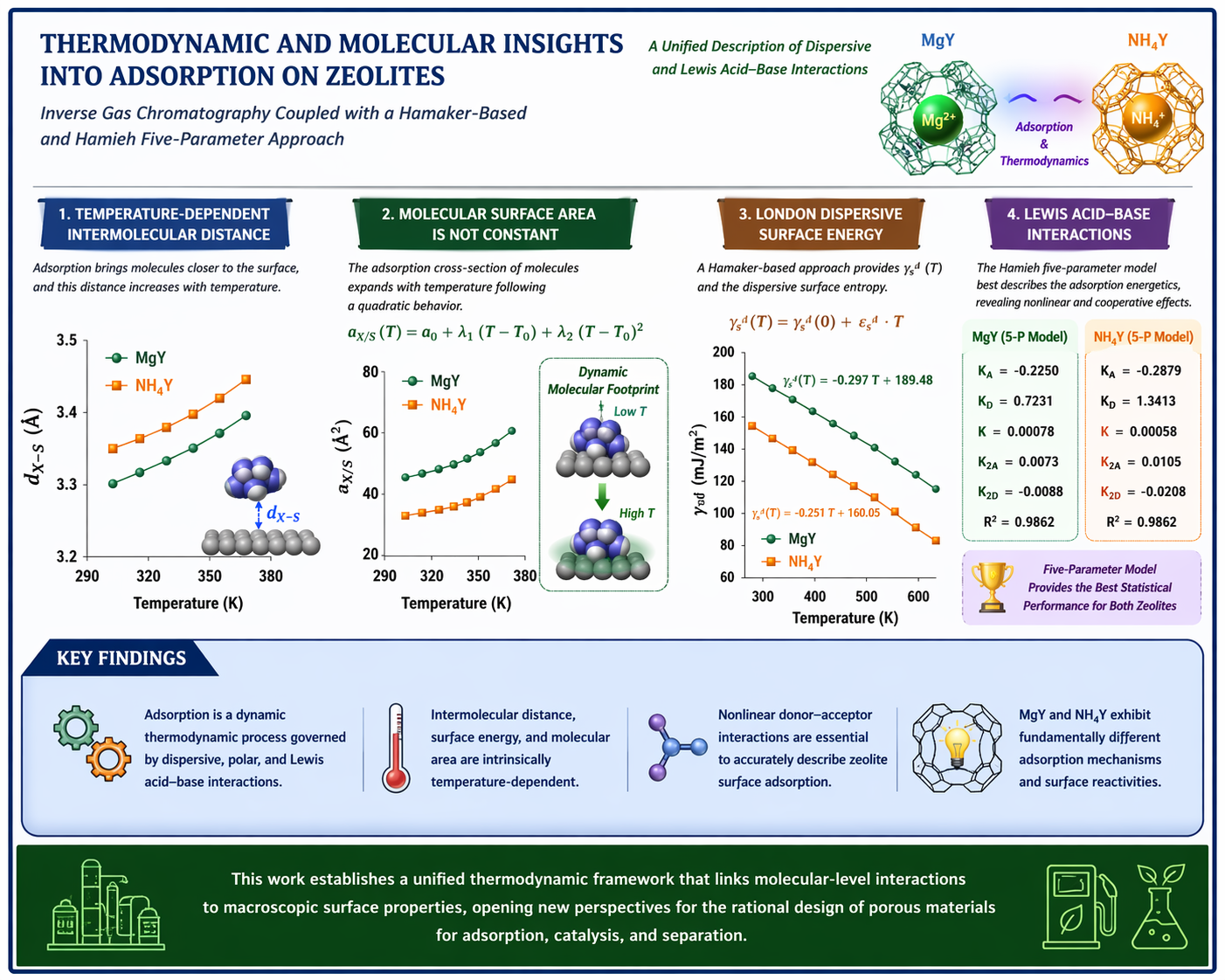
A comprehensive thermodynamic and molecular-level investigation of adsorption on MgY and NH4Y zeolites is presented using inverse gas chromatography at infinite dilution combined with a Hamaker-based formalism and an extended five-parameter Lewis acid–base model. The study establishes a unified framework that integrates dispersive, polar, and donor–acceptor interactions while explicitly accounting for temperature-dependent intermolecular geometry. The results demonstrate that adsorption is governed by a dynamic interplay between London dispersion forces, specific acid–base interactions, and thermal effects, rather than by static or purely additive contributions. The London dispersive surface energy decreases linearly with temperature, reflecting the progressive weakening of electronic correlation forces, while the inter-molecular separation distance exhibits a well-defined linear expansion, enabling the determination of intrinsic contact distances at 0 K. A major finding of this work is that the molecular surface area of adsorbed probes is not a constant geometric property but a thermodynamic quantity that follows a quadratic temperature dependence, revealing the adaptive nature of adsorption geometry. The comparison between MgY and NH4Y highlights two distinct adsorption regimes: MgY is characterized by a structured and strongly dispersive interaction field associated with Mg2+ cations, whereas NH4Y exhibits enhanced polarity, stronger specific interactions, and increased molecular flexibility driven by hydrogen bonding and protonic effects. The thermodynamic analysis of Lewis acid–base interactions shows that classical linear models are insufficient to describe adsorption on zeolite surfaces. A rigorous statistical evaluation demonstrates that the Hamieh five-parameter model provides the most accurate and physically meaningful description, capturing nonlinear donor–acceptor interactions and amphoteric coupling effects. Overall, this work introduces a novel thermodynamic methodology that links macroscopic surface energetics to microscopic interaction parameters, providing new insights into adsorption mechanisms and offering a powerful tool for the rational design of porous materials in catalysis, separation, and energy-related applications.