Submitted:
22 April 2026
Posted:
23 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction and Clinical Significance
2. Materials and Methods
2.1. Study Design
2.2. Focused Literature Review
2.3. Clinical Workflow and Selection Considerations
- -
- confirm the presence and degree of tumour vascularity (tumour blush, early venous drainage);
- -
- identify arterial feeders potentially amenable to superselective catheterisation;
- -
- evaluate risks related to perforators, dangerous anastomoses, and “en passage” supply.
3. Case Presentation
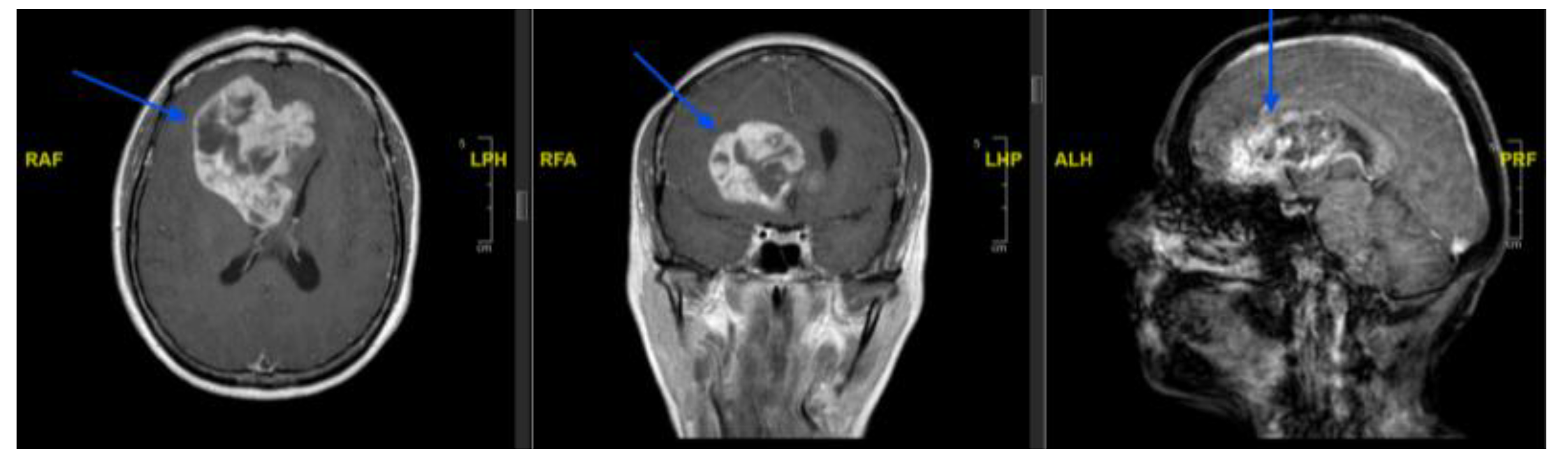
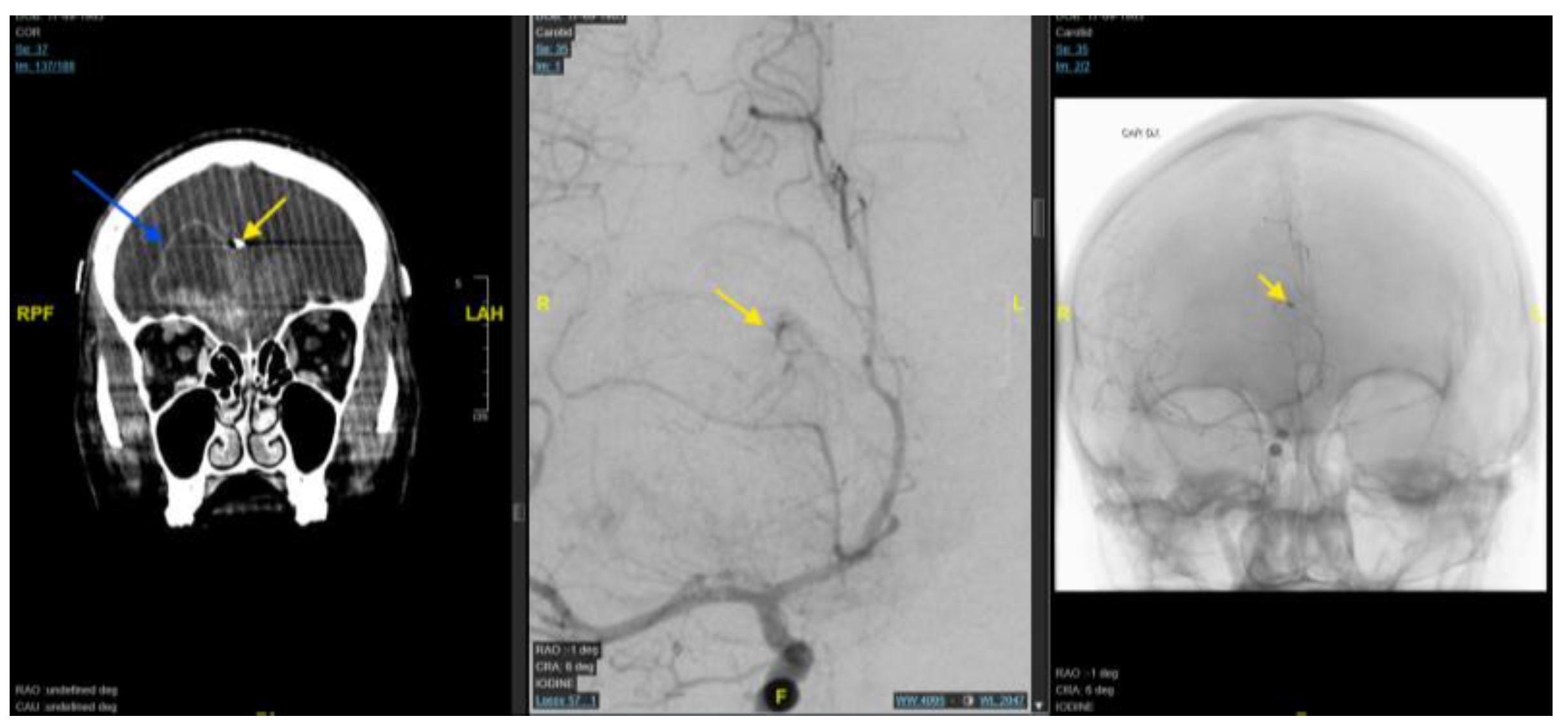
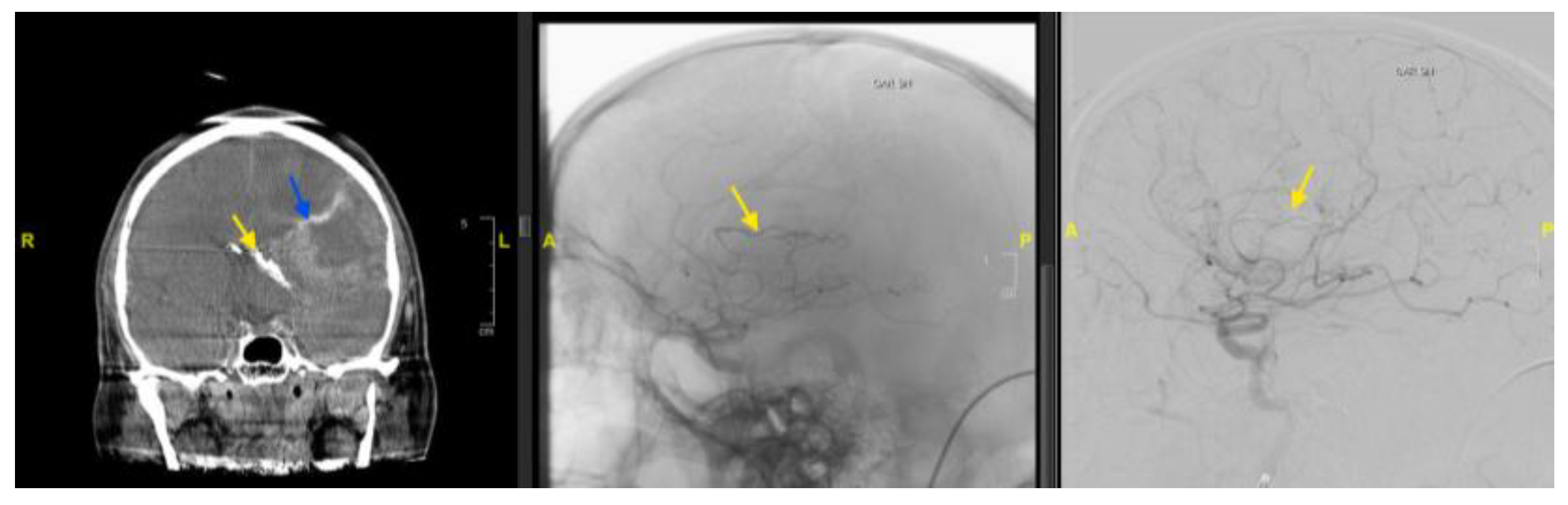
3.1. Case 1
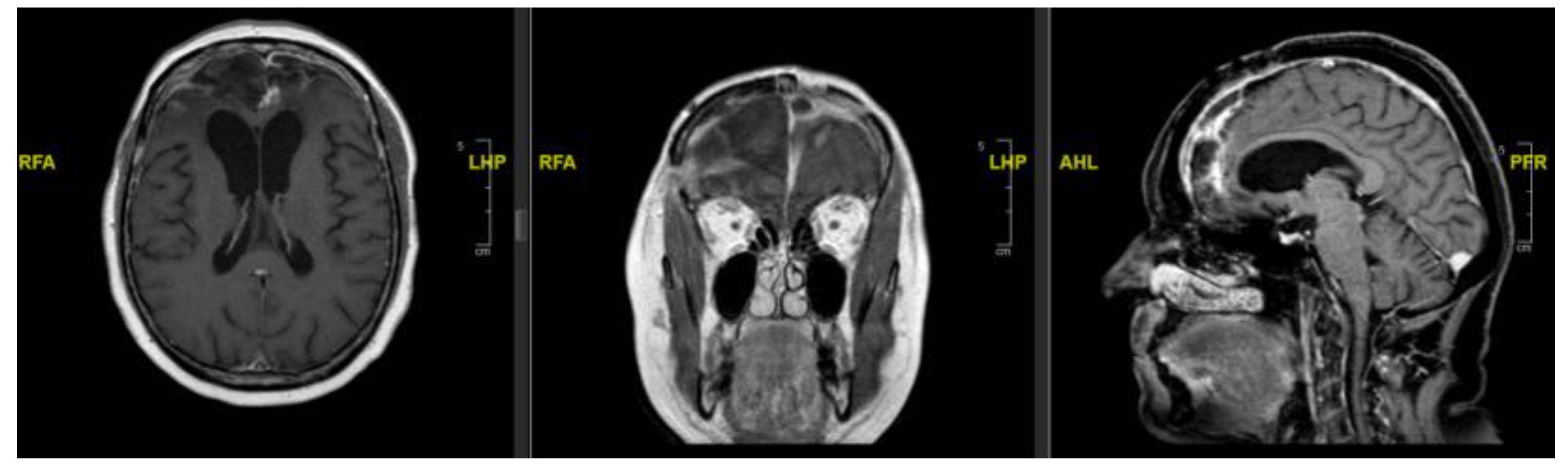
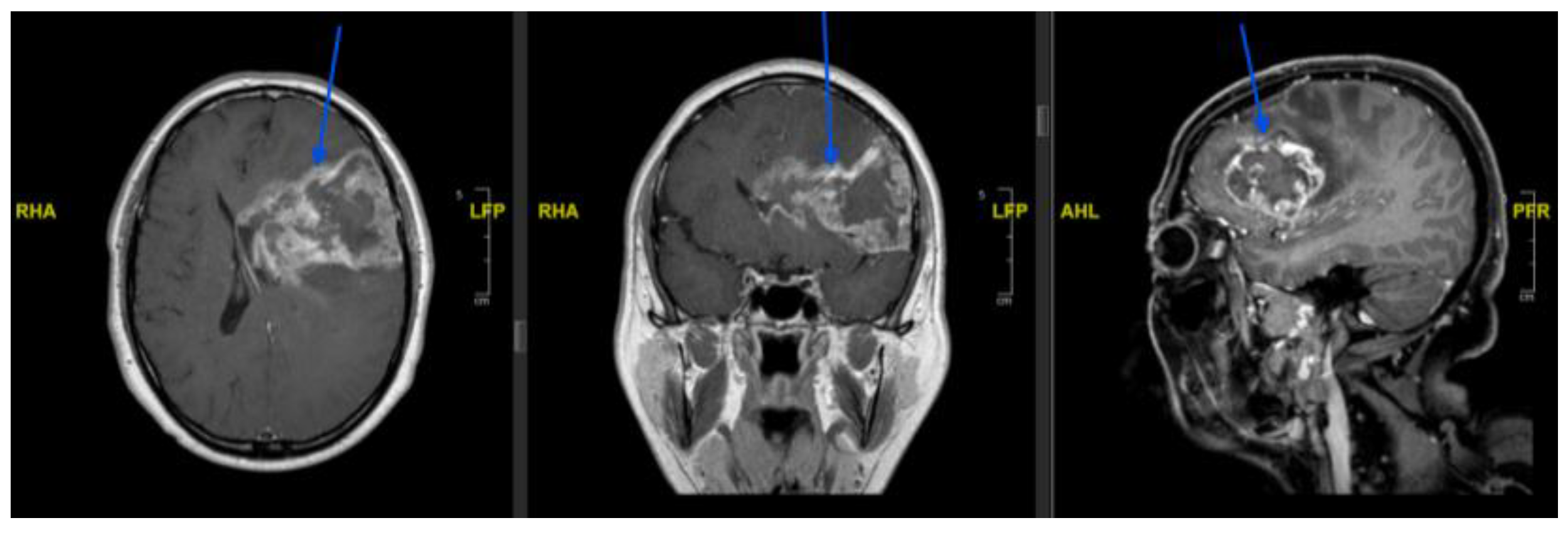
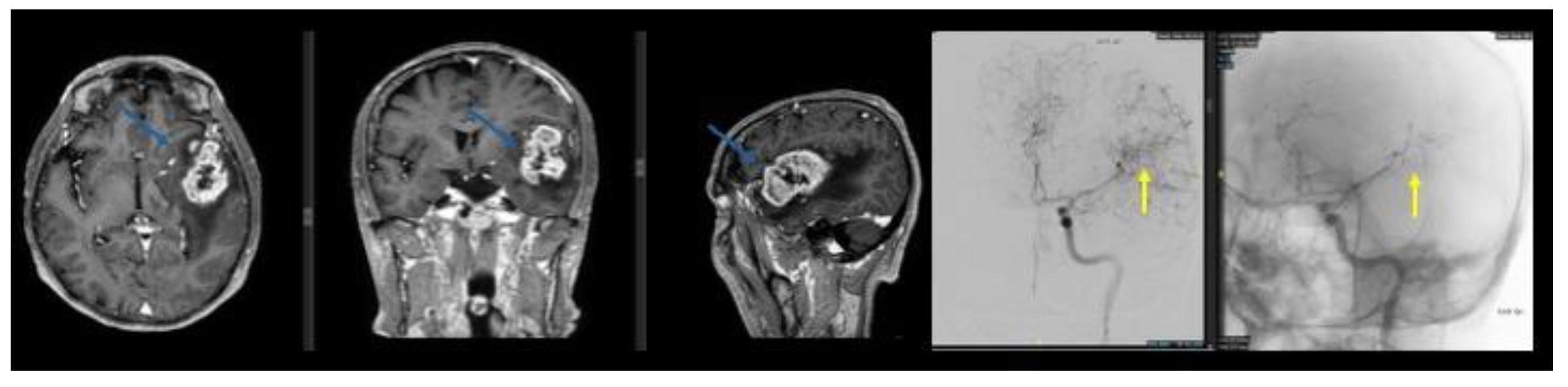
3.2. Case 2
3.3. Case 3
4. Discussion
4.1. Role of DSA in Selected Suspected Glioblastomas
4.2. Feasibility of Embolisation and Technical Considerations
4.3. Potential Clinical Benefit
4.4. Safety Considerations
4.5. Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ACA | anterior cerebral artery |
| AChA | anterior choroidal artery |
| CC | corpus callosum |
| CT | computed tomography |
| DSA | digital subtraction angiography |
| NBCA | N-butyl cyanoacrylate |
| MRI | magnetic resonance imaging |
References
- Achi-Arteaga J, Flores-Vazquez JG, Fuentes-Calvo I, Gonzalez-Salido J, Wong-Achi X. Preoperative embolization of brain, head, and neck tumors: Single center experience and literature review. J Cerebrovasc Endovasc Neurosurg. 2025 Sep;27(3):195-211. [CrossRef] [PubMed] [PubMed Central]
- Imai T, Ohshima T, Nishizawa T, Shimato S, Kato K. Successful Preoperative Endovascular Embolization of an Extreme Hypervascular Glioblastoma Mimicking an Arteriovenous Malformation. World Neurosurg. 2016 Feb;86:512.e1-4. [CrossRef] [PubMed]
- Sakata T, Tanikawa M, Yamada H, Fujinami R, Nishikawa Y, Yamada S, Mase M. Minimally invasive treatment for glioblastoma through endoscopic surgery including tumor embolization when necessary: a technical note. Front Neurol. 2023 Apr 21;14:1170045. [CrossRef] [PubMed] [PubMed Central]
- Ashour R, Aziz-Sultan A. Preoperative tumor embolization. Neurosurg Clin N Am. 2014 Jul;25(3):607-17. [CrossRef] [PubMed]
- Duffis EJ, Gandhi CD, Prestigiacomo CJ, Abruzzo T, Albuquerque F, Bulsara KR, Derdeyn CP, Fraser JF, Hirsch JA, Hussain MS, Do HM, Jayaraman MV, Meyers PM, Narayanan S; Society for Neurointerventional Surgery. Head, neck, and brain tumor embolization guidelines. J Neurointerv Surg. 2012 Jul;4(4):251-5. [CrossRef] [PubMed] [PubMed Central]
- Pal A, Blanzy J, Gómez KJR, Preul MC, Vernon BL. Liquid Embolic Agents for Endovascular Embolization: A Review. Gels. 2023 May 4;9(5):378. [CrossRef] [PubMed] [PubMed Central]
| CASE | AGE/SEX | LOCATION | DSA FINDINGS | EMBOLISATION FEASIBLE | TARGET VESSEL(S) | EMBOLIC AGENT | ANGIOGRAPHIC RESULT | POSTOPERATIVE COURSE |
|---|---|---|---|---|---|---|---|---|
| 1 | 61/F | Right frontal | Tumour blush; discrete feeder | Yes | Frontal branch of right ACA | Coils | Partial devascularisation | No new deficit/complication |
| 2 | 53/M | Left temporo-fronto-insular + CC | Tumour supply from AChA | Yes | AChA tumour feeders | NBCA + Lipiodol | Targeted devascularisation | No new deficit/complication |
| 3 | 77/M | Left temporo-parietal | Capillary blush; no catheterisable feeders | No | — | — | — | No new deficit/complication |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




