Submitted:
23 April 2026
Posted:
24 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Characterization of Nutritional Supplements
2.2. Characterization of Subjects and Clinical Data Collection
2.3. Biological Sampling and Biochemical Analysis
- BDNF determined in plasma by the ELISA method (Enzyme-Linked Immunosorbent Assay) (R&D Systems®, R&D Systems a biotechnical brand, Minneapolis, USA);
- ACTH and cortisol were determined in plasma by the microparticle chemiluminescence method (ACTH: CMIA Immulite 2000 XPi®, Siemens Healthcare Diagnostics Inc., Newark, DE, USA; Cortisol: CMIA Centaur XP®, Siemens Healthcare Diagnostics Inc., Newark, DE, USA);
- TSH, PTH, vitamin D—25OH, vitamin B12, and folic acid were determined in serum by microparticle chemiluminescence method (CMIA Architect®, ABBOTT Park Inc., IL, USA);
- High-sensitive C-reactive protein (hs-CRP) determined in serum by the nephelometry method (BN II®, Siemens Healthcare Diagnostics Inc., Newark, DE, USA);
- IL-1β, IL-6, and TNF-α were determined in plasma by the ELISA method (BD OptEIATM®, BD Biosciences, San Jose, California, USA).
2.4. Statistical Analysis
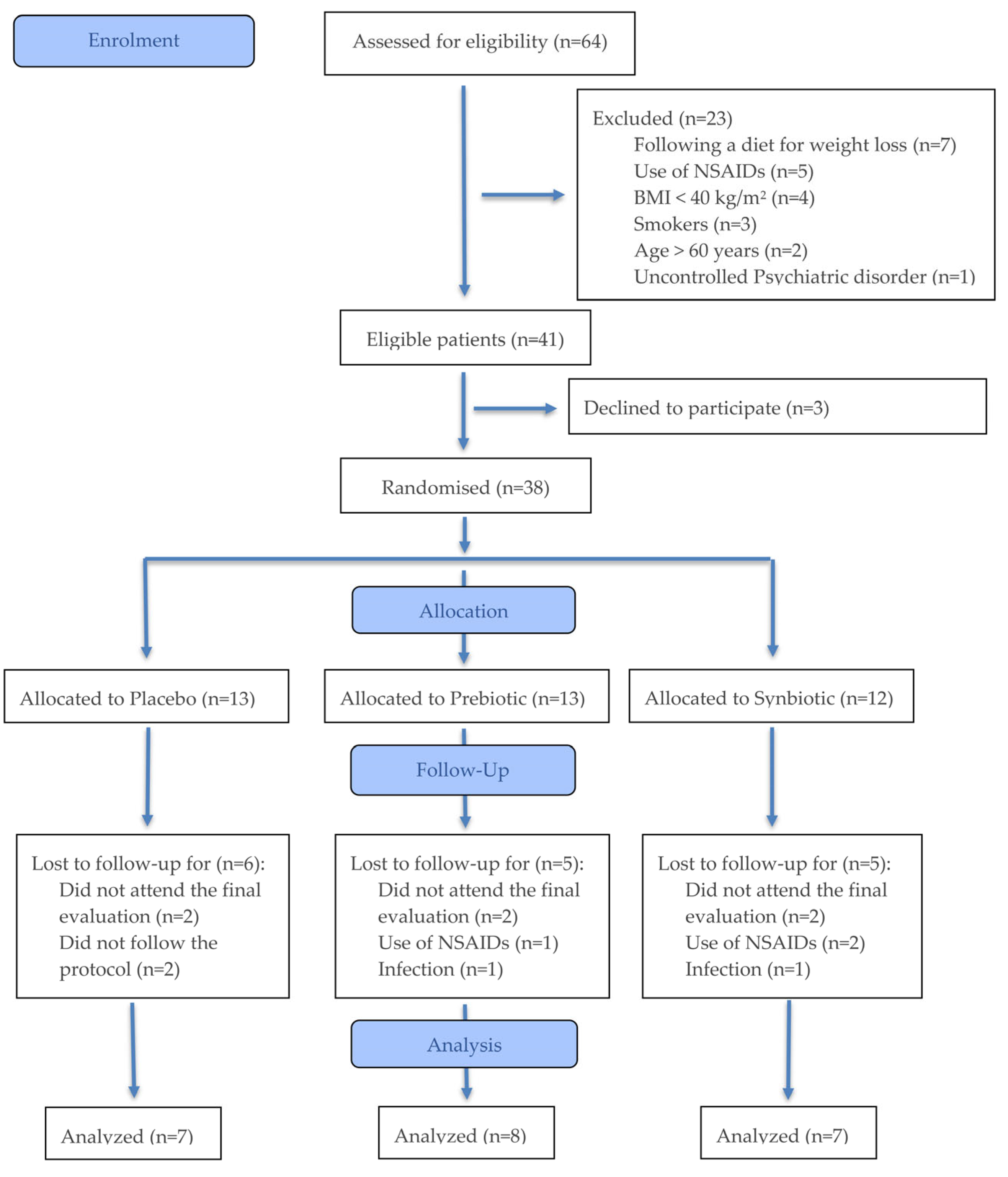
3. Results
Biochemical Variations Between Study Moments
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Schachter, J.; Martel, J.; Lin, C.S.; Chang, C.J.; Wu, T.R.; Lu, C.C.; et al. Effects of obesity on depression: a role for inflammation and the gut microbiota. Brain Behav. Immun. 2018, 69, 1–8. [Google Scholar] [CrossRef]
- Almada, L.F.; Borges, M.F.; Machado, S.E.C. Considerações neurobiológicas sobre a depressão maior. Encontro Rev. De. Psicol. 2014, 17, 111–124. [Google Scholar]
- Palazidou, E. The neurobiology of depression. Br. Med. Bull. 2012, 101, 127–145. [Google Scholar] [CrossRef]
- Zarouna, S.; Wozniak, G.; Papachristou, A.I. Mood disorders: a potential link between ghrelin and leptin on human body? World J. Exp. Med. 2015, 5, 103–109. [Google Scholar] [CrossRef]
- Mikkelsen, K.; Hallam, K.; Stojanovska, L.; Apostolopoulos, V. Yeast based spreads improve anxiety and stress. J. Funct. Foods 2018, 40, 471–476. [Google Scholar] [CrossRef]
- Vargas-Upegui, C.D.; Noreña-Rengifo, D. Déficit de vitamina B12 en la práctica psiquiátrica. Iatreia 2016, 30, 391–403. [Google Scholar] [CrossRef]
- Mollard, E.; Bilek, L.; Waltman, N. Emerging evidence on the link between depressive symptoms and bone loss in postmenopausal women. Int. J. Women’s Health 2018, 10, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Yilmaz, B.A.; Toruner, F.B.; Degertekin, C.K.; Lyidir, O.T.; Kaya, B.; Çakir, N. Neuropsychological Changes and Health-related Quality of Life in Patients with Asymptomatic Primary Hyperparathyroidism. Turk. J. Endocrinol. Metab. 2017, 21, 9–14. [Google Scholar] [CrossRef]
- Cao, J.; Ma, J.; Zha, X.; Bian, X.; Wang, W.; Liu, X. Gut Microbiota Dysbiosis in Depression: Pathological Correlations, Molecular Pathways, and Therapeutic Interventions. Int. J. Mol. Sci. 2026, 27(3), 1530. [Google Scholar] [CrossRef]
- Lin, S.K.K.; Chen, H.C.; Chen, I.M.; et al. Dysbiosis and depression: A study of gut microbiota alterations and functional pathways in antidepressant-naïve mood disorder patients. Transl. Psychiatry 2025, 15, 290. [Google Scholar] [CrossRef]
- Mora-Martínez, C.; Molina-Mendoza, G.; Cenit, M.C.; Medina-Rodríguez, E.M.; Larroya-García, A.; Sanchez-Carro, Y.; Gonzalez-Blanco, L.; Bobes, J.; Lopez-Garcia, P.; Zandio-Zorrilla, M.; Lahortiga-Ramos, F.; Gili, M.; Garcia-Toro, M.; Barcelo, B.; Ibarra, O.; Sanz, Y. Gut microbiome signatures associated with depression and obesity. ASM Journals—mSystems 2026, 11, e01263-25. [Google Scholar] [CrossRef] [PubMed]
- Bruce-Keller, A.J.; Salbaum, J.M.; Berthoud, H.R. Harnessing Gut Microbes for Mental Health: Getting From Here to There. Biol. Psychiatry 2018, 83, 214–223. [Google Scholar] [CrossRef]
- Lang, U.E.; Walter, M. Depression in the Context f Medical Disorders: New Pharmaclogical Pathways Revisited. Neurosignals 2017, 25, 54–73. [Google Scholar] [CrossRef]
- Ambrósio, G.; Kaufmann, F.N.; Manosso, L.; Platt, N.; Ghisleni, G.; Rodrigues, A.L.S.; et al. Depression and peripheral inflammatory profile of patients with obesity. Psychoneuroendocrinology 2018, 91, 132–141. [Google Scholar] [CrossRef]
- Hing, B.; Sathyaputri, L.; Potash, J.B. A comprehensive review of genetic and epigenetic mechanisms that regulate BDNF expression and function with relevance to major depressive disorder. Am. J. Med. Genet. 2018, 177B, 143–167. [Google Scholar] [CrossRef]
- Barde, Y. A. The physiopathology of brain-derived neurotrophic Factor. Physiol. Rev. 2025, 105(4), 2073–2140. [Google Scholar] [CrossRef]
- Cryan, J.F.; O’Mahony, S.M. The microbiome-gut-brain axis: from bowel to behavior. Neurogastroenterol. Motil. 2011, 23, 187–192. [Google Scholar] [CrossRef]
- Neufeld, K.M.; Kang, N.; Bienenstock, J.; Foster, J.A. Effects of intestinal microbiota on anxiety-like behavior. Commun. Integr. Biol. 2011, 4(4), 492–494. [Google Scholar] [CrossRef]
- Rickhman, P.P. Human Experimentation. Code of Ethics of the World Medical Association. Declaration of Helsinki. Br. Med. J. 1964, 2, 177. [Google Scholar] [CrossRef]
- Brasil. Resolução nº 466, de 12 de dezembro de 2012; Aprova diretrizes e normas regulamentadoras de pesquisas envolvendo seres humanos. Diário Oficial da União: Brasília; Brasília, DF, 2013.
- Hopewell, S.; Chan, A.W.; Collins, G.S.; Hróbjartsson, A.; Moher, D.; Schulz, K.F.; et al. CONSORT 2025 statement: updated guideline for reporting randomised trials. BMJ 2025, 388, e081123. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Expert committee on physical status: the use and interpretation of anthropometry. Physical status: the use and interpretation of anthropometry. Report of a WHO expert committee. WHO technical report series, 854; WHO: Geneva, 1995. [Google Scholar]
- World Health Organization. Waist Circumference and Waist–Hip Ratio. Report of a WHO Expert Consultation; WHO: Geneva, 2008. [Google Scholar]
- Lacy, B.E.; Mearin, F.; Chang, L.; Chey, W.D.; Lembo, A.J.; Simren, M.; et al. Bowel Disorders. Gastroenterology 2016, 150, 1393–1407. [Google Scholar] [CrossRef]
- Lewis, S.J.; Heaton, K.W. Stool form scale as a useful guide to intestinal transit time. Scand. J. Gastroenterol. 1997, 32, 920–924. [Google Scholar] [CrossRef]
- Molendijk, M.L.; Spinhoven, P.; Polak, M.; Bus, B.A.A.; Penninx, B.W.J.H.; Elzinga, B.M. Serum BDNF concentration as peripheral manifestations of depression: evidence from a systematic review and meta-analyses on 179 associations (N=9484). Mol. Psychiatry 2014, 19, 791–800. [Google Scholar] [CrossRef]
- Mietus-Snyder, M.J.; Lustig, R.H. Childhood Obesity: Adrift in the “Limbic Triangle”. Annu. Rev. Med. 2008, 59, 147–162. [Google Scholar] [CrossRef]
- Wedrychowicz, A.; Zajac, A.; Pilecki, M.; Koscielniak, B.; Tomasik, P.J. Peptides from adipose tissue in mental disorders. World J. Psychiatry 2014, 4(4), 103–111. [Google Scholar] [CrossRef] [PubMed]
- Krogh, J.; Videbech, P.; Renvillard, S.G.; Garde, A.H.; Jorgensen, M.B.; Nordentoft, M. Cognition and HPA axis reactivity in mildly to moderately depressed outpatients. A case-control study. Nord. J. Psychiatry 2012, 66(6), 414–421. [Google Scholar] [CrossRef] [PubMed]
- Mocking, R.J.T.; Verburg, H.F.; Westerink, A.M.; Assies, J.; Vaz, F.M.; Koeter, M.W.J.; et al. Fatty acid metabolism and its longitudinal relationship with the hypothalamic-pituitary-adrenal axis in major depression: Associations with prospective antidepressant response. Psychoneuroendocrinology 2015, 59, 1–13. [Google Scholar] [CrossRef]
- Otte, C.; Wingenfeld, K.; Kuehl, L.H.; Kaczmarczyk, M.; Richter, S.; Quante, A.; et al. Mineralocorticoid receptor stimulation improves cognitive function and decreases cortisol secretion in depressed patients and healthy individuals. Neuropsychopharmacology 2015, 40(2), 386–393. [Google Scholar] [CrossRef]
- Ruhe, H.G.; Khoenkhoen, S.J.; Ottenhof, K.W.; Koeter, M.W.; Mocking, R.J.T.; Schene, A.H. Longitudinal effects of the SSRI paroxetine on salivary cortisol in Major Depressive Disorder. Psychoneuroendocrinology 2015, 52, 261–271. [Google Scholar] [CrossRef] [PubMed]
- Ataka, K.; Asakawa, A.; Nagaishi, K.; Kaimoto, K.; Sawada, A.; Hayakawa, Y.; et al. Bone marrow-derived microglia infiltrate into the paraventricular nucleus of chronic psychological stress-loaded mice. PLoS ONE 2013, 8(11), e81744. [Google Scholar] [CrossRef]
- Bocchio-Chiavetto, L.; Bargnardi, V.; Zanardini, R.; Molteni, R.; Nielsen, M.G.; Placentino, A.; et al. Serum and plasma BDNF levels in major depression: a replication study and meta-analyses. World J. Biol. Psychiatry 2010, 11(6), 763–773. [Google Scholar] [CrossRef]
- Molteni, R.; Wu, A.; Vaynman, S.; Ying, Z.; Barnard, R.J.; Gómez-Pinilla, F. Exercise reverses the harmful effects of consumption of a high-fat diet on synaptic and behavioral plasticity associated to the action of brain-derived neurotrophic factor. Neuroscience 2004, 123(2), 429–440. [Google Scholar] [CrossRef]
- Pistell, P.J.; Morrison, C.D.; Gupta, S.; Knight, A.G.; Keller, J.N.; Ingram, D.K.; et al. Cognitive impairment following high fat diet consumption is associated with brain inflammation. J. Neuroimmunol. 2010, 219(1-2), 25–32. [Google Scholar] [CrossRef]
- Sharma, S.; Fulton, S. Diet-induced obesity promotes depressive-like behaviour that is associated with neural adaptations in brain reward circuitry. Int. J. Obes. 2013, 37, 382–389. [Google Scholar] [CrossRef] [PubMed]
- Burokas, A.; Arboleya, S.; Moloney, R.D.; Peterson, V.L.; Murphy, K.; Clarke, G.; et al. Targeting the Microbiota-Gut-Brain Axis: Prebiotics Have Anxiolytic and Antidepressant-like Effects and Reverse the Impact of Chronic Stress in Mice. Biol. Psychiatry 2017, 82(7), 472–487. [Google Scholar] [CrossRef]
- Jašarević, E.; Morrison, K. E.; Bale, T. L. Sex differences in the gut microbiome-brain axis across the lifespan. Philos. Trans. R. Soc. London. Ser. B Biol. Sci. 2016, 371(1688), 20150122. [Google Scholar] [CrossRef]
- Holingue, C.; Budavari, A. C.; Rodriguez, K. M.; Zisman, C. R.; Windheim, G.; Fallin, M. D. Sex Differences in the Gut-Brain Axis: Implications for Mental Health. Curr. Psychiatry Rep. 2020, 22(12), 83. [Google Scholar] [CrossRef]
- Ait-Belgnaoui, A.; Colom, A.; Braniste, V.; Ramalho, L.; Marrot, A.; Cartier, C.; et al. Probiotic gut effect prevents the chronic psychological stress-induced brain activity abnormality in mice. Neurogastroenterol. Motil. 2014, 26(4), 510–520. [Google Scholar] [CrossRef]
- Maier, H.; Helms, S.; Toto, S.; Moschny, N.; Sperling, W.; Hillemarcher, T.; et al. S100B, Homocysteine, Vitamin B12, Folic Acid, and Procalcitonin Serum Levls in Remitters to Eletroconvulsive Therapy: A Pilot Study. Dis. Markers 2018, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Callegari, E.T.; Reavley, N.; Gorelik, A.; Garland, S.M.; Wark, J.D. Serum 25-hydroxyvitamin D and mental health in young Australian women: Results from the Safe-D study. J. Affect. Disord. 2017, 224, 48–55. [Google Scholar] [CrossRef] [PubMed]
- Almeida, O.P.; Ford, A.H.; Flincker, L. Systematic review and meta-analysis of randomized placebo-controlled trials of folate and vitamin b12 for depression. Int. Psychogeriatr. 2015, 27, 727–737. [Google Scholar] [CrossRef] [PubMed]
- Magnano, M.; Valastro, I.; Rampello, L.; Vacante, M.; Malaguarnera, M.; Alessandria, I. The gut-brain axis. Effect of probiotics on anxiety. Acta Medica Mediterr. 2012, 28(3), 231–235. [Google Scholar]
- Rios, A.C.; Maurya, P.K.; Pedrini, M.; Zeni-Graiff, M.; Asevedo, E.; Mansur, R.B.; et al. Microbiota abnormalities and the therapeutic potential of probiotics in the treatment of mood disorders. Rev. Neurosci. 2017, 28(7), 739–749. [Google Scholar] [CrossRef] [PubMed]
- Clarke, G.; Grenham, S.; Scully, P.; Fitzgerald, P.; Moloney, R.D.; Shanahan, F.; et al. The microbiome-gut-brain axis during early life regulates the hippocampal serotonergic system in a sexdependent manner. Mol. Psychiatry 2013, 18, 666–673. [Google Scholar] [CrossRef]
- Al-Asmakh, M.; Hedin, L. Microbiota and the control of blood-tissue barriers. Tissue Barriers 2015, 3(3), e1039691. [Google Scholar] [CrossRef]
- Braniste, V.; Al-Asmakh, M.; Kowal, C.; Anuar, F.; Abbaspour, A.; Tóth, M.; et al. The gut microbiota influences blood-brain barrier permeability in mice. Sci. Transl. Med. 2014, 6(263), 263ra158. [Google Scholar] [CrossRef]
- Spor, A.; Koren, O.; Ley, R. Unravelling the effects of the environment and host genotype on the gut microbiome. Nat. Rev. Microbiol. 2011, 9, 279–290. [Google Scholar] [CrossRef]
- Messaoudi, M. Assessment of psychotropic-like properties of a probiotic formulation (Lactobacillus helveticus R0052 and Bifidobacterium longum R0175) in rats and human subjects. Br. J. Nutr. 2011, 105, 755–764. [Google Scholar] [CrossRef]
- Steenbergen, L. A randomized controlled trial to test the effect of multispecies probiotics on cognitive reactivity to sad mood. Brain Behav. Immun. 2015, 48, 258–264. [Google Scholar] [CrossRef]
- Kato-Kataoka, A.; Nishida, K.; Takada, M. Fermented milk containing Lactobacillus casei strain Shirota preserves the diversity of the gut microbiota and relieves abdominal dysfunction in healthy medical students exposed to academic stress. Appl. Env. Microbiol. 2016, 82, 3649–3658. [Google Scholar] [CrossRef]
- McLoughlin, R.F.; Berthon, B.S.; Jensen, M.E.; Baines, K.J.; Wood, L.G. Short-chain fatty acids, prebiotics, synbiotics, and systemic inflammation: a Systematic Review and Meta-Analysis. Am. J. Clin. Nutr. 2017, 106(3), 930–945. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, R.; Rosario, V.A.; Mocellin, M.C.; Kuntz, M.G.F.; Trindade, E.M.S.M. Effects of Inulin-type fructans, galacto-oligosaccharides and related synbiotics on inflammatory markers in adult patients with overweight or obesity: A systematic review. Clin. Nutr. 2017, 36(5), 1197–1206. [Google Scholar] [CrossRef] [PubMed]
| Characteristics | Study Groups | P value | ||
|---|---|---|---|---|
| Placebo (n=7) | Prebiotic (n=8) | Synbiotic (n=7) | ||
| Age (years) | 43.9 ± 10.0 | 40.5 ± 10.3 | 41.3 ± 4.9 | 0.753a |
| Gender (Male/ Female) | 2/5 | 0/8 | 4/3 | 0.057b |
| Body weight (kg) | 135.3 ± 28.4 | 119.9 ± 17.9 | 129.0 ± 25.4 | 0.473a |
| Body Mass Index (kg/ m²) | 50.9 ± 9.4 | 46.6 ± 5.3 | 46.0 ± 5.2 | 0.363a |
| Waist circumference (cm) | 137.3 ± 13.1 | 126.3 ± 11.6 | 136.1 ± 14.4 | 0.216a |
| Functional constipation (n/ %)* | 0 (0) | 2 (25.0) | 0 (0) | 0.582b |
| Stool consistency and shape (Bristol Scale) | 1.000b | |||
| Types 1-3 or 5-7 | 5 (71.4) | 5 (75.0) | 5 (71.4) | |
| Type 4 | 2 (28.6) | 2 (25.0) | 2 (28.6) | |
| Menopause (n/ %) | 2 (28.6) | 2 (25.0) | 0 (0) | 0.582b |
| Associated comorbidities (n/ %) | ||||
| Systemic arterial hypertension | 4 (57.1) | 1 (12.5) | 3 (42.9) | 0.119b |
| Anxiety/ Depression | 4 (57.1) | 1 (12.5) | 2 (28.6) | 0.282b |
| Type 2 Diabetes Mellitus | 1 (14.3) | 0 (0) | 2 (28.6) | 0.303b |
| Dyslipidaemia | 1 (14.3) | 0 (0) | 1 (14.3) | 0.500b |
| Others** | 1 (14.3) | 3 (37.5) | 4 (57.1) | 0.266b |
| Drugs for continuous use (n/ %) | ||||
| Antihypertensives | 4 (57.1) | 1 (12.5) | 3 (42.9) | 0.119b |
| Antidepressants | 4 (57.1) | 1 (12.5) | 2 (28.6) | 0.282b |
| Oral hypoglycaemic agents | 1 (14.3) | 0 (0) | 2 (28.6) | 0.303b |
| Statins | 0 (0) | 0 (0) | 0 (0) | 1.000b |
| Others*** | 1 (14.3) | 2 (25.0) | 1 (14.3) | 1.000b |
| Outcomes | Study groups | P valuea | ||
|---|---|---|---|---|
| Placebo | Prebiotic | Synbiotic | ||
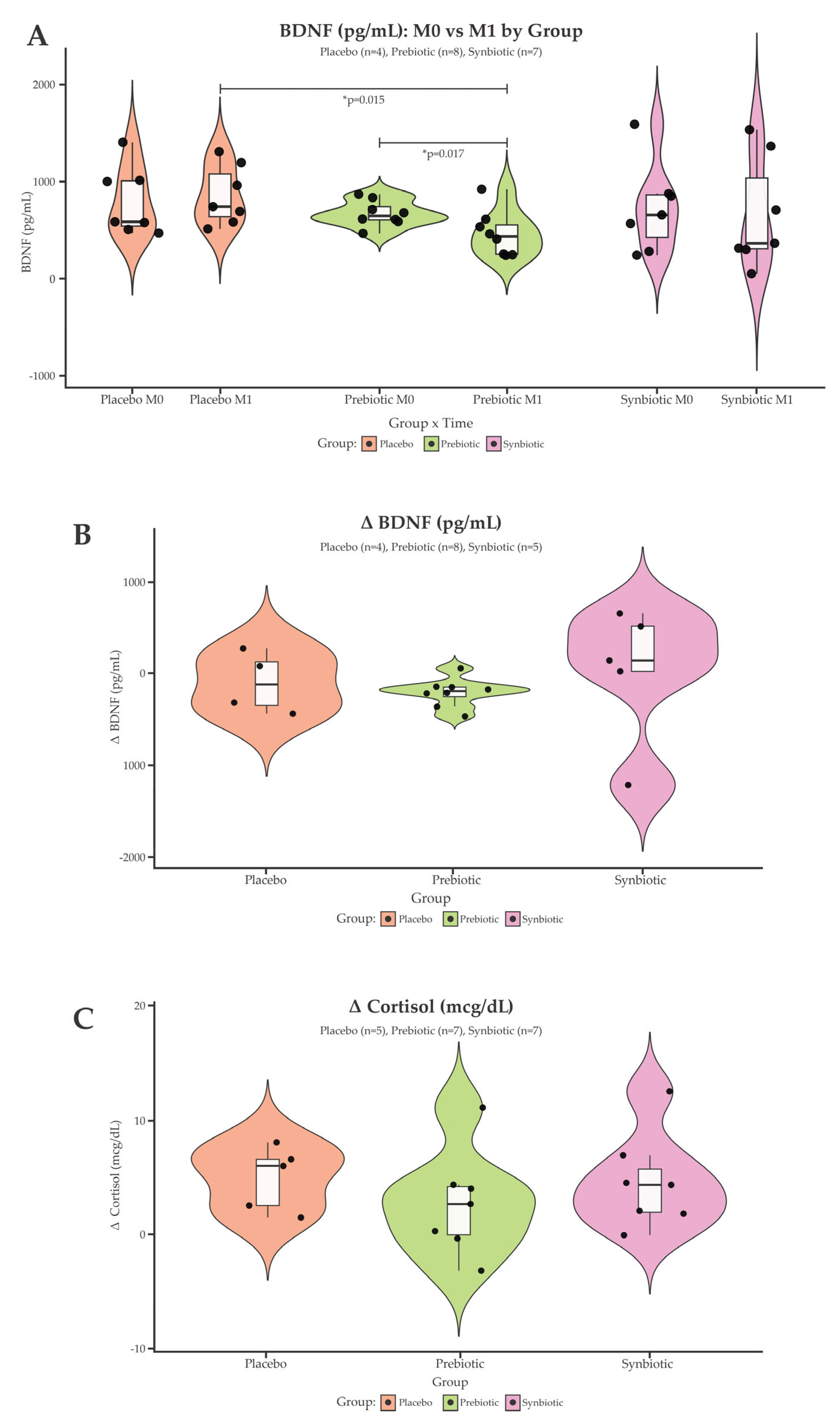
| BDNF (pg/ mL) | (n=7) | (n=8) | (n=7) | |
| Baseline | 575.2 (496.6; 1001.7) | 635.8 (588.3; 761.7) | 645.3 (271.1; 864.7) | 0.959 |
| Endline | 731.2 (572.8; 1182.7) | 424.7 (241.0; 563.0) | 355.2 (291.9; 1350.9) | 0.098 |
| P value (paired test)b | 0.735 | 0.017 | 0.735 | |
| Difference between moments | 76.16 (-319.9; 305.2) | -194.6 (-291.5; -148.3) | 71.9 (-603.2; 514.2) | 0.226 |
| ACTH (pg/ mL) | (n=7) | (n=8) | (n=7) | |
| Baseline | 13.9 (9.5; 17.2) | 12.7 (11.2; 15.0) | 19.0 (7.5; 35.6) | 0.759 |
| Endline | 12.5 (7.9; 15.9) | 17.6 (12.6; 19.3) | 21.5 (7.0; 42.6) | 0.368 |
| P value (paired test)b | 0.671 | 0.092 | 0.612 | |
| Difference between moments | -0.7 (-3.8; 3.0) | 3.3 (1.0; 7.1) | 1.2 (-4.5; 9.6) | 0.343 |
| Cortisol (µg/ dL) | (n=7) | (n=8) | (n=7) | |
| Baseline | 9.4 (4.8; 11.7) | 7.7 (5.5; 9.3) | 6.2 (5.7; 7.3) | 0.223 |
| Endline | 10.8 (8.4; 12.4) | 9.6 (6.9; 11.2) | 10.2 (8.6; 12.9) | 0.751 |
| P value (paired test)b | 0.398 | 0.092 | 0.028 | |
| Difference between moments | 2.5 (-4.7; 6.5) | 3.3 (-0.0; 4.1) | 4.3 (1.8; 6.9) | 0.678 |
| TSH (µUI/ mL) | (n=6) | (n=7) | (n=7) | |
| Baseline | 2.34 (1.6; 2.4) | 2.08 (1.7; 3.2) | 1.56 (1.1; 2.3) | 0.375 |
| Endline | 2.14 (1.8; 2.9) | 2.52 (1.7; 4.1) | 2.12 (1.8; 2.5) | 0.422 |
| P value (paired test)b | 0.600 | 0.236 | 0.310 | |
| Difference between moments | 0.09 (-0.1; 0.2) | 0.45 (-0.4; 1.2) | 0.18 (-0.07; -0.5) | 0.710 |
| PTH (pg/ mL) | (n=6) | (n=8) | (n=7) | |
| Baseline | 56.3 (45.5; 62.3) | 67.1 (61.5; 108.0) | 82.8 (48.7; 86.6) | 0.212 |
| Endline | 51.2 (27.4; 64.4) | 64.0 (58.2; 79.2) | 80.8 (46.0; 98.8) | 0.141 |
| P value (paired test)b | 0.173 | 0.123 | 0.398 | |
| Difference between moments | -8.9 (-14.9; 2.9) | -5.95 (-15.9; 1.7) | 11.7 (-5.2; 14.5) | 0.201 |
| Vitamin D (ng/ mL) | (n=7) | (n=8) | (n=7) | |
| Baseline | 19.8 (13.7; 23.1) | 23.0 (15.9; 25.1) | 25.2 (14.8; 31.0) | 0.362 |
| Endline | 19.3 (11.7; 23.8) | 23.2 (19.7; 25.3) | 27.6 (24.0; 33.5) | 0.079 |
| P value (paired test)b | 0.865 | 0.327 | 0.120 | |
| Difference between moments | -1.3 (-2.9; 4.0) | 0.7 (-1.0; 2.8) | 2.5 (-0.7; 2.8) | 0.429 |
| Vitamin B12 (pg/ mL) | (n=7) | (n=8) | (n=7) | |
| Baseline | 419.0 (298.0; 627.0) | 348.0 (289.5; 446.5) | 546.0 (345.0; 603.0) | 0.338 |
| Endline | 325.0 (309.0; 588.0) | 347.0 (289.0; 431) | 516.0 (375.0; 610.0) | 0.247 |
| P value (paired test)b | 0.865 | 0.888 | 0.671 | |
| Difference between moments | 8.0 (-76.0; 105.0) | 4.0 (-93.5; 57.5) | 28.0 (-30.0; 48.0) | 0.918 |
| Folic acid (ng/ mL) | (n=7) | (n=8) | (n=7) | |
| Baseline | 11.8 (9.1; 21.7) | 13.1 (10.2; 14.2) | 15.7 (11.6; 24.1) | 0.323 |
| Endline | 11.5 (7.8; 15.1) | 14.1 (7.9; 16.3) | 12.9 (11.8; 16.4) | 0.700 |
| P value (paired test)b | 0.499 | 0.674 | 0.176 | |
| Difference between moments | -1.8 (-8.6; 5.1) | 1.0 (-3.0; 4.9) | -3.1 (-7.6; 2.1) | 0.408 |
| hs-CRP (mg/ L) | (n=7) | (n=8) | (n=7) | |
| Baseline | 8.9 (3.5; 10.5) | 11.2 (7.9; 19.8) | 9.0 (2.5; 10.8) | 0.339 |
| Endline | 5.8 (5.5; 22.8) | 8.6 (4.1; 13.4) | 6.7 (4.1; 10.5) | 0.957 |
| P value (paired test)b | 0.671 | 0.207 | 0.735 | |
| Difference between moments | -1.2 (-3.2; 2.9) | -1.9 (-5.5; 1.3) | 1.5 (-3.1; 1.6) | 0.606 |
| IL-1β (pg/ mL) | (n=7) | (n=8) | (n=7) | |
| Baseline | 7.7 (2.2; 8.1) | 6.5 (5.5; 7.7) | 3.0 (0; 9.5) | 0.502 |
| Endline | 7.9 (6.6; 10.0) | 8.2 (6.3; 10.7) | 5.9 (0; 7.2) | 0.097 |
| P value (paired test)b | 0.310 | 0.262 | 0.799 | |
| Difference between moments | 1.9 (-2.2; 7.0) | 1.6 (-0.7; 4.1) | 0 (-3.6; 3.2) | 0.547 |
| IL-6 (pg/ mL) | (n=7) | (n=8) | (n=7) | |
| Baseline | 8.7 (6.2; 11.2) | 10.7 (8.5; 12.4) | 6.6 (4.5; 10.5) | 0.103 |
| Endline | 9.8 (5.6; 12.2) | 11.3 (6.7; 12.0) | 7.7 (4.6; 9.1) | 0.495 |
| P value (paired test)b | 0.499 | 0.483 | 0.612 | |
| Difference between moments | 1.1 (-1.1; 2.6) | -0.7 (-1.8; 1.2) | 0.09 (-1.4; 2.5) | 0.510 |
| TNF-α (pg/ mL) | (n=7) | (n=8) | (n=7) | |
| Baseline | 4.0 (2.6; 8.6) | 4.4 (3.4; 5.8) | 4.9 (0; 10.7) | 0.962 |
| Endline | 5.0 (1.9; 13.8) | 7.0 (3.6; 11.5) | 6.7 (3.6; 11.8) | 0.927 |
| P value (paired test)b | 0.735 | 0.035 | 0.350 | |
| Difference between moments | 0.7 (-2.6; 5.5) | 2.9 (0.4; 5.3) | 1.3 (-1.6; 6.8) | 0.645 |
| GROUPS | BDNF versus Vit. B12 | BDNF versus IL-6 | BDNF versus ACTH | ACTH versus IL-6 |
|---|---|---|---|---|
| Placebo | -0.0357 | -0.2143 | -0.6071 | 0.7857 |
| Prebiotic | -0.0476 | 0.0952 | -0.3095 | 0.3095 |
| Synbiotic | 0.8929 | -0.7143 | -0.3571 | -0.0714 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




