Submitted:
23 April 2026
Posted:
27 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Methodology
3. Development of Waste–Free Technology
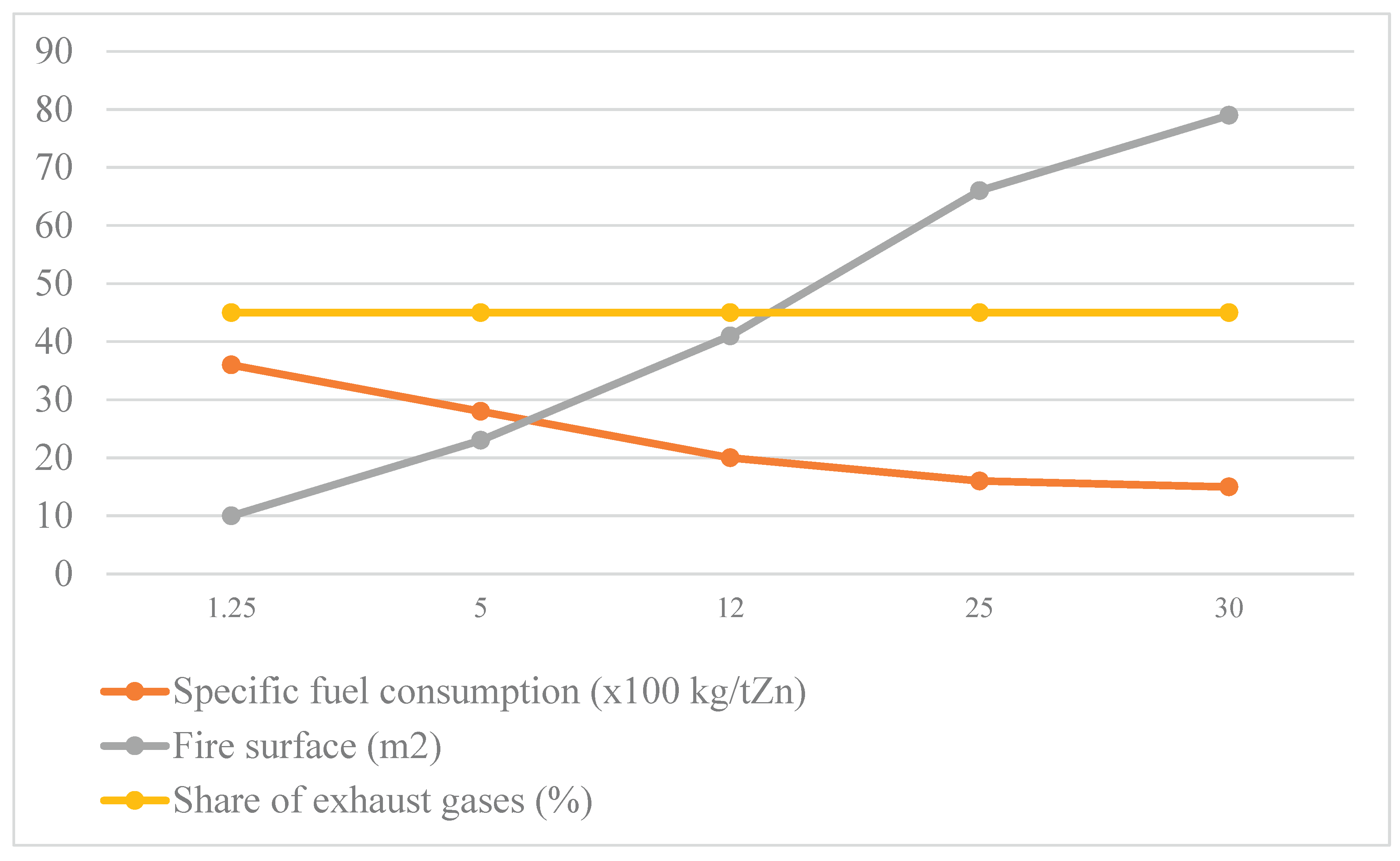
3.1. Calculation Method for Industrial Melting Reactor Characteristics
3.2. Experiments on the Extraction of Zinc and Germanium from Fluxed Ash–Slag
3.3. Experiment Results of Cast Stone Production
3.4. Experiment Results Discussion
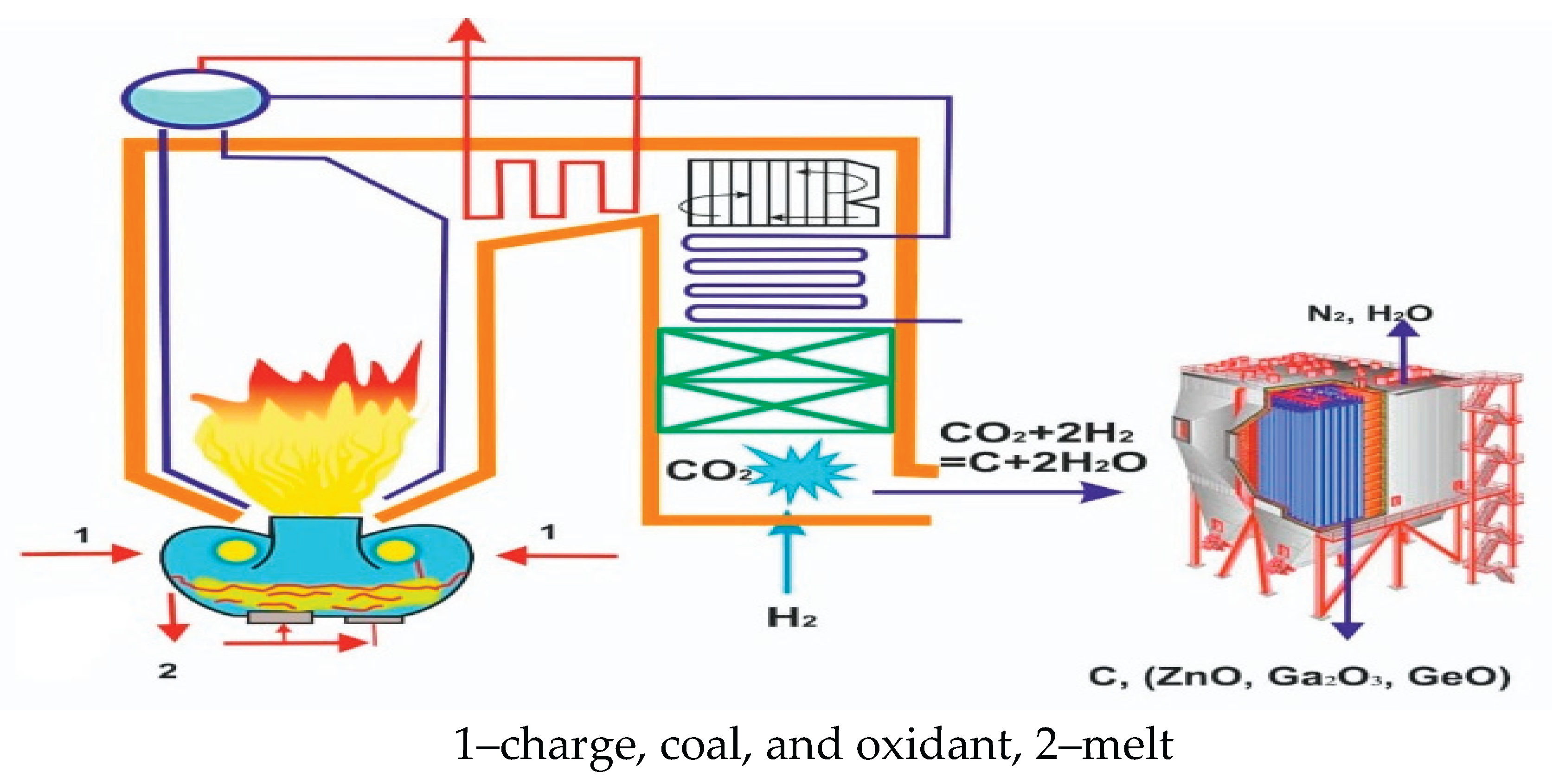
4. Development of Carbon–Free Technology
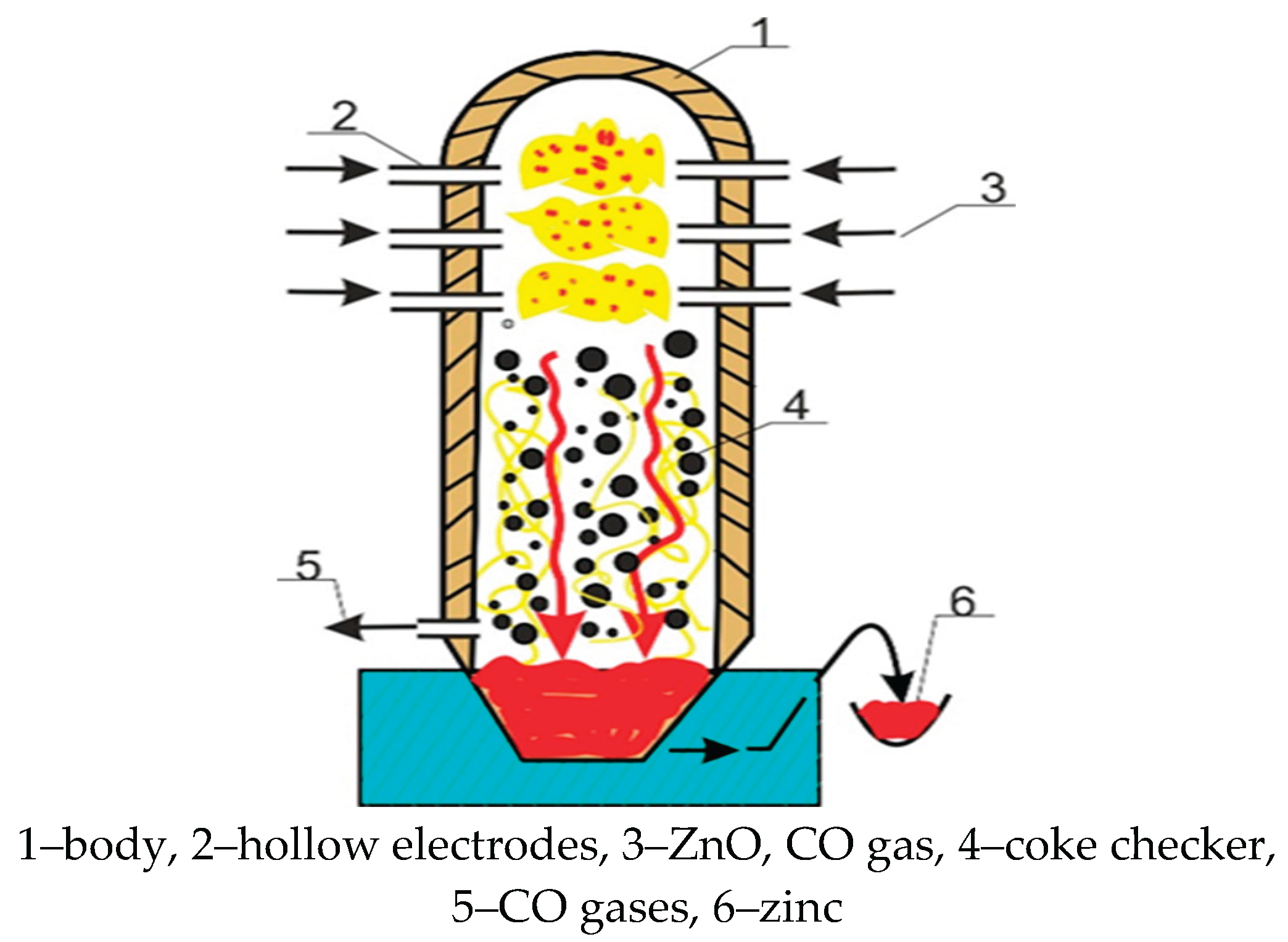
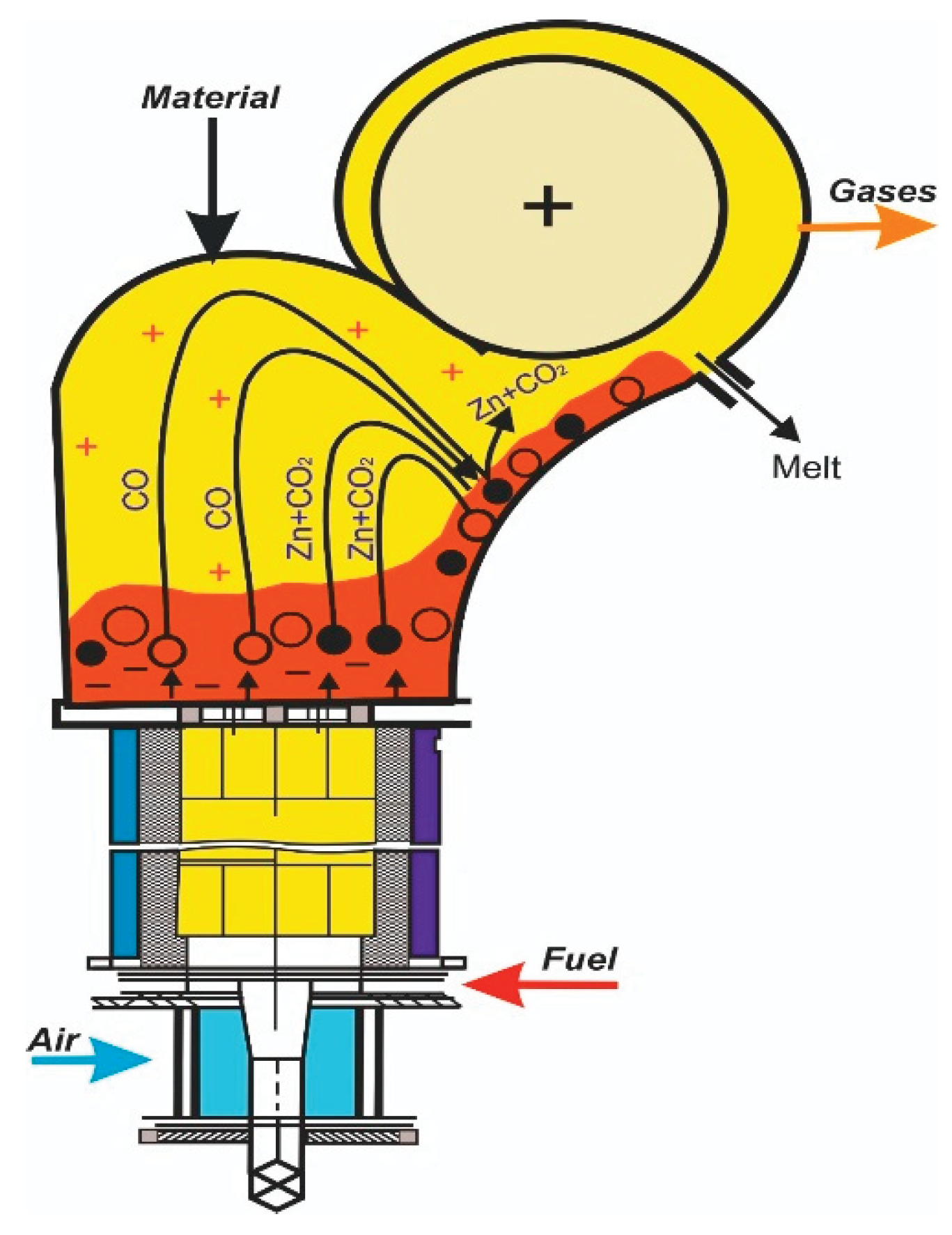
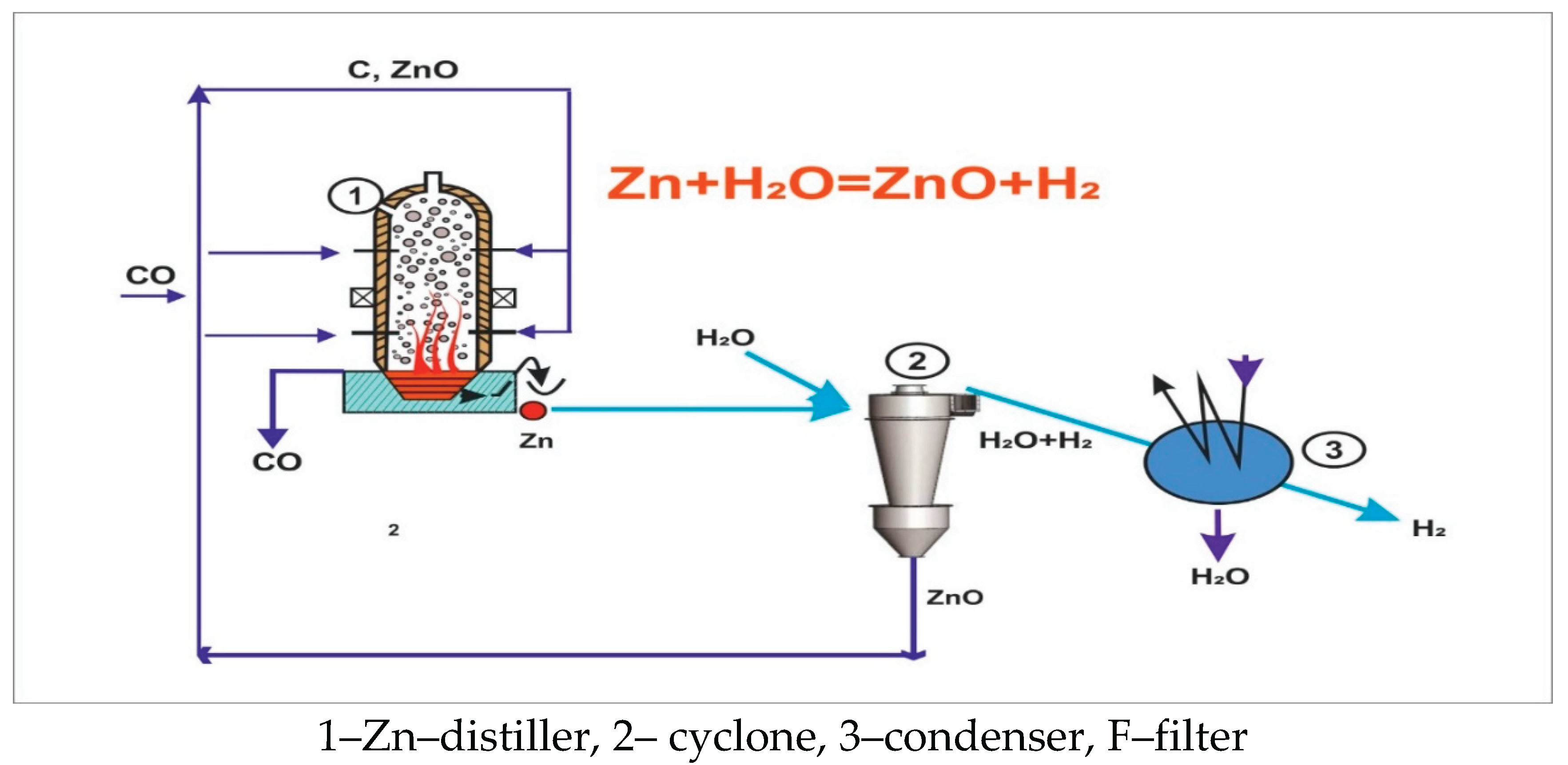
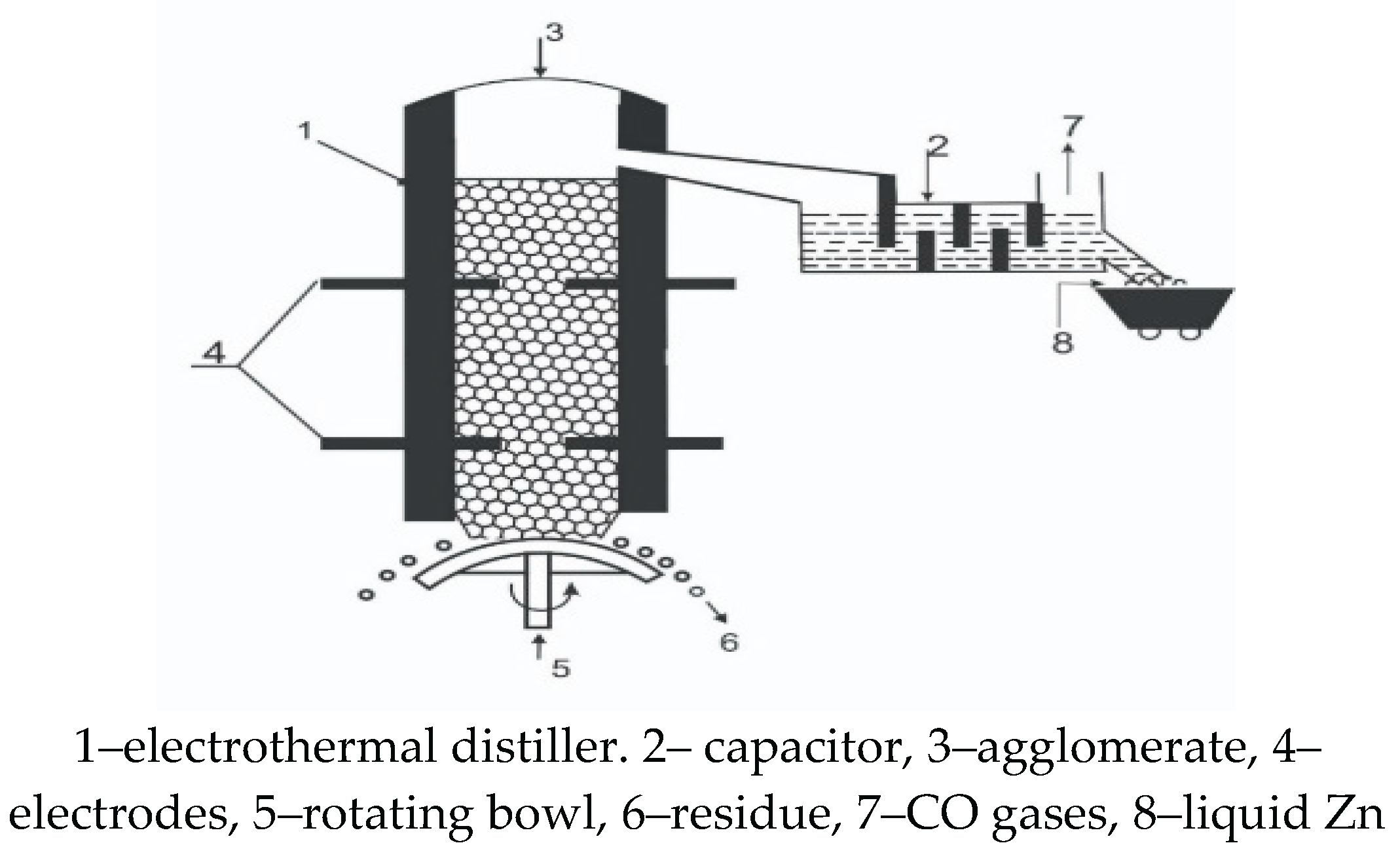
4.1. Estimation of Specific Electrical Energy Consumption for a Zinc Distiller
| # | Heat input | Q, kJ | % | # | Heat output | Q, kJ | % |
|---|---|---|---|---|---|---|---|
| 1 | 819 | 91.57 | 1 | 2790 | 317.9 | ||
| 2 | 75.4 | 8.43 | 2 | With purge “CO” gas |
29 | 3.3 | |
| 3 | Total | 894.4 (0.2484 kWh) |
100 | 3 | 250 | 28.48 | |
| Zn | 4 | –2011 | -229.15 | ||||
| 5 | The difference in the heat of incoming and outgoing zinc consumed in the distiller |
–504 | -57.43 | ||||
| 6 | The difference in the heat of incoming and outgoing “CO” consumed in the distiller |
–46.4 | -5.28 | ||||
| 7 | With liquid zinc |
200 | 22.79 | ||||
| 8 | With slag |
20 | 2.27 | ||||
| 9 | Losses to the environment through distiller surfaces, ~17% | 150 | 17.1 | ||||
| Total | 877.6 | 100 | |||||
4.2. Assessment of the Possibility of Complete Neutralization of CO2 Gas in the Proposed System
5. Assessment of Economic Indicators of the Proposed Technology
6. Conclusions
- (1)
- A novel smelting reactor has been proposed for the combustion of Ekibastuz coal in power plant boilers, utilizing a combined "ideal mixing–ideal displacement" approach. This method reduces energy consumption by two to three times compared to existing technologies.
- (2)
- Experiments on a smelting reactor in continuous mode have demonstrated the possibility of waste-free processing of coal ash, with a degree of extraction of germanium and zinc above 70%, and the processing of the silicate portion of the slag into stone casting.
- (3)
- A new method for calculating the parameters of a melting reactor has been developed. Based on this, it was established that with an increase in reactor productivity from 1.25 to 30 t/h, the specific fuel consumption will decrease by 2.3 times.
- (4)
- Based on the new method of “counter colliding dust and gas jets”, a new distiller design has been proposed, where distillation and condensation of zinc are carried out in one chamber. A tenfold reduction in energy consumption in the distiller compared to a traditional zinc distiller is estimated.
- (5)
- A novel zinc method technology, for hydrogen production, and using it for carbon reduction from its dioxide, has been developed. This approach enables a reduction of specific energy consumption for H2 to one-third that of the electrolysis process. Additionally, the feasibility of supplementary hydrogen processing from distiller-associated CO gas through the water-gas displacement reaction has been proposed.
- (6)
- A comparison of the thermodynamic characteristics of hydrogen reduction of iron from Fe2O3 in the Midrex (USA) and Corex (Austria) processes with the proposed method of hydrogen reduction of carbon from CO2 leads to the conclusion that this method can be implemented.
- (7)
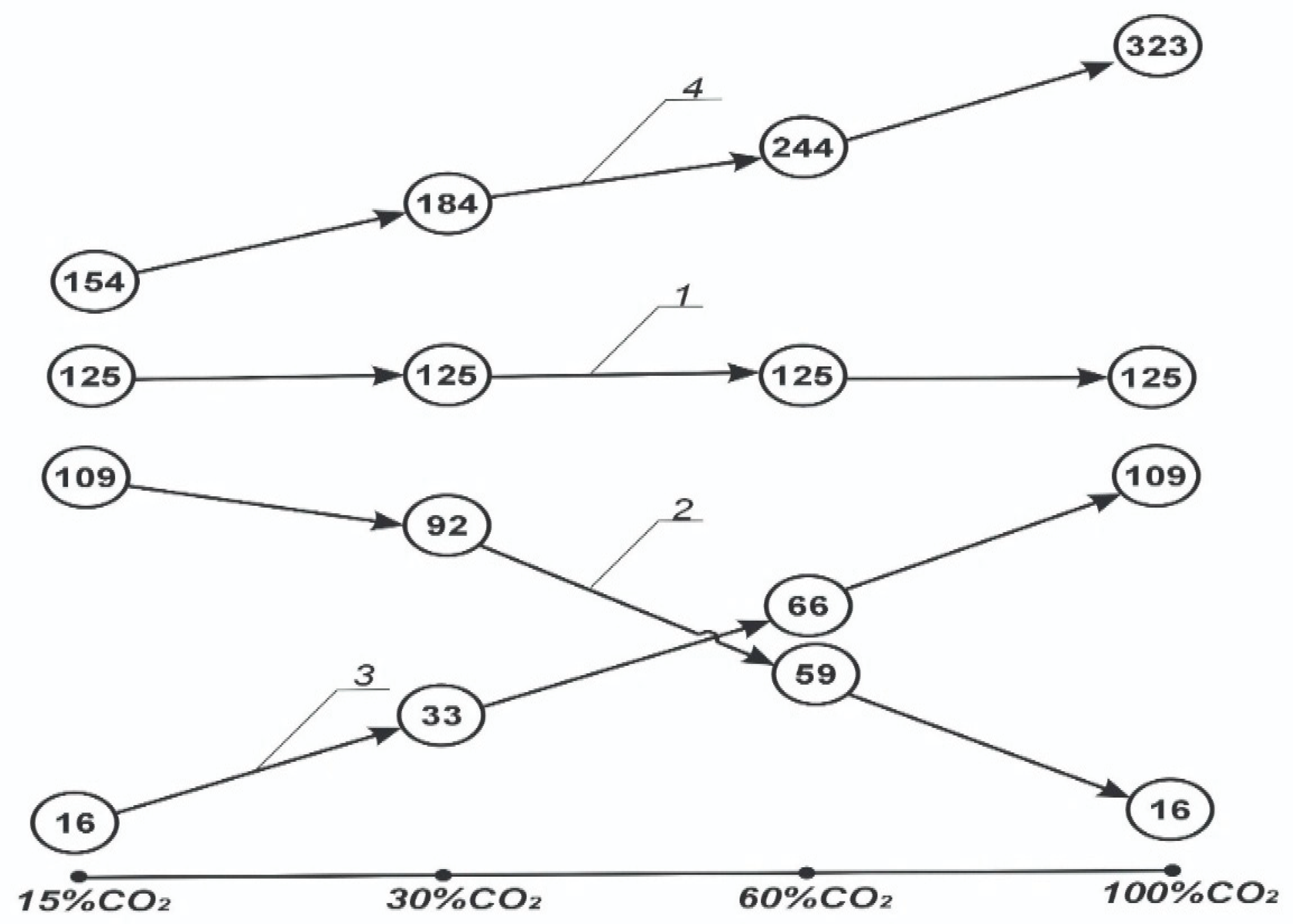
- For the BKZ–420 boiler, analysis indicates that attaining carbon neutrality by 2060 while sustaining a power output of 125 MW will require increasing the boiler’s capacity to offset energy losses in the distiller. Specifically, during the period from 2030 to 2060, the necessary boiler power must be raised from 154 MW (representing a 1.23-fold increase) to 323 MW (a 2.58-fold increase).
- (8)
- An assessment of the economic efficiency of processing Ekibastuz coal with 15% decarbonization of gases shows that the average payback period of the proposed system can be ~8 years.
- (9)
- Implementing this technology allows coal-fired plants to keep existing infrastructure, eliminating the need for new nuclear facilities or costly conversions to natural gas. It reduces the immediate pressure to shift fully to renewables by improving both efficiency and environmental performance, ensuring reliable power and avoiding major infrastructure expenses.
Conflicts of Interest
Nomenclature
| Nomenclature |
|
number of kilomoles in combustion products per 1 kilomole of initial carbon (km/km); carbon activity in the system,–; – number of moles of carbon in coal (km/km); (km/km); –number of oxygen moles in the oxidizer, (km/km); –total number of moles of nitrogen reacted (km/km); – pyrometric coefficient –; P–reactor’s productivity for charge (kg/h); – gas pressure in the reactor (mm w.c.); – temperature in the melt bath (); mass of the bath (kg); – the productivity of the reactor (kg/h); – ratio of the momentum of gases in the nozzles of the purge grate to the weight of bath–; – ratio of the initial and final concentration of zinc in the slag–; – ratio of the initial and final content of germanium in the slag–; E – the degree of extraction of zinc and germanium from the slag (%); the consumption of propane in the reactor inversion phase (m3/h); – air consumption (m3/h); – oxygen consumption for the process (m3/h); ΔG – the change of Gibbs (free) energy (kJ); – initial and final particle concentration of the zinc in the melt–; τ – sublimation time of zinc in the distiller (min); |
| Acronyms |
| BKZ–420 – steam boiler with productivity 420t/h; DAC – direct air capture; OG – outgoingt gases; |
References
- Sumer, V., Zhanaltay, Z., & Parkhomchik, L. (2019). Renewable Energy in Kazakhstan: Potential and Challenges. Renewable Energy. [CrossRef]
- Ahmad, S., Nadeem, A., Akhanova, G., Houghton, T., & Muhammad–Sukki, F. (2017). Multi–criteria evaluation of renewable and nuclear resources for electricity generation in Kazakhstan. Energy, 141, 1880–1891. [CrossRef]
- Blyda I. A., Slyusarenko L. I., Abisheva Z. “Zoloshlakovie othody energetiki – syrie dly proizvodstva redkih metallov i glinozema. Kompleksnoe ispolzovanie miniralnogo Syria. [Ash and slag waste from the power industry – raw material for the production of rare metals and alumina. Complex use of mineral raw materials]”. no. 4. (2008): 39–51. (In Russian).
- Delitsyn, L., Kulumbegov, R., Klimenko, N., Popel’, O., Sulman, M., Sinelschikov, V., Sychev, G., & Petropavlovsky, K. (2024). Enrichment of Iron–containing Concentrate with Ammonium Hydrofluoride. Ecology and Industry of Russia. [CrossRef]
- Fanchi, John R., Fanchi, Christopher J. “Energy in the 21st Century”. World Scientific Publishing Co Inc., (2016): 350. ISBN 978–981–314–480–4.
- Birch, E. L. “A Review of “Climate Change 2014: Impacts, Adaptation, and Vulnerability” and “Climate Change 2014: Mitigation of Climate Change.” Journal of the American Planning Association, no 80(2). (2014): 184–185. [CrossRef]
- Rhodes, J. S., Keith, D. W. “Biomass with capture: negative emissions within social and environmental constraints: an editorial comment”. Climatic Change, no 87(3–4) (2008): 321–328. [CrossRef]
- Werner, C., Schmidt, H.–P., Gerten, D., Lucht, W., Kammann, C. “Biogeochemical potential of biomass pyrolysis systems for limiting global warming to 1.5 °C”. Environmental Research Letters, 13(4), (2018): 044036. [CrossRef]
- Tomasz Czakiert, Jaroslaw Krzywanski, Anna Zylka Wojciech Nowak. Chemical Looping Combustion: A Brief Overview. Energies 2022, 15(4), 1563. [CrossRef]
- Wadim Strielkowski, Anna Sherstobitova, Patrik Rovny and Tatiana Evteeva. Increasing Energy Efficiency and Modernization of Energy Systems in Russia: A Review. Energies. May 2021Energies 14(11):3164. [CrossRef]
- Raid J. Hassiba, Patrick Linke. On the simultaneous integration of heat and carbon dioxide in industrial parks. Applied Thermal Engineering. Volume 127, 25 December 2017, Pages 81-94.
- Fang, Z., & Gesser, H. (1996). Recovery of gallium from coal fly ash. Hydrometallurgy, 41, 187–200. [CrossRef]
- Bai, R., Huang, Y., Li, S., Su, C., Li, H., Cui, Q., Liu, W., Wei, H., & Si, Y. (2025). Study on Extraction of Gallium from Fly Ash by Sodium Fluoride Calcination–Organic Acid Leaching. Sustainability. [CrossRef]
- Zhao, Z., Cui, L., Guo, Y., Jianming, G., Li, H., & Cheng, F. (2021). A stepwise separation process for selective recovery of gallium from hydrochloric acid leach liquor of coal fly ash. Separation and Purification Technology, 265, 118455. [CrossRef]
- Junjun Xu, Haibin Zuo, Guangwei Wang, Jianliang Zhang, Ke Guo, Wang Liang. Gasification mechanism and kinetics analysis of coke using distributed activation energy model (DAEM) Applied Thermal Engineering Volume 152, April 2019, Pages 605–614. [CrossRef]
- Kaan Büyükkanber, Hanzade Haykiri–Acma, Serdar Yaman. Calorific value prediction of coal and its optimization by machine learning based on limited samples in a wide range. Energy. Volume 277, 15 August 2023, 127666. [CrossRef]
- Outokumpu HSC Chemistry for Windows. Chemical Reaction and Equilibrium Software with Extensive Thermochemical Database. Version 5.1. October 31, 2012.
- Dikhanbaev A. B., Dikhanbaev B. I., Koshumbaev M. B., Ybrai S. B. On the issue of converting the exhaust gases of the phase inversion reactor into artificial gas. Bulletin of Toraigyrs University. ISSN 2710–3420. Energy series. No. 1. 2022. Republic of Kazakhstan, Pavlodar. (In Russian). [CrossRef]
- Dikhanbaev B.I., Dikhanbaev A.B. Monograph. Energy–efficient technologies for processing industrial waste. Aktaulova's LLP. ISBN 978–601–257–438–8. – Astana. – 2024. – 445p.
- Vagin V. V., Kileso V. V., Scotarekko Z. G. Products of ferrous metallurgy. Industry catalogue. Stone casting. –Moscow. –2008.–6p. (in Russian).
- Dikhanbaev, Bayandy, Chandima Gomes, and Aristan Bayandievich Dikhanbaev. "Design and implementation of an energy–saving melting reactor." Case Studies in Thermal Engineering 26 (2021): 101003. [CrossRef]
- Dikhanbaev B., Dikhanbaev A., Chandima G. “Energy–saving method for technogenic waste processing”. Journal PLoS ONE, no 12(12): – San Francisco, California, USA. (2017): 1–16. [CrossRef]
- Sumida, K., Rogow, D. L., Mason, J. A., McDonald, T. M., Bloch, E. D., Herm, Z. R., ... & Long, J. R. (2012). Carbon dioxide capture in metal–organic frameworks. Chemical reviews, 112(2), 724–781. [CrossRef]
- Habib Mohamed A, Nemitallah Medhat A. Design of an ion transport membrane.
- reactor for application in fire tube boilers. Energy 2015;81:787–801. [CrossRef]
- Remiorz Leszek, Wiciak Grzegorz, Grzywnowicz Krzysztof. Novel concept of supporting the membrane separation of CO2 in power plants by thermoacoustic dehumidification. Energy Peltola, P., Saari, J., Tynjälä, T., & Hyppänen, T. (2020). Process integration of chemical looping combustion with oxygen uncoupling in a biomass–fired combined heat and power plant. Energy, 210, 118550. [CrossRef]
- H. Wang, Z.G. Qu, W. Zhang, Y.X. Chang, Y.L. He. Experimental and numerical study of CO2 adsorption on Ni/DOBDC metal-organic framework. Applied Thermal Engineering. Volume 73, Issue 2, 22 December 2014, Pages 1501-1509.
- Durakovic Goran del Granado, Crespo Pedro, Tomasgard Asgeir. Are green and blue hydrogen competitive or complementary? Insights from a decarbonized European power system analysis. Energy November 2023;282:128282. [CrossRef]
- Matus, E. V., Ismagilov, I. Z., Yashnik, S. A., Ushakov, V. A., Prosvirin, I. P., Kerzhentsev, M. A., & Ismagilov, Z. R. (2020). Hydrogen production through autothermal reforming of CH4: Efficiency and action mode of noble (M= Pt, Pd) and non-noble (M= Re, Mo, Sn) metal additives in the composition of Ni-M/Ce0. 5Zr0. 5O2/Al2O3 catalysts. International Journal of Hydrogen Energy, 45(58), 33352-33369.
- Shchukin AA. Gas and furnace facilities of factories. M: – Publishing house "Energy;1973. p. 232p (in Russian).
- Marchenko, N. V., E. P. Vershinina, E. M. Hildebrandt. Metallurgy of heavy non–ferrous metals. ISBN 978–5–7638–1776–8. Krasnoyarsk: IPK SFU, 2009. 394 p. (in Russian).
- Sidenko P.M.. Grinding in the chemical industry. Moscow. Publishing house "Chemistry" –1968. 384s. (in Russian).

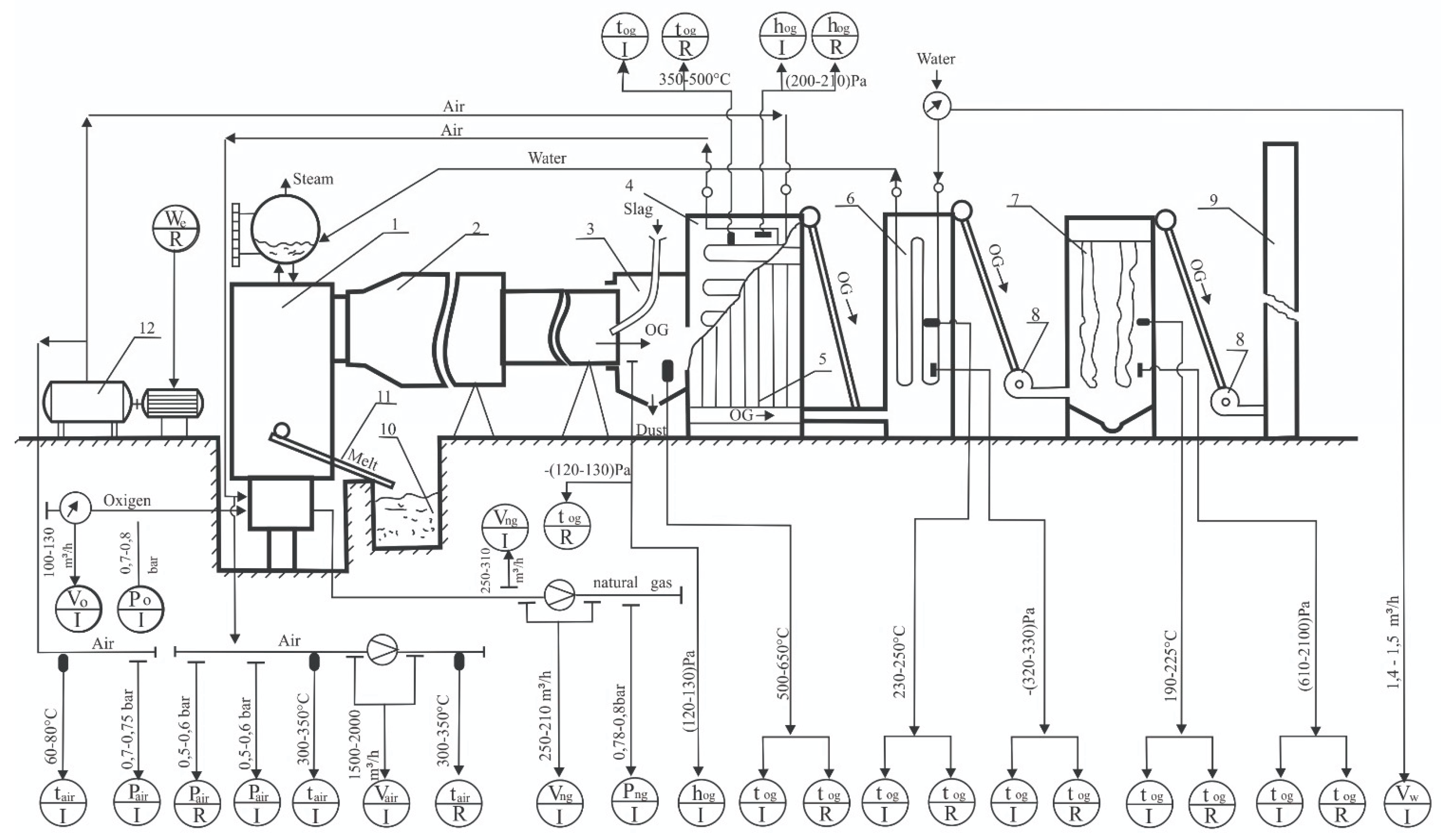
| Measured Value | Limit of Error | Name, Type | Measuring Range |
| Natural gas consumption |
At maximum gas flow - (±1)%, at minimum - (±2)% | Explosion-Proof Vortex Flow Meter. SUP-FVX480. HANGZHOU SUPMEA INTERNATIONAL TRADING CO.,LTD. Process Connection and Body Material: HG/T20592 Flange Connection, 304SS; Accuracy: Class 1.5; Compensation Method: Temperature and Pressure Compensation; Output, Display, and Power Supply: Pulse + Two-wire 4-20mA, Display, 24VDC; Heat Resistance Temperature: -40-250℃; | From 200m3/h to 300m3/h |
| Oxygen consumption | From (±1)% to (±2)% | Explosion-proof Vortex Flow meter.SUP-FVX480. HANGZHOU SUPMEA INTERNATIONAL TRADING CO.,LTD. Process Connection and Body Material: HG/T20592 Flange Connection, 304SS; Accuracy: Class 1.5; Compensation Method: Temperature and Pressure Compensation; Output, Display, and Power Supply: Pulse + Two-wire 4-20mA, Display, 24VDC; Heat Resistance Temperature: -40-250℃; |
From 100m3/h to 200m3/h |
| Air consumption | ±0.15% | Explosion-proof Vortex Flow Meter. SUP-FVX480. HANGZHOU SUPMEA INTERNATIONAL TRADING CO.,LTD. Measuring Medium: air; Accuracy: Class 1.5; Compensation Method: Temperature and Pressure Compensation; Output, Display, and Power Supply: Pulse + Two-wire 4-20mA, Display, 24VDC; Heat Resistance Temperature: -40-250℃; |
From 900m3/h to 2000m3/h |
| Collection of analogue signals from temperature, pressure, rarefaction sensors, their conversion and transfer to a computer | - | Programmable microprocessor controllers: -ADAM -4018, 8-channel analogue input module, 2pcs. - ADAM-4520, RS-232, RS-485 interface signal converter, 1 pc. |
One to 16 input channels |
| Flue gas temperature downstream of the rotary kiln, air heater, and scrubber | 0.01 of measured medium temperature | Thermoelectric converters Metran, Russia. Type TXA-0292K, 4 pieces | From (-400°C) to (+1,000°C) |
| The pressure of natural gas and blast air in front of the furnace | From (±0.25)% to (±0.5)% | Overpressure sensors Sapphire 22M-DI, Russia, model 2140, 2 pcs. | 0~250 kPa |
| The composition of the combustion products behind the reactor, rotary kiln, air heater and scrubber | From (±5%) to (±1%) | Manual automatic gas analyzer with display and printer, "Crowcon Sprint", Kane International Ltd. | CO2-(0.0÷15%); CO-(0.0-15%); O2(0-30%) Maximum sample temperature 800°C |
| Melt temperature in the melting reactor | ±1°C | Optical pyrometer " Smotrych-5P-01" |
From 800°C to 1,500°C |
| Parameters | 1 Experiment | 2 Experiment |
|---|---|---|
| , mm water column. | 50 – 100 | 50 – 100 |
| , °C | 1,300 – 1,400 | 1,300 – 1,400 |
| 200 | 240 | |
| 1200 | 1500 | |
| 0.36 | 0.42 | |
| 2.15/0.43 | 2.5/0.5 | |
| , gr/t | 76/15 | 80/16 |
| 80 | 79 | |
| 270 | 280 | |
|
|
1350 110 |
1380 120 |
| , °C | 300 | 290 |
| , in sublimates,% | 27 | 20 |
| 1 | Melt temperature at the exit from the tap hole,°C | 1300 – 1350 |
| 2 | Melt viscosity, poise | 8 – 7 |
| 3 | Specific heat flux through water–cooled reactor caissons, kW/m2 | 130 – 140 |
| 4 | Specific gravity, g/cm3 | 2.2 – 2.5 |
| 5 | Water absorption‚% | 0.13 |
| 6 | Compressive strength, kgf/cm2 | 2700 |
| 7 | Abrasion loss, g/cm2 | 0.21 – 0.24 |
| 8 | Softening temperature, °C | 1200 |
| 9 | Acid resistance in hydrochloric acid, % | 96.7 |
| 10 | Grain (crystal) size, microns | 80 – 150 |
| 11 | Heat resistance, heat changes | 20 – 25 |
| 2H2+CO2=2H2O+C | ||
| T | ΔG | K |
| C | kJ | |
| 100 | –55,752 | 6,382E+007 |
| 200 | –45,922 | 1,175E+005 |
| 300 | –35,718 | 1,801E+003 |
| 9H2 +3Fe2O3=6Fe+ 9H2O | ||
| T | deltaG | K |
| C | kJ | |
| 700 | -50,028 | 4,848E+002 |
| 800 | -74,737 | 4,346E+003 |
| 900 | -99,217 | 2,618E+004 |
| H2O+COH2+CO2 | ||
| T | deltaG | K |
| C | kJ | |
| 200 | -21,469 | 2,346E+002 |
| 300 | -17,618 | 4,034E+001 |
| 400 | -13,928 | 1,205E+001 |
| № | Name quantities |
Units | Degree of decarbonization |
|||
|---|---|---|---|---|---|---|
| 15%CO2 | 30%CO2 | 60%CO2 | 100%CO2 | |||
| 1 | Electrical energy | MWh | 154.0 | 184.52 | 244.13 | 323.33 |
| 2 | Ekibastuz coal | t/h | 90.56 | 101.64 | 123.3 | 152.1 |
| 3 | Yield of CO2 from boiler | t/h | 41.36 | 84.1 | 168.2 | 280.18 |
| 4 | Reusable zinc | t/h | 122.2 | 248.43 | 497.05 | 827.65 |
| 5 | Hydrogen for carbon recovery from CO2 | t/h | 3.76 | 7.37 | 15.3 | 25.47 |
| 6 | Carbon for a distiller as a checker | t/h | 45.28 | 50.82 | 61.65 | 76.05 |
| 7 | Melt suitable for the production of construction products | t/h | 65.65 | 73.69 | 89.39 | 110.27 |
| 8 | Zinc oxide in sublimates (ZnO-2% in ash) | t/h | 0.905 | 1.016 | 1.233 | 1.521 |
| 9 | Germanium oxides in sublimates (GeO2, Ge2O3-100 g/t) | kg/h | 4.528 | 5.082 | 6.165 | 7.605 |
| 10 | Carbon monoxide (CO) from a distiller | t/h | 105.65 | 118.58 | 143.86 | 177.45 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




