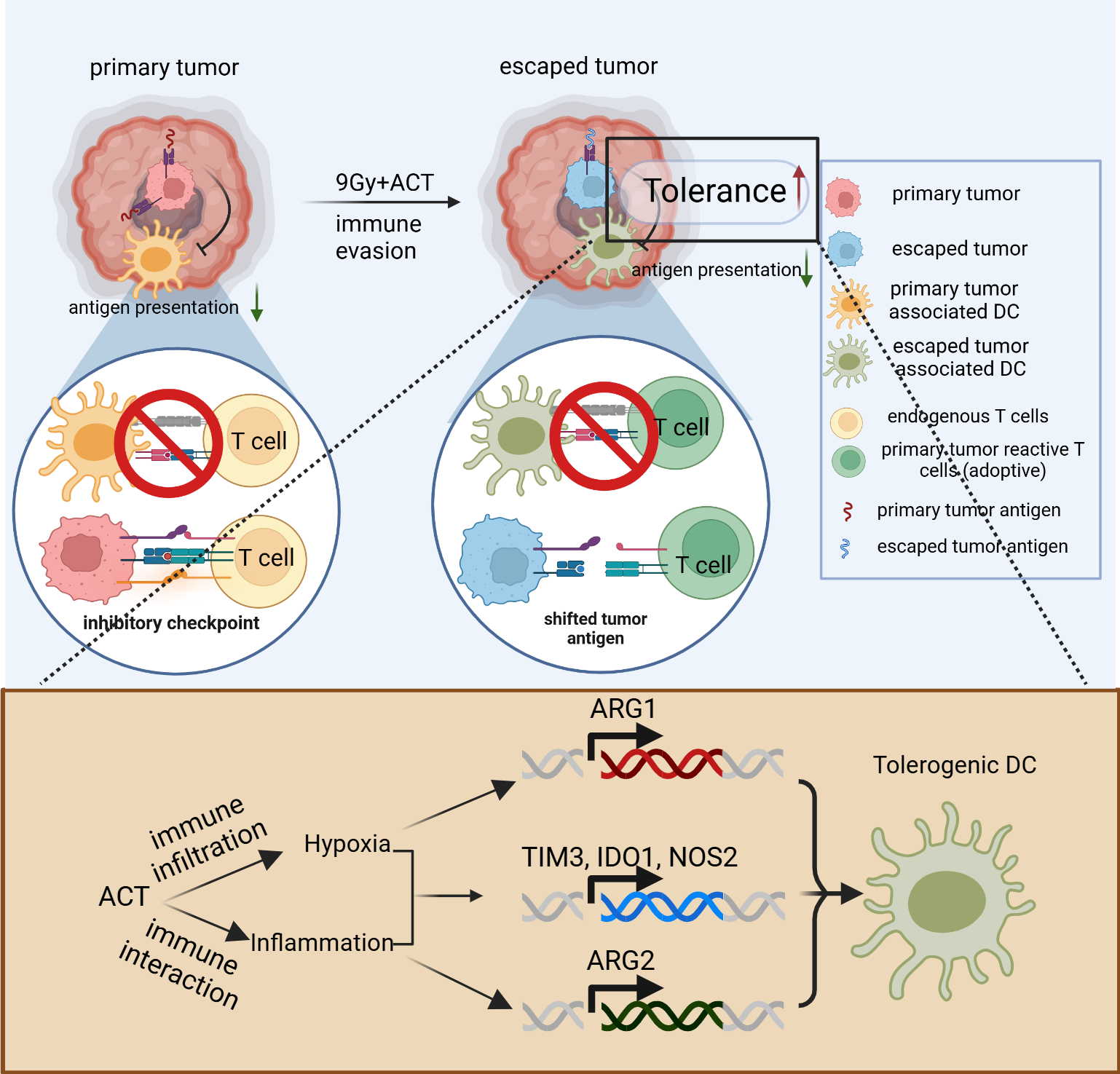
Background/Objectives: Glioblastoma (GBM) remains a lethal primary CNS malignancy with limited response to immunotherapy. Adoptive cellular therapy (ACT) improves survival in preclinical models, yet tumors ultimately recur. While T cell exhaustion is a common mechanism of resistance, the contribution of dendritic cell (DC) dysfunction remains unclear. We aimed to define mechanisms of immune escape following ACT, focusing on DC function and the role of hypoxia. Methods: Using a murine glioma model (KR158B), mice were treated with ACT consisting of tumor RNA–pulsed DC vaccines and adoptively transferred T cells. Tumor-infiltrating immune populations were analyzed by flow cytometry. DC function was assessed using T cell activation assays. Bulk RNA sequencing and gene set enrichment analysis were performed on sorted DCs. Hypoxia was modeled in vitro, and HIF1α was perturbed using CRISPR-mediated knockout. Results: ACT significantly increased survival but did not prevent tumor recurrence. Escaped tumors contained abundant cytotoxic, non-exhausted T cells, indicating that T cell dysfunction was not the primary driver of resistance. Instead, tumor-associated DCs exhibited impaired T cell activation despite preserved antigen uptake. Transcriptomic analyses revealed reduced antigen presentation and co-stimulatory signaling, alongside increased expression of tolerogenic factors. ACT-treated tumors demonstrated heightened hypoxia pathway activation, with elevated HIF1α expression in DCs. Hypoxia induced DC tolerogenic programs and reduced their ability to activate T cells, an effect partially reversed by HIF1α disruption. Increased immune infiltration and inflammation following ACT further amplified hypoxia signaling. Conclusions: DC dysfunction is a key mechanism of immune escape following ACT in glioma. Hypoxia-driven tolerization of DCs impairs sustained anti-tumor immunity, highlighting the hypoxia–DC axis as a promising therapeutic target to enhance immunotherapy efficacy.