3. Discussion
A liver abscess is defined as a collection of suppurative material enclosed within the liver parenchyma. It may be infected by bacteria, fungi, or parasites. Multiple risk factors are linked to increased mortality. These risk factors include diabetes mellitus, cirrhosis, immunocompromised status, prolonged use of proton pump inhibitors (PPIs), gender, and age over 57 years. Individuals with compromised immune systems due to chemotherapy, immunosuppressive therapy, or inherited or acquired immunodeficiency syndromes are at are higher risk for liver abscesses caused by fungi and opportunistic microorganisms [
1,
2,
5].
The primary mechanism is hematogenous infection via the hepatic artery during severe septic processes, such as metastatic-pyemic liver abscess, or through the portal vein in cases of liver abscess associated with portal vascular thrombosis. Occasionally, infection spreads through the umbilical vein (as in omphalophlebitis) or via the biliary tract and may also result from parasitic invasion or the presence of foreign bodies. Additionally, liver abscess can develop from the extension of an inflammatory process from adjacent structures, direct liver injury, intrahepatic hematoma, or postoperative complications [
2].
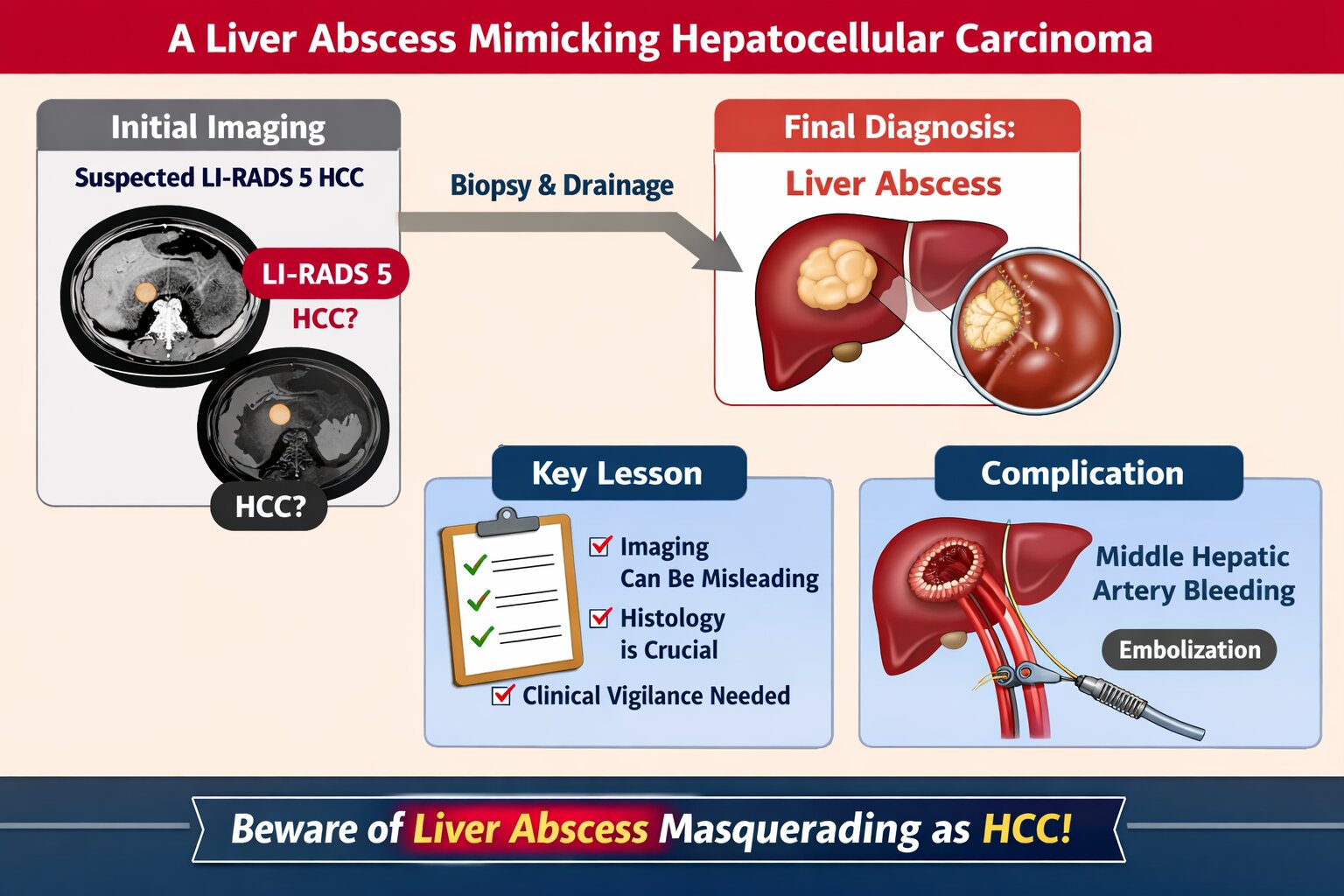
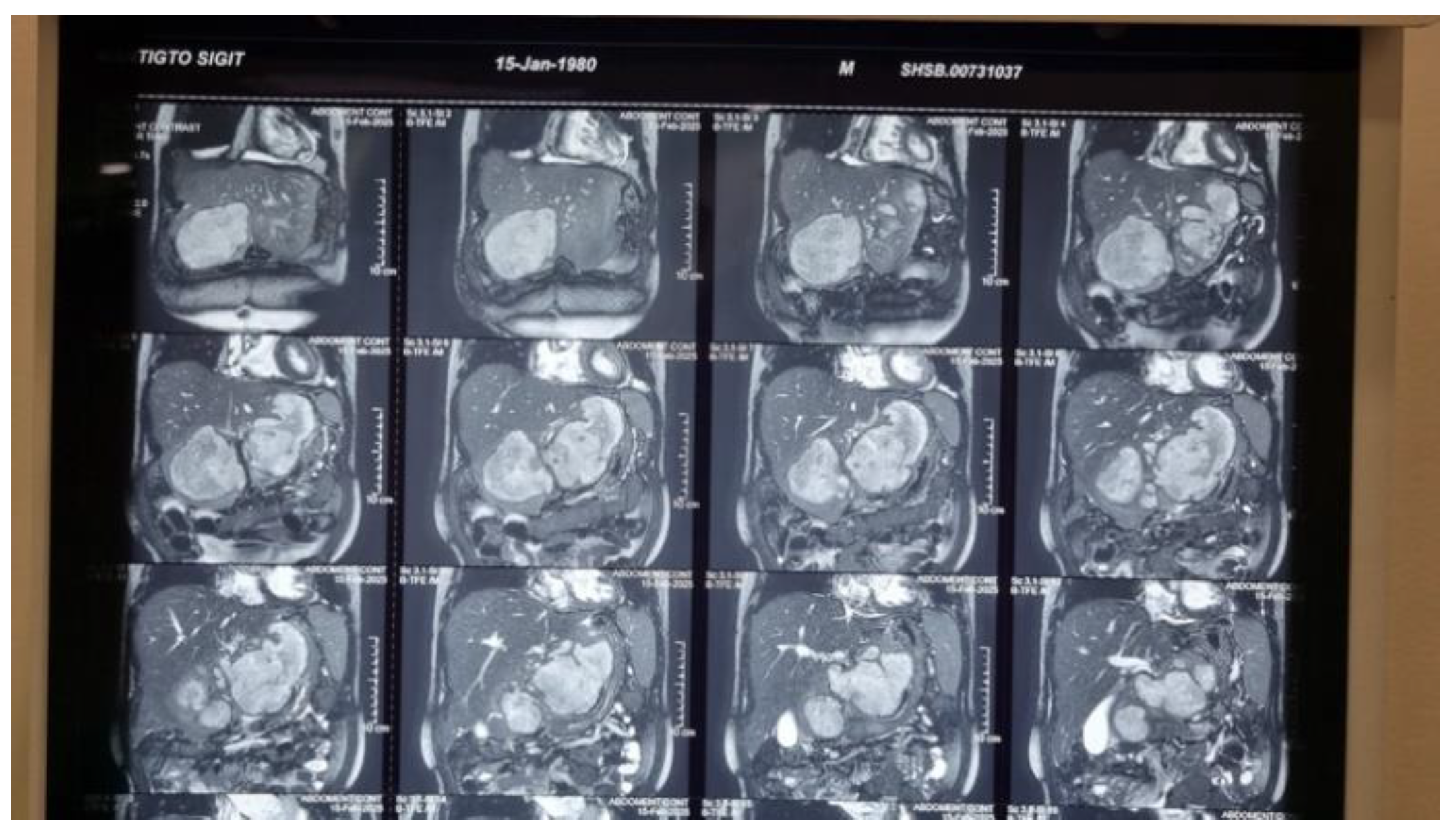
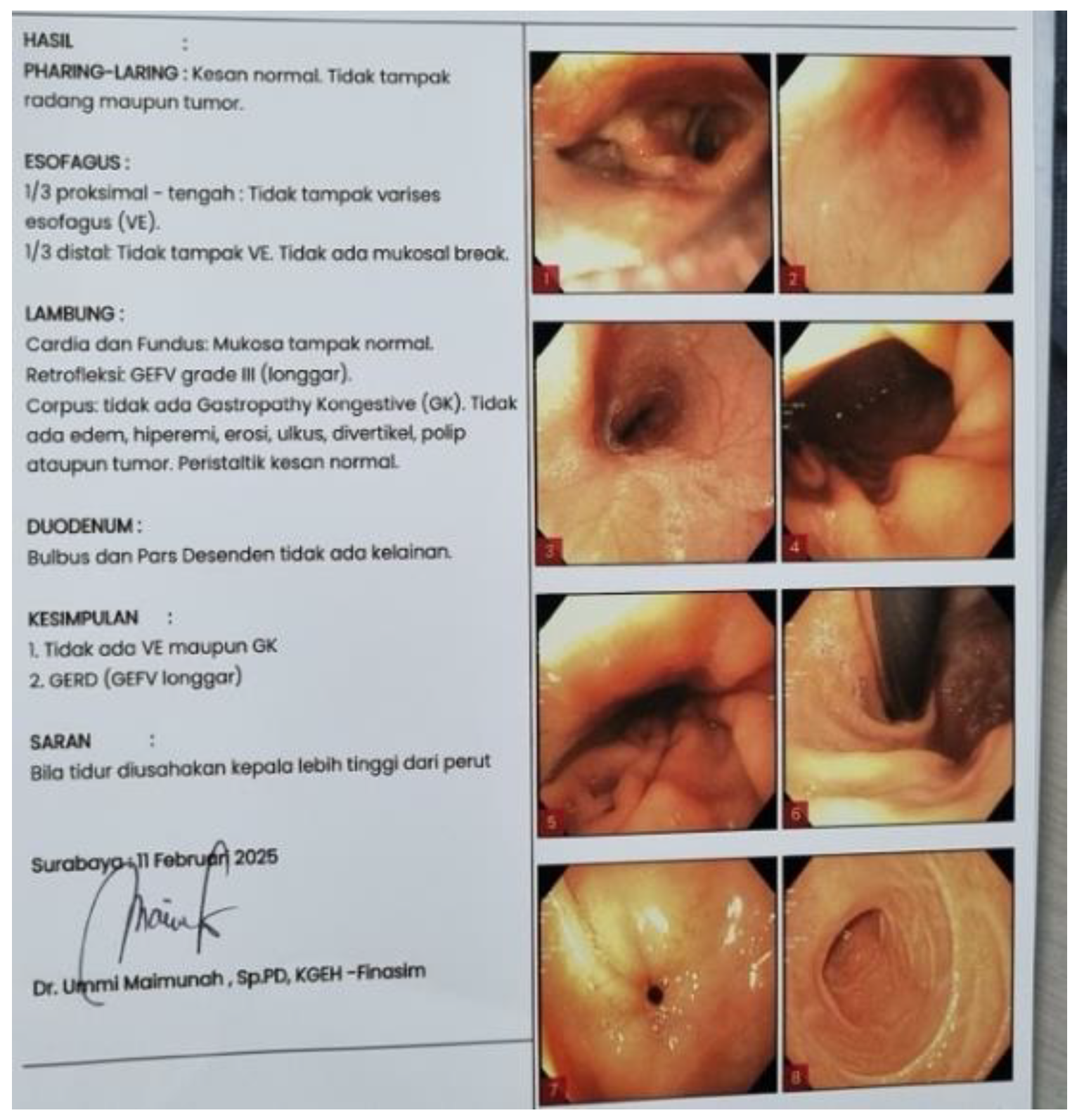
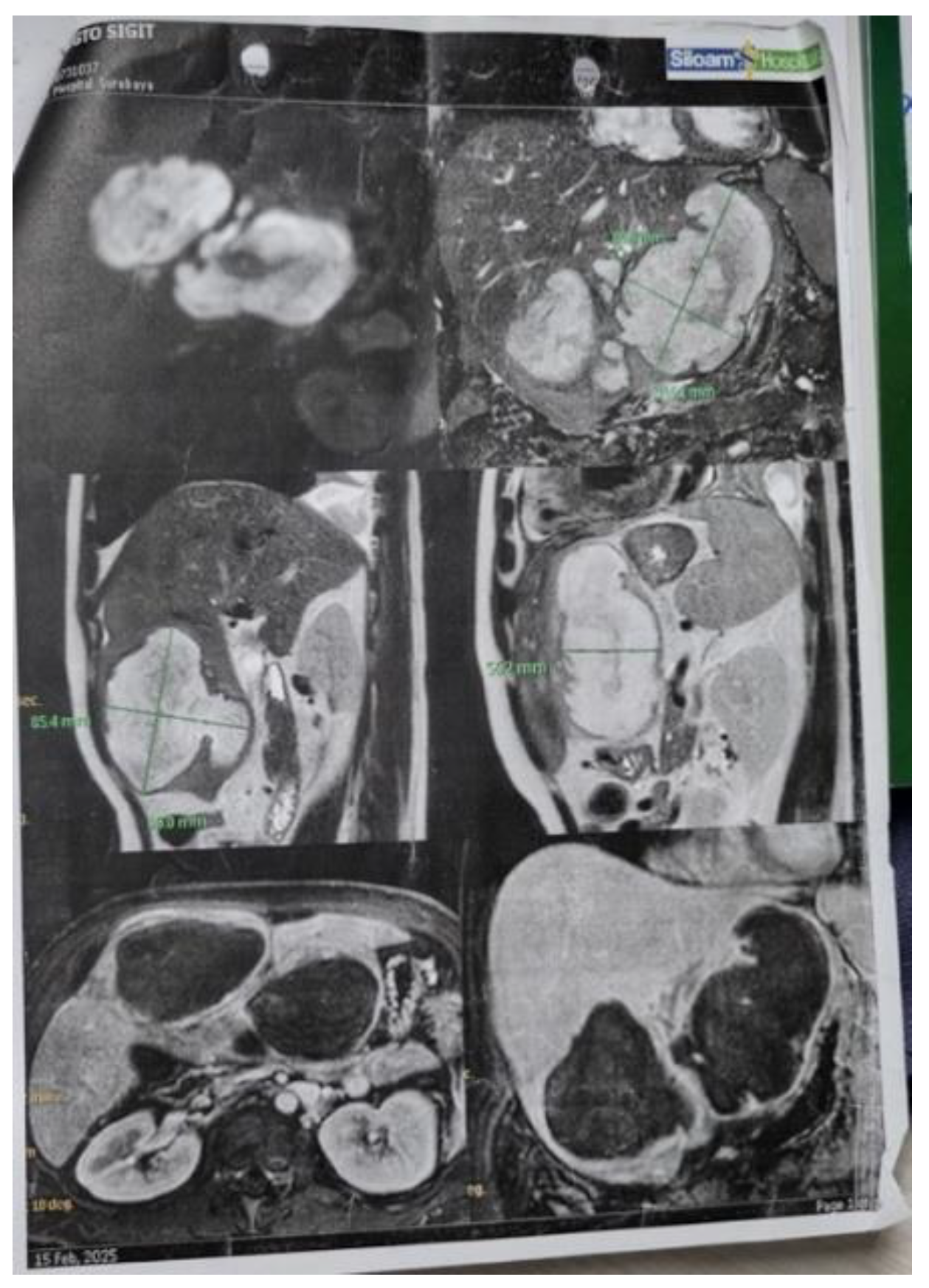
Liver abscesses are classified into three categories: infectious, malignant, and iatrogenic. Malignant abscesses are further subdivided into secondary infection of primary liver tumors, secondary infection of metastatic liver lesions, and spontaneous necrotic superinfection. Primary hepatocellular carcinoma (HCC) may develop spontaneously in areas of central necrosis, which are susceptible to bacterial infection. HCC can also result in biliary obstruction, potentially leading to ascending cholangitis and subsequent liver abscess formation. In some cases, liver abscess may serve as an early manifestation of HCC. Patients with this presentation frequently have a poorer prognosis, as the diagnosis of liver abscess can delay identification of the underlying HCC. Several imaging features have been reported to support the identification of superinfected malignancy, including thickened walls, septation, aerobilia, portal vein thrombosis, and gas within the abscess1. In the present case, non-contrast MRI of the abdomen suggested either (1) hepatic lymphoma or (2) atypical HCC with cystic or necrotic degeneration. The mass was located adjacent to the intrahepatic bile duct (IHBD) with mild distal IHBD dilatation. Additional findings included minimal right and left-sided pleural effusion (maximum thickness 3 cm) and a cyst in the lower pole of the right kidney (Bosniak II).
The most commonly observed clinical manifestations are fever, abdominal pain, and hypotension. The proportion of patients presenting with each symptom varies considerably, indicating substantial heterogeneity in clinical presentation. The challenge in establishing a diagnosis is further demonstrated by the average delay of 1 week between symptom onset and diagnosis. In the present case, the patient exhibited right upper quadrant abdominal pain and stiffness, accompanied by nausea and vomiting persisting for ten days prior to admission. On examination, his temperature was recorded at 37.8°C.
Laboratory findings in patients with liver abscesses are generally nonspecific. The most frequent abnormalities include elevated leukocyte count, increased C-reactive protein (CRP), hypoalbuminemia, elevated aspartate aminotransferase and alanine aminotransferase, elevated alkaline phosphatase, increased gamma-glutamyl transpeptidase, elevated bilirubin, and an increased international normalized ratio (INR). Although laboratory testing alone is insufficient for a definitive diagnosis, these abnormalities typically prompt further imaging, which is essential for confirming the presence of a liver abscess1. In the present case, the laboratory results demonstrated leukocytosis, hypoalbuminemia, elevated ALT, alkaline phosphatase, gamma GT, direct bilirubin, and total bilirubin, which are consistent with the findings commonly reported in the literature.
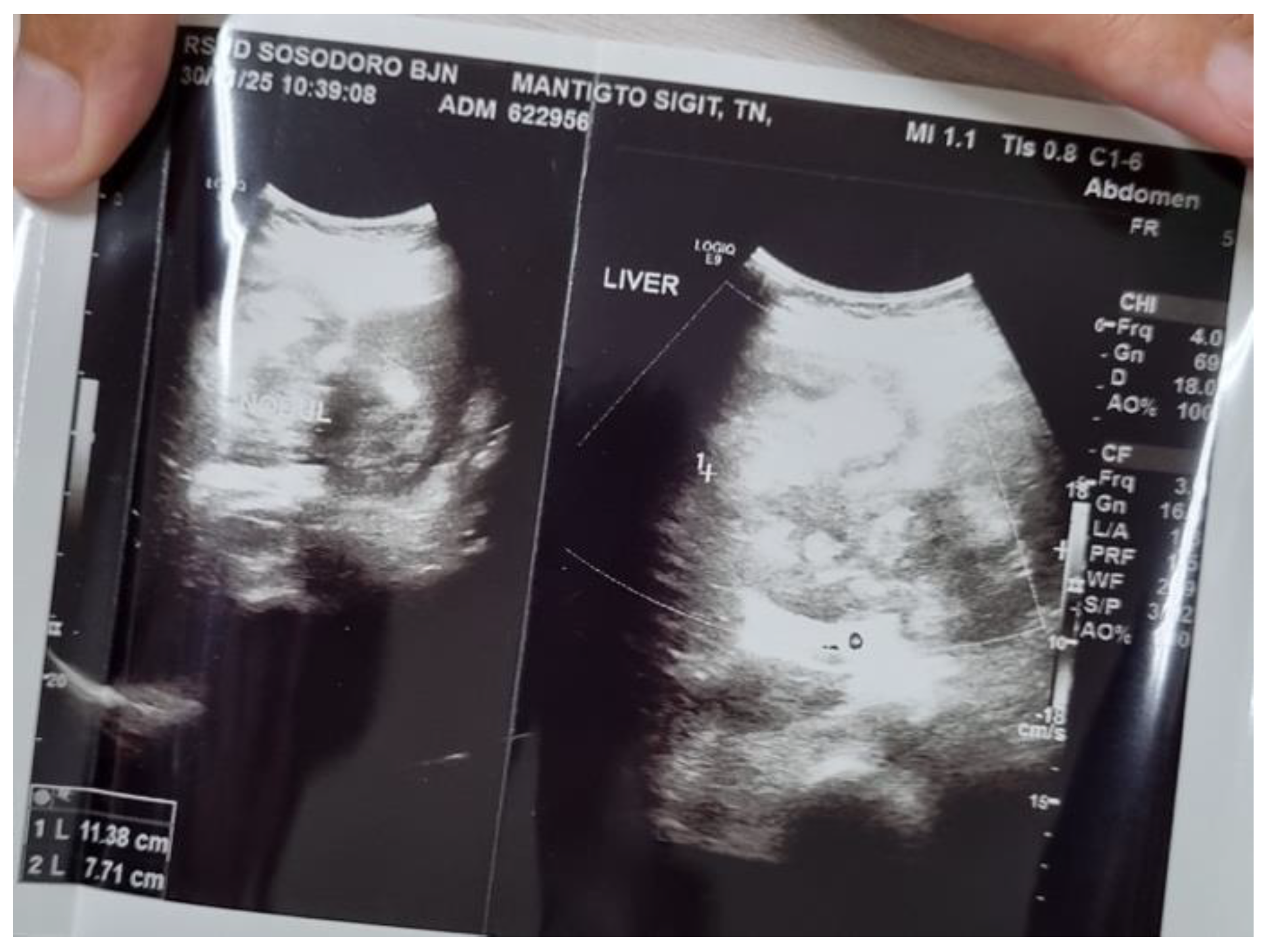
Imaging establishes the diagnosis of a liver abscess in approximately 90% of cases and can also help identify the underlying etiology. The primary diagnostic modalities include conventional ultrasonography (US) and computed tomography (CT), both with sensitivities of 96% to 100% for detecting liver abscesses. US typically reveals hypoechoic lesions with variable internal echogenicity, influenced by the presence of septations or gas. If US is nondiagnostic, CT, magnetic resonance imaging (MRI), or contrast-enhanced ultrasound (CEUS) should be employed. On noncontrast CT, liver abscesses exhibit lower attenuation than normal hepatic parenchyma. Following intravenous (IV) administration of iodinated contrast, CT scans may demonstrate peripheral enhancement and enhanced internal septations. On MRI, liver abscesses are hyperintense on T2-weighted images and hypointense on noncontrast T1-weighted images, although some may appear hyperintense on noncontrast T1-weighted images depending on protein content. After gadolinium administration, enhancement patterns are similar to those observed on CT scans [
1].
Imaging techniques, including US-guided needle aspiration and CT scan, are used to confirm the diagnosis of liver abscess, as the material obtained can be analyzed to identify the etiologic agent. These procedures also serve a therapeutic function in percutaneous drainage, which will be addressed subsequently. Imaging studies provide information on the location, size, number, consistency, and presence of gas within abscesses. The right lobe of the liver is the most frequent site, accounting for 68.7% of cases in one study. Solitary abscesses are observed more frequently than multiple abscesses. In terms of consistency, 58% of abscesses are solid, while 42% are cystic. Gas is detected in approximately 17% of cases [
1].
Liver abscesses demonstrate a spectrum of radiographic features, ranging from well-defined cystic lesions with rim enhancement to heterogeneously enhancing mass-like lesions that may be difficult to distinguish from hepatic neoplasms [
4]. Although the imaging characteristics of liver abscesses vary depending on the stage of maturation and internal composition, it remains uncommon for a liver abscess to closely mimic a malignant liver tumor such as hepatocellular carcinoma (HCC) [
4,
6,
7]. Previous reports have documented cases in which liver abscesses presented with imaging findings similar to HCC, sometimes in the absence of clinical or laboratory indicators of infection [
4,
7]. In certain cases, additional findings, such as portal vein thrombosis or elevated inflammatory markers, have helped differentiate abscess from malignancy [
8].
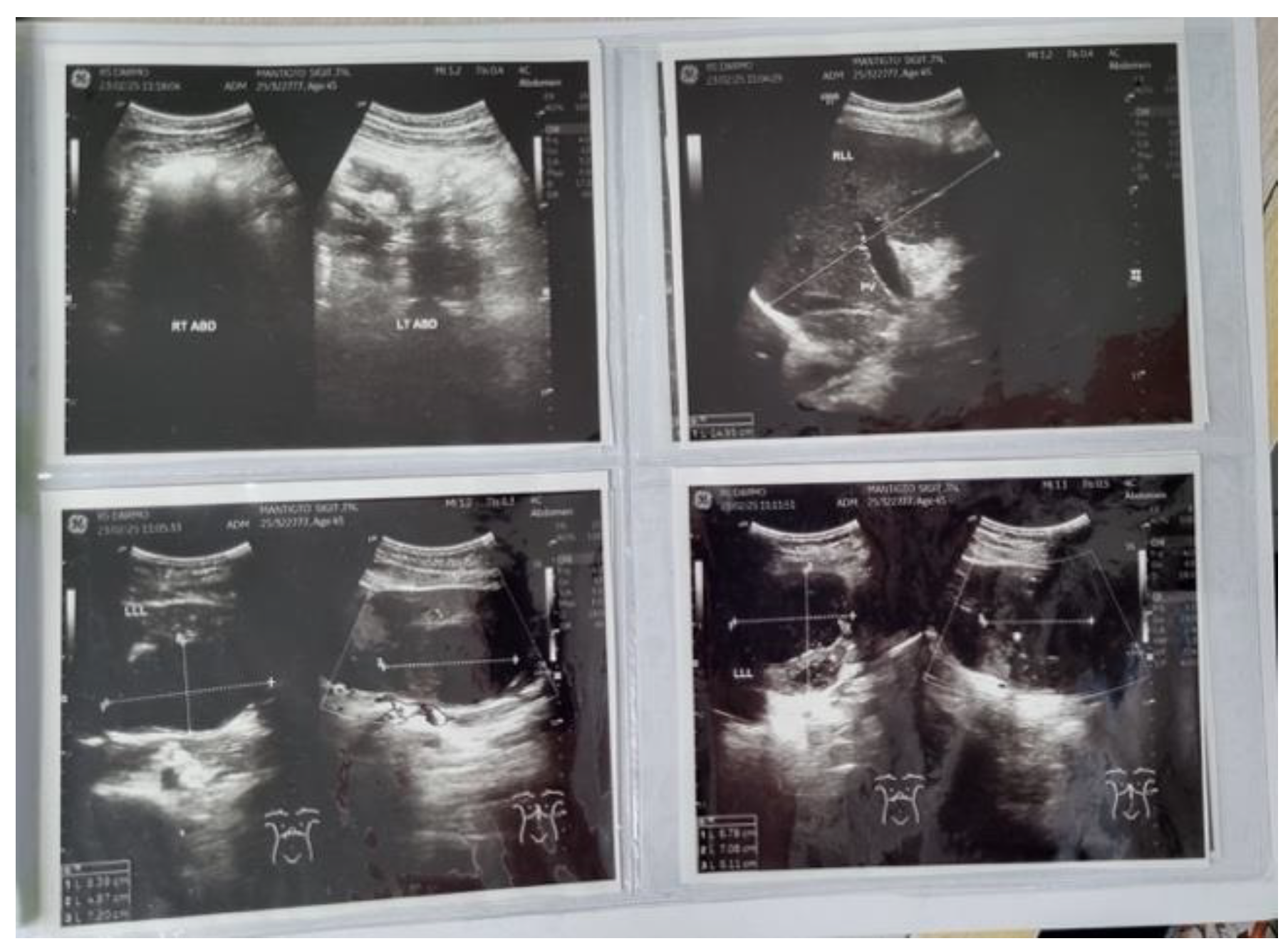
However, unlike previous reports, our case was considerably complex to distinguish between liver abscess and HCC due to the lack of clinical presentation suggesting an abscess. Initially, our case was diagnosed with HCC BCLC stage C based on findings from abdominal ultrasound, abdominal CT scan, and abdominal MRI without contrast. However, a biopsy revealed a liver abscess. Our patient did not undergo CRP examination. Abdominal CT revealed hepatomegaly accompanied by multiple solid, lobulated exophytic masses in segments 2, 3, 4a, and 4b (the largest size was 9.8 x 12.7 x 9.2 cm), which showed early enhancement in the arterial phase and washout in the venous and delayed phases, tending towards hepatocellular carcinoma according to LI-RADS 5.
The Liver Imaging Reporting and Data System (LI-RADS), established by the American College of Radiology, standardizes terminology and criteria for interpreting and reporting liver CT and MR imaging findings in patients at risk for hepatocellular carcinoma (HCC). Observations are categorized from LR-1 (definitely benign) to LR-5 (definitely HCC). LI-RADS incorporates both primary and secondary criteria. Primary criteria include lesion size (10–19 mm or≥20 mm), contrast enhancement on multiphase imaging (arterial, portal venous, and/or delayed phases), the presence of a capsule, and growth over 6 months. Secondary criteria comprise additional features that may prompt an upgrade or downgrade of the observation. The relative weight of each feature remains undetermined [
9,
10]. Previous studies of LI-RADS versions 2014 and 2017 have demonstrated that LR-5 criteria on CT and/or MRI provide high specificity (85%–100%) for HCC, with moderate to fair sensitivity (50%–80%). For small HCCs (10–19 mm), the sensitivity of the LR-5 criteria in versions 2014 and 2017 was even lower (23%–67%), although high specificity was maintained (89%–98%). Chen et al. reported that, using LI-RADS v2018, LR-5 had a sensitivity of 70% and specificity of 95% for small HCCs (10–19 mm) across both readers. There were two false positive results for reader 1 (one histologically confirmed hepatocellular adenoma and one benign hepatocellular nodule based on imaging and follow-up) and two false positive results for reader 2 (two benign hepatocellular nodules based on imaging and follow-up) [
12]. Alhasan et al. reported a sensitivity of 53.7% and specificity of 97.3% for LI-RADS 5 in the diagnosis of HCC [13]. Although LR-5 indicates a 95% probability of HCC, there remains a 5% likelihood that the lesion is not HCC [
11].
The management of liver abscesses was initially limited to open surgical drainage. In recent years, percutaneous drainage has become increasingly utilized. For smaller abscesses, conservative management with antibacterial therapy alone has been adopted. Evidence indicates that antibiotic therapy alone is effective for abscesses measuring 3–5 cm or less. In one study, 100% success was observed in 8 patients with unilocular abscesses measuring less than 3 cm treated solely with antibiotics. A larger study involving 176 patients reported a success rate of 81.2% following antibiotic administration. Antibiotic therapy should be commenced promptly after obtaining blood samples for organism identification, beginning with intravenous antibiotics for approximately 3 weeks, followed by oral antibiotics for 1 to 2 months. The duration of therapy is determined by the clinical response, as assessed by repeat ultrasound imaging and the resolution of fever and leukocytosis. Recommended broad-spectrum antibiotics include a third-generation cephalosporin combined with metronidazole or piperacillin/tazobactam. It should be noted that certain common pathogens associated with liver abscesses demonstrate resistance to ampicillin and fluoroquinolones [
1].
Treatment is complicated by the increasing incidence of hyperresistant K. pneumoniae in some parts of the world. Percutaneous drainage is the most common option for first-line treatment of liver abscesses. Liver abscesses can be drained by needle aspiration or by inserting a pigtail catheter drain under ultrasound or CT scan guidance. Several studies have found percutaneous catheter drainage to be more effective than percutaneous needle aspiration, due to its higher success rate. Percutaneous drainage has many advantages, including minimally invasive procedures, avoiding the need for general anesthesia, a lower risk of adhesion formation and contamination, and a relatively lower cost compared to surgical drainage. Percutaneous drainage has been reported to fail in 15%–36% of cases due to multilocular liver abscesses or those containing thick fluid and necrotic tissue1. Surgery is indicated as the initial management for ruptured liver abscesses, peritonitis, difficult anatomical access, and pathologies requiring surgery. Initial surgical management may also be indicated for larger abscesses measuring >3–5 cm in diameter [
1].
Our patient underwent drainage of an abscess. The patient received oral metronidazole TID. A liver biopsy was scheduled. Ultrasound-guided FNAB of the liver revealed chronic, suppurative inflammation (abscess) with negative acid-fast bacilli on Ziehl-Neelsen staining. Pus cultures showed no bacterial growth.
It has been reported that 15.7% of patients develop complications from liver abscesses namely septic metastasis leading to extrahepatic complications, such as endophthalmitis, septic pulmonary embolism, lung, central nervous system, and eye infections. Abscess rupture is another reported complication, with spontaneous rupture occurring in 6.1% of cases. There is a reported higher incidence of liver abscess rupture in Klebsiella-infected abscesses than in those infected with other bacteria. Liver abscesses can also erode the diaphragm, causing pleural effusion, empyema, pneumonia, pericarditis, bronchopleural fistula, or duodenobronchofistula. Multiorgan failure can also occur as a result of liver abscesses [
1].
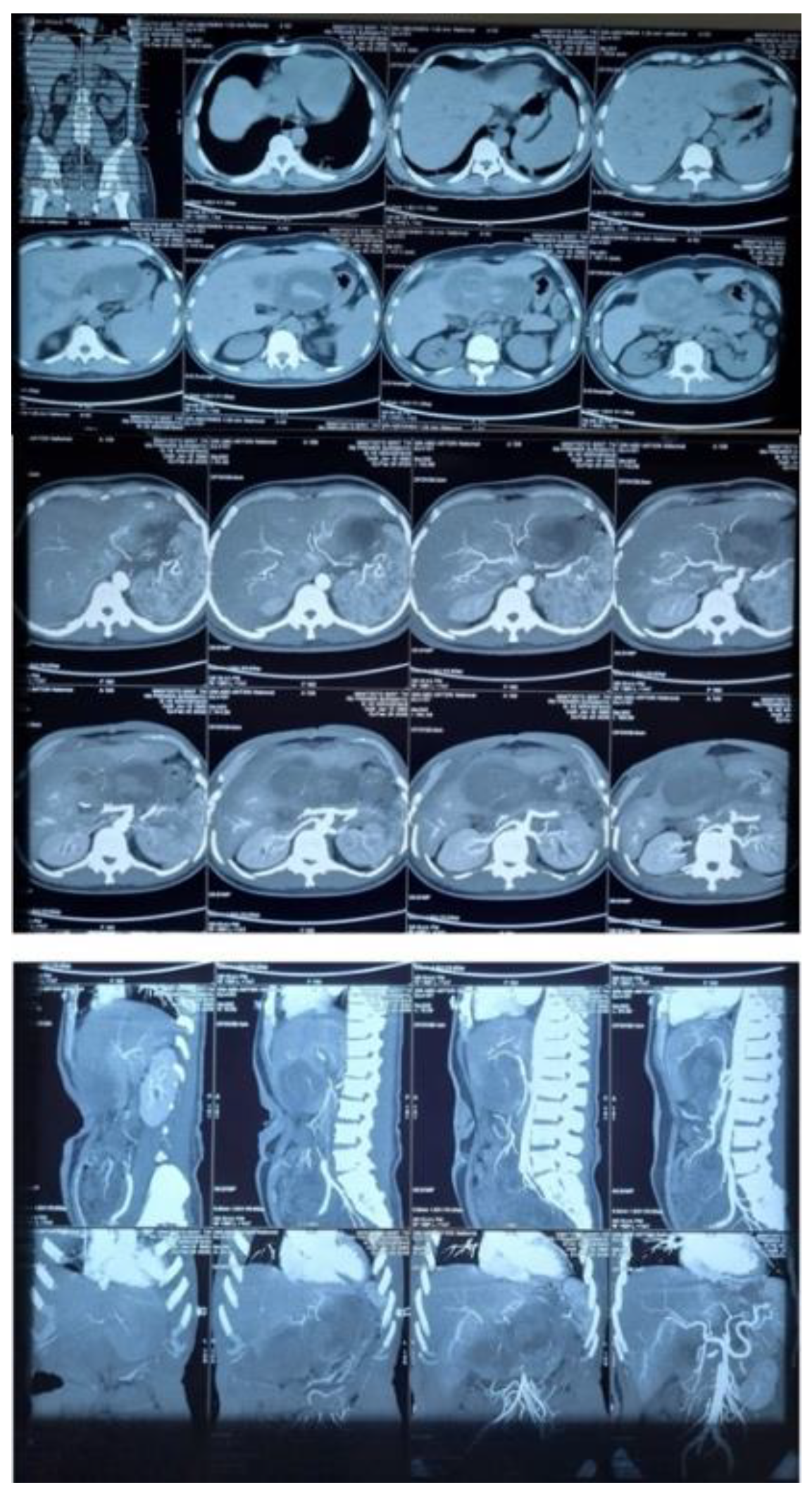
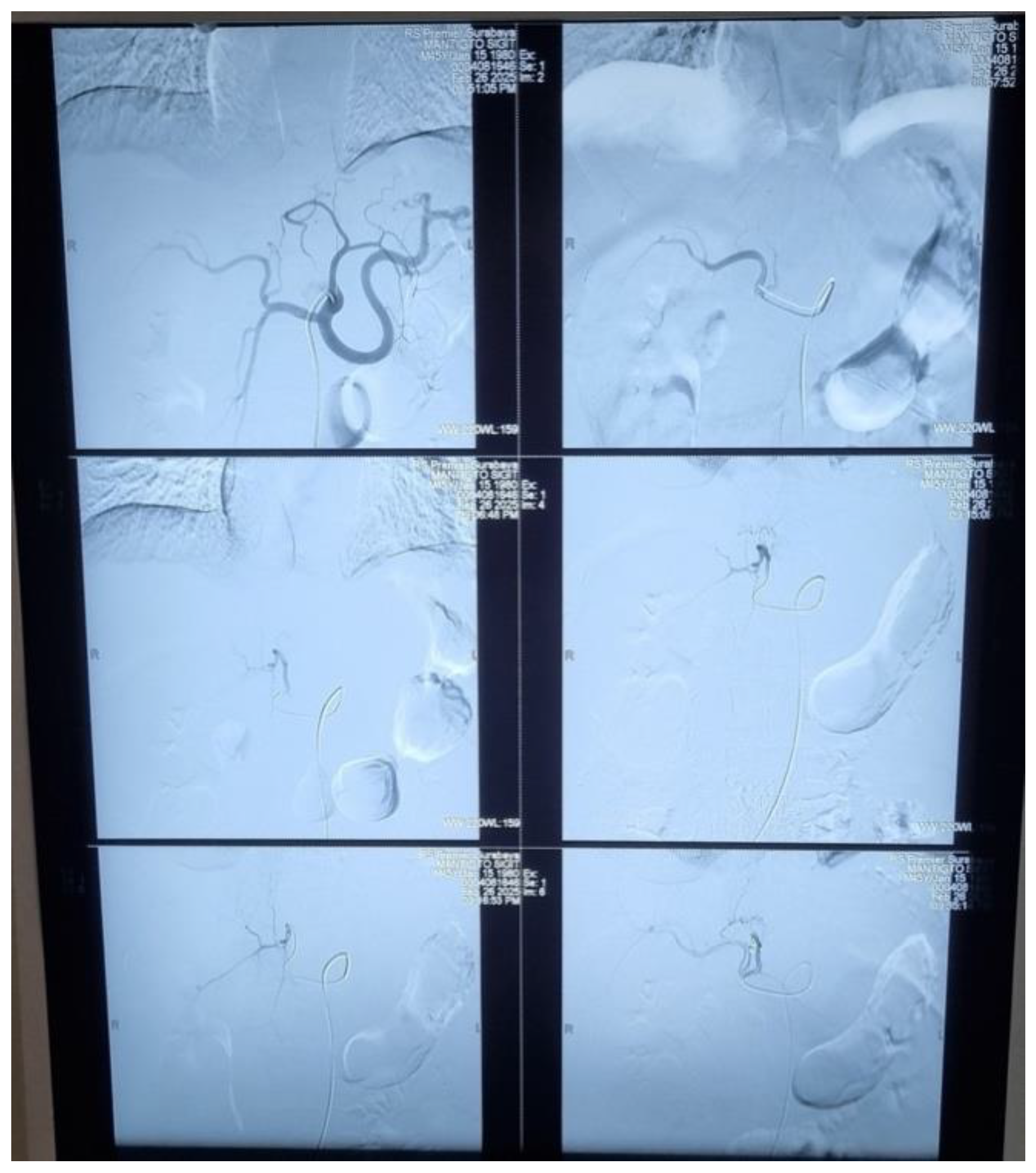
The patient developed a complication characterized by an infected hemorrhagic cyst with differential diagnosis of abscess and associated hematoma. MSCT of the upper abdomen, including non-contrast and triphasic dynamic intravenous contrast studies, revealed lobulated cystic lesions in liver segments I, II, and IV, containing hematoma, 11.9 x 6.7 x 8.8 cm in size. Prominent branches of the middle hepatic artery were observed entering the cystic lesion in segment IV during the arterial phase. Middle hepatic artery embolization was performed, and the patient was discharged two days later.
Although mortality from liver abscess has declined over time, it remains a consequential issue until now. Multiple associated factors and comorbidities contribute to the risk of death from liver abscess. Chen et al. conducted a study involving 134 patients with primary liver abscess, categorizing them into mortality and survivor groups. The study identified several risk factors for increased mortality, including male sex, malignancy, multi-organ failure, and liver abscess rupture. Notably, the mortality rate among patients with malignancy was twice as high as that among cancer-free patients. Additional signs and symptoms associated with the mortality group included respiratory distress, hypotension, jaundice, and extrahepatic involvement such as endophthalmitis [
1].