status as a major challenge in the treatment of patients with amyloidosis: A systematic review. Nutrition 2026, 141, 112930. [Google Scholar] [CrossRef]
- Emery, A.; Moore, S.; Crowe, J.; Murray, J.; Peacock, O.; Thompson, D.; Betts, F.; Rapps, S.; Ross, L.; Rothschild-Rodriguez, D.; et al. The effects of short-term, progressive exercise training on disease activity in smouldering multiple myeloma and monoclonal gammopathy of undetermined significance: a single-arm pilot study. BMC Cancer 2024, 24, 174. [Google Scholar] [CrossRef]
- Jeevanantham, D.; Rajendran, V.; Tremblay, L.; Larivière, C.; Knight, A. Evidence-based guidelines for physiotherapy management of patients with multiple myeloma: study protocol. Syst. Rev. 2018, 7, 118. [Google Scholar] [CrossRef]
- Jeevanantham, D.; Rajendran, V.; McGillis, Z.; Tremblay, L.; Larivière, C.; Knight, A. Mobilization and Exercise Intervention for Patients With Multiple Myeloma: Clinical Practice Guidelines Endorsed by the Canadian Physiotherapy Association. Phys. Ther. 2021, 101, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Larsen, R.F.; Jarden, M.; Minet, L.R.; Frølund, U.C.; Möller, S.; Abildgaard, N. Physical function in patients newly diagnosed with multiple myeloma; a Danish cohort study. BMC Cancer 2020, 20, 169. [Google Scholar] [CrossRef]
- Land, J.; Hackett, J.; Sidhu, G.; Heinrich, M.; McCourt, O.; Yong, K.L.; Fisher, A.; Beeken, R.J. Myeloma patients’ experiences of a supervised physical activity programme: a qualitative study. Support. Care Cancer 2022, 30, 6273–6286. [Google Scholar] [CrossRef] [PubMed]
- Dapunt, U.; Ehret, P.; Paratte, J.-L.; Kuehl, R.M.; Wiskemann, J.; Jäger, D.; Müller-Tidow, C.; Raab, M.-S.; Goldschmidt, H. A precision-based exercise program for patients with multiple myeloma. Eur. J. Haematol. 2023, 111, 930–937. [Google Scholar] [CrossRef]
- Park, S.-S.; Kim, E.-S.; Youn, J.-C.; Jung, M.-H.; Park, J.; Eun Nam, K.; Min, C.-K.; Armenian, S.H.; Lee, K. Risk Stratification Strategies and Implementation of Exercise Training in Patients with Multiple Myeloma. Curr. Oncol. Rep. 2025, 27, 1448–1463. [Google Scholar] [CrossRef]
- Larsen, R.F.; Jarden, M.; Minet, L.R.; Frølund, U.C.; Abildgaard, N. Supervised and home-based physical exercise in patients with newly diagnosed multiple myeloma—a randomized controlled feasibility study. Pilot Feasibility Stud. 2019, 5, 130. [Google Scholar] [CrossRef] [PubMed]
- Larsen, R.F.; Jarden, M.; Minet, L.R.; Frølund, U.C.; Hermann, A.P.; Breum, L.; Möller, S.; Abildgaard, N. Exercise in newly diagnosed patients with multiple myeloma: A randomized controlled trial of effects on physical function, physical activity, lean body mass, bone mineral density, pain, and quality of life. Eur. J. Haematol. 2024, 113, 298–309. [Google Scholar] [CrossRef] [PubMed]
- Koutoukidis, D.A.; Land, J.; Hackshaw, A.; Heinrich, M.; McCourt, O.; Beeken, R.J.; Philpott, S.; DeSilva, D.; Rismani, A.; Rabin, N.; et al. Fatigue, quality of life and physical fitness following an exercise intervention in multiple myeloma survivors (MASCOT): an exploratory randomised Phase 2 trial utilising a modified Zelen design. Br. J. Cancer 2020, 123, 187–195. [Google Scholar] [CrossRef] [PubMed]
- Nicol, J.L.; Woodrow, C.; Cunningham, B.J.; Mollee, P.; Weber, N.; Smith, M.D.; Nicol, A.J.; Gordon, L.G.; Hill, M.M.; Skinner, T.L. An Individualized Exercise Intervention for People with Multiple Myeloma—Study Protocol of a Randomized Wait list-Controlled Trial. Curr. Oncol. 2022, 29, 901–923. [Google Scholar] [CrossRef]
- Nicol, J.L.; Chong, J.E.; McQuilten, Z.K.; Mollee, P.; Hill, M.M.; Skinner, T.L. Safety, Feasibility, and Efficacy of Exercise Interventions for People With Multiple Myeloma: A Systematic Review. Clin. Lymphoma Myeloma Leuk. 2022, 23, 86–96. [Google Scholar] [CrossRef]
- Dreyling, E.; Räder, J.; Möller, M.-D.; Ihorst, G.; Wenger, S.; Pahl, A.; Arends, J.; Herget, G.; Deibert, P.; Wäsch, R.; et al. A Randomized Controlled ‘REAL-FITNESS’ Trial to Evaluate Physical Activity in Patients With Newly Diagnosed Multiple Myeloma. J. Cachexia Sarcopenia Muscle 2025, 16, e13793. [Google Scholar] [CrossRef]
- Spiliopoulou, P.; Rousakis, P.; Panteli, C.; Eleutherakis-Papaiakovou, E.; Migkou, M.; Kanellias, N.; Ntanasis-Stathopoulos, I.; Malandrakis, P.; Theodorakakou, F.; Fotiou, D.; et al. Effects of Exercise Training on the Bone Marrow Immune Microenvironment and Minimal Residual Disease in Multiple Myeloma Patients Following First-Line Treatment. Scand. J. Med. Sci. Sports 2025, 35, e70020. [Google Scholar] [CrossRef]
- Zhao, Q.; Chen Hong, Z.; Wei, T.T.; Jiang, Y.T.; Zeng, H.; Miao, G. Effects of aerobic exercise on cognitive function in older adults with mild cognitive impairment: a systematic review and meta-analysis. Front. Psychiatry 2026, 16, 1741998. [Google Scholar] [CrossRef]
- Li, J.; Peng, Y.; Zhan, D.; Zhang, Y.; Yu, S. Exercise interventions in patients with multiple myeloma: a scoping review. BMC Sports Sci. Med. Rehabil. 2025, 17, 148. [Google Scholar] [CrossRef]
- Ludwig, H.; San Miguel, J.; Dimopoulos, M.A.; Palumbo, A.; García Sanz, R.; Powles, R.; Lentzsch, S.; Ming Chen, W.; Hou, J.; Jurczyszyn, A.; et al. International Myeloma Working Group recommendations for global myeloma care. Leukemia 2014, 28, 981–992. [Google Scholar] [CrossRef]
- George, L.L.; Malik, M.N.; Miller, E.J.; Hicks, K.; Khanam, R.; Saterehaseman, A.; McNichol, M.; Anwer, F. Special Considerations for Supportive Care and Management of Complications in Elderly Patients With Multiple Myeloma. Clin. Lymphoma Myeloma Leuk. 2021, 12, P812–P822. [Google Scholar] [CrossRef] [PubMed]
- Terpos, E.; Morgan, G.; Dimopoulos, M.A.; Drake, M.T.; Lentzsch, S.; Raje, N.; Sezer, O.; Garcia-Sanz, R.; Shimizu, K.; Turesson, I.; et al. International Myeloma Working Group Recommendations for the Treatment of Multiple Myeloma–Related Bone Disease. J. Clin. Oncol. 2013, 31, 2347–2357. [Google Scholar] [CrossRef]
- Ríos-Tamayo, R.; Sánchez Pérez, M.J.; Sánchez Rodríguez, D.; García Ruiz, A.; Garrido Collado, P.; Salas, G.; Muñoz Ballester, J.; Hernández Mohedo, F.; Mesa, Z.; Leyva Ferrer, R.; et al. The prognostic impact of pathologic fracture as myeloma-defining event in newly diagnosed multiple myeloma. Hemasphere 2019, 3(S1), 277. [Google Scholar] [CrossRef]
- Ríos-Tamayo, R.; Sainz, J.; Jurado, M. Comparative Baseline Health-Related Quality of Life in Real-Life Patients with Monoclonal Gammopathies. J. Leuk. 2015, 3, 4. [Google Scholar] [CrossRef]
- Ludwig, H.; Bailey, A.L.; Marongiu, A.; Khela, K.; Milligan, G.; Carlson, K.B.; Rider, A.; Seesaghur, A. Patient-reported pain severity and health-related quality of life in patients with multiple myeloma in real world clinical practice. Cancer Rep. 2021, e1429. [Google Scholar] [CrossRef]
- Bhowmik, D.; Hines, D.M.; Intorcia, M.; Wade, R.L. Economic burden of skeletal-related events in patients with multiple myeloma: analysis of US commercial claims database. J. Med. Econ. 2018, 21, 622–628. [Google Scholar] [CrossRef] [PubMed]
- Anderson, K.; Ismaila, N.; Flynn, P.J.; Halabi, S.; Jagannath, S.; Ogaily, M.S.; Omel, J.; Raje, N.; Roodman, G.D.; Yee, G.C.; Kyle, R.A. Role of Bone-Modifying Agents in Multiple Myeloma: American Society of Clinical Oncology Clinical Practice Guideline Update. J. Clin. Oncol. 2018, 36, 812–818. [Google Scholar] [CrossRef] [PubMed]
- Kyriakou, C.; Molloy, S.; Vrionis, F.; Alberico, R.; Bastian, J.L.; Zonder, J.A.; Giralt, S.; Raje, N.; Kyle, R.A.; Roodman, D.G.D.; et al. The role of cement augmentation with percutaneous vertebroplasty and balloon kyphoplasty for the treatment of vertebral compression fractures in multiple myeloma: a consensus statement from the International Myeloma Working Group (IMWG). Blood Cancer J. 2019, 9, 27. [Google Scholar] [CrossRef]
- Abbas, S.; Sadia Adil, H.; Raza, S.; Basit, J.; Arul, K.; Anwer, F.; Hamza Habib, M. Advances in Supportive Care for Multiple Myeloma-Related Bone Disease—A Review. Cancers 2025, 17, 2166. [Google Scholar] [CrossRef]
- Massat, B.; Stiff, P.; Esmail, F.; Gauto-Mariotti, E.; Hagen, P. Preventing Skeletal-Related Events in Newly Diagnosed Multiple Myeloma. Cells 2025, 14, 1263. [Google Scholar] [CrossRef]
- Cantley, M.D.; Trainor, L.J.; Cheney, E. A.-J.; Watt, S.M.; Vandyke, K. Skeletal health in the precursor stages of multiple myeloma: fracture risk and bone phenotypes in monoclonal gammopathy of undetermined significance and smouldering myeloma. Pathology 2026, 58, 199–213. [Google Scholar] [CrossRef]
- Fernández-Lázaro, D.; Seco-Casares, M.; López Llorente, A.; Hernández-Burgos, N.; Santamaría Gómez, G. Impact of the therapeutic exercise in people with multiple myeloma on biomarkers of physical, psychological, and biological health status: A systematic review. Phys. Act. Exerc. Cancer 2025, 2, 63–84. [Google Scholar] [CrossRef]
- Na, X.; Yang, Y.; Yang, H.; Chen, Z.; Qu, X.; Zhang, J.; Chen, M.; Wang, D.; Breuille, D.; Yu, K.; et al. The effect of non-pharmacological interventions on bone health among patients with low bone mass: a systematic review and meta-analysis. Front. Endocrinol. 2025, 16, 1612739. [Google Scholar] [CrossRef]
- Filis, P.; Markozannes, G.; Chan, D.S.M.; Mauri, D.; Foukakis, T.; Matikas, A.; Droufakou, S.; Pentheroudakis, G.; Tsilidis, K. Grading the evidence for physical activity and any outcome in cancer survivors: An Umbrella review of 740 meta-analytic associations. Crit. Rev. Oncol. 2025, 207, 104602. [Google Scholar] [CrossRef]
- Park, S.S.; Kim, E.S.; Youn, J.C.; Jung, M.H.; Park, J.; Nam, K.E.; Min, C.K.; Armenian, S.H.; Lee, K. Risk Stratification Strategies and Implementation of Exercise Training in Patients with Multiple Myeloma. Curr. Oncol. Rep. 2025, 27, 1448–1463. [Google Scholar] [CrossRef]
- Rock, C.L..; Thomson, C.A..; Sullivan, K.R..; Howe, C.L.; Kushi, L.H.; Caan, B.J.; Neuhouser, M.L.; Bandera, E.V.; Wang, Y.; Robien, K.; et al. American Cancer Society nutrition and physical activity guideline for cancer survivors. CA Cancer J. Clin. 2022, 72, 230–262. [Google Scholar] [CrossRef]
- Baughman, C.; Norman, K.; Mukamal, K. Adherence to American Cancer Society Nutrition and Physical Activity Guidelines Among Cancer Survivors. JAMA Oncol. 2024, 10, 789–792. [Google Scholar] [CrossRef] [PubMed]
- Heymsfield, S.B.; Shapses, S.A. Guidance on Energy and Macronutrients across the Life Span. N. Engl. J. Med. 2024, 390, 1299–1310. [Google Scholar] [CrossRef] [PubMed]
- Maslana, K.E.; Skogerboe, G.E.; Sborov, D.W.; Coletta, A.M. Exercise and diet studies among patients living with multiple myeloma: current evidence and considerations of patient preferences. Front. Hematol. 2025, 4, 1550681. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.H.; Fung, T.T.; Tabung, F.K.; Marinac, C.R.; Devore, E.E.; Rosner, B.A.; Ghobrial, I.M.; Colditz, G.A.; Giovannucci, E.L.; Birmann, B.M. Prediagnosis dietary pattern and survival in patients with multiple myeloma. Intern. J. Cancer 2020, 147, 1823–1830. [Google Scholar] [CrossRef]
- Malik, M.A.; Nathan, W.; Sweeney, N.W.; Jafri, M.; Derkach, A.; Chmielewski, C.; Adintori, P.A.; Mailankody, S.; Korde, N.; Tan, C. R.; Hassoun, H.; et al. Nutrition perceptions, needs and practices among patients with plasma cell disorders. Blood Cancer J. 2022, 12, 70. [Google Scholar] [CrossRef]
- Zhang, L.; Chen, S.; Huang, M.; Wang, W.; Liang, Y.; Wang, Y. Prognostic value of the nutritional risk index in patients with newly diagnosed multiple myeloma. Ann. Hematol. 2023, 102, 125–132. [Google Scholar] [CrossRef]
- Zhang, L.; Chen, S.; Wang, W.; Wang, Y.; Liang, Y. Inflammatory and Nutritional Scoring System for Predicting Prognosis in Patients with Newly Diagnosed Multiple Myeloma. J. Infl. Res. 2023, 16, 7–17. [Google Scholar] [CrossRef]
- Zhang, C.; Miao, D.; Zhang, Y.; Xu, Y. nutritional index model for predicting resistance and prognosis in multiple myeloma following autologous stem cell transplantation. Discov. Oncol. 2026, 17, 261. [Google Scholar] [CrossRef]
- Nie, Y.; Zhang, Z.; Tang, X. Prognostic and clinicopathological value of prognostic nutritional index in patients with multiple myeloma: a meta-analysis. Front. Oncol. 2025, 15, 1545096. [Google Scholar] [CrossRef] [PubMed]
- Xu, P.; Liu, Y.; Wang, Y.; Feng, Q.; Zhang, H.; Cheng, H.; Qi, K.; Xu, K.; Li, Z. Nutritional status indices on the prognosis of patients with relapsed and refractory multiple myeloma treated with CAR-T cell immunotherapy. Front. Nutr. 2025, 12, 1654407. [Google Scholar] [CrossRef]
- Duvenci Birben, O.; Yenibertiz, D.; Akkurt, E. S.; Darcın, T.; Kes, Y. D.; Dal, M.S. Evaluating pre-transplant nutritional indices as predictors of pneumonia and mortality in multiple myeloma patients post-autologous stem cell transplantation. BMC Pulm. Med. 2025, 11(25), 517. [Google Scholar] [CrossRef] [PubMed]
- Neupane, K.; Singstock, M.; Shah, D.; Dahal, R.; Mian, H.; Smith, F.; Chakraborty, R.; Al Hadidi, S.; Mainou, M.; Grajales-Cruz, A.; et al. Inconsistent definitions of transplant ineligibility in multiple myeloma: A systematic review. Br. J. Haematol. 2026, 208, 829–836. [Google Scholar] [CrossRef]
- McCourt, O.; Fisher, A.; Ramdharry, G.; Roberts, A.L.; Land, J.; Rabin, N.; Yong, K. PERCEPT myeloma: a protocol for a pilot randomised controlled trial of exercise prehabilitation before and during autologous stem cell transplantation in patients with multiple myeloma. BMJ Open. 2020, 10, e033176. [Google Scholar] [CrossRef] [PubMed]
- Mawson, S.; Keen, C.; Skilbeck, J.; Ross, H.; Smith, L.; Dixey, J.; Walters, S.J.; Simpsond, R.; Greenfield, D.M.; Snowden, J.A. Feasibility and benefits of a structured prehabilitation programme prior to autologous stem cell transplantation (ASCT) in patients with myeloma; a prospective feasibility study. Physiotherapy 2021, 113, 88–99. [Google Scholar] [CrossRef]
- Möller, M.-D.; Ihorst, G.; Pahl, A.; Scheubeck, S.; Barsch, F.; Dold, S.M.; Bertz, H.; Arends, J.; Wäsch, R.; Engelhardt, M. Physical activity is associated with less comorbidity, better treatment tolerance and improved response in patients with multiple myeloma undergoing stem cell transplantation. J. Geriatr. Oncol. 2021, 12, 521–530. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.; Nathwani, N.; Shamunee, J.; Lindenfeld, L.; F. Wong, L.; Krishnan, A.; Armenian, S. Telehealth exercise to Improve Physical function and frailty in patients with multiple myeloma treated with autologous hematopoietic Stem cell transplantation (TIPS): protocol of a randomized controlled trial. Trials 2022, 23, 921. [Google Scholar] [CrossRef]
- Land, J.; Luong, M.K.; Longden, A.; Rabin, N.; Kyriakou, C.; Sive, J.; Fisher, A.; Yong, K.; McCourt, O. Real-world evaluation of physiotherapist-led exercise prehabilitation and rehabilitation during autologous stem cell transplantation in myeloma: a single-centre experience. BMJ Open. Qual. 2025, 14, e002936. [Google Scholar] [CrossRef]
- Li, X.; Li, S.; Chen, J. Fatigue as main symptom in an elderly multiple myeloma patient: a Case Report. Front. Med. 2025, 12, 1635403. [Google Scholar] [CrossRef]
- Dimopoulos, M.A.; Terpos, E.; Chanan-Khan, A.; Leung, N.; Ludwig, H.; Jagannath, S.; Niesvizky, R.; Giralt, S.; Fermand, J.-P.; Bladé, J.; et al. Renal Impairment in Patients With Multiple Myeloma: A Consensus Statement on Behalf of the International Myeloma Working Group. J. Clin. Oncol. 2010, 28, 4976–4984. [Google Scholar] [CrossRef] [PubMed]
- Dimopoulos, M.A.; Merlini, G.; Bridoux, F.; Leung, N.; Mikhael, J.; Harrison, S.J.; Kastritis, E.; Garderet, L.; Gozzetti, A.; van de Donk, N.W.C.J.; et al. Management of multiple myeloma-related renal impairment: recommendations from the International Myeloma Working Group. Lancet Oncol. 2023, 24, e293–e311. [Google Scholar] [CrossRef]
- Fotiou, D.; Theodorakakou, F.; Solia, E.; Spiliopoulou, V.; Ntanasis-Stathopoulos, I.; Malandrakis, P.; Psimenou, E.; Kanellias, N.; Roussou, M.; Migkou, M.; et al. Outcomes of Newly Diagnosed Multiple Myeloma Patients Requiring Dialysis. Clin. Lymphoma Myeloma Leuk. 2025, 25, 532–538. [Google Scholar] [CrossRef]
- Ling, X.; Yu, M. Application of individualized low-protein diet nursing for managing multiple myeloma patients with renal impairment. Medicine 2025, 104, e46087. [Google Scholar] [CrossRef]
- Ríos-Tamayo, R. Monoclonal Gammopathies. In Comprehensive Hematology and Stem Cell Research, 1st ed.; Rezaei, N., Ed.; Elsevier: Amsterdam, The Netherlands, 2024; pp. 358–374. [Google Scholar] [CrossRef]
- Chaudhry, H.M.; Mauermann, M.L.; Rajkumar, S.V. Monoclonal Gammopathy–Associated Peripheral Neuropathy: Diagnosis and Management. Mayo Clin. Proc. 2017, 92, 838–850. [Google Scholar] [CrossRef] [PubMed]
- Ropper, A.H.; Gorson, K.C. Neuropathies associated with paraproteinemia. N. Engl. J. Med. 1998, 338, 1601–1607. [Google Scholar] [CrossRef]
- Cheng, Y.; Sun, F.; D’Souza, A.; Dhakal, B.; Pisano, M.; Chhabra, S.; Stolley, M.; Hari, P.; Janz, S. Autonomic nervous system control of multiple myeloma. Blood Rev. 2021, 46, 100741. [Google Scholar] [CrossRef] [PubMed]
- Dispenzieri, A. POEMS syndrome: diagnosis, treatments, and outcomes. Hematol. Am. Soc. Hematol. Educ. Program. 2025, 2025(1), 393–400. [Google Scholar] [CrossRef] [PubMed]
- Davalos, L.; Kissel, J.T.; Barohn, R.J.; Dimachkie, M.M. Guillain-Barré Syndrome and Variants. Neurol. Clin. 2025, 43, 743–760. [Google Scholar] [CrossRef]
- Li, Z.; Huang, F.; Hao, S. Guillain-Barré syndrome in patients with multiple myeloma: three cases report and literature review. BMC Neurol. 2025, 25, 36. [Google Scholar] [CrossRef] [PubMed]
- Morawska, M.; Grzasko, N.; Kostyra, M.; Wojciechowicz, J.; Hus, M. Therapy-related peripheral neuropathy in multiple myeloma patients. Hematol. Oncol. 2015, 33, 113–119. [Google Scholar] [CrossRef]
- Wen, D.; Cao, S.; Feng, Y. Recent advances in the treatment and prevention of peripheral neuropathy after multiple myeloma treatment. Ibrain 2023, 9, 421–430. [Google Scholar] [CrossRef]
- Loureiro, J.; Costa-Pereira, J.T.; Pozza, D.H.; Tavares, I. The Power of Movement: How Exercise Influences Chemotherapy-Induced Peripheral Neuropathy. Biomedicines 2025, 13, 1103. [Google Scholar] [CrossRef]
- Yates, S.J.; Abdallah, N.H.; Christofyllakis, K.; VanderVeen, B.N.; Pardee, T.S.; Houston, D.K.; Kritchevsky, S.B.; Klepin, H.D. Sarcopenia in Older Adults with Hematologic Malignancies: A Comprehensive Review of Epidemiology, Prognosis, and Interventions. Cancers 2026, 18, 503. [Google Scholar] [CrossRef]
- Delaire, L.; Courtay-Breuil, A.; Humblot, J.; Vidal, H.; Bonnefoy, M.; Meugnier, E. Influence of Resistance Training Variables to Improve Muscle Mass Outcomes in Sarcopenia: A Systematic Review With Meta-Regressions. J. Cachexia Sarcopenia Muscle 2025, 16, e70162. [Google Scholar] [CrossRef]
- Araújo, L.P.; Figueiredo Godoy, A.C.; Fortes Frota, F.; Barbalho Lamas, C.; Quesada, K.; Rucco Penteado Detregiachi, C.; Cressoni Araújo, A.; Miglino, M.A.; Landgraf Guiguer, E.; Santos de Argollo Haber, R.; et al. Sarcopenia in the Aging Process: Pathophysiological Mechanisms, Clinical Implications, and Emerging Therapeutic Approaches. Int. J. Mol. Sci. 2025, 26, 12147. [Google Scholar] [CrossRef]
- Gertz, M.A. Immunoglobulin Light Chain Amyloidosis: 2026 Update on Diagnosis, Prognosis, and Treatment. Am. J. Hematol. 2026, 101, 1056–1069. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.K.; Callander, N.S.; Adekola, K.; Anderson, L.D.; Baljevic, M.; Baz, R.; Campagnaro, E.; Costello, C.; D’Angelo, C.; Derman, B.; et al. Multiple Myeloma, Version 5.2026. J. Natl. Compr. Canc. Netw. 2026, 24, e260001. [Google Scholar] [CrossRef]
- Ríos-Tamayo, R. Systemic Light Chain Amyloidosis: An Always Challenging Landscape, 1st ed.; Nova: New York, USA, 2026. [Google Scholar] [CrossRef]
- Barret, E.M.; Hussey, J.; Darker, C.D. Feasibility of a physical activity pathway for Irish primary care physiotherapy services. Physiotherapy 2017, 103, 106–112. [Google Scholar] [CrossRef] [PubMed]
- Galan-Martin, M.A.; Montero-Cuadrado, F.; Luch-Girbes, E.; Coca-López, M.C.; Mayo-Iscar, A.; Cuesta-Vargas, A. Pain Neuroscience Education and Physical Therapeutic Exercise for Patients with Chronic Spinal Pain in Spanish Physiotherapy Primary Care: A Pragmatic Randomized Controlled Trial. J. Clin. Med. 2020, 9, 1201. [Google Scholar] [CrossRef]
- Zhang, K.V.; Vallabhaneni, S.; Alvarez-Cardona, J.A.; Krone, R.J.; Mitchell, J.D.; Lenihan, D.J. Cardiac Amyloidosis for the Primary Care Provider: A Practical Review to Promote Earlier Recognition of Disease. Am. J. Med. 2021, 134, 587–595. [Google Scholar] [CrossRef]
- Mikhael, J.; Bhutani, M.; Cole, C.E. Multiple Myeloma for the Primary Care Provider: A Practical Review to Promote Earlier Diagnosis Among Diverse Populations. Am. J. Med. 2023, 136, 33–41. [Google Scholar] [CrossRef]
- Kugler, H.L.; Brusco, N.K.; Taylor, N.F. Physiotherapists may influence hospital patient physical activity levels indirectlythrough workforce training. Physiother. Theory Pract. 2025, 41, 1181–1190. [Google Scholar] [CrossRef]
- Mayer, K.P.; Johnson, A.M.; Smith, D.J.; Crandall, C.M.; Johnson, M.D.; Fresenko, L.E.; Robinson, C.M.; Robinson, L.E.; Kaplan, S.L.; Kumble, S.; et al. A Core Set of Outcome Measures to Assess Physical Function for Adults Participating in Physical Therapist Treatment in the Hospital: A Clinical Practice Guideline. Phys. Ther. 2025, 105, pzaf076. [Google Scholar] [CrossRef] [PubMed]
- Damien Guinault, D.; Canet, E.; Huart, A.; Jaccard, A.; Ribes, D.; Lavayssiere, L.; Venot, M.; Cointault, O.; Roussel, M.; Nogier, M.-B.; et al. Short- and long-term outcomes of AL amyloidosis patients admitted into intensive care units. Br. J. Haematol. 2016, 174, 868–875. [Google Scholar] [CrossRef]
- Nair, R.; Patel, K. What the Intensivists Need to Know About Critically Ill Myeloma Patients. Oncol.-Crit. Care 2019, 9, 1513–1530. [Google Scholar] [CrossRef]
- Hamazaki, N.; Ogura, K. Implementation and Challenges of Early-Phase Physiotherapy During Cardiovascular Intensive Care. Circ. Rep. 2026, 8, 21–26. [Google Scholar] [CrossRef] [PubMed]
- Lewko, A.; Eggmann, S.; Ragna Amundadóttir, O.; Astrup, K.; Hugo Azevedo, V.; Battaglini, D.; Benko Mestrovic, S.; Dowds, J.; Duska, F.; Eriksson, F.; Hästbacka, J.; Hernández-Méndez, B.; et al. Physiotherapy provision and practice: A EuropeAn Intensive CarE Unit (ICU) survey (PEAiCE) – A profile of ICU physiotherapy in Europe. Intens. Crit. Care Nur. 2026, 95, 104390. [Google Scholar] [CrossRef]
- Hayes, C.; Conneely, M.; McNamara, R.; Quinn, R.; French, H.; Desmeules, F.; Kilcawley, R.; McCreesh, K.; Galvin, R. What is the role of advanced physiotherapy practice for adults in urgent care and emergency department settings? A scoping review protocol. HRB Open. Res. 2025, 8, 116. [Google Scholar] [CrossRef]
- Habib, M.H.; Tiger, Y.K.R.; Dima, D.; Schlögl, M.; McDonald, A.; Mazzoni, S.; Khouri, J.; Williams, L.; Anwer, F.; Raza, S. Role of Palliative Care in the Supportive Management of AL Amyloidosis—A Review. J. Clin. Med. 2024, 13, 1991. [Google Scholar] [CrossRef] [PubMed]
- Samala, R.V.; Valent, J.; Noche, N.; Lagman, R. Palliative Care in Patients with Multiple Myeloma. J. Pain. Symptom Manag. 2019, 58, 1113–1118. [Google Scholar] [CrossRef]
- Bigi, F.; Fiacchini, M.; Restuccia, R.; Masci, S.; Mazzocchetti, G.; Talarico, M.; Puppi, M.; Sacchetti, I.; Manzato, E.; Iezza, M.; et al. Ten-year experience on home care for patients with plasma cell disorders: bringing optimal therapy home. Clin. Exp. Med. 2025, 25, 179. [Google Scholar] [CrossRef]
- Boyer, P.; Burns, D.; Whyne, C. Evaluation of at-home physiotherapy. Bone Jt. Res. 2023, 12, 165–177. [Google Scholar] [CrossRef]
- Purdy, G.M.; Sobierajski, F.M.; Al Onazi, M.M.; Effa, C.J.; Venner, C.P.; Tandon, P.; McNeely, M.L. Exploring participant perceptions of a virtually supported home exercise program for people with multiple myeloma using a novel eHealth application: a qualitative study. Support. Care Cancer 2023, 25(31), 298. [Google Scholar] [CrossRef]
- Herold, F.; Theobald, P.; Gronwald, T.; Kaushal, N.; Zou, L.; de Bruin, E.D.; Bherer, L.; Müller, N.G. The Best of Two Worlds to Promote Healthy Cognitive Aging: Definition and Classification Approach of Hybrid Physical Training Interventions. JMIR Aging 2024, 7, e56433. [Google Scholar] [CrossRef]
- Schmitz, K.H.; Demanelis, K.; Crisafio, M.E.; Kennedy, M.A.; Schwartz, A.L.; Campbell, A.; Gorzelitz, J.; Wood, K.C.; Wilson, C.M.; Scalise, R.L.; et al. Proximity to cancer rehabilitation and exercise oncology by geography, race, and socioeconomic status. Cancer 2025, e35515. [Google Scholar] [CrossRef] [PubMed]
- Bickton, F.M.; Manifield, J.R.; Limbani, F.; Dixon, J.; Holland, A.E.; Taylor, R.S.; Calderwood, C.; Wittich, W.; Gregson, C.L.; Heine, M.; et al. Protocol for the development and validation of a Core Set for exercise-based rehabilitation of adults with multiple long-term conditions (multimorbidity) based on the World Health Organization’s International Classification of Functioning, Disability,and Health (ICF) framework. J. Multimorb. Comorb. 2025, 15, 1–14. [Google Scholar] [CrossRef]
- Shapiro, Y.N.; Peppercorn, J.M.; Yee, A.J.; Branagan, A.R.; Raje, N.S.; O’Donnell, E.K. Lifestyle considerations in multiple myeloma. Blood Cancer J. 2021, 11, 172. [Google Scholar] [CrossRef]
- Kisner, C.; Colby, L.A.; Borstad, J. Therapeutic Exercise: Foundations and Techniques, 7th ed; Davis Company: Philadelphia, USA, 2017. [Google Scholar]
- Brody, L.T. Effective Therapeutic Exercise Prescription: The Right Exercise at the Right Dose. J. Hand Ther. 2012, 25, 220–232. [Google Scholar] [CrossRef] [PubMed]
- Couto, D.S.; Lopes, I.; Oliveira, M.I.; Schmidt, C.; Magalhaes, S.; Dores, H.; Ribeiro, F.; Santos, M. Exercise intensity prescription in heart failure: A comparison of different physiological parameters. Rev. Port. Cardiol. 2025, 44, 361–371. [Google Scholar] [CrossRef]
- Levinger, I.; Giuliano, C.; Holland, D.J.; Taylor, J.L.; Howden, E.J.; Naylor, L.; Pearson, M.J.; Williams, A.D.; Green, D.J.; Smart, N.A.; et al. Exercise & Sports Science Australia Position Statement on exercise training in patients with chronic heart failure: 2025 update. J. Sci. Med. Sport 2026. [Google Scholar] [CrossRef]
- Mendes Wefelnberg, M.; Niels, T.; Holtick, U.; Jundt, F.; Scheid, C.; Baumann, F.T. Clinical exercise therapy program with multiple myeloma patients: Impacts on feasibility, adherence and efficacy. Support. Care Cancer 2022, 30, 9615–9623. [Google Scholar] [CrossRef]
- De Sena Anchieta Rodrigues, M.; de Melo Atanasio, L.; Kelly dos Santos, I.; Gomes da Silva, J.C.; de Araujo Tinoco Cabral, B.G.; Moreira Silva Dantas, P. Efficacy of Interventions to Promote Exercise Adherence in People With Overweight or Obesity: A Systematic Review. J.Obes. 2025, 4164477. [Google Scholar] [CrossRef]
- Cesnik, R.; Kunstler, B.; Toohey, K.; Freene, N.; Semple, S. Barriers to physical activity levels in people with cancer who are undergoing chemotherapy: a narrative systematic review with mapping to the capability, opportunity, motivation behaviour (COM-B) model. Support. Care Cancer 2025, 34, 20. [Google Scholar] [CrossRef] [PubMed]
- D’Souza, A.; Szabo, A.; Akinola, I.; Finkel, K.M.; Flyn, K.E. A cross-sectional study of patient-reported outcomes and symptom burden using PROMIS and PRO-CTCAE measures in light chain amyloidosis. Qual. Life Res. 2023, 32, 1807–1817. [Google Scholar] [CrossRef]
- Dittrich, T.; Weinert, L.; D’Souza, A. Patient-reported outcomes - the missing link to advancing light chain (AL) amyloidosis clinical research. Blood Rev. 2025, 74, 101303. [Google Scholar] [CrossRef] [PubMed]
- Niscola, P.; Scaramucci, L.; Efficace, F. Towards the integration of patient-reported outcomes into the global clinical management of multiple myeloma. Expert Rev. Hematol. 2019, 12, 703–705. [Google Scholar] [CrossRef] [PubMed]
- Shah, D.; Sparano, F.; Shah, V.; Krepper, D.; Baldi, T.; Chakraborty, R.; Efficace, F. Quality of Patient-Reported Outcomes in Multiple Myeloma Randomized Controlled Trial Protocols and Publications According to the SPIRIT-PRO and CONSORT-PRO guidelines: A Systematic Review. Eur. J. Haematol. 2025, 115, 454–464. [Google Scholar] [CrossRef]
- Abdallah, N.; Bohra, A.; Mammadzadeh, A.; Buadi, F.; Kapoor, P.; Dispenzieri, A.; Gertz, M.; Hayman, S.; ElHaj, M.; Dingli, D.; et al. Prognostic impact of patient-reported symptoms in multiple myeloma. Blood Cancer J. 2025, 9, 884–892. [Google Scholar] [CrossRef]
- Dwyer Orr, L.; Lin, D.; Wu, B.; LeBlanc, T.W.; Faiman, B.; Ahlstrom, J.; Yung, M.; Deering, K.L.; Kulbokas, V.; Feldman, J.L.; et al. Patient, Care Partner, and Physician Voices in Treatment Decision-Making for Multiple Myeloma. Patient Prefer. Adherence 2024, 18, 2147–2158. [Google Scholar] [CrossRef]
- Ballan-Haj, M.; Midlej, K.; Silverman, B.; Draliuk, R.; Mishchenko, E.; Valkovsky, O.; Perek, S.; Warwar, A.; Zoabi, I.; Preis, M. Assessing the correlation between patient and family reported outcome measures and multiple myeloma clinical parameters. Sci. Rep. 2025, 15, 45172. [Google Scholar] [CrossRef]
- Talbot, A.; Delcour, B.; Gillot, L.; Arnulf, B.; Avet Loiseau, H.; Boccaccio, C.; Frenzel, L.; Karlin, L.; Kraeuter, K.; Macro, M.; et al. Patient-Centered Education Through A Massive Open Online Course (MOOC) for Patients With Multiple Myeloma and Caregivers: Descriptive Study of Knowledge Gains by French Association of Patients With Multiple Myeloma (AF3M) and French-Speaking Myeloma Intergroup (IFM). JMIR Form. Res. 2026, 10, e81225. [Google Scholar] [CrossRef] [PubMed]
- Duan, L.; Cai, P.; Zhang, Z.; Yang, Y.; Wen, X.; Chu, X. Research trends on exercise and cancer pain relationships: a bibliometric analysis over the past three decades. Support. Care Cancer 2025, 34, 7. [Google Scholar] [CrossRef]
- Le Berre, M.; Yousefi, F.; Sowanou, A.; Laberge, M. Patients’ and physiotherapists’ perspectives on group-based interventions: a mixed systematic review. Arch. Phys. Med. Rehabil. 2026. [Google Scholar] [CrossRef]
- Li, Y.; Jiang, X.; Zhu, M.; Li, X.; Wang, Y. Efficacy of music therapy on mild cognitive impairment in the aging population. A systematic review and meta-analysis. Medicine 2026, 105, 3. [Google Scholar] [CrossRef]
- Mulia, G.J.; Chiang, Y.-H.; Maringka, S.G.; Wu, J.C.-C.; Ma, H.-P.; Ou, J.-C.; Chen, K.-Y. Cognitive enhancement through music therapy: meta-analytic evidence across clinical population. Front. Public Health 2026, 13, 1735470. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.; Gao, B.; Zhou, R. Meta-analysis of the effects of dance- and movement-based kinesthetic games on cognitive function in older adults with cognitive impairment (CI) under different intervention periods. Aging Clin. Exp. Res. 2025, 37, 343. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Sun, Y.; Han, Q.; Sun, Q.; Jiang, Z.; Ma, W. The effects of virtual reality-based interventions on cognitive function, depressive symptoms, and daily functioning in older adults with mild cognitive impairment: a systematic review and meta-analysis of randomized controlled trials. Front. Public Health 2026, 13, 1682781. [Google Scholar] [CrossRef]
- Soong, R.Y.; Low, C.E.; Ong, V.; Sim, I.; Lee, C.; Lee, F.; Chew, L.; Yau, C.E.; Lee, A.; Chen, M.Z. Exercise interventions for depression, anxiety, and quality of life in older adults with cancer: a systematic review and meta-analysis. JAMA Netw. Open. 2025, 8, e2457859. [Google Scholar] [CrossRef]
- Liu, R.; Kywe Moe, A.A.; Zheng, Z.; Alanazi, H.; Zoghi, M.; Jaberzadeh, S. Effects of laser acupuncture on pain and motor function in musculoskeletal disorders: A systematic review and meta-analysis. Complement. Ther. Med. 2026, 96, 103323. [Google Scholar] [CrossRef]
- Wu, C.-H.; Chang, C.-N.; Chang, C.-F.; Lin, M.-H.; Chen, H.-Y.; Chen, Y.-C. Clinical Usability of Exercise Prescription Apps for Professional Use: Systematic Review and Multidimensional Evaluation. JMIR Mhealth Uhealth 2026, 14, e77616. Available online: https://mhealth.jmir.org/2026/1/e77616. [CrossRef]
- Febbraio, M.A.; Klarlund Pedersen, B. Exercise as a therapeutic intervention for long-lasting and chronic diseases. Cell. Metab. 2026. [Google Scholar] [CrossRef]
- Nazha, A.; Elemento, O.; Ahuja, S.; Lam, B.; Miles, M.; Shouval, R.; McWeeney, S.; Sirhan, S.; Srisuwananukorn, A.; Haferlach, T. Artificial intelligence in hematology. Blood 2025, 146, 2283–2292. [Google Scholar] [CrossRef] [PubMed]
- Yan, W.; Chen, J.; He, T.; Chen, J.; Liu, X.; Zhong, L.; Li, Y.; Sun, C.; Wang, X.; Gao, L.; et al. Development of an artificial intelligence-based early diagnostic system for light-chain amyloidosis. Ann. Hematol. 2025, 104, 6315–6326. [Google Scholar] [CrossRef]
- Wu, D.; Liu, X.; Li, X.; Chen, J.; Lin, X.; Fang, L.; Chen, W. Beyond NT-proBNP and troponin: How machine learning redefines light-chain cardiac amyloidosis risk assessment. BMC Med. Inform. Decis. Mak. 2025, 25, 367. [Google Scholar] [CrossRef]
- Wan, X.; Ge, L.; Tian, J.; Ding, G.; Gao, B.; Geng, H.; Ge, C.; Song, X. Integrating Artificial Intelligence Into Digital Health-Optimized Therapeutics for the Remote Cardiac Rehabilitation. Clin. Med. Insights Cardiol. 2026, 20, 11795468261421344. [Google Scholar] [CrossRef] [PubMed]
- Bland, K.A.; Catalá-Vilaplana, I.; Nunez, J.J.; Capozzi, L.C.; Campbell, K.L. Artificial Intelligence Meets Cancer Rehabilitation: Emerging Evidence for Exercise and Physical Activity Interventions. Cancer Control 2026, 33, 10732748261432280. [Google Scholar] [CrossRef]
- Heo, S.; Choi, T.; Choi, W. Clinical Validation of an On-Device AI-Driven Real-Time Human Pose Estimation and Exercise Prescription Program; Prospective Single-Arm Quasi-Experimental Study. Healthcare 2026, 14, 482. [Google Scholar] [CrossRef]
- Enichen, E.J.; Young, C.C.; Frates, E.P. The Potential of AI to Create Personalized Exercise Plans. Health Promot. Pract. 2025, 15248399251394695, Online ahead of print. [Google Scholar] [CrossRef]
- Eckardt, J.-N.; Walter, W.; Middeke, J.M. Artificial intelligence for risk assessment and outcome prediction in malignant haematology. Br. J. Haematol. 2026, 208, 25–38. [Google Scholar] [CrossRef]
- Hussain, Z.; De Brouwer, E.; Boiarsky, R.; Setty, S.; Gupta, N.; Liu, G.; Li, C.; Srimani, J.; Zhang, J.; Labotka, R.; et al. Joint AI-driven event prediction and longitudinal modeling in newly diagnosed and relapsed multiple myeloma. Npj Digit. Med. 2024, 7, 200. [Google Scholar] [CrossRef]
- Canzone, A.; Belmonte, G.; Patti, A.; Vicari, D.S.S.; Rapisarda, F.; Giustino, V.; Drid, P.; Bianco, A. The multiple uses of artificial intelligence in exercise programs: a narrative review. Front. Public Health 2025, 13, 1510801. [Google Scholar] [CrossRef]
- Fernando, M.; Lange, E. New ways of working as a physiotherapist in the digital era: A qualitative study. Physiother. Pract. Res. 2025, 46, 103–110. [Google Scholar] [CrossRef]
- Fernandes, M.; Souto-Mirnada, S.; Pires, D.; Jácome, C.; Ribeiro, F.; Cordeiro, N.; Cruz, E. The Use of Electronic Health Records in Physiotherapy Practice: Protocol for a Scoping Review. J. Eval. Clin. Pract. 2025, 31, e70324. [Google Scholar] [CrossRef] [PubMed]
- Krittanawong, C.; Sagar Maitra, N.; Hassan Virk, H.U.; Fogg, S.; Wang, Z.; Kaplin, S.; Gritsch, D.; Storch, E.A.; Tobler, P.N.; Charney, D.S.; et al. Association of Optimism with Cardiovascular Events and All-Cause Mortality: Systematic Review and Meta-Analysis. Am. J. Med. 2022, 135, 856–863. [Google Scholar] [CrossRef] [PubMed]
- Mantzari, E.; Brain, K.; Bessems, K.M.H.H.; Bigaard, J.; Bouaoun, L.; D’Souza, E.; Espina, C.; Fonseca, C.; Feliu, A.; Petrova, D.; et al. Optimising the European Code Against Cancer, 5th edition, to increase awareness of avoidable cancer risks in all socioeconomic groups. Mol. Oncol. 2026, 20, 154–169. [Google Scholar] [CrossRef] [PubMed]
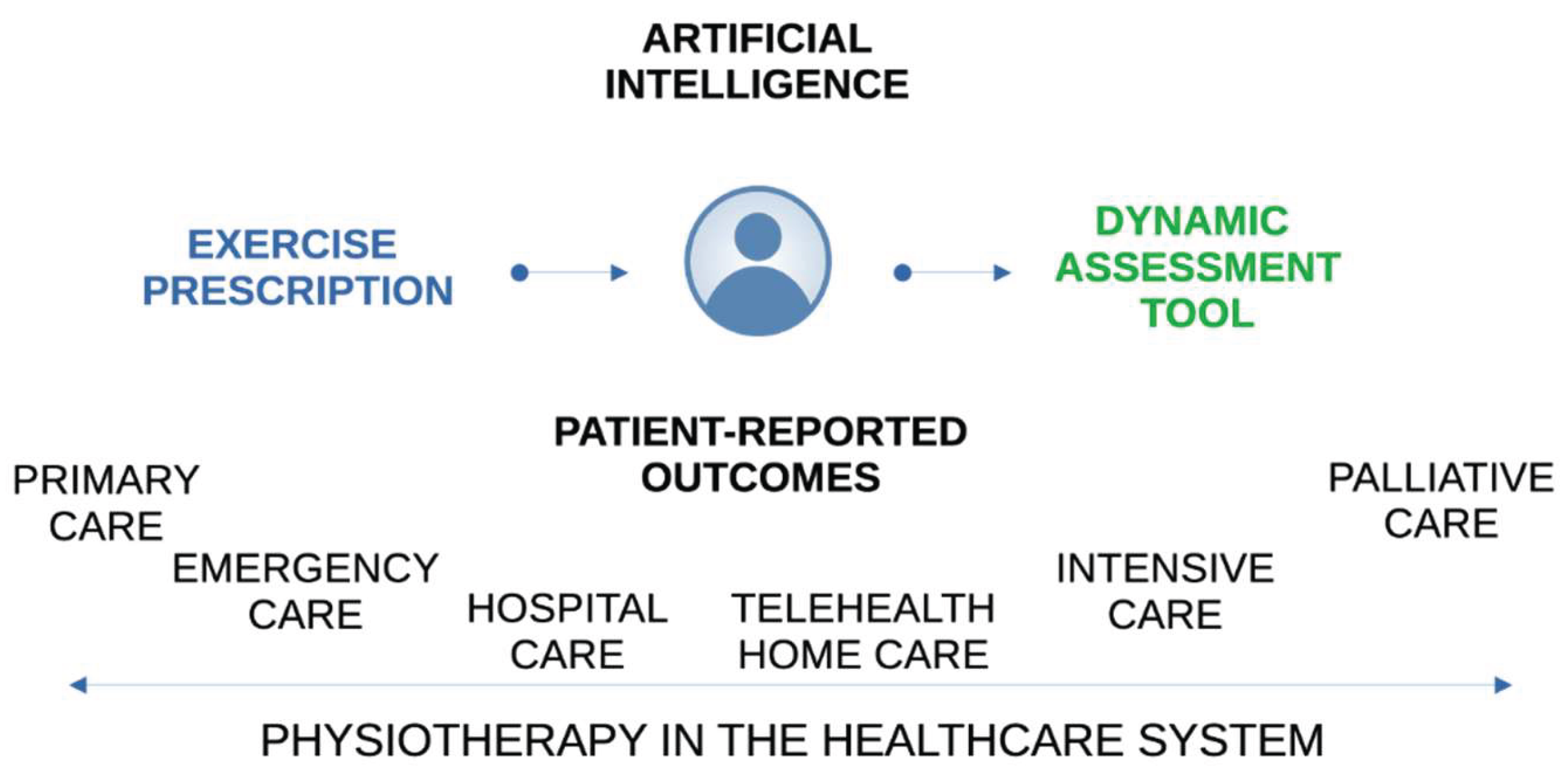
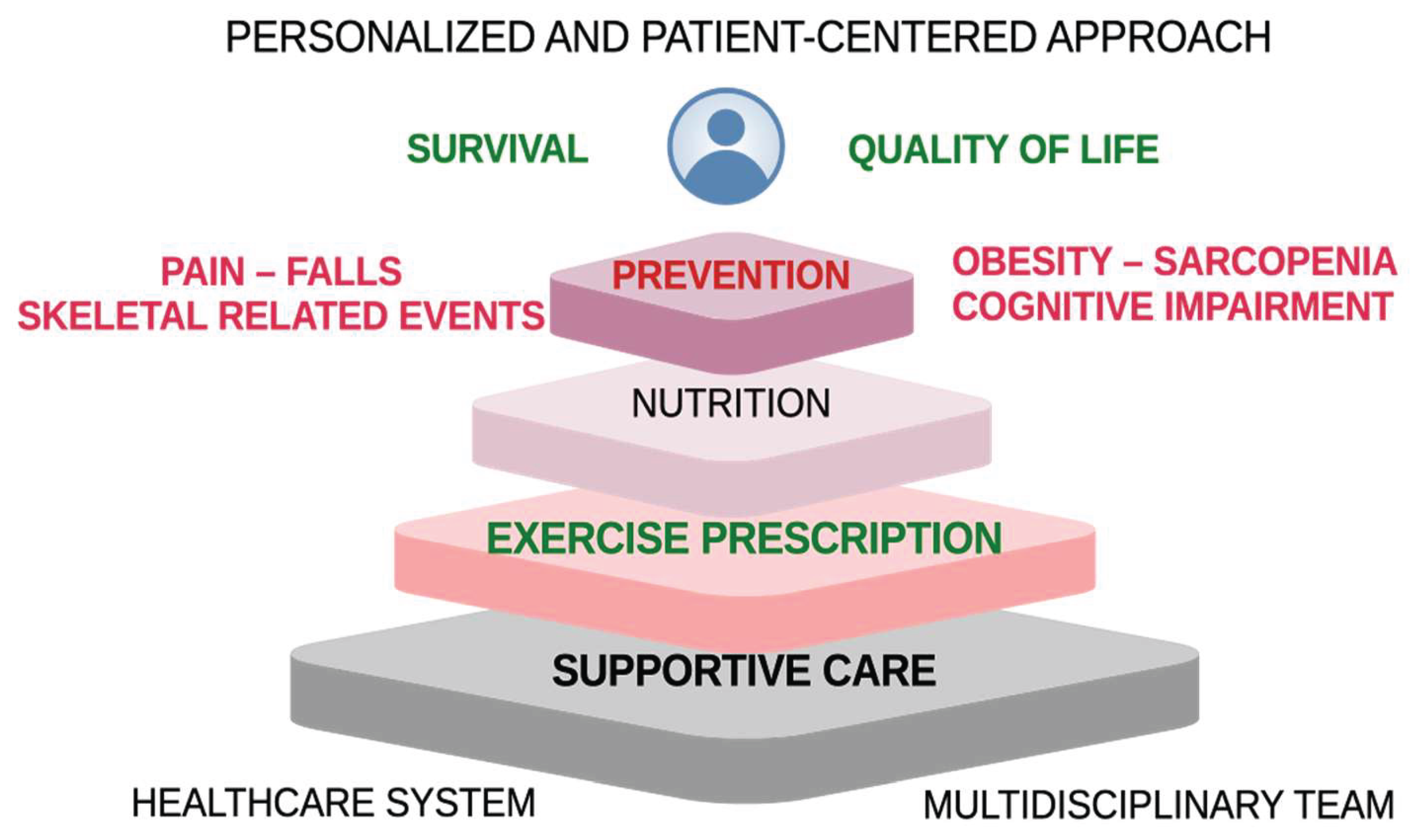
| AL | MM | |
|---|---|---|
| Scope of healthcare | M | M |
| Type of healthcare | M | M |
| Requesting person | M | M |
| Level of urgency | M | M |
| Clinical target | M | M |
| Patient location | M | M |
| Native language | M | M |
| Age | M | M |
| Sex | M | M |
| Weight, height, BMI | M | M |
| ECOG-PS | M | M |
| Comorbidities | M | M |
| Staging | M | M |
| Hemoglobin | M | M |
| Serum calcium | M | M |
| eGFR | M | M |
| Imaging | M | M |
| Current therapy | M | M |
| Clinical trial | M | M |
| Six-minute walk test | R | R |
| HRQoL | R | R |
| Nutrition score | R | R |
| Frailty score | R | R |
| Issues | Present | Future |
|---|---|---|
| Scope | Hospital-based | Full spectrum |
| Setup | Department | Multidisciplinary unit |
| Focus | Treatment | Prevention |
| Approach | Clinical | Holistic AI-based |
| Imaging | On demand | On-site |
| Baseline study | Heterogeneity | Standardization |
| Follow-up | Variable | Dynamic |
| Geriatric assessment | Recommended | Integrated |
| Frailty score | Punctual | Systematic |
| Nutritional score | Punctual | Systematic |
| Comorbidity | Variable | Standardized |
| HRQoL | Old tools | PROs, e-platforms |
| Patient-staff interaction | Physical visit | Real-time tele-health |
| Exercise prescription | Adapted | Personalized |
| Outcome measures | Variable | Standardized |
| AL Research | Limited | Collaborative trials |
| Contextualized PT | Heterogeneity | Optimized |
| Patient education | Weak | Strong |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




