Submitted:
22 April 2026
Posted:
23 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
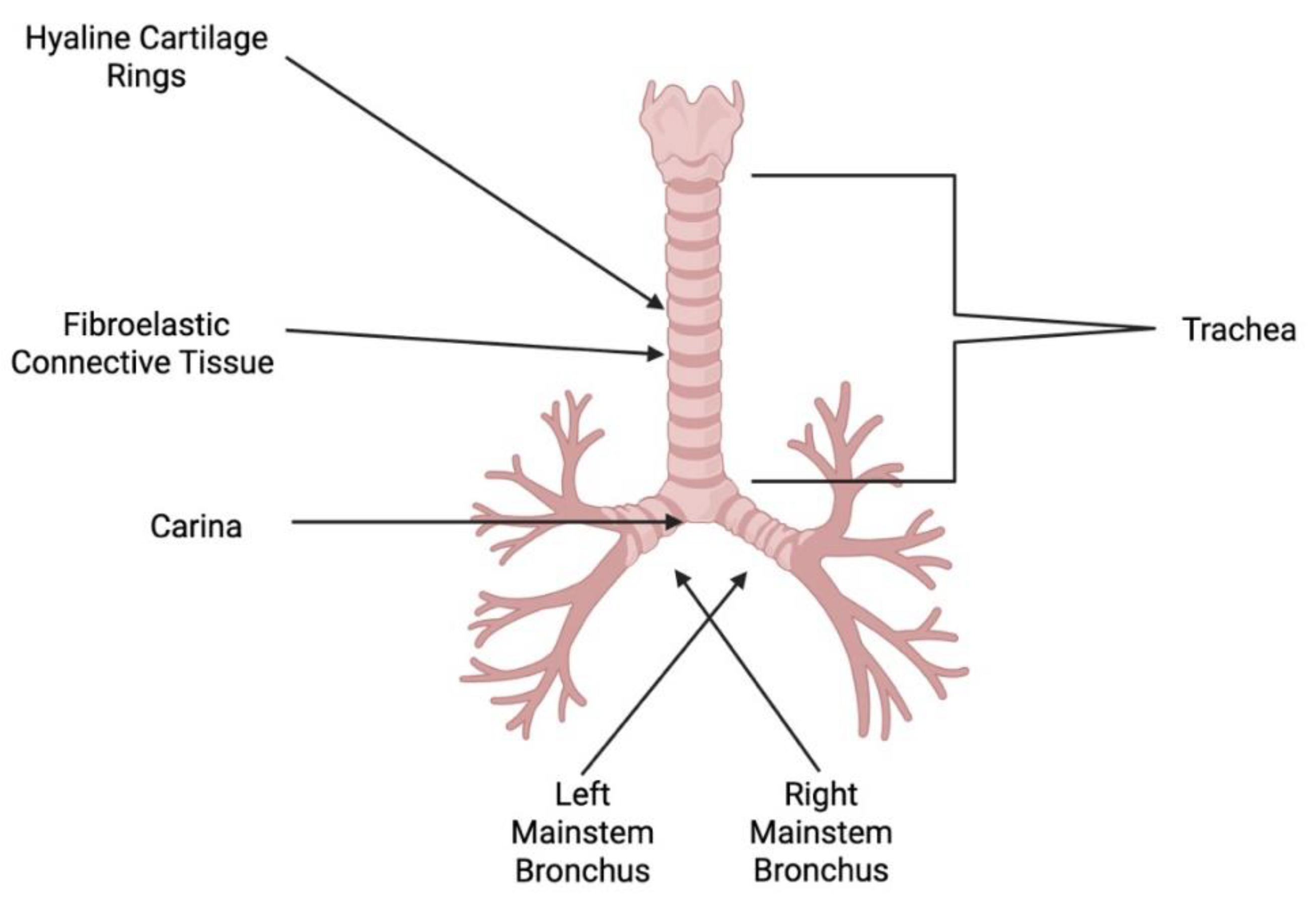
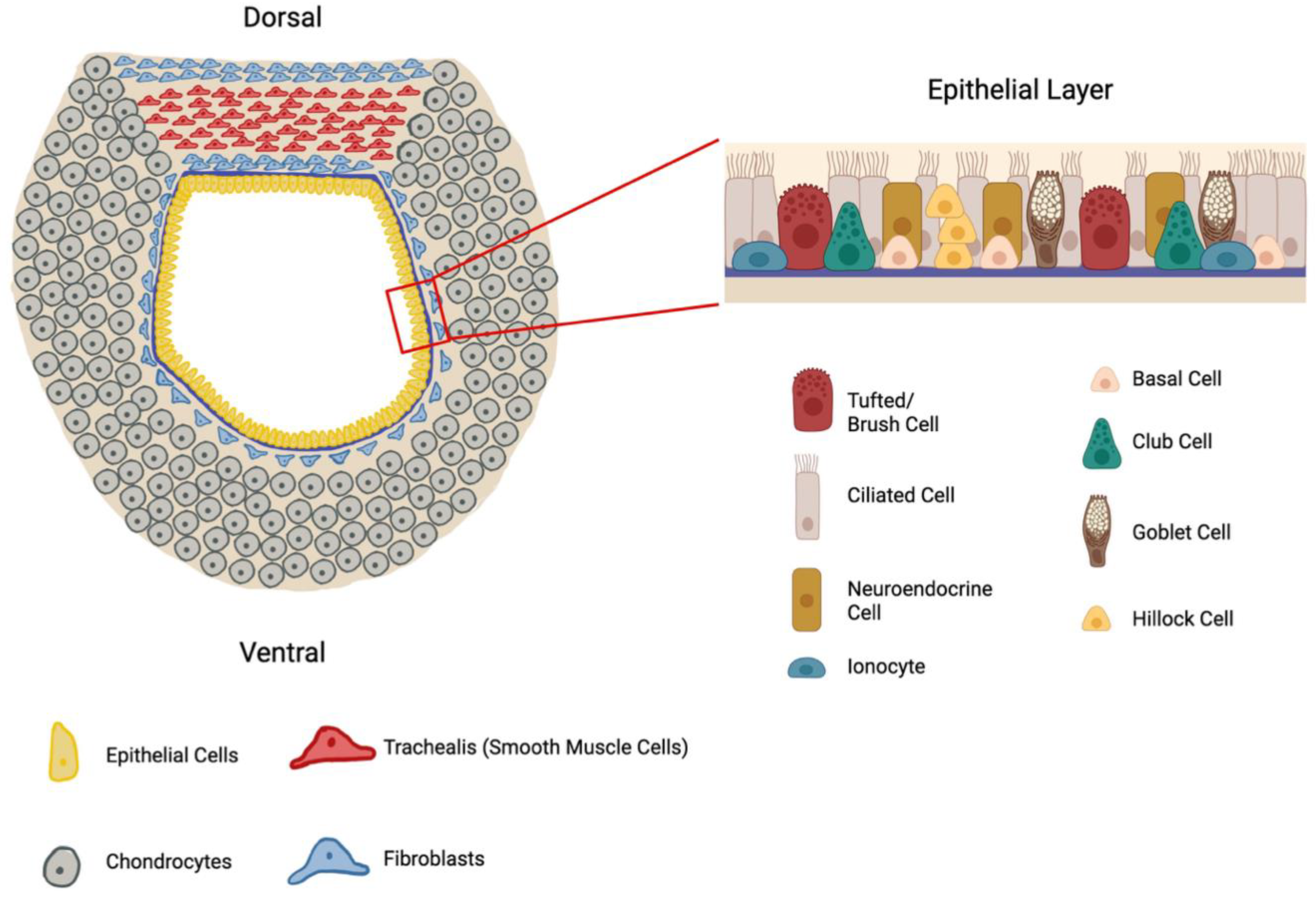
2. Tracheal Embryogenesis, Anatomy and Regeneration: A Brief Overview
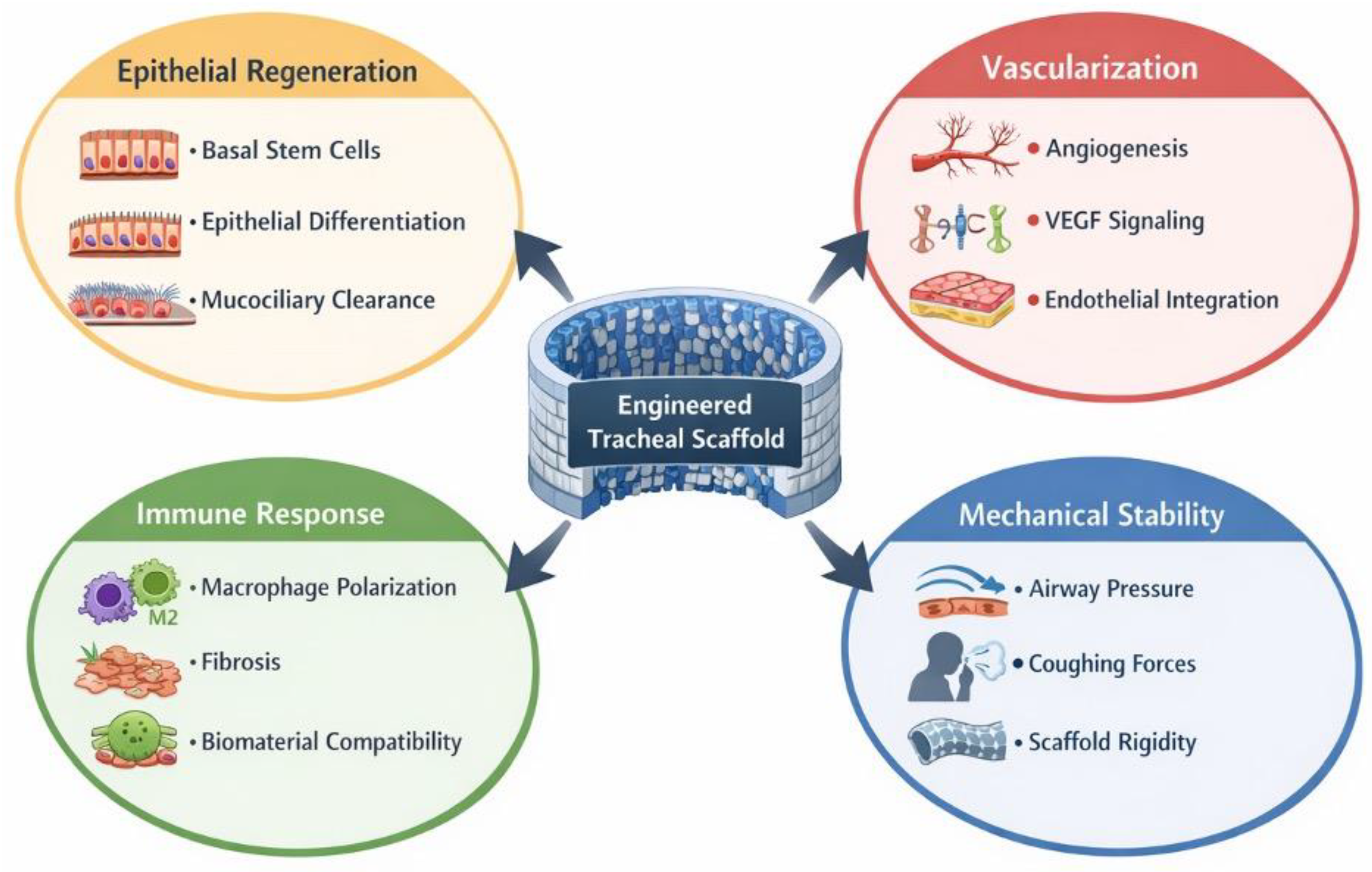
3. Main Barriers to Regeneration
4. Leveraging Innate Repair Mechanisms in the Trachea
5. Cell Types Used in Tracheal Tissue Engineering and Regeneration
6. Biomaterials Used in Designing Scaffolds for Tracheal Tissue Engineering and Regeneration
7. Specific Challenges with Engineering Tracheal Tissue
8. The Future of Tracheal Regeneration
9. Conclusion
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| TEF | Tracheoesophageal Fistula |
| VEGF | Vascular Endothelial Growth Factors |
| TGF-β | Transforming growth factor-beta |
| MSC | Mesenchymal Stem Cell |
| iPSCs | Induced pluripotent stem cells |
| PLA | Polylactic Acid |
| PCL | Polycaprolactone |
| PC | Polycarbonate |
| TPU | Thermoplastic polyurethane |
| PGA | Polyglycolic acid |
| PLGA | Poly(lactic-co-glycolic) acid |
| PPE | Polyphosphoesters |
| PET | Polyethylene terephthalate |
| HDPE | High-density polyethylene |
| bFGF | Basic fibroblast growth factor |
| ECM | Extracellular matrix |
| hECM | Human extracellular matrix |
| pECM | Porcine extracellular matrix |
| IL-4 | Interleukin-4 |
| IL-13 | Interleukin-13 |
| 3D | Three-dimension |
References
- Herrera, M.A.; Tintinago, L.F.; Morales, W. Victoria; Ordoñez, C.A.; Parra, M.W.; Betancourt-Cajiao, M.; Caicedo, Y.; Guzmán-Rodríguez, M.; Gallego, L.M.; González Hadad, A.; et al. Damage control of laryngotracheal trauma: the golden day. Colomb Med (Cali) 2020, 51(4), p. e4124599. [Google Scholar] [CrossRef]
- Grewal, H.S.; Dangayach, N.S.; Ahmad, U.; Ghosh, S.; Gildea, T.; Mehta, A.C. Treatment of Tracheobronchial Injuries: A Contemporary Review. Chest 2019, 155(3), 595–604. [Google Scholar] [CrossRef] [PubMed]
- Boutros, J.; Marquette, C.H.; Ichai, C.; Leroy, S.; Benzaquen, J. Multidisciplinary management of tracheobronchial injury. Eur Respir Rev 2022, 31(163). [Google Scholar] [CrossRef] [PubMed]
- De Jong, R.; Hohman, M.H.; Farahani, C. Laryngeal Clefts.; StatPearls [Internet], 2025. [Google Scholar]
- Asnaghi, A.; Macchiarini, P.; Mantero, S. Tissue engineering toward organ replacement: a promising approach in airway transplant. Int J Artif Organs 2009, 32(11), 763–8. [Google Scholar] [CrossRef] [PubMed]
- Auchincloss, H.G.; Wright, C.D. Complications after tracheal resection and reconstruction: prevention and treatment. J Thorac Dis 2016, 8 (Suppl 2), S160–7. [Google Scholar]
- Tapias, L.F.; Mathisen, D.J. Prevention and management of complications following tracheal resections-lessons learned at the Massachusetts General Hospital. Ann Cardiothorac Surg 2018, 7(2), 237–243. [Google Scholar] [CrossRef]
- Wei, S.; Zhang, Y.; Luo, F.; Duan, K.; Li, M.; Lv, G. Tissue-engineered tracheal implants: Advancements, challenges, and clinical considerations. Bioengineering & Translational Medicine 2024, 9(4), p. e10671. [Google Scholar]
- Sadreameli, S.C.; McGrath-Morrow, S.A. Respiratory Care of Infants and Children with Congenital Tracheo-Oesophageal Fistula and Oesophageal Atresia. Paediatr Respir Rev 2016, 17, 16–23. [Google Scholar] [CrossRef]
- Mieczkowski, B. S.B.F., Anatomy, Head and Neck, Trachea.; StatPearls [Internet], 2023. [Google Scholar]
- Edwards, N.A.; Shacham-Silverberg, V.; Weitz, L.; Kingma, P.S.; Shen, Y.; Wells, J.M.; Chung, W.K.; Zorn, A.M. Developmental basis of trachea-esophageal birth defects. Dev Biol 2021, 477, 85–97. [Google Scholar] [CrossRef]
- Faure, S.; de Santa Barbara, P. Molecular embryology of the foregut. J Pediatr Gastroenterol Nutr 2011, 52 Suppl 1(Suppl 1), S2–3. [Google Scholar] [CrossRef]
- Jacobsen, B.; VanKampen, N.; Ashurst, J.V. Anatomy, Head and Neck, Thyrohyoid Membrane, in StatPearls; StatPearls Publishing Copyright © 2025, StatPearls Publishing LLC.; Treasure Island (FL), 2025. [Google Scholar]
- Shamji, F.M. Factors Favoring and Impairing Healing of Tracheal Anastomosis. Thorac Surg Clin 2018, 28(2), 211–218. [Google Scholar] [CrossRef] [PubMed]
- Kishimoto, K.; Morimoto, M. Mammalian tracheal development and reconstruction: insights from in vivo and in vitro studies. Development 2021, 148(13). [Google Scholar] [CrossRef] [PubMed]
- Safshekan, F.; Tafazzoli-Shadpour, M.; Abdouss, M.; Behgam Shadmehr, M.; Ghorbani, F. Investigation of the Mechanical Properties of the Human Tracheal Cartilage. Tanaffos 2017, 16(2), 107–114. [Google Scholar] [PubMed]
- Di Cicco, M.; Kantar, A.; Masini, B.; Nuzzi, G.; Ragazzo, V.; Peroni, D. Structural and functional development in airways throughout childhood: Children are not small adults. Pediatr Pulmonol 2021, 56(1), 240–251. [Google Scholar] [CrossRef] [PubMed]
- Szpinda, M.; Daroszewski, M.; Woźniak, A.; Szpinda, A.; Mila-Kierzenkowska, C. Tracheal dimensions in human fetuses: an anatomical, digital and statistical study. Surg Radiol Anat 2012, 34(4), 317–23. [Google Scholar] [CrossRef]
- Luscan, R.; Leboulanger, N.; Fayoux, P.; Kerner, G.; Belhous, K.; Couloigner, V.; Garabedian, E.N.; Simon, F.; Denoyelle, F.; Thierry, B. Developmental changes of upper airway dimensions in children. Paediatr Anaesth 2020, 30(4), 435–445. [Google Scholar] [CrossRef]
- Liu, L.; Dharmadhikari, S.; Pouliot, R.A.; Li, M.M.; Minneci, P.M.; Tan, Z.; Shontz, K.; Johnson, J.; Reynolds, S.D.; Breuer, C.K.; et al. Modulation of Synthetic Tracheal Grafts with Extracellular Matrix Coatings. Bioengineering (Basel) 2021, 8(8). [Google Scholar] [CrossRef]
- Naik, S.S.; Dutta, N.K.; Sukumaran, N. Kiran; Choudhury, N.R. Critical advances in biofabrication and biomaterial strategies in tracheal tissue engineering: A comprehensive overview. Adv Colloid Interface Sci 2025, 345, 103604. [Google Scholar] [CrossRef]
- DeStefano, V.; Khan, S.; Tabada, A. Applications of PLA in modern medicine. Eng Regen 2020, 1, 76–87. [Google Scholar] [CrossRef]
- Imaizumi, M.; Nomoto, Y.; Sato, Y.; Sugino, T.; Miyake, M.; Wada, I.; Nakamura, T.; Omori, K. Evaluation of the use of induced pluripotent stem cells (iPSCs) for the regeneration of tracheal cartilage. Cell Transplant 2013, 22(2), 341–53. [Google Scholar] [CrossRef]
- Eenjes, E.; Tibboel, D.; Wijnen, R.M.H.; Rottier, R.J. Lung epithelium development and airway regeneration. Front Cell Dev Biol 2022, 10, 1022457. [Google Scholar] [CrossRef] [PubMed]
- Lin, B.; Shah, V.S.; Chernoff, C.; Sun, J.; Shipkovenska, G.G.; Vinarsky, V.; Waghray, A.; Xu, J.; Leduc, A.D.; Hintschich, C.A.; et al. Airway hillocks are injury-resistant reservoirs of unique plastic stem cells. Nature 2024, 629(8013), 869–877. [Google Scholar] [CrossRef] [PubMed]
- Rock, J.R.; Randell, S.H.; Hogan, B.L. Airway basal stem cells: a perspective on their roles in epithelial homeostasis and remodeling. Dis Model Mech 2010, 3(9-10), 545–56. [Google Scholar] [CrossRef] [PubMed]
- Butler, C.R.; Hynds, R.E.; Gowers, K.H.; Lee Ddo, H.; Brown, J.M.; Crowley, C.; Teixeira, V.H.; Smith, C.M.; Urbani, L.; Hamilton, N.J.; et al. Rapid Expansion of Human Epithelial Stem Cells Suitable for Airway Tissue Engineering. Am J Respir Crit Care Med 2016, 194(2), 156–68. [Google Scholar] [CrossRef]
- Bustamante-Marin, X.M.; Ostrowski, L.E. Cilia and Mucociliary Clearance. Cold Spring Harb Perspect Biol 2017, 9(4). [Google Scholar] [CrossRef]
- Rogers, D.F. The airway goblet cell. Int J Biochem Cell Biol 2003, 35(1), 1–6. [Google Scholar] [CrossRef]
- Ruysseveldt, E.; Martens, K.; Steelant, B. Airway Basal Cells, Protectors of Epithelial Walls in Health and Respiratory Diseases. Front Allergy 2021. 2, 787128. [Google Scholar] [CrossRef]
- Davis, J.D.; Wypych, T.P. Cellular and functional heterogeneity of the airway epithelium. Mucosal Immunol 2021, 14(5), 978–990. [Google Scholar] [CrossRef]
- Thakur, A.; Mei, S.; Zhang, N.; Zhang, K.; Taslakjian, B.; Lian, J.; Wu, S.; Chen, B.; Solway, J.; Chen, H.J. Pulmonary neuroendocrine cells: crucial players in respiratory function and airway-nerve communication. Front Neurosci 2024, 18, 1438188. [Google Scholar] [CrossRef]
- Hollenhorst, M.I.; Husnik, T.; Zylka, M.; Duda, N.; Flockerzi, V.; Tschernig, T.; Maxeiner, S.; Krasteva-Christ, G. Human airway tuft cells influence the mucociliary clearance through cholinergic signalling. Respir Res 2023, 24(1), p. 267. [Google Scholar] [CrossRef]
- Chen, Y.; Tang, H.; Zhang, Y.; Wang, L.; Zhu, J.; Wang, L.; Li, A.; Zeng, X.; Yin, B.; Liang, Y. Multiplexed self-adaptable Janus hydrogels rescue epithelial malfunction to promote complete trachea repair. Nature Communications 2025, 16(1), p. 5734. [Google Scholar] [CrossRef]
- Rogers, D.F. Airway goblet cells: responsive and adaptable front-line defenders. Eur Respir J 1994, 7(9), 1690–706. [Google Scholar] [CrossRef] [PubMed]
- Margiana, R.; Markov, A.; Zekiy, A.O.; Hamza, M.U.; Al-Dabbagh, K.A.; Al-Zubaidi, S.H.; Hameed, N.M.; Ahmad, I.; Sivaraman, R.; Kzar, H.H.; et al. Clinical application of mesenchymal stem cell in regenerative medicine: a narrative review. Stem Cell Res Ther 2022, 13(1), p. 366. [Google Scholar] [CrossRef] [PubMed]
- Hernández, R.; Jiménez-Luna, C.; Perales-Adán, J.; Perazzoli, G.; Melguizo, C.; Prados, J. Differentiation of Human Mesenchymal Stem Cells towards Neuronal Lineage: Clinical Trials in Nervous System Disorders. Biomol Ther (Seoul) 2020, 28(1), 34–44. [Google Scholar] [CrossRef] [PubMed]
- Shin, Y.S.; Choi, J.W.; Park, J.K.; Kim, Y.S.; Yang, S.S.; Min, B.H.; Kim, C.H. Tissue-engineered tracheal reconstruction using mesenchymal stem cells seeded on a porcine cartilage powder scaffold. Ann Biomed Eng 2015, 43(4), 1003–13. [Google Scholar] [CrossRef]
- Xie, X.; Miao, B.; Yao, J.; Chen, Z. Silk fibroin-hydroxyapatite scaffolds promote the proliferation of adipose-derived mesenchymal stem cells by activating the ERK signal. J Biomater Appl 2023, 37(10), 1767–1775. [Google Scholar] [CrossRef]
- Aboul-Soud, M.A.M.; Alzahrani, A.J.; Mahmoud, A. Induced Pluripotent Stem Cells (iPSCs)-Roles in Regenerative Therapies, Disease Modelling and Drug Screening. Cells 2021, 10(9). [Google Scholar] [CrossRef]
- Ikeda, M.; Imaizumi, M.; Yoshie, S.; Otsuki, K.; Miyake, M.; Hazama, A.; Wada, I.; Omori, K. Regeneration of tracheal epithelium using mouse induced pluripotent stem cells. Acta Otolaryngol 2016, 136(4), 373–8. [Google Scholar] [CrossRef]
- Nomoto, M.; Nomoto, Y.; Tada, Y.; Tani, A.; Otsuki, K.; Suzuki, R.; Nakamura, T.; Omori, K. Bioengineered trachea using autologous chondrocytes for regeneration of tracheal cartilage in a rabbit model. Laryngoscope 2013, 123(9), 2195–201. [Google Scholar] [CrossRef]
- Khalid, T.; Soriano, L.; Lemoine, M.; Cryan, S.A.; O'Brien, F.J.; O'Leary, C. Development of tissue-engineered tracheal scaffold with refined mechanical properties and vascularisation for tracheal regeneration. Front Bioeng Biotechnol 2023, 11, 1187500. [Google Scholar] [CrossRef]
- Wei, S.; Zhang, Y.; Luo, F.; Duan, K.; Li, M.; Lv, G. Tissue-engineered tracheal implants: Advancements, challenges, and clinical considerations. Bioeng Transl Med 2024, 9(4), p. e10671. [Google Scholar] [CrossRef]
- Gomez-Florit, M.; Pardo, A.; Domingues, R.M.A.; Graça, A.L.; Babo, P.S.; Reis, R.L.; Gomes, M.E. Natural-Based Hydrogels for Tissue Engineering Applications. Molecules 2020, 25(24), 5858. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Wang, Z.; Hua, Y.; Zhu, X.; Wang, Y.; Duan, L.; Zhu, L.; Jiang, G.; Xia, H.; She, Y. Photocrosslinked natural hydrogel composed of hyaluronic acid and gelatin enhances cartilage regeneration of decellularized trachea matrix. Mater Sci Eng C Mater Biol Appl 2021, 120, 111628. [Google Scholar] [CrossRef] [PubMed]
- Nematollahi, Z.; Tafazzoli-Shadpour, M.; Zamanian, A.; Seyedsalehi, A.; Mohammad-Behgam, S.; Ghorbani, F.; Mirahmadi, F. Fabrication of Chitosan Silk-based Tracheal Scaffold Using Freeze-Casting Method. Iran Biomed J 2017, 21(4), 228–39. [Google Scholar] [CrossRef] [PubMed]
- Dai, Y.; Liu, G.; Ma, L.; Wang, D.; Gao, C. Cell-free macro-porous fibrin scaffolds for in situ inductive regeneration of full-thickness cartilage defects. Journal of Materials Chemistry B 2016, 4(25), 4410–4419. [Google Scholar] [CrossRef]
- Luo, Y.; Lode, A.; Gelinsky, M. Direct Plotting of Three-Dimensional Hollow Fiber Scaffolds Based on Concentrated Alginate Pastes for Tissue Engineering. Advanced Healthcare Materials 2013, 2(6), 777–783. [Google Scholar] [CrossRef]
- Cao, Y.; Cheng, P.; Sang, S.; Xiang, C.; An, Y.; Wei, X.; Shen, Z.; Zhang, Y.; Li, P. Mesenchymal stem cells loaded on 3D-printed gradient poly(ε-caprolactone)/methacrylated alginate composite scaffolds for cartilage tissue engineering. Regenerative Biomaterials 2021, 8(3). [Google Scholar] [CrossRef]
- Fares, M.M.; Shirzaei Sani, E.; Portillo Lara, R.; Oliveira, R.B.; Khademhosseini, A.; Annabi, N. Interpenetrating network gelatin methacryloyl (GelMA) and pectin-g-PCL hydrogels with tunable properties for tissue engineering. Biomaterials Science 2018, 6(11), 2938–2950. [Google Scholar] [CrossRef]
- Novosel, E.C.; Kleinhans, C.; Kluger, P.J. Vascularization is the key challenge in tissue engineering. Adv Drug Deliv Rev 2011, 63(4-5), 300–11. [Google Scholar] [CrossRef]
- Gandha, P.; Surve, T.; Kandasubramanian, B. Mozafari, M., Chauhan, N.P. Singh, Eds.; 15 - Polycaprolactone as biomaterial. In Handbook of Polymers in Medicine; Woodhead Publishing, 2023; pp. 425–443. [Google Scholar]
- Artham, T.; Doble, M. Biodegradation of aliphatic and aromatic polycarbonates. Macromol Biosci 2008, 8(1), 14–24. [Google Scholar] [CrossRef]
- Tatekawa, Y.; Kawazoe, N.; Chen, G.; Shirasaki, Y.; Komuro, H.; Kaneko, M. Tracheal defect repair using a PLGA-collagen hybrid scaffold reinforced by a copolymer stent with bFGF-impregnated gelatin hydrogel. Pediatr Surg Int 2010, 26(6), 575–80. [Google Scholar] [CrossRef]
- Reyes, C.D.; Petrie, T.A.; García, A.J. Mixed extracellular matrix ligands synergistically modulate integrin adhesion and signaling. J Cell Physiol 2008, 217(2), 450–8. [Google Scholar] [CrossRef] [PubMed]
- Pouliot, R.A.; Link, P.A.; Mikhaiel, N.S.; Schneck, M.B.; Valentine, M.S.; Kamga Gninzeko, F.J.; Herbert, J.A.; Sakagami, M.; Heise, R.L. Development and characterization of a naturally derived lung extracellular matrix hydrogel. J Biomed Mater Res A 2016, 104(8), 1922–35. [Google Scholar] [CrossRef] [PubMed]
- Villegas-Alvarez, F.; González-Zamora, J.F.; González-Maciel, A.; Soriano-Rosales, R.; Pérez-Guille, B.; Padilla-Sánchez, L.; Reynoso-Robles, R.; Ramos-Morales, A.; Zenteno-Galindo, E.; Pérez-Torres, A.; et al. Fibrocollagen-covered prosthesis for a noncircumferential segmental tracheal replacement. J Thorac Cardiovasc Surg 2010, 139(1), 32–7. [Google Scholar] [CrossRef] [PubMed]
- Young, B.M.; Shankar, K.; Allen, B.P.; Pouliot, R.A.; Schneck, M.B.; Mikhaiel, N.S.; Heise, R.L. Electrospun Decellularized Lung Matrix Scaffold for Airway Smooth Muscle Culture. ACS Biomater Sci Eng 2017, 3(12), 3480–3492. [Google Scholar] [CrossRef]
- Anderson, J.M.; Rodriguez, A.; Chang, D.T. Foreign body reaction to biomaterials. Semin Immunol 2008, 20(2), 86–100. [Google Scholar] [CrossRef]
- Liang, Y.; Wei, S.; Zhang, A. Bioengineered tracheal graft with enhanced vascularization and mechanical stability for functional airway reconstruction. Regen Ther 2025, 29, 364–380. [Google Scholar] [CrossRef]
- Majumder, N.; Roy, C.; Doenges, L.; Martin, I.; Barbero, A.; Ghosh, S. Covalent Conjugation of Small Molecule Inhibitors and Growth Factors to a Silk Fibroin-Derived Bioink to Develop Phenotypically Stable 3D Bioprinted Cartilage. ACS Appl Mater Interfaces 2024, 16(8), 9925–9943. [Google Scholar] [CrossRef]
| Challenge | Biological / Engineering Issue | Current Strategies |
| Vascularization | Poor blood supply leads to scaffold ischemia and graft failure | Growth factor delivery (VEGF), pre-vascularized scaffolds, in vivo bioreactors 20 |
| Mechanical Stability | Airway must withstand airflow pressure, coughing, and neck motion | Synthetic polymers, reinforced scaffolds 21 |
| Immune Response | Foreign body reactions and fibrosis can cause scaffold rejection | Immunomodulatory biomaterials, cytokine delivery 22 |
| Epithelial Regeneration | Functional airway epithelium required for mucociliary clearance | Stem cell seeding, epithelial progenitor cells 23 |
| Tracheal Growth | Airway growth requires adaptable scaffolds | Biodegradable and growth-permitting biomaterials 22 |
| Cell Name | Cell Function |
| Ciliated (Brush) | Mucociliary clearance – the cilia (hair-like projections) that beat in a wave-like movement to move mucus and debris out of the airway 28 |
| Goblet | Production of mucus that traps debris such as dust and pathogens from further entering the respiratory tract 29 |
| Basal | Multipotent stem cell – responsible for regenerating and repairing the epithelium by differentiating into ciliated and goblet cells 30 |
| Club (Clara) | Secretes proteins that reduce inflammation (uteroglobin) and regulate immune properties while having the capability to de-differentiate when basal cells are injured 31 |
| Pulmonary Neuroendocrine (PNECS) | Sensory and paracrine signaling that regulate airway tone, mucociliary clearance, and hyperresponsiveness to pathogens and airway irritants 32 |
| Tuft | Regulates the mucociliary clearance in response to chemical irritants that enter the respiratory tract 33 |
| Cell Type | Advantages | Limitations | Representative Study |
| Mesenchymal Stem Cells (MSCs) | Immunomodulatory, anti-inflammatory, multipotent, easy to isolate | Limited differentiation toward airway epithelium in some models | Shin et al. 38 |
| Induced Pluripotent Stem Cells (iPSCs) | High differentiation potential, can generate airway epithelial cells | Tumorigenicity concerns and complex differentiation protocols | Ikeda et al. 41 |
| Autologous Chondrocytes | Promote cartilage regeneration and structural support | Limited proliferation capacity and donor tissue requirements | Nomoto et al. 42 |
| Airway Basal Stem Cells | Native airway progenitors capable of regenerating epithelium | Difficult isolation and expansion | Lin et al. 25 |
| Endothelial Cells | Promote vascularization of scaffolds | Require supportive microenvironment for stability | Khalid et al. 43 |
| Material | Type | Strengths | Limitations | Representative Study |
| Alginate | Natural | Biocompatible, supports cell adhesion and hydrogel formation | Weak mechanical strength, rapid degradation | Luo et al. 49 |
| Chitosan | Natural | Antimicrobial properties, promotes cell attachment | Limited mechanical stability | Nematollahi et al. 47 |
| Collagen | Natural | Mimics extracellular matrix, excellent cell compatibility | Rapid degradation and poor structural strength | Xu et al. 46 |
| Fibrin | Natural | Supports cell infiltration and angiogenesis | Weak mechanical properties, fast degradation | Dai et al. 48 |
| Gelatin | Natural | Promotes cell adhesion and proliferation | Low mechanical stability | Fares et al. 51 |
| Hyaluronic Acid | Natural | Supports cartilage regeneration and ECM signaling | Limited structural strength | Xu et al. 46 |
| Soy Protein | Natural | Biodegradable and supportive for cell growth | Limited studies in airway applications | Naik et al. 21 |
| HDPE | Synthetic | High mechanical strength and durability | Poor biodegradability and limited bioactivity | Naik et al. 21 |
| PLA | Synthetic | Biodegradable, tunable degradation rate | Can produce acidic degradation byproducts | DeStefano et al. 22 |
| PCL | Synthetic | Excellent mechanical strength, slow degradation | Hydrophobic surface limits cell attachment | Gandha et al. 53 |
| PC | Synthetic | Strong structural support | Limited biodegradability | Artham et al. 54 |
| PET | Synthetic | High durability and stability | Limited biocompatibility in regenerative applications | Naik et al. 21 |
| PGA | Synthetic | Biodegradable with good mechanical strength | Rapid degradation may compromise structural support | Naik et al. 21 |
| PLGA | Synthetic | Tunable degradation and widely used in tissue engineering | Degradation may produce acidic environment | Tatekawa et al. 55 |
| PPE | Synthetic | Tunable degradation and polymer properties | Limited long-term airway studies | Naik et al. 21 |
| TPU | Synthetic | Elastic and mechanically durable | Poor bioactivity for cell attachment | Naik et al. 21 |
| Abbreviations: HDPE - high-density polyethylene; PLA - polylactic acid; PCL – polycaprolactone; PC – polycarbonate; PET - polyethylene terephthalate; PGA - polyglycolic acid; PLGA - poly(lactic-co-glycolic) acid; PPE – polyphosphoesters; TPU - thermoplastic polyurethane | ||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




