Submitted:
21 April 2026
Posted:
23 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
Patients
Treatment
Data Collection and Ethics
Statistical Analysis
3. Results
3.1. Clinical Characteristics and Therapy
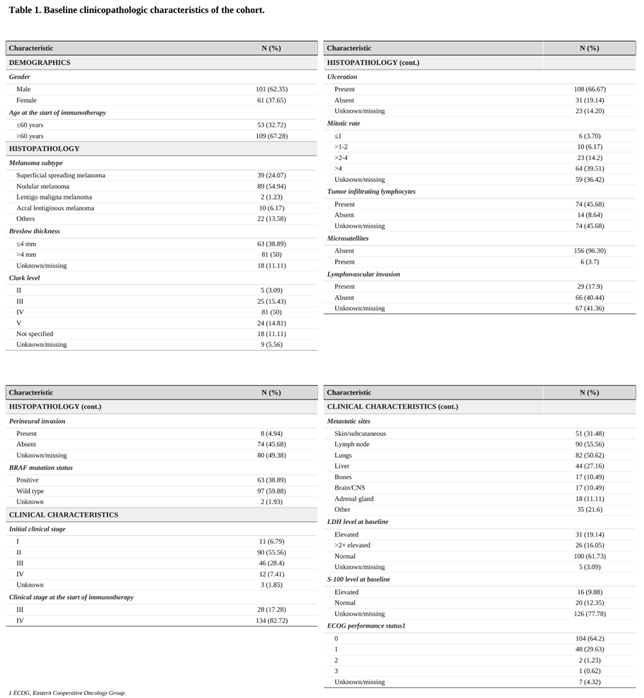
3.2. ROC analyses and cut-off values of inflammatory biomarkers
3.3. Inflammatory Biomarkers and Clinical Characteristics
3.4. Inflammatory Biomarkers and Survival Analyses
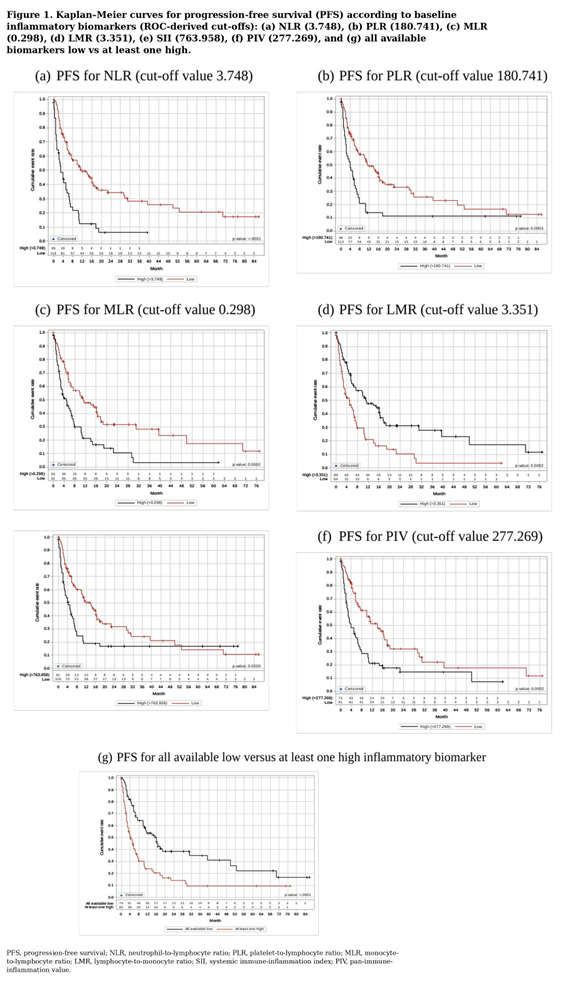
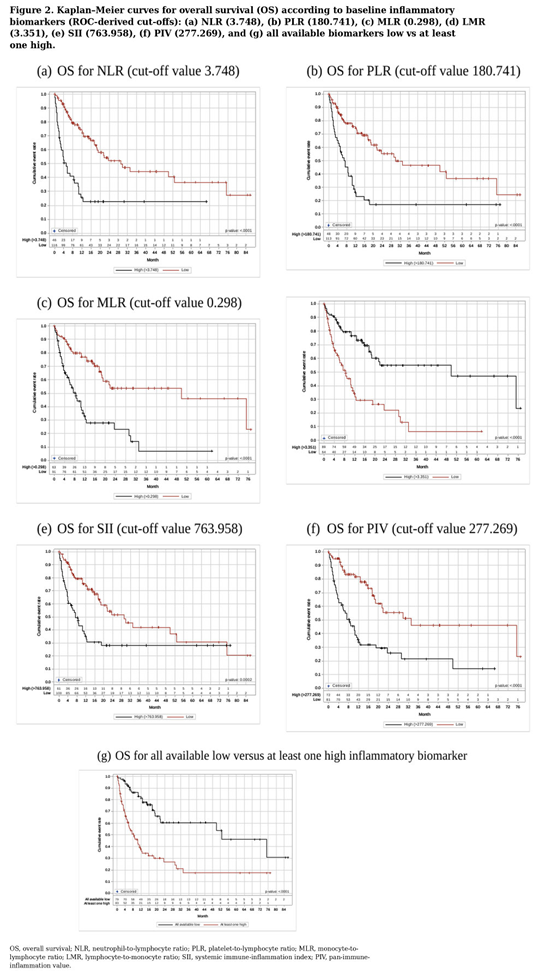
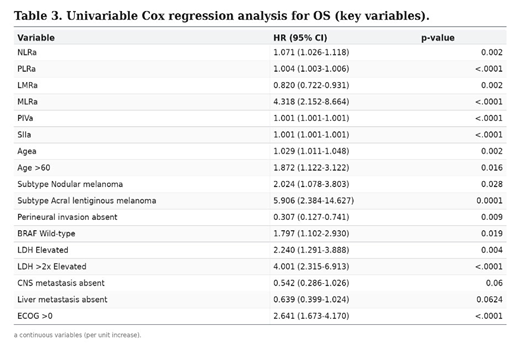
3.5. Cox Regression Analysis

4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Arnold, M.; Singh, D.; Laversanne, M.; et al. Global Burden of Cutaneous Melanoma in 2020 and Projections to 2040. JAMA Dermatol 2022, 158, 495–503. [Google Scholar] [CrossRef]
- Garbe, C.; Amaral, T.; Peris, K.; et al. European consensus-based interdisciplinary guideline for melanoma. Part 1: Diagnostics—Update 2024. Eur J Cancer 2025, 215, 115152. [Google Scholar] [CrossRef] [PubMed]
- Robert, C.; Ribas, A.; Schachter, J.; et al. Pembrolizumab versus ipilimumab in advanced melanoma (KEYNOTE-006): post-hoc 5-year outcomes from an open-label, multicentre, randomised, controlled, phase 3 study. Lancet Oncol 2019, 20, 1239–51. [Google Scholar] [CrossRef] [PubMed]
- Robert, C.; Long, G.V.; Brady, B.; et al. Five-Year Outcomes With Nivolumab in Patients With Wild-Type BRAF Advanced Melanoma. J Clin Oncol 2020, 38, 3937–46. [Google Scholar] [CrossRef] [PubMed]
- Larkin, J.; Chiarion-Sileni, V.; Gonzalez, R.; et al. Five-year survival with combined nivolumab and ipilimumab in advanced melanoma. N Engl J Med 2019, 381, 1535–46. [Google Scholar] [CrossRef]
- Robert, C.; Schachter, J.; Long, G.V.; et al. Pembrolizumab versus Ipilimumab in Advanced Melanoma. N Engl J Med 2015, 372, 2521–32. [Google Scholar] [CrossRef]
- Nebhan, C.A.; Johnson, D.B. Predictive biomarkers of response to immune checkpoint inhibitors in melanoma. Expert Rev Anticancer Ther 2020, 20, 137–45. [Google Scholar] [CrossRef]
- Baltussen, J.C.; Welters, M.J.P.; Verdegaal, E.M.E.; et al. Predictive Biomarkers for Outcomes of Immune Checkpoint Inhibitors (ICIs) in Melanoma: A Systematic Review. Cancers (Basel) 2021, 13, 6366. [Google Scholar] [CrossRef]
- Poletto, S.; Paruzzo, L.; Nepote, A.; et al. Predictive Factors in Metastatic Melanoma Treated with Immune Checkpoint Inhibitors: From Clinical Practice to Future Perspective. Cancers (Basel) 2023, 16, 101. [Google Scholar] [CrossRef]
- Templeton, A.J.; McNamara, M.G.; Šeruga, B.; et al. Prognostic role of neutrophil-to-lymphocyte ratio in solid tumors: a systematic review and meta-analysis. J Natl Cancer Inst 2014, 106, dju124. [Google Scholar] [CrossRef]
- Dolan, R.D.; Lim, J.; McSorley, S.T.; et al. The role of the systemic inflammatory response in predicting outcomes in patients with operable cancer: Systematic review and meta-analysis. Sci Rep 2017, 7, 16717. [Google Scholar] [CrossRef]
- Mantovani, A.; Allavena, P.; Sica, A.; Balkwill, F. Cancer-related inflammation. Nature 2008, 454, 436–44. [Google Scholar] [CrossRef]
- Ou, Y.; Liang, S.; Gao, Q.; et al. Prognostic value of inflammatory markers NLR, PLR, LMR, dNLR, ANC in melanoma patients treated with immune checkpoint inhibitors: a meta-analysis and systematic review. Front Immunol 2024, 15, 1482746. [Google Scholar] [CrossRef]
- Zhou, X.; Du, Y.; Huang, Z.; et al. Prognostic value of PLR in various cancers: a meta-analysis. PLoS One 2014, 9, e101119. [Google Scholar] [CrossRef] [PubMed]
- Capone, M.; Giannarelli, D.; Mallardo, D.; et al. Baseline neutrophil-to-lymphocyte ratio (NLR) and derived NLR could predict overall survival in patients with advanced melanoma treated with nivolumab. J Immunother Cancer 2018, 6, 74. [Google Scholar] [CrossRef]
- Ferrucci, P.F.; Gandini, S.; Battaglia, A.; et al. Baseline neutrophil-to-lymphocyte ratio is associated with outcome of ipilimumab-treated metastatic melanoma patients. Br J Cancer 2015, 112, 1904–10. [Google Scholar] [CrossRef]
- Giannakeas, V.; Kotsopoulos, J.; Brooks, J.D.; et al. Platelet Count and Survival after Cancer. Cancers (Basel) 2022, 14, 549. [Google Scholar] [CrossRef]
- Fucà, G.; Guarini, V.; Antoniotti, C.; et al. The Pan-Immune-Inflammation Value is a new prognostic biomarker in metastatic colorectal cancer: results from a pooled-analysis of the Valentino and TRIBE first-line trials. Br J Cancer 2020, 123, 403–9. [Google Scholar] [CrossRef] [PubMed]
- Lin, F.; Zhang, L.P.; Xie, S.Y.; et al. Pan-Immune-Inflammation Value: A New Prognostic Index in Operative Breast Cancer. Front Oncol 2022, 12, 830138. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Gong, L.; Gu, P.; et al. Pan-immune-inflammation and its dynamics: predictors of survival and immune-related adverse events in patients with advanced NSCLC receiving immunotherapy. BMC Cancer 2023, 23, 944. [Google Scholar] [CrossRef] [PubMed]
- Eisenhauer, E.A.; Therasse, P.; Bogaerts, J.; et al. New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur J Cancer 2009, 45, 228–47. [Google Scholar] [CrossRef] [PubMed]
- Susok, L.; Said, S.; Reinert, D.; et al. The pan-immune-inflammation value and systemic immune-inflammation index in advanced melanoma patients under immunotherapy. J Cancer Res Clin Oncol 2022, 148, 3103–8. [Google Scholar] [CrossRef]
- Fuca, G.; Beninato, T.; Bini, M.; et al. The Pan-Immune-Inflammation Value in Patients with Metastatic Melanoma Receiving First-Line Therapy. Target Oncol 2021, 16, 529–36. [Google Scholar] [CrossRef]
- Kuang, T.; Qiu, Z.; Wang, K.; et al. Pan-immune inflammation value as a prognostic biomarker for cancer patients treated with immune checkpoint inhibitors. Front Immunol 2024, 15, 1326083. [Google Scholar] [CrossRef]
- Van Wilpe, S.; Koornstra, R.; Den Brok, M.; et al. Lactate dehydrogenase: a marker of diminished antitumor immunity. Oncoimmunology 2020, 9, 1731942. [Google Scholar] [CrossRef]
- Fischer, K.; Hoffmann, P.; Voelkl, S.; et al. Inhibitory effect of tumor cell-derived lactic acid on human T cells. Blood 2007, 109, 3812–9. [Google Scholar] [CrossRef]
- Chatziioannou, E.; Higuita, L.M.S.; Kreft, S.; et al. Nomogram for predicting survival after first-line anti-PD-1-based immunotherapy in unresectable stage IV melanoma: a multicenter international study. ESMO Open 2024, 9, 103661. [Google Scholar] [CrossRef]
- Amaral, T.; Ottaviano, M.; Arance, A.; et al. Cutaneous melanoma: ESMO Clinical Practice Guideline for diagnosis, treatment and follow-up. Ann Oncol 2025, 36, 10–30. [Google Scholar] [CrossRef]
- Zhao, J.; Jin, J. Neutrophil extracellular traps: New players in cancer research. Front Immunol 2022, 13, 937565. [Google Scholar] [CrossRef] [PubMed]
- Liao, K.; Zhang, X.; Liu, J.; et al. The role of platelets in the regulation of tumor growth and metastasis: the mechanisms and targeted therapy. MedComm (2020) 2023, 4, e350. [Google Scholar] [CrossRef] [PubMed]
- Filippelli, A.; Del Gaudio, C.; Simonis, V.; et al. Scoping Review on Platelets and Tumor Angiogenesis: Do We Need More Evidence or Better Analysis? Int J Mol Sci 2022, 23, 13401. [Google Scholar] [CrossRef]
- Labelle, M.; Begum, S.; Hynes, R.O. Direct signaling between platelets and cancer cells induces an epithelial-mesenchymal-like transition and promotes metastasis. Cancer Cell 2011, 20, 576–90. [Google Scholar] [CrossRef] [PubMed]
- Labelle, M.; Begum, S.; Hynes, R.O. Platelets guide the formation of early metastatic niches. Proc Natl Acad Sci U S A 2014, 111, E3053-61. [Google Scholar] [CrossRef] [PubMed]
- Postow, M.A.; Chasalow, S.D.; Kuk, D.; et al. Absolute lymphocyte count as a prognostic biomarker for overall survival in patients with advanced melanoma treated with ipilimumab. Melanoma Res 2020, 30, 71–5. [Google Scholar] [CrossRef] [PubMed]
- Brummel, K.; Eerkens, A.L.; de Bruyn, M.; Nijman, H.W. Tumour-infiltrating lymphocytes: from prognosis to treatment selection. Br J Cancer 2023, 128, 451–8. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



