Submitted:
22 April 2026
Posted:
23 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
| Class | Concentration (kU/L) | Interpretation |
|---|---|---|
| 0 | 0 ≤ sIgE < 0.35 | Negative. No antibodies detected. |
| 1 | 0.35 ≤ sIgE < 0.7 | Very low positive. Very low antibody concentration; clinical symptoms are usually absent upon sensitization. |
| 2 | 0.7 ≤ sIgE < 3.5 | Low positive. Low antibody concentration; sensitization is present, and clinical symptoms frequently occur at the upper limit of this class. |
| 3 | 3.5 ≤ sIgE < 17.5 | Positive. Significant antibody concentration; clinical symptoms frequently occur. |
| 4 | 17.5 ≤ sIgE < 50 | Strong positive. High antibody concentration; clinical symptoms consistently occur. |
| 5 | 50 ≤ sIgE < 100 | Very strong positive. |
| 6 | sIgE ≥ 100 | Extremely strong positive. |
3. Results
3.1. Baseline Characteristics of the Study Population
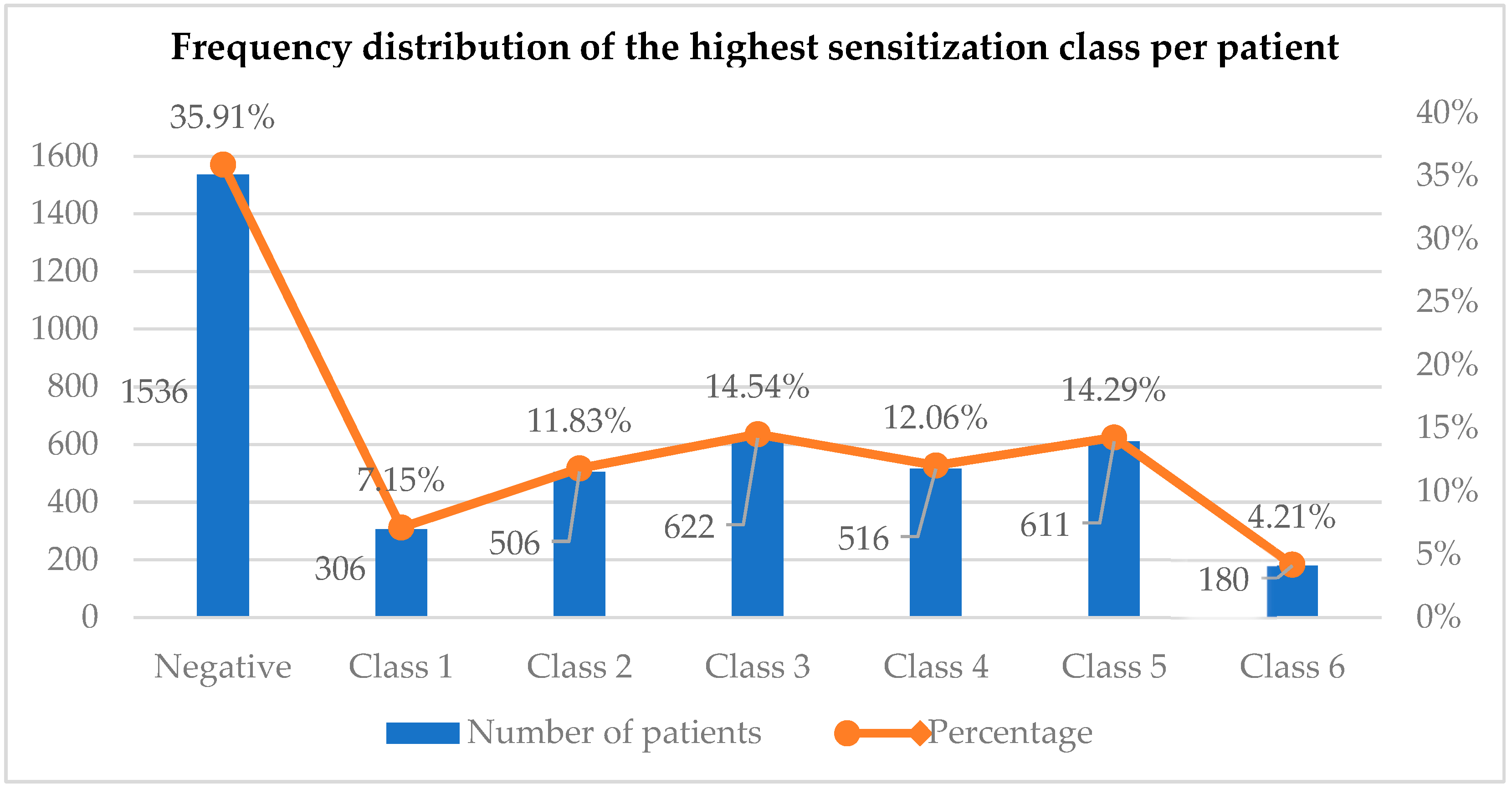
3.2. Characteristics of Allergen-Specific IgE Sensitization
| Allergen | Positive | Class 1 | Class 2 | Class 3 | Class 4 | Class 5 | Class 6 |
| Egg white | 301 (7.0%) | 75 (1.8%) | 104 (2.4%) | 60 (1.4%) | 40 (0.9%) | 22 (0.5%) | 0 (0.0%) |
| Egg yolk | 55 (1.3%) | 18 (0.4%) | 21 (0.5%) | 6 (0.1%) | 7 (0.2%) | 3 (0.1%) | 0 (0.0%) |
| Codfish | 19 (0.4%) | 8 (0.2%) | 2 (0.0%) | 6 (0.1%) | 1 (0.0%) | 2 (0.0%) | 0 (0.0%) |
| Shrimp | 161 (3.8%) | 15 (0.4%) | 36 (0.8%) | 32 (0.7%) | 39 (0.9%) | 38 (0.9%) | 1 (0.0%) |
| Tuna | 6 (0.1%) | 2 (0.0%) | 2 (0.0%) | 2 (0.0%) | 0 (0.0%) | 0 (0.0%) | 0 (0.0%) |
| Sardine | 96 (2.2%) | 58 (1.4%) | 30 (0.7%) | 7 (0.2%) | 1 (0.0%) | 0 (0.0%) | 0 (0.0%) |
| Wheat | 211 (4.9%) | 189 (4.4%) | 14 (0.3%) | 5 (0.1%) | 3 (0.1%) | 0 (0.0%) | 0 (0.0%) |
| Rye | 303 (7.1%) | 97 (2.3%) | 178 (4.2%) | 25 (0.6%) | 3 (0.1%) | 0 (0.0%) | 0 (0.0%) |
| Barley | 412 (9.6%) | 91 (2.1%) | 141 (3.3%) | 174 (4.1%) | 5 (0.1%) | 1 (0.0%) | 0 (0.0%) |
| Oat | 61 (1.4%) | 32 (0.7%) | 18 (0.4%) | 6 (0.1%) | 4 (0.1%) | 1 (0.0%) | 0 (0.0%) |
| Rice | 318 (7.4%) | 251 (5.9%) | 55 (1.3%) | 7 (0.2%) | 3 (0.1%) | 2 (0.0%) | 0 (0.0%) |
| Soybean | 77 (1.8%) | 51 (1.2%) | 15 (0.4%) | 8 (0.2%) | 1 (0.0%) | 2 (0.0%) | 0 (0.0%) |
| Maize (corn) | 277 (6.5%) | 137 (3.2%) | 119 (2.8%) | 16 (0.4%) | 4 (0.1%) | 1 (0.0%) | 0 (0.0%) |
| Gluten | 59 (1.4%) | 26 (0.6%) | 10 (0.2%) | 5 (0.1%) | 9 (0.2%) | 8 (0.2%) | 1 (0.0%) |
| Peanut | 204 (4.8%) | 70 (1.6%) | 99 (2.3%) | 15 (0.4%) | 14 (0.3%) | 6 (0.1%) | 0 (0.0%) |
| Hazelnut | 63 (1.5%) | 44 (1.0%) | 12 (0.3%) | 4 (0.1%) | 2 (0.0%) | 1 (0.0%) | 0 (0.0%) |
| Almond | 439 (10.3%) | 110 (2.6%) | 140 (3.3%) | 175 (4.1%) | 9 (0.2%) | 5 (0.1%) | 0 (0.0%) |
| Cow’s milk | 199 (4.7%) | 75 (1.8%) | 75 (1.8%) | 34 (0.8%) | 14 (0.3%) | 1 (0.0%) | 0 (0.0%) |
| α-lactalbumin | 182 (4.3%) | 92 (2.2%) | 69 (1.6%) | 17 (0.4%) | 4 (0.1%) | 0 (0.0%) | 0 (0.0%) |
| β-lactoglobulin | 258 (6.0%) | 98 (2.3%) | 104 (2.4%) | 44 (1.0%) | 10 (0.2%) | 2 (0.0%) | 0 (0.0%) |
| Casein | 142 (3.3%) | 58 (1.4%) | 60 (1.4%) | 15 (0.4%) | 8 (0.2%) | 1 (0.0%) | 0 (0.0%) |
| Chocolate | 123 (2.9%) | 63 (1.5%) | 53 (1.2%) | 7 (0.2%) | 0 (0.0%) | 0 (0.0%) | 0 (0.0%) |
| Goat milk | 50 (1.2%) | 22 (0.5%) | 17 (0.4%) | 8 (0.2%) | 2 (0.0%) | 1 (0.0%) | 0 (0.0%) |
| Tomato | 317 (7.4%) | 66 (1.5%) | 217 (5.1%) | 31 (0.7%) | 1 (0.0%) | 2 (0.0%) | 0 (0.0%) |
| Lemon | 182 (4.3%) | 109 (2.5%) | 59 (1.4%) | 9 (0.2%) | 5 (0.1%) | 0 (0.0%) | 0 (0.0%) |
| Orange | 263 (6.1%) | 171 (4.0%) | 76 (1.8%) | 4 (0.1%) | 9 (0.2%) | 3 (0.1%) | 0 (0.0%) |
| Strawberry | 302 (7.1%) | 104 (2.4%) | 183 (4.3%) | 13 (0.3%) | 1 (0.0%) | 1 (0.0%) | 0 (0.0%) |
| Apple | 204 (4.8%) | 157 (3.7%) | 24 (0.6%) | 5 (0.1%) | 8 (0.2%) | 10 (0.2%) | 0 (0.0%) |
| Pineapple | 229 (5.4%) | 111 (2.6%) | 85 (2.0%) | 15 (0.4%) | 12 (0.3%) | 6 (0.1%) | 0 (0.0%) |
| Pork (cooked) | 39 (0.9%) | 14 (0.3%) | 8 (0.2%) | 9 (0.2%) | 6 (0.1%) | 2 (0.0%) | 0 (0.0%) |
| Beef | 88 (2.1%) | 34 (0.8%) | 34 (0.8%) | 10 (0.2%) | 6 (0.1%) | 4 (0.1%) | 0 (0.0%) |
| Chicken | 27 (0.6%) | 14 (0.3%) | 7 (0.2%) | 4 (0.1%) | 1 (0.0%) | 1 (0.0%) | 0 (0.0%) |
| Brewer’s yeast | 101 (2.4%) | 27 (0.6%) | 41 (1.0%) | 25 (0.6%) | 8 (0.2%) | 0 (0.0%) | 0 (0.0%) |
| Baker’s yeast | 52 (1.2%) | 16 (0.4%) | 25 (0.6%) | 6 (0.1%) | 5 (0.1%) | 0 (0.0%) | 0 (0.0%) |
| Latex | 464 (10.8%) | 80 (1.9%) | 289 (6.8%) | 71 (1.7%) | 16 (0.4%) | 8 (0.2%) | 0 (0.0%) |
| D. pteronyssinus | 1098 (25.7%) | 140 (3.3%) | 155 (3.6%) | 184 (4.3%) | 170 (4.0%) | 367 (8.6%) | 82 (1.9%) |
| D. farinae | 1322 (30.9%) | 168 (3.9%) | 228 (5.3%) | 214 (5.0%) | 164 (3.8%) | 400 (9.4%) | 148 (3.5%) |
| Blomia tropicalis | 863 (20.2%) | 185 (4.3%) | 204 (4.8%) | 194 (4.5%) | 167 (3.9%) | 105 (2.5%) | 8 (0.2%) |
| Cat dander | 192 (4.5%) | 15 (0.4%) | 25 (0.6%) | 30 (0.7%) | 49 (1.1%) | 62 (1.4%) | 11 (0.3%) |
| Dog dander | 82 (1.9%) | 7 (0.2%) | 8 (0.2%) | 13 (0.3%) | 11 (0.3%) | 31 (0.7%) | 12 (0.3%) |
| Chicken feathers | 19 (0.4%) | 7 (0.2%) | 9 (0.2%) | 2 (0.0%) | 1 (0.0%) | 0 (0.0%) | 0 (0.0%) |
| Penicillium notatum | 82 (1.9%) | 39 (0.9%) | 28 (0.7%) | 13 (0.3%) | 2 (0.0%) | 0 (0.0%) | 0 (0.0%) |
| Cladosporium herbarum | 24 (0.6%) | 14 (0.3%) | 5 (0.1%) | 2 (0.0%) | 2 (0.0%) | 0 (0.0%) | 1 (0.0%) |
| Aspergillus fumigatus | 40 (0.9%) | 13 (0.3%) | 15 (0.4%) | 5 (0.1%) | 4 (0.1%) | 2 (0.0%) | 1 (0.0%) |
| Candida albicans | 192 (4.5%) | 72 (1.7%) | 62 (1.4%) | 32 (0.7%) | 19 (0.4%) | 7 (0.2%) | 0 (0.0%) |
| Alternaria alternata | 59 (1.4%) | 24 (0.6%) | 24 (0.6%) | 10 (0.2%) | 1 (0.0%) | 0 (0.0%) | 0 (0.0%) |
| Grass pollen | 632 (14.8%) | 95 (2.2%) | 131 (3.1%) | 309 (7.2%) | 72 (1.7%) | 24 (0.6%) | 1 (0.0%) |
| Dandelion pollen | 312 (7.3%) | 130 (3.0%) | 156 (3.6%) | 21 (0.5%) | 2 (0.0%) | 3 (0.1%) | 0 (0.0%) |
| Honey bee venom | 754 (17.6%) | 122 (2.9%) | 240 (5.6%) | 267 (6.2%) | 90 (2.1%) | 35 (0.8%) | 0 (0.0%) |
| Wasp venom | 649 (15.2%) | 199 (4.7%) | 194 (4.5%) | 138 (3.2%) | 90 (2.1%) | 26 (0.6%) | 2 (0.0%) |
| Fire ant | 1029 (24.1%) | 210 (4.9%) | 313 (7.3%) | 230 (5.4%) | 160 (3.7%) | 107 (2.5%) | 9 (0.2%) |
| Mosquito | 85 (2.0%) | 26 (0.6%) | 39 (0.9%) | 11 (0.3%) | 5 (0.1%) | 4 (0.1%) | 0 (0.0%) |
| Cockroach | 837 (19.6%) | 190 (4.4%) | 263 (6.1%) | 201 (4.7%) | 146 (3.4%) | 37 (0.9%) | 0 (0.0%) |
3.3. Factors Associated with Clinically Relevant sIgE Sensitization (Class ≥3)
| Characteristics | Class ≥3 n (%) |
Class <3 n (%) |
OR | 95% CI | p-value |
|---|---|---|---|---|---|
| Sex | |||||
| Male | 849 (51.05) | 841 (48.95) | 1 | - | - |
| Female | 1080 (41.32) | 1534 (58.68) | 0.67 | 0.60 – 0.76 | <0.01 |
| Age group | |||||
| <12 years | 562 (47.31) | 626 (52.69) | 1 | - | - |
| 12–35 years | 899 (55.12) | 732 (44.88) | 1.37 | 1.18 – 1.59 | <0.01 |
| 36–59 years | 383 (33.36) | 765 (66.64) | 0.56 | 0.47 – 0.66 | <0.01 |
| ≥60 years | 85 (27.42) | 225 (72.58) | 0.42 | 0.32 – 0.55 | <0.01 |
| Residence | |||||
| Ho Chi Minh City | 938 (43.71) | 1208 (56.29) | 1 | - | - |
| Other regions | 991 (46.50) | 1140 (53.50) | 1.12 | 0.99 – 1.26 | 0.07 |
| Diagnosis | |||||
| Psoriasis | 15 (60.00) | 10 (40.00) | 1 | - | - |
| Urticaria | 620 (42.97) | 823 (57.03) | 0.51 | 0.43 – 0.60 | 0.001 |
| Atopic dermatitis | 1095 (48.45) | 1165 (51.55) | 0.63 | 0.28 – 1.40 | 0.32 |
| Contact dermatitis | 199 (36.25) | 350 (63.75) | 0.38 | 0.28 – 0.53 | <0.01 |
| Comorbidity | |||||
| Yes | 254 (47.57) | 280 (52.43) | 1 | - | - |
| No | 1675 (44.75) | 2068 (55.25) | 1.12 | 0.93 – 1.34 | 0.22 |
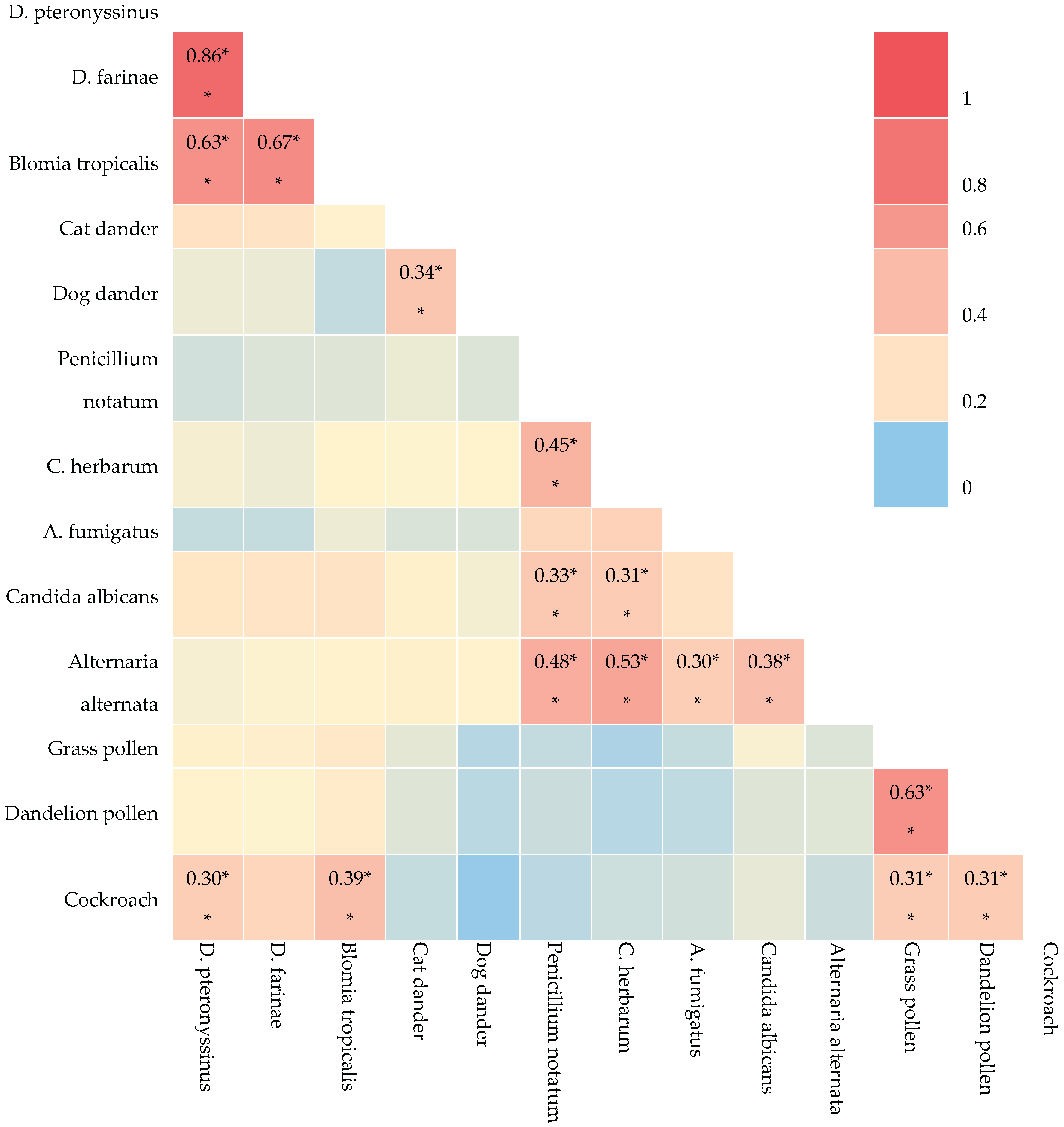
3.4. Association Patterns of Aeroallergen Co-Sensitization
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| HCMC | Ho Chi Minh City |
Appendix A
Appendix A.1
| Characteristics | Number | Percentage |
|---|---|---|
| Age group | ||
| <12 years | 1188 | 27.78 |
| 12–35 years | 1631 | 38.13 |
| 36–59 years | 1148 | 26.84 |
| ≥60 years | 310 | 7.25 |
| Sex | ||
| Male | 1663 | 38.88 |
| Female | 2614 | 61.12 |
| Residence | ||
| Ho Chi Minh City | 2146 | 50.18 |
| Other regions | 2131 | 49.82 |
| Diagnosis | ||
| Urticaria | 1443 | 33.74 |
| Psoriasis | 25 | 0.58 |
| Atopic dermatitis | 2260 | 52.84 |
| Allergic contact dermatitis | 549 | 12.84 |
| Comorbidities | ||
| Urticaria | 138 | 3.23 |
| Seborrheic dermatitis | 52 | 1.22 |
| Infectious dermatitis | 187 | 4.37 |
| Allergic contact dermatitis | 157 | 3.67 |
| None | 3743 | 87.51 |
References
- Valenta, R.; Karaulov, A.; Niederberger, V.; et al. Molecular Aspects of Allergens and Allergy. Adv Immunol 2018, 2018, 195–256. [Google Scholar] [CrossRef]
- Ansotegui, I.J.; Melioli, G.; Canonica, G.W.; et al. IgE allergy diagnostics and other relevant tests in allergy, a World Allergy Organization position paper. World Allergy Organ J 2020, 2020, 100080. [Google Scholar] [CrossRef] [PubMed]
- Chang, M.L.; Cui, C.; Liu, Y.H.; et al. Analysis of total immunoglobulin E and specific immunoglobulin E of 3,721 patients with allergic disease. Biomedical Reports 2015, 2015, 573–577. [Google Scholar] [CrossRef] [PubMed]
- Zuberbier, T.; Abdul Latiff, A.H.; Abuzakouk, M.; et al. The international EAACI/GA²LEN/EuroGuiDerm/APAAACI guideline for the definition, classification, diagnosis, and management of urticaria. Allergy 2022, 2022 Mar, 734–766. [Google Scholar] [CrossRef] [PubMed]
- Pham Nguyen Hai Ho; Tran Nguyen Du; Do Hoang Long. The prevalenceof allergens detected byimmunoblotting assay and some related factorsamong allergy patients at Can Tho Hospital of Dermato–Venereology in 2020. Can Tho Journal of Medicine and Pharmacy 2021, 41, 167–175.
- Vu Le Ngoc Lan; Cao Thi Binh; Uong Nguyen Duc Ninh; et, a. Total serum IgE and the relationship between total serum IgE and allergen - specific IgE on allergic patients at LAM Department, Pasteur Institute in Ho Chi Minh City, 2020 - 2021. VietNam Journal of Preventive Medicine 2022, 32, 169–178. [CrossRef]
- Zuberbier, T.; Aberer, W.; Asero, R.; et al. The EAACI/GA²LEN/EDF/WAO guideline for the definition, classification, diagnosis and management of urticaria. Allergy 2018, 2018 Jul, 1393–1414. [Google Scholar] [CrossRef] [PubMed]
- Xu-De, Z.; Bei-Bei, G.; Xi-Juan, W.; et al. Serum IgE Predicts Difference of Population and Allergens in Allergic Diseases: Data from Weifang City, China. Mediators of Inflammation 2021, 2021, 6627087. [Google Scholar] [CrossRef] [PubMed]
- Pawankar, R.; Canonica, G.W.; Holgate, S.T.; et al. WAO White Book on Allergy; World Allergy Organization, 2013. [Google Scholar]
- Gutiérrez-Brito, J.A.; Lomelí-Nieto, J.Á.; Muñoz-Valle, J.F.; et al. Sex hormones and allergies: exploring the gender differences in immune responses. Front Allergy 2025, 2025 Jan 7, 1483919. [Google Scholar] [CrossRef] [PubMed]
- King, F.; Kaczmarczyk, R.; Zink, A.; Biedermann, T.; Brockow, K. A new way forward? Examining the potential of quantitative analysis of IgE datasets. Allergy, Asthma & Clinical Immunology 2022, 18, 75. [Google Scholar] [CrossRef] [PubMed]
- Mediaty, A.; Neuber, K. Total and specific serum IgE decreases with age in patients with allergic rhinitis, asthma and insect allergy but not in patients with atopic dermatitis. Immunity & Ageing 2005, 2, 9. [Google Scholar] [CrossRef] [PubMed]
- Shi, L.; Liu, C.; Xiong, H.; Shi, D. Elevation of IgE in patients with psoriasis: Is it a paradoxical phenomenon? Frontiers in Medicine 2022, 9. [Google Scholar] [CrossRef] [PubMed]
- Chew; Yi; Chua; Fernandez, C.; Arruda; Chapman; Lee. Allergenic differences between the domestic mites Blomia tropicalis and Dermatophagoides pteronyssinus. Clinical & Experimental Allergy 1999, 29, 982–988. [Google Scholar] [CrossRef] [PubMed]
- Yeoh, S.M.; Kuo, I.C.; Wang, D.Y.; Liam, C.K.; Sam, C.K.; de Bruyne, J.A.; Lee, B.W.; Cheong, N.; Chua, K.Y. Sensitization Profiles of Malaysian and Singaporean Subjects to Allergens from Dermatophagoides pteronyssinus and Blomia tropicalis. International Archives of Allergy and Immunology 2003, 132, 215–220. [Google Scholar] [CrossRef] [PubMed]
- Lam, H.T.; Ekerljung, L.; Bjerg, A.; et al. Sensitization to airborne allergens among adults and its impact on allergic symptoms: a population survey in northern Vietnam. Clinical and Translational Allergy 2014, 4, 6. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




