Submitted:
23 April 2026
Posted:
24 April 2026
You are already at the latest version
Abstract
Keywords:
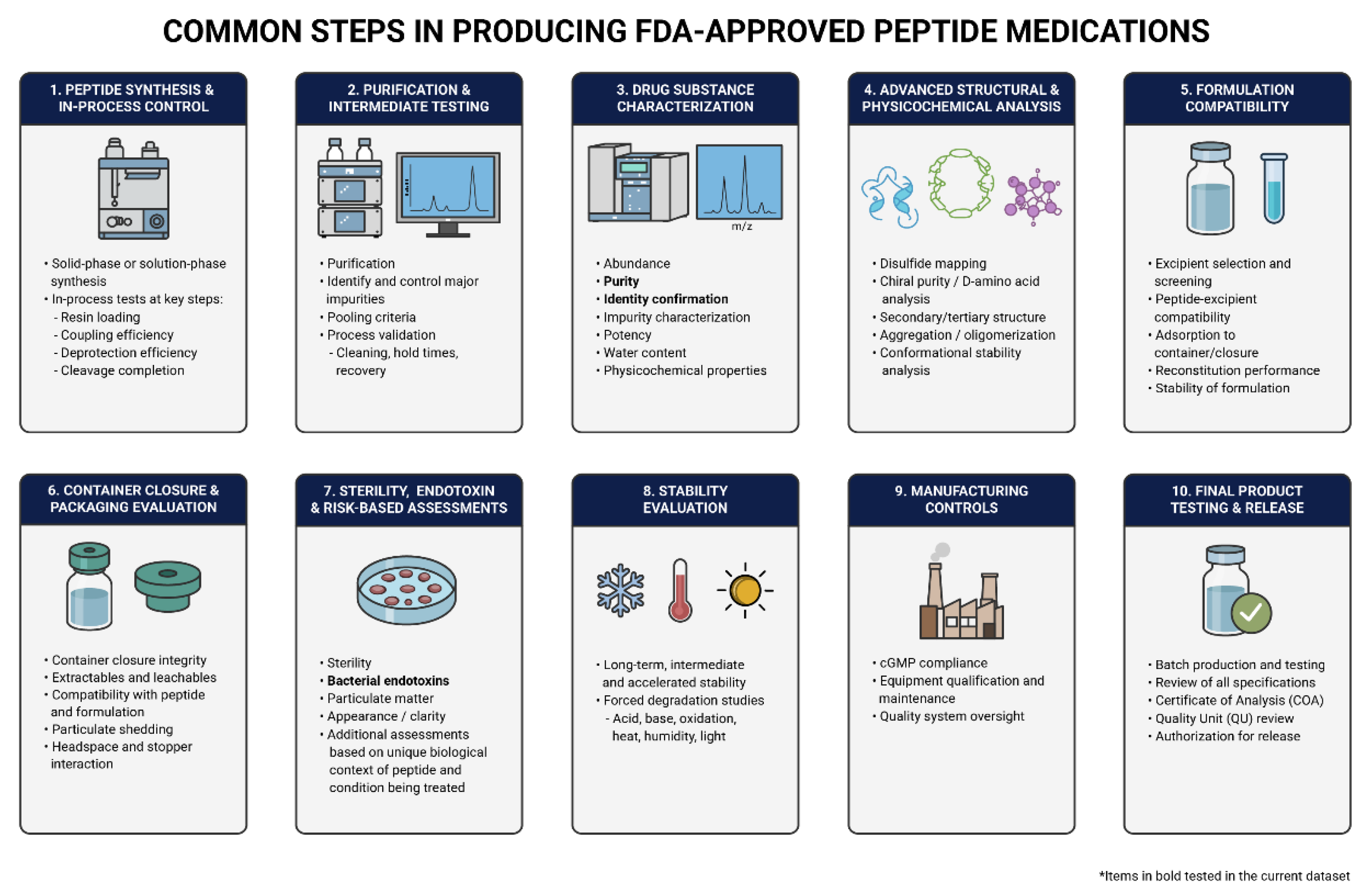
1. Introduction
2. Materials and Methods
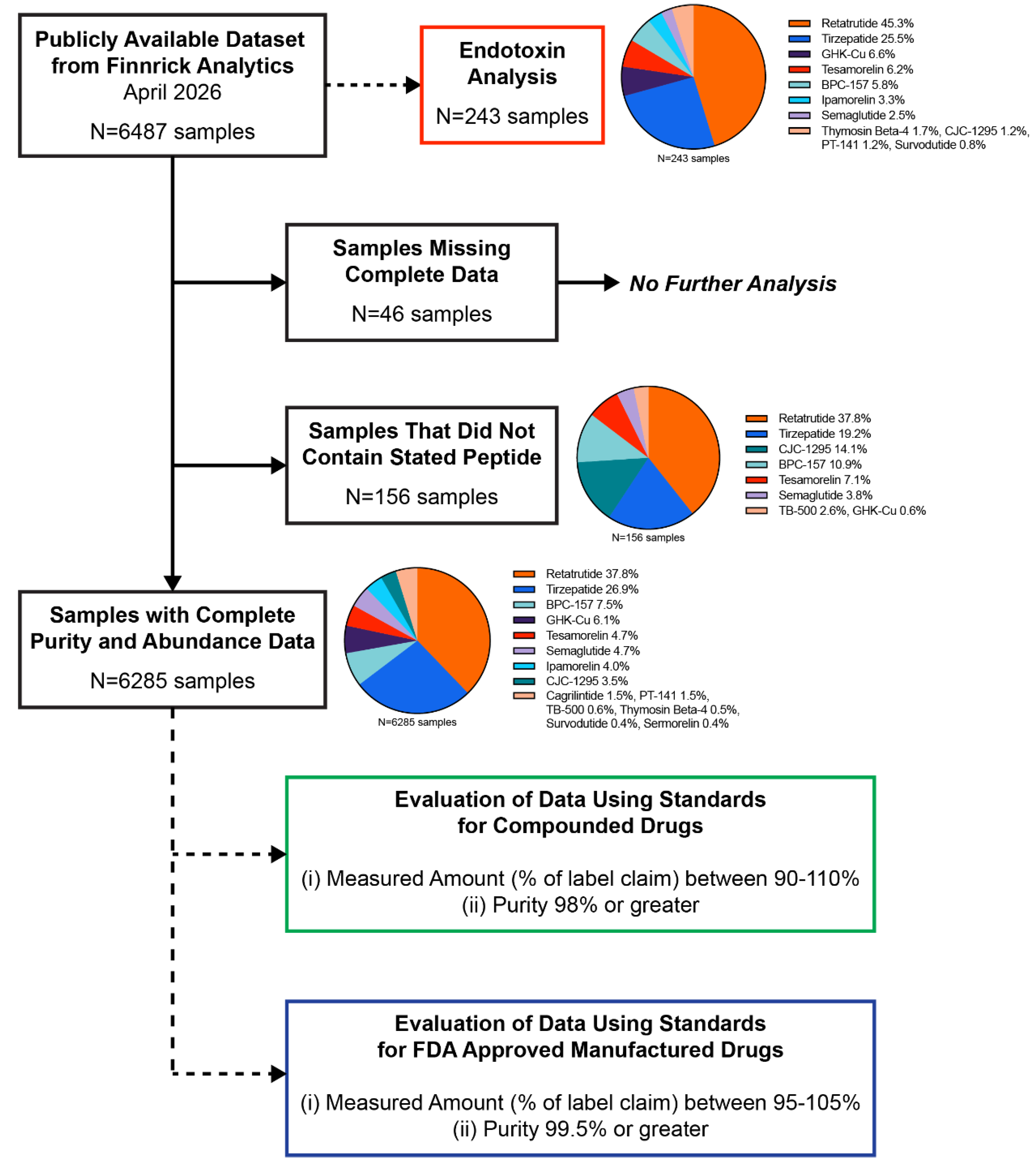
2.1. Data Source
2.2. Quality Acceptance Criteria and Model Development
2.3. Cost Comparisons
2.4. Data Analysis
3. Results
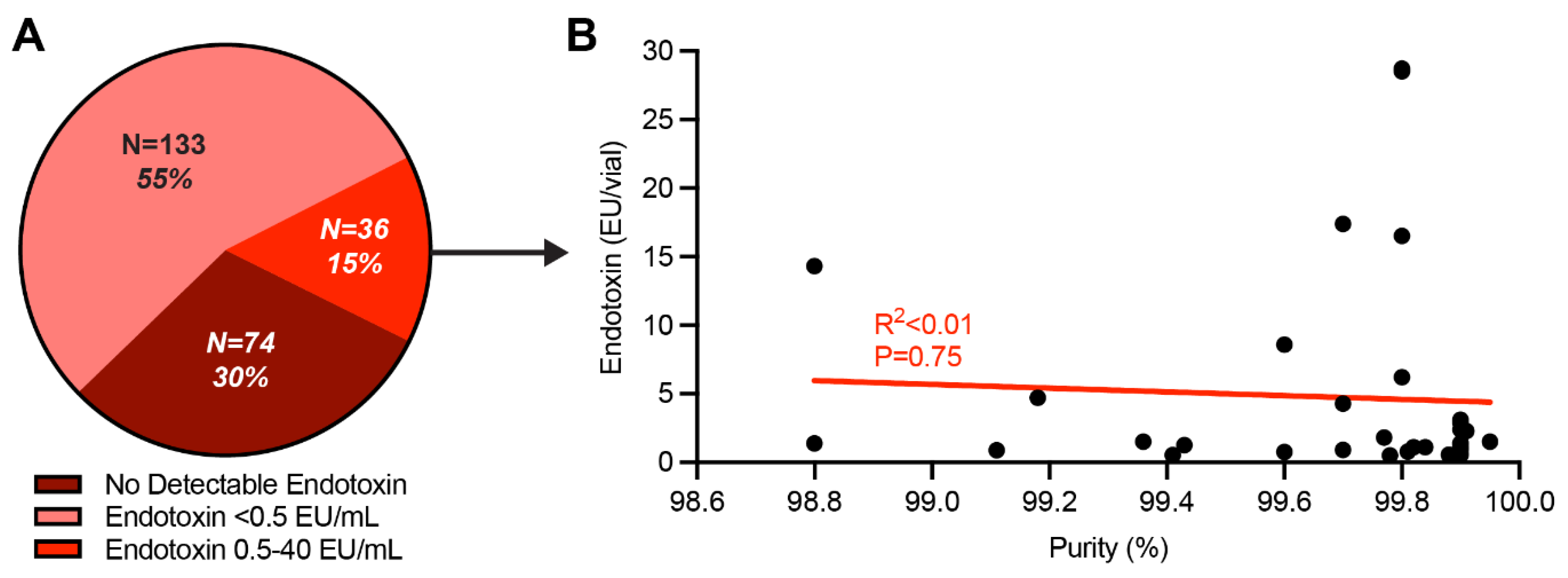
3.1. Endotoxin
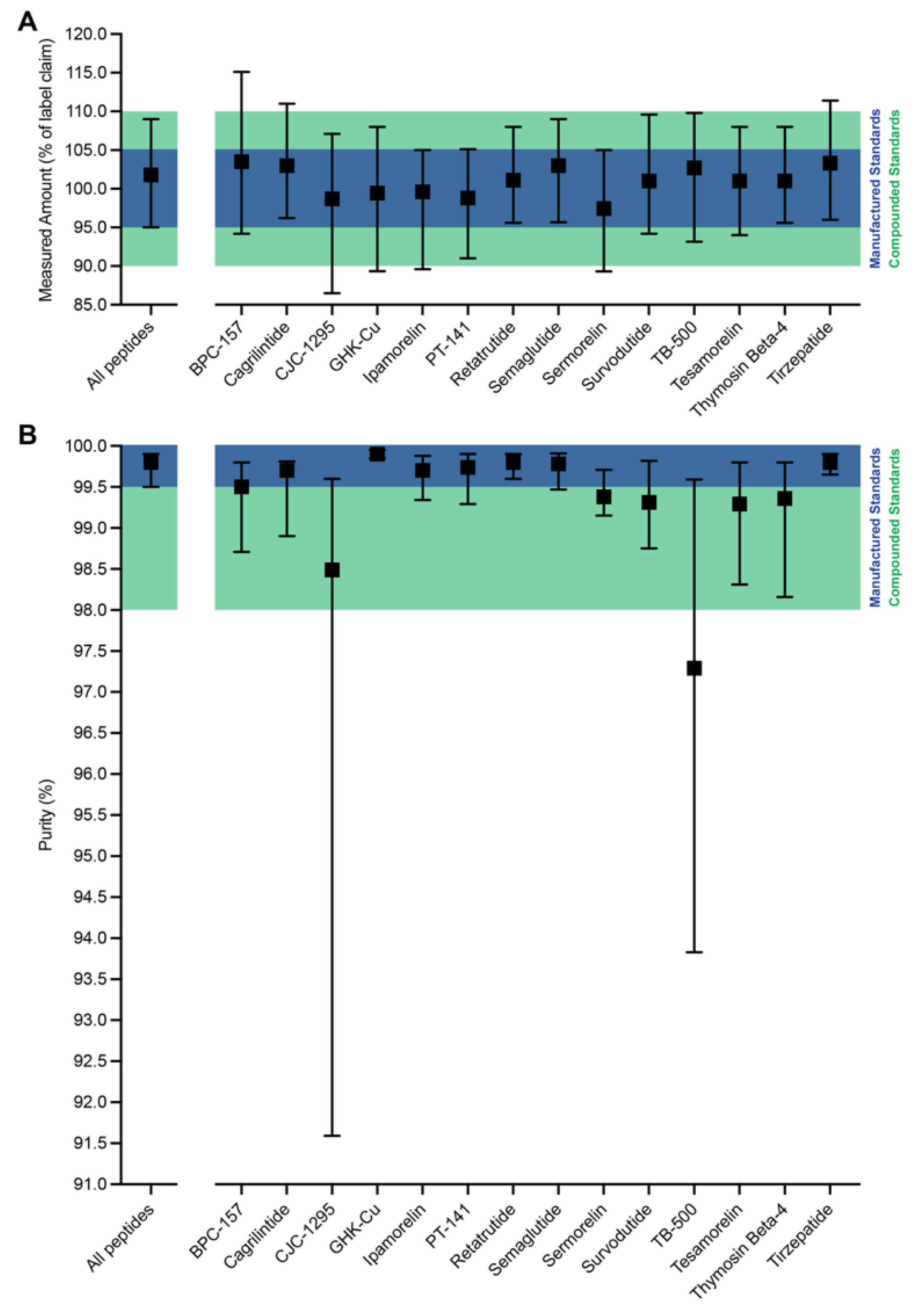
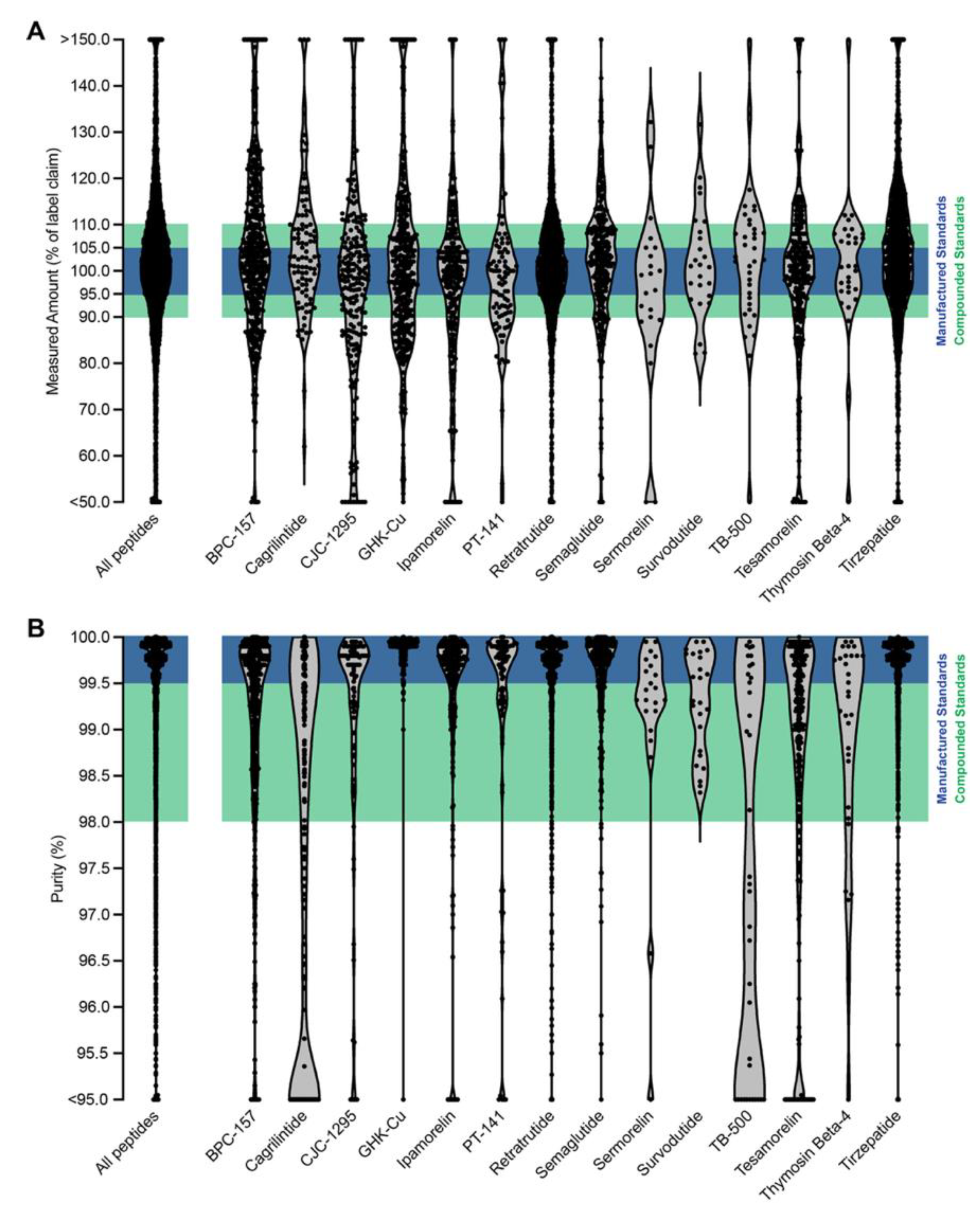
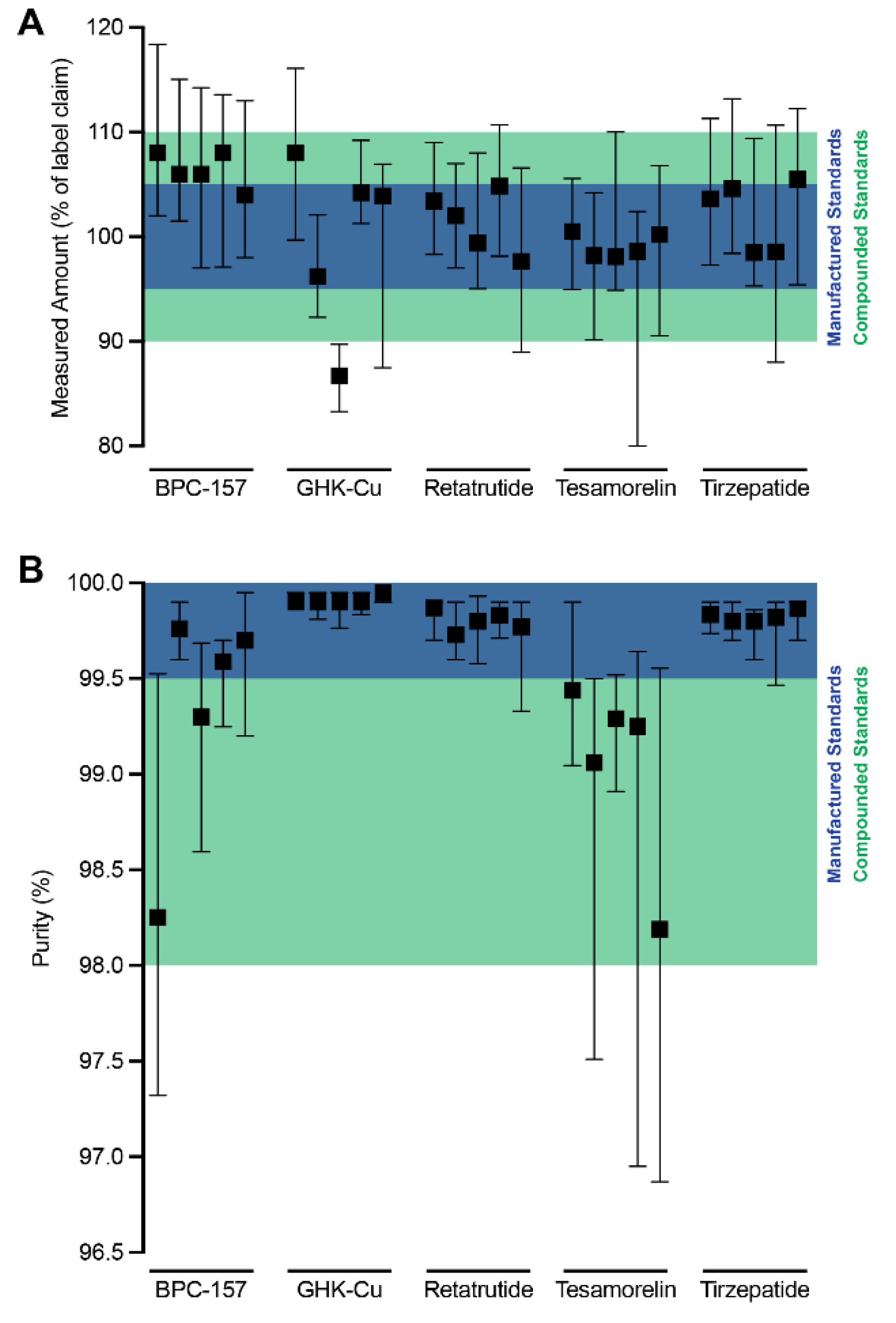
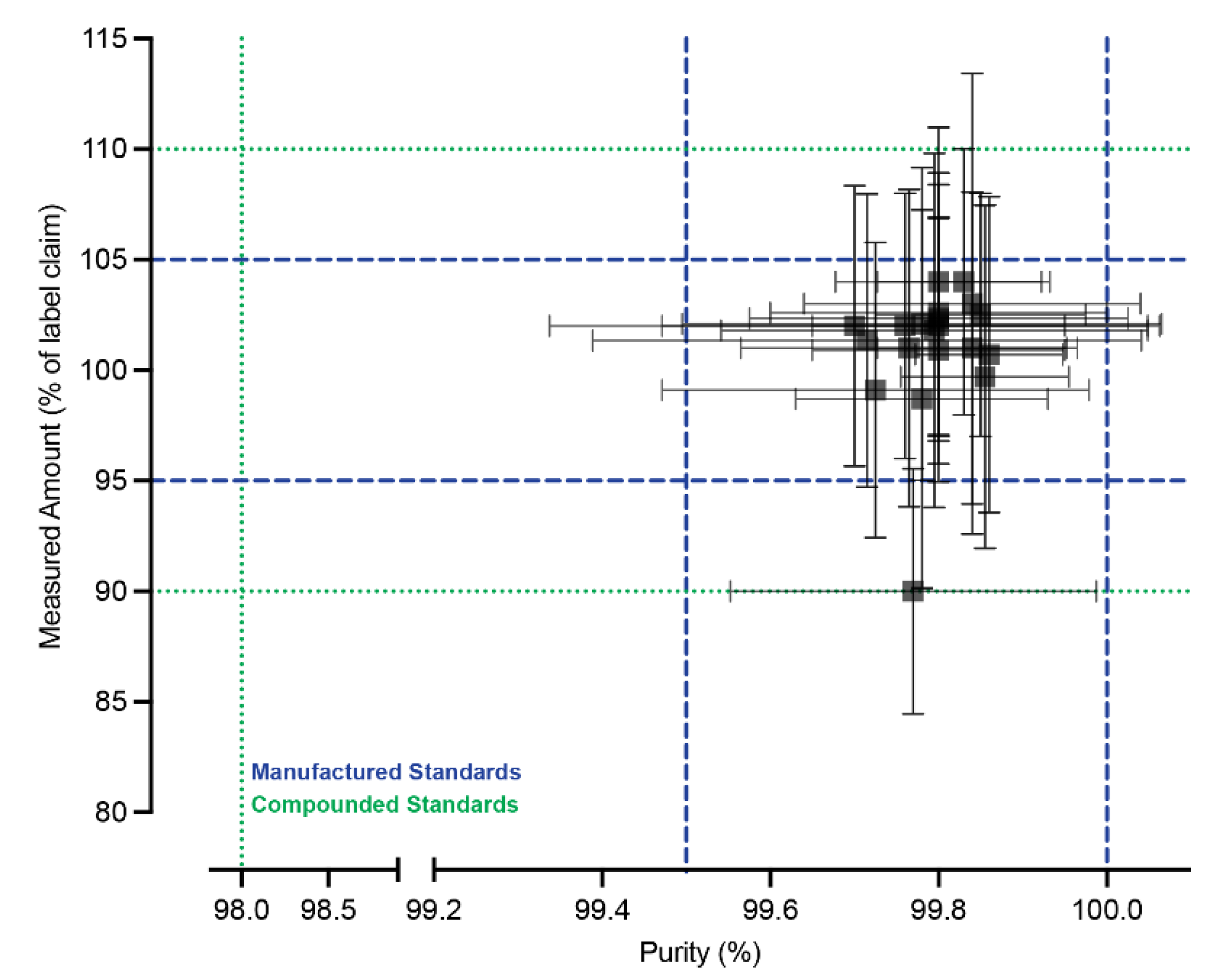
3.2. Measured Abundance and Purity
3.3. Inter-Vendor Variability
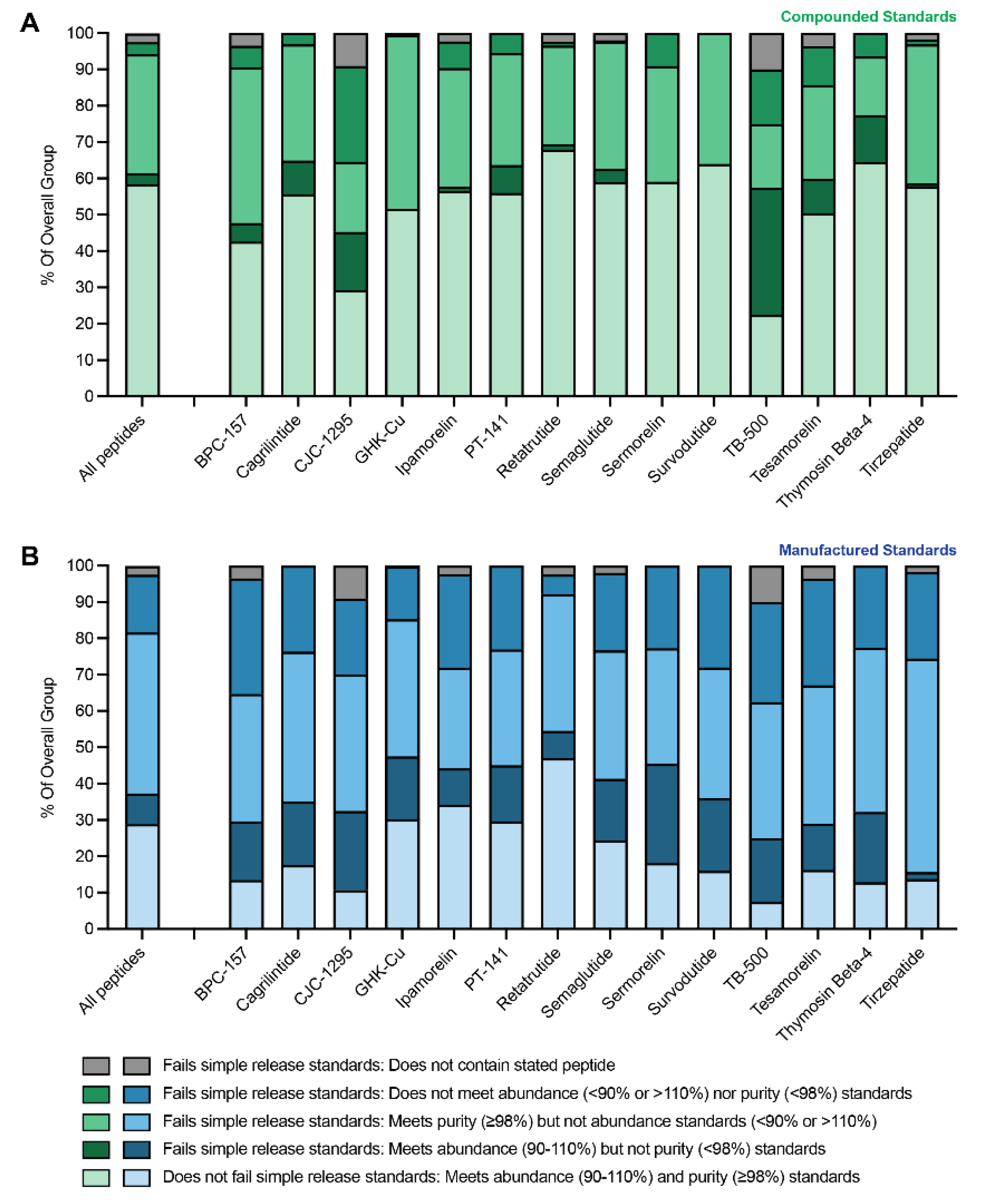
3.4. Composite Release Score
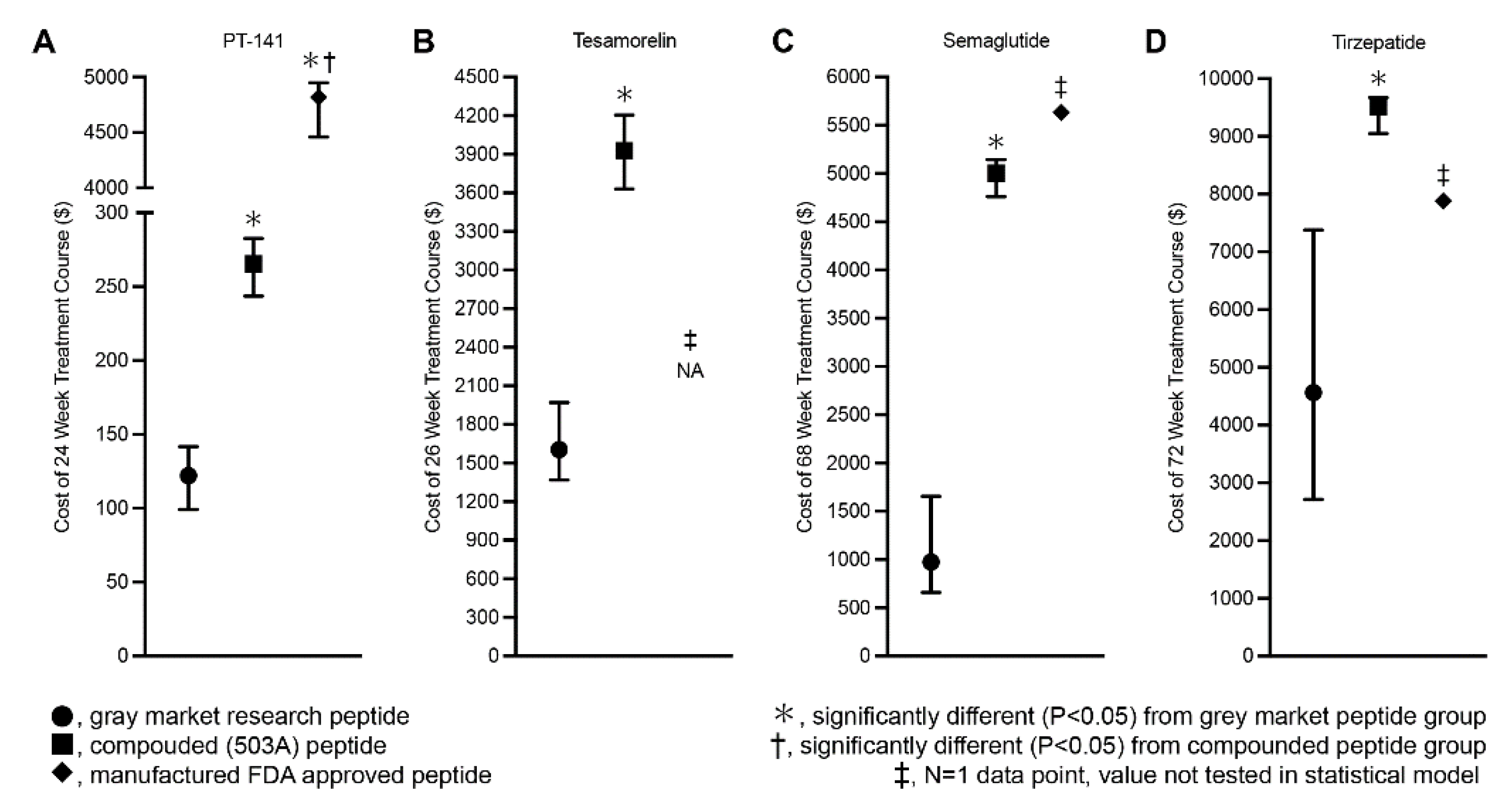
3.5. Cost Comparisons Between Gray Market, Compounded and FDA-Approved Peptides
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ACTH | adrenocorticotropic hormone |
| ANDA | abbreviated new drug application |
| ANOVA | analysis of variance |
| BPC-157 | body protection compound 157 |
| CagriSema | cagrilintide plus semaglutide |
| CJC-1295 | peptide CJC-1295 |
| DAC | Drug Affinity Complex |
| DPP-4 | dipeptidyl peptidase 4 |
| EGF | epidermal growth factor |
| EU | endotoxin units |
| F-actin | filamentous actin |
| FAK | focal adhesion kinase |
| FDA | U.S. Food and Drug Administration |
| G-actin | globular actin |
| GCGR | glucagon receptor |
| GH | growth hormone |
| GHK-Cu | glycyl-L-histidyl-L-lysine copper |
| GHRH | growth hormone-releasing hormone |
| GHRHR | growth hormone-releasing hormone receptor |
| GHRP-2 | growth hormone-releasing peptide 2 |
| GHRP-6 | growth hormone-releasing peptide 6 |
| GHSR-1a | growth hormone secretagogue receptor 1a |
| GI | gastrointestinal |
| GIP | glucose-dependent insulinotropic polypeptide |
| GIPR | glucose-dependent insulinotropic polypeptide receptor |
| GLP-1 | glucagon-like peptide 1 |
| GLP-1R | glucagon-like peptide 1 receptor |
| HIV | human immunodeficiency virus |
| HPLC | high performance liquid chromatography |
| IGF-1 | insulin-like growth factor 1 |
| IQR | interquartile range |
| ILK | integrin-linked kinase |
| LAL | Limulus amebocyte lysate |
| MC3R | melanocortin 3 receptor |
| MC4R | melanocortin 4 receptor |
| MDMA | 3,4-methylenedioxymethamphetamine |
| MMP | matrix metalloproteinase |
| MMP-1 | matrix metalloproteinase 1 |
| MMP-2 | matrix metalloproteinase 2 |
| NDA | new drug application |
| NO | nitric oxide |
| PKG | protein kinase G |
| PT-141 | bremelanotide |
| R2 | coefficient of determination |
| rhGH | recombinant human growth hormone |
| TB-500 | thymosin beta-4 fragment 17-23 |
| TIMP | tissue inhibitor of metalloproteinase |
| USP | United States Pharmacopeia |
| VEGF | vascular endothelial growth factor |
| alpha-MSH | alpha-melanocyte-stimulating hormone |
| cGMP | cyclic guanosine monophosphate |
References
- Otvos, L.; Wade, J.D. Big peptide drugs in a small molecule world. Front. Chem. 2023, 11, 1302169. [Google Scholar] [CrossRef]
- Mendias, C.L.; Awan, T.M. Safety and Efficacy of Approved and Unapproved Peptide Therapies for Musculoskeletal Injuries and Athletic Performance. Sports Med. 2026, 1–15. [Google Scholar] [CrossRef]
- Wang, L.; Wang, N.; Zhang, W.; Cheng, X.; Yan, Z.; Shao, G.; Wang, X.; Wang, R.; Fu, C. Therapeutic peptides: current applications and future directions. Signal Transduct. Target. Ther. 2022, 7, 1–27. [Google Scholar] [CrossRef] [PubMed]
- Rastogi, S.; Shukla, S.; Kalaivani, M.; Singh, G.N. Peptide-based therapeutics: quality specifications, regulatory considerations, and prospects. Drug Discov. Today 2019, 24, 148–162. [Google Scholar] [CrossRef] [PubMed]
- Vergote, V.; Burvenich, C.; Van de Wiele, C.; De Spiegeleer, B. Quality specifications for peptide drugs: a regulatory-pharmaceutical approach. J. Pept. Sci. 2009, 15, 697–710. [Google Scholar] [CrossRef] [PubMed]
- US Food and Drug Administration. ANDAs for Certain Highly Purified Synthetic Peptide Drug Products That Refer to Listed Drugs of rDNA Origin. In US Food and Drug Administration; 2021. [Google Scholar]
- De Groot, A.S.; Roberts, B.J.; Mattei, A.; Lelias, S.; Boyle, C.; Martin, W.D. Immunogenicity risk assessment of synthetic peptide drugs and their impurities. Drug Discov. Today 2023, 28, 103714. [Google Scholar] [CrossRef]
- D’hondt, M.; Bracke, N.; Taevernier, L.; Gevaert, B.; Verbeke, F.; Wynendaele, E.; De Spiegeleer, B. Related impurities in peptide medicines. J. Pharm. Biomed. Anal. 2014, 101, 2–30. [Google Scholar] [CrossRef]
- Elsayed, Y.Y.; Kühl, T.; Imhof, D. Regulatory Guidelines for the Analysis of Therapeutic Peptides and Proteins. J. Pept. Sci. 2025, 31, e70001. [Google Scholar] [CrossRef]
- Turnock, L.; Gibbs, N. Click, click, buy: The market for novel synthetic peptide hormones on mainstream e-commerce platforms in the UK. Perform. Enhanc. Heal. 2023, 11. [Google Scholar] [CrossRef]
- Turnock, L.A.; Hearne, E. Novel wellbeing and repair peptide use in the UK: Netnographic findings. Perform. Enhanc. Heal. 2024, 13. [Google Scholar] [CrossRef]
- Finnrick Analytics [Online]. Finnrick Analytics. 2026. Available online: https://www.finnrick.com (accessed on 13 April 2026).
- International Council for Harmonisation. ICH Q6A: Specifications: Test Procedures and Acceptance Criteria for New Drug Substances and New Drug Products - Chemical Substances; International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use: Geneva, 1999. [Google Scholar]
- International Council for Harmonisation. ICH Q6B: Specifications: Test Procedures and Acceptance Criteria for Biotechnological/Biological Products; International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use: Geneva, 1999. [Google Scholar]
- Bercu, J.; Berlam, S.C.; Berridge, J.; Cherney, B.; Cowley, D.; Laughton, H.W.; McLoughlin, D.; McMahon, M.; Moore, C.M.V.; Murti, C.; et al. Establishing Patient Centric Specifications for Drug Substance and Drug Product Impurities. J. Pharm. Innov. 2018, 14, 76–89. [Google Scholar] [CrossRef]
- United States Pharmacopeia. Water Determination; United States Pharmacopeial Convention: Rockville, MD, 2024. [Google Scholar]
- McCarthy, D.; Han, Y.; Carrick, K.; Schmidt, D.; Workman, W.; Matejtschuk, P.; Duru, C.; Atouf, F. Reference Standards to Support Quality of Synthetic Peptide Therapeutics. Pharm. Res. 2023, 40, 1317–1328. [Google Scholar] [CrossRef] [PubMed]
- United States Pharmacopeia. Strength and Stability Testing for Compounded Preparations. United States Pharmacopeial Convention, 2014. [Google Scholar]
- United States Pharmacopeia. Bacterial Endotoxins Test; United States Pharmacopeial Convention: Rockville, MD, 2023. [Google Scholar]
- International Council for Harmonisation. ICH Q3A(R2): Impurities in New Drug Substances; International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use: Geneva, 2006. [Google Scholar]
- International Council for Harmonisation. ICH Q3B(R2): Impurities in New Drug Products; International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use: Geneva, 2006. [Google Scholar]
- International Council for Harmonisation. ICH Q3C(R8): Impurities: Guideline for Residual Solvents; International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use: Geneva, 2021. [Google Scholar]
- Kupiec, T.C.; Skinner, R.; Lanier, L. Stability Versus Potency Testing: The Madness is in the Method. Int. J. Pharm. Compd. 2008, 12, 50–3. [Google Scholar] [PubMed]
- United States Pharmacopeia. Pharmaceutical Compounding — Sterile Preparations; United States Pharmacopeial Convention: Rockville, MD, 2023. [Google Scholar]
- United States Pharmacopeia. Sterility Tests; United States Pharmacopeial Convention: Rockville, MD, 2023. [Google Scholar]
- Capen, R.; Christopher, D.; Forenzo, P.; Ireland, C.; Liu, O.; Lyapustina, S.; O’nEill, J.; Patterson, N.; Quinlan, M.; Sandell, D.; et al. On the Shelf Life of Pharmaceutical Products. Aaps Pharmscitech 2012, 13, 911–918. [Google Scholar] [CrossRef]
- Simon, J.A.; Thorp, J.; Millheiser, L. Flibanserin for Premenopausal Hypoactive Sexual Desire Disorder: Pooled Analysis of Clinical Trials. J. Women's Heal. 2019, 28, 769–777. [Google Scholar] [CrossRef]
- Kingsberg, S.A.; Clayton, A.H.; Portman, D.; Williams, L.A.; Krop, J.; Jordan, R.; Lucas, J.; Simon, J.A. Bremelanotide for the Treatment of Hypoactive Sexual Desire Disorder. Obstet. Gynecol. 2019, 134, 899–908. [Google Scholar] [CrossRef]
- US Food and Drug Administration. Vyleesi (bremelanotide) Injection Prescribing Information. US Food and Drug Administration, 2021. [Google Scholar]
- US Food and Drug Administration. Egrifta SV (tesamorelin) Injection Prescribing Information. In US Food and Drug Administration; 2024. [Google Scholar]
- Falutz, J.; Allas, S.; Blot, K.; Potvin, D.; Kotler, D.; Somero, M.; Berger, D.; Brown, S.; Richmond, G.; Fessel, J.; et al. Metabolic Effects of a Growth Hormone–Releasing Factor in Patients with HIV. New Engl. J. Med. 2007, 357, 2359–2370. [Google Scholar] [CrossRef]
- Wilding, J.P.H.; Batterham, R.L.; Calanna, S.; Davies, M.; Van Gaal, L.F.; Lingvay, I.; McGowan, B.M.; Rosenstock, J.; Tran, M.T.; Wadden, T.A.; et al. Once-Weekly Semaglutide in Adults with Overweight or Obesity. N. Engl. J. Med. 2021, 384, 989–1002. [Google Scholar] [CrossRef]
- Jastreboff, A.M.; Aronne, L.J.; Ahmad, N.N.; Wharton, S.; Connery, L.; Alves, B.; Kiyosue, A.; Zhang, S.; Liu, B.; Bunck, M.C.; et al. Tirzepatide Once Weekly for the Treatment of Obesity. New Engl. J. Med. 2022, 387, 205–216. [Google Scholar] [CrossRef]
- Peptide Critic [Online]. 2026. Available online: https://peptidecritic.com/ (accessed on 13 April 2026).
- PeptiPrices [Online]. PeptiPrices: 2026. Available online: https://peptiprices.com (accessed on 13 April 2026).
- THERA patient support program [Online]. THERA patient support program: 2026. Available online: https://www.egriftawr.com/thera-patient-support/how-to-enroll (accessed on 13 April 2026).
- NovoCare Pharmacy [Online]. NovoCare Pharmacy: 2026. Available online: https://www.novocare.com/pharmacy.html (accessed on 13 April 2026).
- Zepbound - Lilly Direct [Online]. Zepbound - Lilly Direct: 2026. Available online: https://www.lilly.com/lillydirect/medicines/zepbound (accessed on 13 April 2026).
- Liu, F. Safety assessment of drug impurities for patient safety: A comprehensive review. Regul. Toxicol. Pharmacol. 2024, 153, 105715. [Google Scholar] [CrossRef]
- Cordeiro, C.F.; Franco, L.L.; Carvalho, D.T.; Bonfilio, R. Impurities in Active Pharmaceutical Ingredients and Drug Products: A Critical Review. Crit. Rev. Anal. Chem. 2024, 56, 55–75. [Google Scholar] [CrossRef] [PubMed]
- Pokar, D.; Rajput, N.; Sengupta, P. Industrial approaches and consideration of clinical relevance in setting impurity level specification for drug substances and drug products. Int. J. Pharm. 2020, 576, 119018. [Google Scholar] [CrossRef] [PubMed]
- Buschmann, H.; Handler, N.; Holzgrabe, U. The quality of drugs and drug products – Always guaranteed? J. Pharm. Biomed. Anal. 2023, 239, 115880. [Google Scholar] [CrossRef] [PubMed]
- Görög, S. Drug safety, drug quality, drug analysis. J. Pharm. Biomed. Anal. 2008, 48, 247–253. [Google Scholar] [CrossRef]
- Cohen, J.S. Dose Discrepancies Between the Physicians' Desk Reference and the Medical Literature, and Their Possible Role in the High Incidence of Dose-Related Adverse Drug Events. Arch. Intern. Med. 2001, 161, 957–964. [Google Scholar] [CrossRef]
- Phillips, J.; Beam, S.; Brinker, A.; Holquist, C.; Honig, P.; Lee, L.Y.; Pamer, C. Retrospective analysis of mortalities associated with medication errors. Am. J. Heal. Pharm. 2001, 58, 1835–1841. [Google Scholar] [CrossRef]
- United States Pharmacopeia. What is a USP Monograph; United States Pharmacopeial Convention: Rockville, MD, 2019. [Google Scholar]
- Mattingly, A.N.; Mattingly, T.J.; Phan, A.L.N.; Negash, K. Categorization and comparisons of drug recalls for manufacturers and compounders. J. Am. Pharm. Assoc. 2022, 62, 1344–1350. [Google Scholar] [CrossRef]
- Watson, C.J.; Whitledge, J.D.; Siani, A.M.; Burns, M.M. Pharmaceutical Compounding: a History, Regulatory Overview, and Systematic Review of Compounding Errors. J. Med Toxicol. 2020, 17, 197–217. [Google Scholar] [CrossRef]
- United States Pharmacopeia. The Pharmacopeia of the United States of America, Thirty-Second Revision and the National Formulary, Twenty-Seventh Edition; United States Pharmacopeial Convention: Rockville, MD, 2009. [Google Scholar]
- Puig, M.; Shubow, S. Immunogenicity of therapeutic peptide products: bridging the gaps regarding the role of product-related risk factors. Front. Immunol. 2025, 16, 1608401. [Google Scholar] [CrossRef]
- Vasireddi, N.; Hahamyan, H.; Salata, M.J.; Karns, M.; Calcei, J.G.; Voos, J.E.; Apostolakos, J.M. Emerging Use of BPC-157 in Orthopaedic Sports Medicine: A Systematic Review. HSS J. ® 2025, 21, 485–495. [Google Scholar] [CrossRef]
- Janvier, S.; Cheyns, K.; Canfyn, M.; Goscinny, S.; De Spiegeleer, B.; Vanhee, C.; Deconinck, E. Impurity profiling of the most frequently encountered falsified polypeptide drugs on the Belgian market. Talanta 2018, 188, 795–807. [Google Scholar] [CrossRef] [PubMed]
- Limbu, Y.B.; Huhmann, B.A. Illicit Online Pharmacies: A Scoping Review. Int. J. Environ. Res. Public Heal. 2023, 20, 5748. [Google Scholar] [CrossRef] [PubMed]
- Mozaffarian, D.; Agarwal, M.; Aggarwal, M.; Alexander, L.; Apovian, C.M.; Bindlish, S.; Bonnet, J.; Butsch, W.S.; Christensen, S.; Gianos, E.; et al. Nutritional priorities to support GLP-1 therapy for obesity: a joint Advisory from the American College of Lifestyle Medicine, the American Society for Nutrition, the Obesity Medicine Association, and The Obesity Society. Am. J. Clin. Nutr. 2025, 122, 344–367. [Google Scholar] [CrossRef] [PubMed]
- Maghsoudi, N.; Tanguay, J.; Scarfone, K.; Rammohan, I.; Ziegler, C.; Werb, D.; Scheim, A.I. Drug checking services for people who use drugs: a systematic review. Addiction 2021, 117, 532–544. [Google Scholar] [CrossRef]
- Józwiak, M.; Bauer, M.; Kamysz, W.; Kleczkowska, P. Multifunctionality and Possible Medical Application of the BPC 157 Peptide—Literature and Patent Review. Pharmaceuticals 2025, 18, 185. [Google Scholar] [CrossRef]
- McGuire, F.P.; Martinez, R.; Lenz, A.; Skinner, L.; Cushman, D.M. Regeneration or Risk? A Narrative Review of BPC-157 for Musculoskeletal Healing. Curr. Rev. Musculoskelet. Med. 2025, 18, 611–619. [Google Scholar] [CrossRef]
- Davies, M.J.; Bajaj, H.S.; Broholm, C.; Eliasen, A.; Garvey, W.T.; le Roux, C.W.; Lingvay, I.; Lyndgaard, C.B.; Rosenstock, J.; Pedersen, S.D. Cagrilintide–Semaglutide in Adults with Overweight or Obesity and Type 2 Diabetes. New Engl. J. Med. 2025, 393, 648–659. [Google Scholar] [CrossRef]
- Garvey, W.T.; Blüher, M.; Contreras, C.K.O.; Davies, M.J.; Lehmann, E.W.; Pietiläinen, K.H.; Rubino, D.; Sbraccia, P.; Wadden, T.; Zeuthen, N.; et al. Coadministered Cagrilintide and Semaglutide in Adults with Overweight or Obesity. New Engl. J. Med. 2025, 393, 635–647. [Google Scholar] [CrossRef]
- Teichman, S.L.; Neale, A.; Lawrence, B.; Gagnon, C.; Castaigne, J.-P.; Frohman, L.A. Prolonged Stimulation of Growth Hormone (GH) and Insulin-Like Growth Factor I Secretion by CJC-1295, a Long-Acting Analog of GH-Releasing Hormone, in Healthy Adults. J. Clin. Endocrinol. Metab. 2006, 91, 799–805. [Google Scholar] [CrossRef]
- A Multicenter, Randomized, Placebo-Controlled, Double-Blind, Phase 2 Study to Evaluate the Efficacy and Safety of CJC 1295 Administered for 12 Weeks in HIV Infected Patients With HIV Associated Visceral Obesity. Available online: https://clinicaltrials.gov/study/NCT00267527 (accessed on 2 February 2026).
- Maquart, F.X.; Bellon, G.; Chaqour, B.; Wegrowski, J.; Patt, L.M.; E Trachy, R.; Monboisse, J.C.; Chastang, F.; Birembaut, P.; Gillery, P. In vivo stimulation of connective tissue accumulation by the tripeptide-copper complex glycyl-L-histidyl-L-lysine-Cu2+ in rat experimental wounds. J. Clin. Investig. 1993, 92, 2368–2376. [Google Scholar] [CrossRef]
- Pickart, L.; Margolina, A. Regenerative and Protective Actions of the GHK-Cu Peptide in the Light of the New Gene Data. Int. J. Mol. Sci. 2018, 19, 1987. [Google Scholar] [CrossRef]
- Pickart, L.; Margolina, A. Skin Regenerative and Anti-Cancer Actions of Copper Peptides. Cosmetics 2018, 5, 29. [Google Scholar] [CrossRef]
- Dou, Y.; Lee, A.; Zhu, L.; Morton, J.; Ladiges, W. The potential of GHK as an anti-aging peptide. Aging Pathobiol. Ther. 2020, 2, 58–61. [Google Scholar] [CrossRef] [PubMed]
- Johansen, P.B.; Nowak, J.; Skjærbæk, C.; Flyvbjerg, A.; Andreassen, T.T.; Wilken, M.; Ørskov, H. Ipamorelin, a new growth-hormone-releasing peptide, induces longitudinal bone growth in rats. Growth Horm. IGF Res. 1999, 9, 106–113. [Google Scholar] [CrossRef] [PubMed]
- Prinz, P.; Stengel, A. Control of Food Intake by Gastrointestinal Peptides: Mechanisms of Action and Possible Modulation in the Treatment of Obesity. J. Neurogastroenterol. Motil. 2017, 23, 180–196. [Google Scholar] [CrossRef]
- Beck, D.E.; Sweeney, W.B.; McCarter, M.D. On behalf of the Ipamorelin 201 Study Group Prospective, randomized, controlled, proof-of-concept study of the Ghrelin mimetic ipamorelin for the management of postoperative ileus in bowel resection patients. Int. J. Color. Dis. 2014, 29, 1527–1534. [Google Scholar] [CrossRef]
- Safarinejad, M.R.; Hosseini, S.Y. Salvage of Sildenafil Failures With Bremelanotide: A Randomized, Double-Blind, Placebo Controlled Study. J. Urol. 2008, 179, 1066–1071. [Google Scholar] [CrossRef]
- Rosenstock, J.; Frias, J.; Jastreboff, A.M.; Du, Y.; Lou, J.; Gurbuz, S.; Thomas, M.K.; Hartman, M.L.; Haupt, A.; Milicevic, Z.; et al. Retatrutide, a GIP, GLP-1 and glucagon receptor agonist, for people with type 2 diabetes: a randomised, double-blind, placebo and active-controlled, parallel-group, phase 2 trial conducted in the USA. Lancet 2023, 402, 529–544. [Google Scholar] [CrossRef]
- Jastreboff, A.M.; Kaplan, L.M.; Frías, J.P.; Wu, Q.; Du, Y.; Gurbuz, S.; Coskun, T.; Haupt, A.; Milicevic, Z.; Hartman, M.L. Triple–Hormone-Receptor Agonist Retatrutide for Obesity — A Phase 2 Trial. New Engl. J. Med. 2023, 389, 514–526. [Google Scholar] [CrossRef]
- NCT06354660: A Phase 3, Randomized, Multicenter, Double-Blind Study to Investigate the Efficacy and Safety of Retatrutide Once Weekly Compared With Placebo in Adult Participants With Type 2 Diabetes and Inadequate Glycemic Control With Diet and Exercise Alone (TRANSCEND-T2D-1). Available online: https://clinicaltrials.gov/study/NCT06354660 (accessed on 13 April 2026).
- NCT05931367: A Phase 3 Study to Investigate the Efficacy and Safety of LY3437943 Once Weekly in Participants Who Have Obesity or Overweight and Osteoarthritis of the Knee: A Randomized, Double-Blind, Placebo-Controlled Trial [Online]. Available online: https://clinicaltrials.gov/study/NCT05931367 (accessed on 13 April 2026).
- Albor, C.; Anyiam, O.; Boyle, L.D.; Makaronidis, J.; Tan, R.; Iftikhar, S.; Burzic, A.; Trempe, F.L.; Stanley, M.; Bhatti, A.; et al. The Role of GLP1 Receptor Agonists and Multi-agonist Incretin Therapies for Specific Obesity-related Health Conditions: Evidence and Rationale for Prioritisation. Curr. Obes. Rep. 2026, 15, 29. [Google Scholar] [CrossRef]
- Bhandarkar, A.; Bhat, S.; Kapoor, N. Effect of GLP-1 receptor agonists on body composition. Curr. Opin. Endocrinol. Diabetes 2025, 32, 279–285. [Google Scholar] [CrossRef] [PubMed]
- Prakash, A.; Goa, K.L. Sermorelin. BioDrugs 1999, 12, 139–157. [Google Scholar] [CrossRef] [PubMed]
- Ishida, J.; Saitoh, M.; Ebner, N.; Springer, J.; Anker, S.D.; von Haehling, S. Growth hormone secretagogues: history, mechanism of action, and clinical development. JCSM Rapid Commun. 2020, 3, 25–37. [Google Scholar] [CrossRef]
- Khorram, O.; Laughlin, G.A.; Yen, S.S.C. Endocrine and Metabolic Effects of Long-Term Administration of[ Nle 27 ]Growth Hormone-Releasing Hormone-(1–29)-NH 2 in Age-Advanced Men and Women 1. J. Clin. Endocrinol. Metab. 1997, 82, 1472–1479. [Google Scholar] [CrossRef] [PubMed]
- Mendias, C.L.; Enselman, E.R.S.; Olszewski, A.M.; Gumucio, J.P.; Edon, D.L.; Konnaris, M.A.; Carpenter, J.E.; Awan, T.M.; Jacobson, J.A.; Gagnier, J.J.; et al. The Use of Recombinant Human Growth Hormone to Protect Against Muscle Weakness in Patients Undergoing Anterior Cruciate Ligament Reconstruction: A Pilot, Randomized Placebo-Controlled Trial. Am. J. Sports Med. 2020, 48, 1916–1928. [Google Scholar] [CrossRef]
- Sanyal, A.J.; Bedossa, P.; Fraessdorf, M.; Neff, G.W.; Lawitz, E.; Bugianesi, E.; Anstee, Q.M.; Hussain, S.A.; Newsome, P.N.; Ratziu, V.; et al. A Phase 2 Randomized Trial of Survodutide in MASH and Fibrosis. New Engl. J. Med. 2024, 391, 311–319. [Google Scholar] [CrossRef]
- le Roux, C.W.; Wharton, S.; Bozkurt, B.; Platz, E.; Bleckert, G.; Hussain, S.A.; Brueckmann, M.; Startseva, E.; Kloer, I.M.; Kaplan, L.M. Survodutide for treatment of obesity: Baseline characteristics of participants in a randomized, double-blind, placebo-controlled, phase 3 trial ( SYNCHRONIZE ™-1). Diabetes, Obes. Metab. 2025, 28, 337–346. [Google Scholar] [CrossRef]
- Sosne, G.; Qiu, P.; Goldstein, A.L.; Wheater, M. Biological activities of thymosin ß 4 defined by active sites in short peptide sequences. FASEB J. 2010, 24, 2144–2151. [Google Scholar] [CrossRef]
- Esposito, S.; Deventer, K.; Goeman, J.; Van der Eycken, J.; Van Eenoo, P. Synthesis and characterization of the N-terminal acetylated 17-23 fragment of thymosin beta 4 identified in TB-500, a product suspected to possess doping potential. Drug Test. Anal. 2012, 4, 733–738. [Google Scholar] [CrossRef]
- Makimura, H.; Feldpausch, M.N.; Rope, A.M.; Hemphill, L.C.; Torriani, M.; Lee, H.; Grinspoon, S.K. Metabolic Effects of a Growth Hormone-Releasing Factor in Obese Subjects with Reduced Growth Hormone Secretion: A Randomized Controlled Trial. J. Clin. Endocrinol. Metab. 2012, 97, 4769–4779. [Google Scholar] [CrossRef]
- Baker, L.D.; Barsness, S.M.; Borson, S.; Merriam, G.R.; Friedman, S.D.; Craft, S.; Vitiello, M.V. Effects of Growth Hormone–Releasing Hormone on Cognitive Function in Adults With Mild Cognitive Impairment and Healthy Older Adults: Results of a Controlled Trial. Arch. Neurol. 2012, 69, 1420–1429. [Google Scholar] [CrossRef] [PubMed]
- Gattu, A.K.; Fourman, L.T. Metabolic dysfunction-associated steatotic liver disease in people with HIV. Curr. Opin. HIV AIDS 2025, 20, 350–358. [Google Scholar] [CrossRef] [PubMed]
- Tuffaha, S.H.; Singh, P.; Budihardjo, J.D.; Means, K.R.; Higgins, J.P.; Shores, J.T.; Salvatori, R.; Höke, A.; Lee, W.A.; Brandacher, G. Therapeutic augmentation of the growth hormone axis to improve outcomes following peripheral nerve injury. Expert Opin. Ther. Targets 2016, 20, 1259–1265. [Google Scholar] [CrossRef] [PubMed]
- Sosne, G.; Kleinman, H.K.; Springs, C.; Gross, R.H.; Sung, J.; Kang, S. 0.1% RGN-259 (Thymosin ß4) Ophthalmic Solution Promotes Healing and Improves Comfort in Neurotrophic Keratopathy Patients in a Randomized, Placebo-Controlled, Double-Masked Phase III Clinical Trial. Int. J. Mol. Sci. 2022, 24, 554. [Google Scholar] [CrossRef]
- Goldstein, A.L.; Kleinman, H.K. Advances in the basic and clinical applications of thymosin β 4. Expert Opin. Biol. Ther. 2015, 15, 139–145. [Google Scholar] [CrossRef]
- Spurney, C.F.; Cha, H.-J.; Sali, A.; Pandey, G.S.; Pistilli, E.; Guerron, A.D.; Gordish-Dressman, H.; Hoffman, E.P.; Nagaraju, K. Evaluation of Skeletal and Cardiac Muscle Function after Chronic Administration of Thymosin β-4 in the Dystrophin Deficient Mouse. PLOS ONE 2010, 5, e8976. [Google Scholar] [CrossRef]
- Guarnera, G.; DeRosa, A.; Camerini, R.; Sites, O.B.O.8E. The effect of thymosin treatment of venous ulcers. Ann. New York Acad. Sci. 2010, 1194, 207–212. [Google Scholar] [CrossRef]
- Treadwell, T.; Kleinman, H.K.; Crockford, D.; Hardy, M.A.; Guarnera, G.T.; Goldstein, A.L. The regenerative peptide thymosin β4 accelerates the rate of dermal healing in preclinical animal models and in patients. Ann. New York Acad. Sci. 2012, 1270, 37–44. [Google Scholar] [CrossRef]
- Di, H.; Huang, J.; Zhang, D.; Ni, F.; Zheng, R.; Geng, H. Thymosin beta 4: An emerging therapeutic candidate for kidney diseases. Peptides 2026, 195, 171467. [Google Scholar] [CrossRef]
- Karacabeyli, D.; Lacaille, D. Glucagon-like peptide-1 receptor agonists in arthritis: current insights and future directions. Nat. Rev. Rheumatol. 2025, 21, 671–683. [Google Scholar] [CrossRef]
- Fuentes-Mendoza, J.M.; Concepción-Zavaleta, M.J.; Mendoza-Godoy, J.J.; Concepción-Urteaga, L.; Paz-Ibarra, J.; Coronado-Arroyo, J.C. Beyond metabolism: sexual dysfunction and weight-loss drugs. Sex. Med. Rev. 2025, 14. [Google Scholar] [CrossRef]
- Patil, S.; Jha, N.; Jha, M.K. Glucagon-like peptide 1 receptor agonists in substance use disorders: A systematic review of ClinicalTrials. Gov. Addict. Behav. Rep. 2026, 23. [Google Scholar] [CrossRef]
- Malhotra, A.; Grunstein, R.R.; Fietze, I.; Weaver, T.E.; Redline, S.; Azarbarzin, A.; Sands, S.A.; Schwab, R.J.; Dunn, J.P.; Chakladar, S.; et al. Tirzepatide for the Treatment of Obstructive Sleep Apnea and Obesity. New Engl. J. Med. 2024, 391, 1193–1205. [Google Scholar] [CrossRef]
| Gray Market Research Peptide Price ($ per mg) | 503A Compounded Peptide Price ($ per mg) | FDA Approved Commercial Peptide Price ($ per mg) | |
|---|---|---|---|
| PT-141 | 3.88 (3.15-4.50) N=86 | 8.50 (7.80-9.05)* N=34 | 153.0 (141.6-157.1) † N=47 |
| Semaglutide | 7.00 (4.74-11.90) N=53 | 36.00 (34.25-37.00)* N=64 | 36.35‡ N=1 |
| Tesamorelin | 6.30 (5.37-7.73) N=81 | 15.42 (14.25-16.50)* N=33 | NA‡ |
| Tirzepatide | 4.90 (2.91-7.93) N=51 | 10.25 (9.75-10.42)* N=47 | 7.48‡ N=1 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




