Submitted:
21 April 2026
Posted:
22 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
Study Group
- Study inclusion criteria:
- Study exclusion criteria:
Statistical Methods
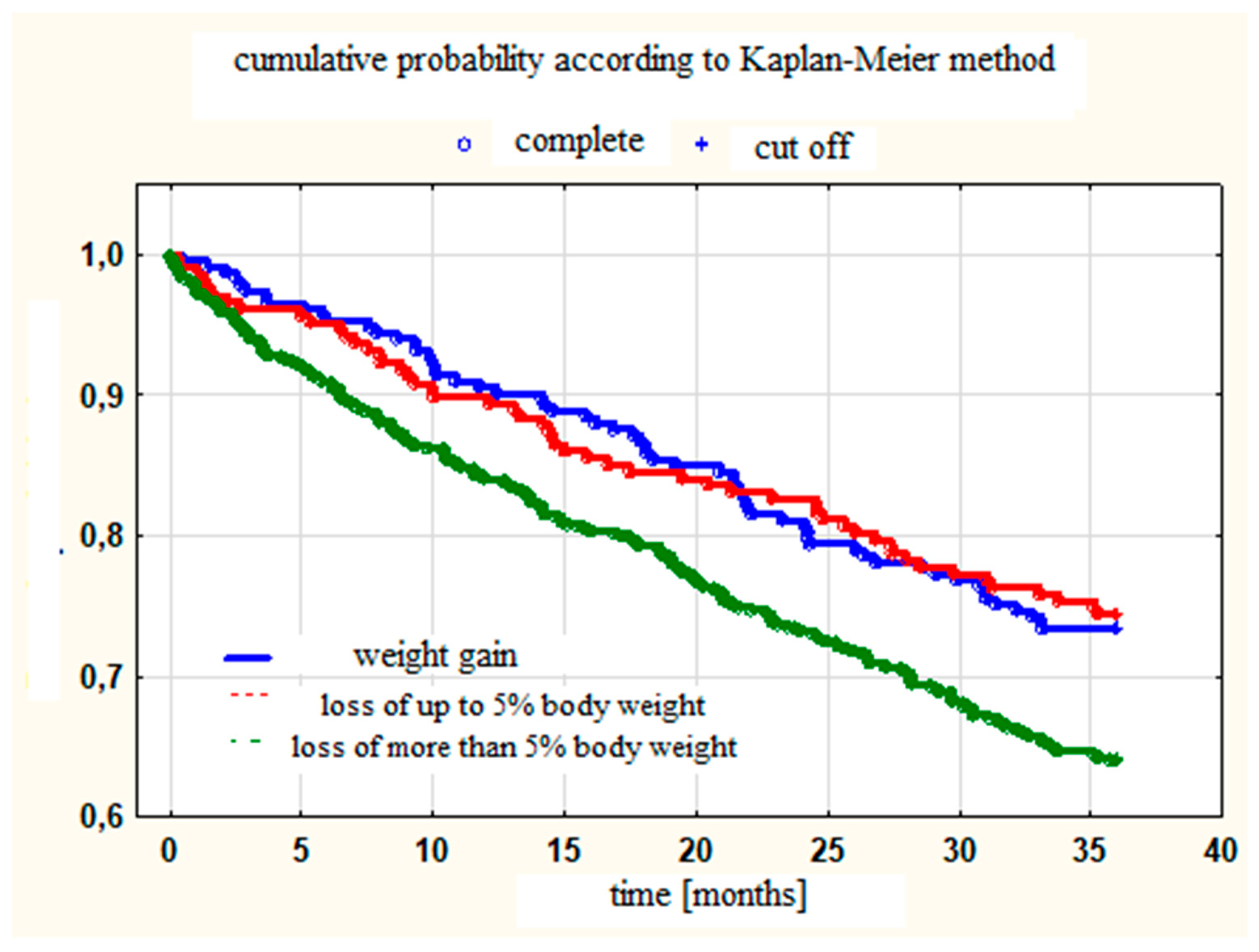
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| BMI | – body mass index, |
| HF | – heart failure, |
| NYHA | - New York Heart Association, |
| LVEF | - left ventricle ejection fraction, |
| ESV | - end-systolic volume, |
| EDV | - end-diastolic volume, |
| HR | - heart rate, |
| MVO2 | - maximal volume of oxygen consumption during treadmill test, |
| SBP | - systolic blood pressure, |
| DBP | - diastolic blood pressure, |
| eGFRMDRD | estimated glomerular filtration rate based on Modification of Diet in Renal Disease Study equation, |
| hsCRP | - high sensitivity C-reactive protein, |
| LDL | - low density lipoprotein, |
| HDL | - high density lipoprotein, |
| NTproBNP | - N-terminal pro brain-type natriuretic peptide, |
| ACEI | - angiotensin converting enzyme inhibitor, |
| BB | - β-receptors antagonists, |
| MRA | - mineralocorticoid receptor antagonists |
References
- Nessler, J.; Kozierkiewicz, A.; Gackowski, A.; Ponikowski, P.; Straburzynska-Migaj, E.; Uchmanowicz, I.; Hoffman, P.; Chlebus, K.; Gielerak, G.; Gąsior, M.; et al. Coordinated heart failure care in Poland: Towards optimal organisation of the health care system. Kardiol. Pol. 2018, 76, 479–487. [Google Scholar] [CrossRef] [PubMed]
- Lelonek, M.; Grabowski, M.; Kasprzak, J.D.; Leszek, P.; Nessler, J.; Pawlak, A.; Rozentryt, P.; Straburzynska-Migaj, E.; Rubiś, P. An expert opinion of the Heart Failure Association of the Polish Cardiac Society on the 2021 European Society of Cardiology guidelines for the diagnosis and treatment of acute and chronic heart failure: Heart failure guidelines from a national perspective. Kardiol. Pol. 2022, 80, 239–246. [Google Scholar] [CrossRef]
- McDonagh, T.A.; Metra, M.; Adamo, M.; Gardner, R.S.; Baumbach, A.; Böhm, M.; Burri, H.; Butler, J.; Čelutkienė, J.; Chioncel, O.; et al. 2021 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure: Developed by the Task Force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC). With the special contribution of the Heart Failure Association (HFA) of the ESC. Eur. J. Heart Fail. 2022, 24, 4–131. [Google Scholar] [CrossRef]
- Chartrand, D.J.; Murphy-Després, A.; Alméras, N.; Lemieux, I.; Larose, E.; Després, J.P. Overweight, Obesity, and CVD Risk: A Focus on Visceral/Ectopic Fat. Curr. Atheroscler. Rep. 2022, 24, 185–195. [Google Scholar] [CrossRef]
- Heath, L.; Jebb, S.A.; Aveyard, P.; Piernas, C. Obesity, metabolic risk and adherence to healthy lifestyle behaviours: Prospective cohort study in the UK Biobank. BMC Med. 2022, 20, 65. [Google Scholar] [CrossRef]
- Niedziela, J.; Hudzik, B.; Rozentryt, P. Epidemiologia zmian masy ciała w przebiegu przewlekłej niewydolności serca. Folia Cardiol. Excerpta 2013, 8, 18–24. [Google Scholar]
- Marcks, N.; Aimo, A.; Januzzi, J.L.; Vergaro, G.; Clerico, A.; Latini, R.; Meessen, J.; Anand, I.S.; Cohn, J.N.; Gravning, J.; et al. Re-appraisal of the obesity paradox in heart failure: A meta-analysis of individual data. Clin. Res. Cardiol. 2021, 110, 1280–1291. [Google Scholar] [CrossRef]
- Horwich, T.B.; Fonarow, G.C.; Clark, A.L. Obesity and the Obesity Paradox in Heart Failure. Prog. Cardiovasc. Dis. 2018, 61, 151–156. [Google Scholar] [CrossRef]
- Carbone, S.; Lavie, C.J.; Arena, R. Obesity and Heart Failure: Focus on the Obesity Paradox. Mayo Clin. Proc. 2017, 92, 266–279. [Google Scholar] [CrossRef] [PubMed]
- Aimo, A.; Januzzi, J.L.; Vergaro, G.; Clerico, A.; Latini, R.; Meessen, J.; Anand, I.S.; Cohn, J.N.; Gravning, J.; Ueland, T.; et al. Revisiting the obesity paradox in heart failure: Per cent body fat as predictor of biomarkers and outcome. Eur. J. Prev. Cardiol. 2019, 26, 1751–1759. [Google Scholar] [CrossRef] [PubMed]
- Thomas, E.; Gupta, P.P.; Fonarow, G.C.; Horwich, T.B. Bioelectrical impedance analysis of body composition and survival in patients with heart failure. Clin. Cardiol. 2019, 42, 129–135. [Google Scholar] [CrossRef]
- Wawrzeńczyk, A.; Anaszewicz, M.; Budzyński, J. Clinical significance of nutritional status in patients with chronic heart failure-a systematic review. Heart Fail. Rev. 2019, 24, 671–700. [Google Scholar] [CrossRef]
- Edqvist, J.; Rawshani, A.; Adiels, M.; Björck, L.; Lind, M.; Svensson, A.M.; Gudbjörnsdottir, S.; Sattar, N.; Rosengren, A. BMI, Mortality, and Cardiovascular Outcomes in Type 1 Diabetes: Findings Against an Obesity Paradox. Diabetes Care 2019, 42, 1297–1304. [Google Scholar] [CrossRef]
- Schetz, M.; De Jong, A.; Deane, A.M.; Druml, W.; Hemelaar, P.; Pelosi, P.; Pickkers, P.; Reintam-Blaser, A.; Roberts, J.; Sakr, Y.; et al. Obesity in the critically ill: A narrative review. Intensive Care Med. 2019, 45, 757–769. [Google Scholar] [CrossRef]
- Czapla, M.; Juárez-Vela, R.; Łokieć, K.; Karniej, P. The Association between Nutritional Status and In-Hospital Mortality among Patients with Heart Failure-A Result of the Retrospective Nutritional Status Heart Study 2 (NSHS2). Nutrients 2021, 13, 1669. [Google Scholar] [CrossRef]
- Karahalios, A.; English, D.R.; Simpson, J.A. Change in body size and mortality: A systematic review and meta-analysis. Int. J. Epidemiol. 2017, 46, 526–546. [Google Scholar] [CrossRef]
- Wleklik, M.; Uchmanowicz, I.; Jankowska-Polańska, B.; Andreae, C.; Regulska-Ilow, B. The Role of Nutritional Status in Elderly Patients with Heart Failure. J. Nutr. Health Aging 2018, 22, 581–588. [Google Scholar] [CrossRef] [PubMed]
- Yasuhara, S.; Maekawa, M.; Bamba, S.; Kurihara, M.; Nakanishi, N.; Yamamoto, T.; Sakai, H.; Yagi, N.; Nakagawa, Y.; Sasaki, M. Energy Metabolism and Nutritional Status in Hospitalized Patients with Chronic Heart Failure. Ann. Nutr. Metab. 2020, 76, 129–139. [Google Scholar] [CrossRef] [PubMed]
- Sze, S.; Pellicori, P.; Zhang, J.; Weston, J.; Clark, A.L. The impact of malnutrition on short-term morbidity and mortality in ambulatory patients with heart failure. Am. J. Clin. Nutr. 2021, 113, 695–705. [Google Scholar] [CrossRef] [PubMed]
- Valentova, M.; Anker, S.D.; von Haehling, S. Cardiac Cachexia Revisited: The Role of Wasting in Heart Failure. Heart Fail. Clin. 2020, 16, 61–69. [Google Scholar] [CrossRef]
- Przewłocki, T.; Gackowski, A. [Is it practical to use measurement of BNP and NT-proBNP levels in patients with acute coronary syndromes?]. Kardiol. Pol. 2009, 67, 1207–1209. [Google Scholar]
- Nessler, J.; Straburzyńska-Migaj, E.; Windak, A.; Solnica, B.; Szmitkowski, M.; Paradowski, M.; Kaźmierczak, J.; Puacz, E.; Rozentryt, P. Expert consensus on the usefulness of natriuretic peptides in heart failure. Kardiol. Pol. 2018, 76, 215–224. [Google Scholar] [CrossRef] [PubMed]
- Hsich, E.M.; Grau-Sepulveda, M.V.; Hernandez, A.F.; Eapen, Z.J.; Xian, Y.; Schwamm, L.H.; Bhatt, D.L.; Fonarow, G.C. Relationship between sex, ejection fraction, and B-type natriuretic peptide levels in patients hospitalized with heart failure and associations with inhospital outcomes: Findings from the Get With The Guideline-Heart Failure Registry. Am. Heart J. 2013, 166, 1063–1071.e1063. [Google Scholar] [CrossRef] [PubMed]
- Das, S.R.; Drazner, M.H.; Dries, D.L.; Vega, G.L.; Stanek, H.G.; Abdullah, S.M.; Canham, R.M.; Chung, A.K.; Leonard, D.; Wians, F.H.; et al. Impact of body mass and body composition on circulating levels of natriuretic peptides: Results from the Dallas Heart Study. Circulation 2005, 112, 2163–2168. [Google Scholar] [CrossRef] [PubMed]
- Arora, P.; Reingold, J.; Baggish, A.; Guanaga, D.P.; Wu, C.; Ghorbani, A.; Song, Y.; Chen-Tournaux, A.; Khan, A.M.; Tainsh, L.T.; et al. Weight loss, saline loading, and the natriuretic peptide system. J. Am. Heart Assoc. 2015, 4, e001265. [Google Scholar] [CrossRef]
- Wang, T.J.; Larson, M.G.; Levy, D.; Benjamin, E.J.; Leip, E.P.; Wilson, P.W.; Vasan, R.S. Impact of obesity on plasma natriuretic peptide levels. Circulation 2004, 109, 594–600. [Google Scholar] [CrossRef]
| Weight Change Categories | p-Value | |||
|
Group 1 N = 233 (23%) Weight Gain |
Group 2 N = 207 (20%) Stable Weight [%] |
Group 3 N = 589 (57%) Weight Loss |
||
| Clinical Parameters | ||||
| Age [years] | 54; [49–58] | 55; [48–59] | 54; [48–59] | p = 0.59 |
| Gender [% of women] | 18.5 | 12.6 | 12.2 | p = 0.06 |
| Etiology HF [% ischemic] | 63.1 | 66.7 | 61.5 | p = 0.41 |
| Height [m] | 1.7; [1.7–1.8] | 1.7; [1.7–1.8] | 1.7; [1.7–1.8] | p = 0.60 |
| BMI before the development of HF symptoms [kg/m2] | 26.0; [24.0–29.1] | 27.7; [25.2–30.5] B | 28.7; [25.8–32.0] Y,$ | p < 0.0001 |
| BMI at index date [kg/m2] | 28.7; [26.1–31.8] | 27.0; [24.8–29.7] C | 24.8; [25.8–32.0] Z,$ | p < 0.0001 |
| Weight change [%] | 7.8; [14.6–4.2] | 2.6; [0.4–3.6] C | 12.4; [8.6–17.3] Z,$ | p < 0.0001 |
| HF duration [months] | 46.1; [22.8–88.5] | 33.5; [14.2–64.3] B | 31.1; [11.0–66.7] % | p < 0.001 |
| NYHA class | 3.0; [2.0–3.0] | 2.0; [2.0–3.0] A | 3.0; [2.0–3.0] $ | p < 0.0001 |
| NYHA class (I/II/III/IV) [%] | 8.7/40.8/47.2/3.4 | 11.1/42.0/40.1/6.8 $ | 3.7/32.9/50.6/12.7 | p < 0.0001 |
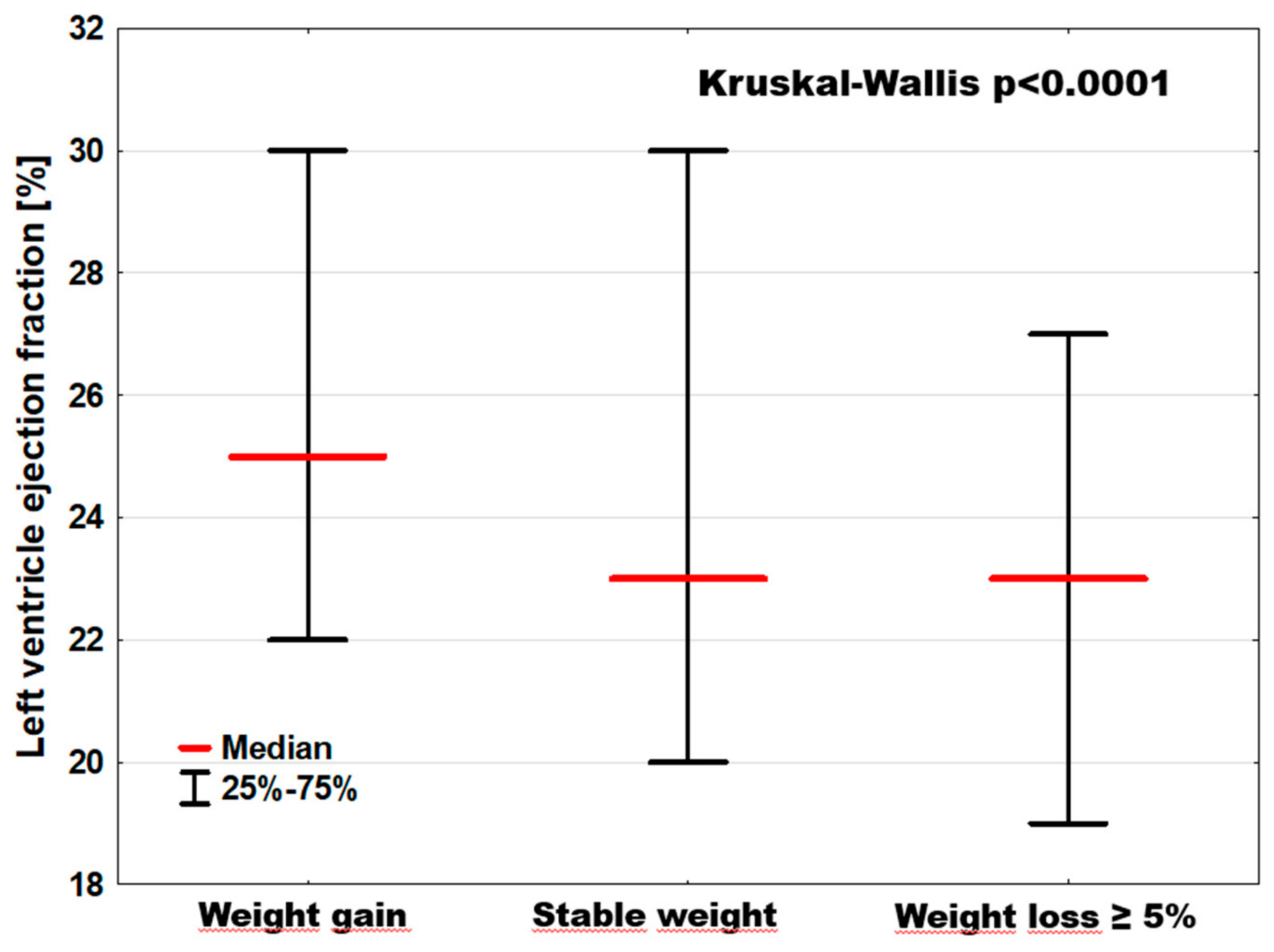
| LVEF [%] | 25; [22–30] | 23; [20–30] B | 23.0; [19.0–27.0] $ | p < 0.0001 |
| ESV [mm] | 55; [48–62] | 57; [48–65] | 57; [51–65] $ | p = 0.0138 |
| EDV [mm] | 69; [63–75] | 70; [64–76] | 69; [64–76] | p = 0.31 |
| HR [ud/min] | 79; [72–90] | 79; [73–88] | 79; [71–91] | p = 0.85 |
| MVO2 [mL/kg*min] | 14.3; [11.3–17.5] | 14.3; [11.3–17.6] | 14.0; [11.4–17.0] | p = 0.73 |
| SBP [mmHg] | 110; [100–125] | 110; [100–120] C | 105; [90–115] $ | p < 0.0001 |
| DBP [mmHg] | 70; [65–80] | 70; [70–80] A | 70; [60–70] % | p < 0.0001 |
| Biochemical parameters | ||||
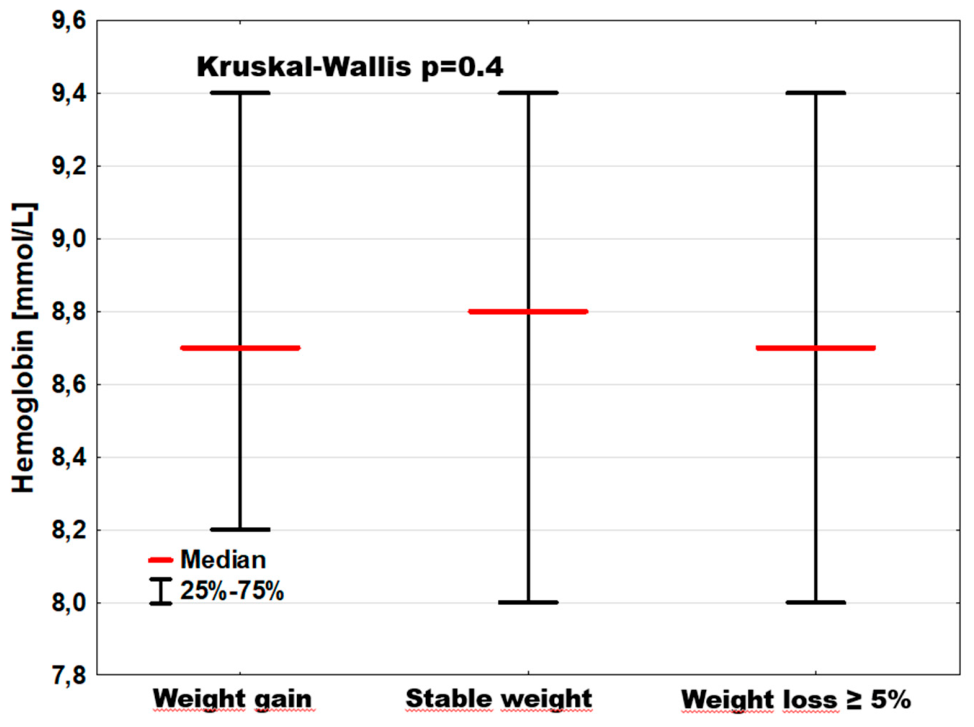
| Hemoglobin [mmol/L] | 8.7; [8.2–9.4] | 8.8; [8.0–9.4] | 8.7; [8.0–9.4] | p = 0.43 |
| Creatinine [µmoll/l] | 86; [71–106] | 84; [74–104] | 88; [74–110] | p = 0.31 |
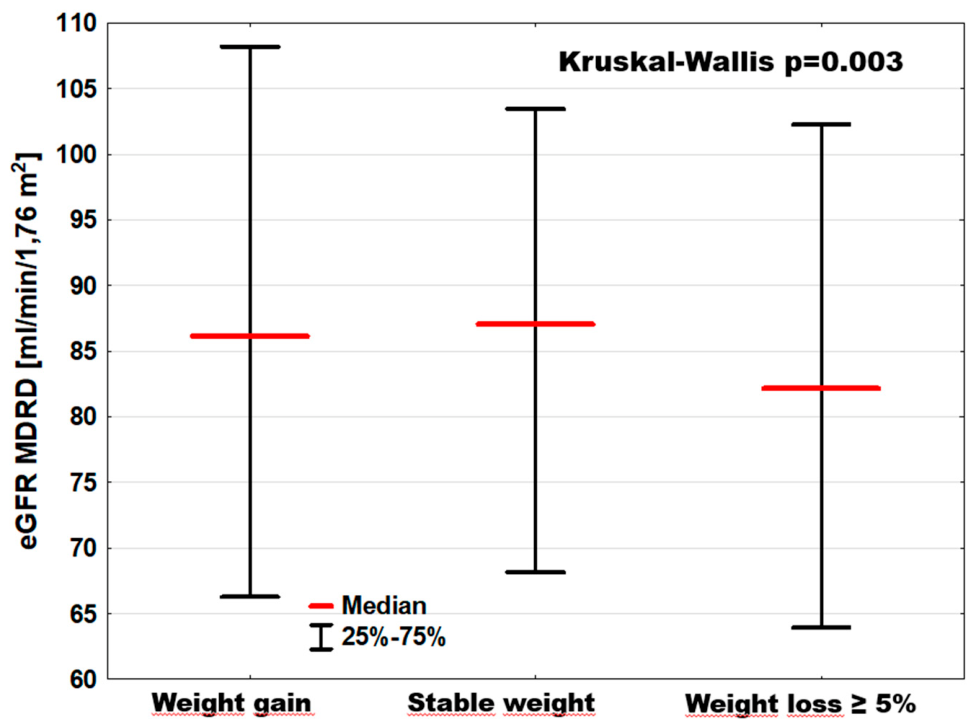
| eGFRMDRD [mL/min] | 86; [66–108] | 87; [68–103] | 82; [64–102] | p = 0.38 |
| Uric acid [mmol/L] | 409; [337–504] | 402; [329–493] | 413; [337–519] | p = 0.66 |
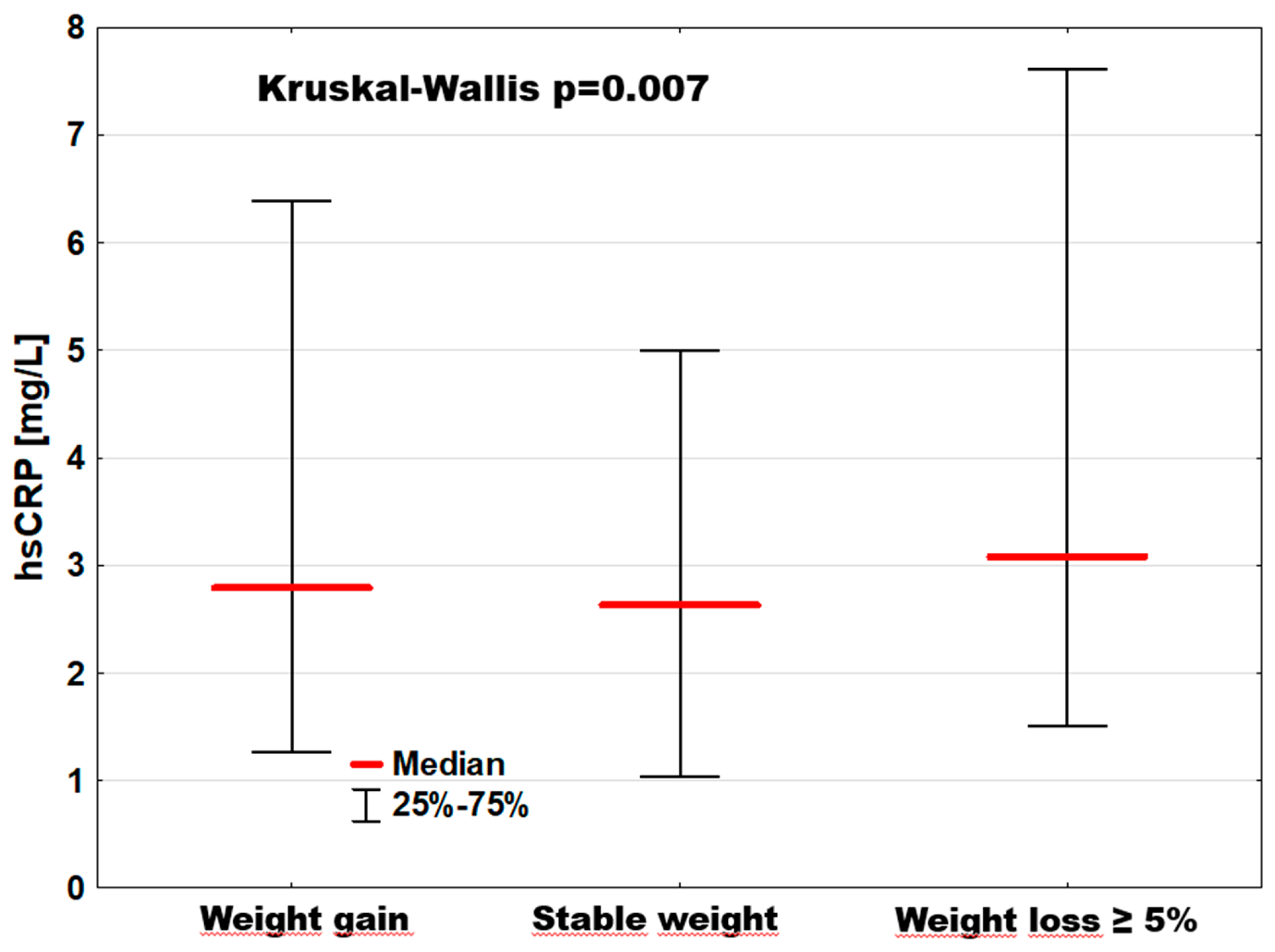
| hsCRP [mg/L] | 2.8; [1.3–6.4] | 2.6; [1.0–5.0] | 3.1; [1.5–7.6] | p = 0.0066 |
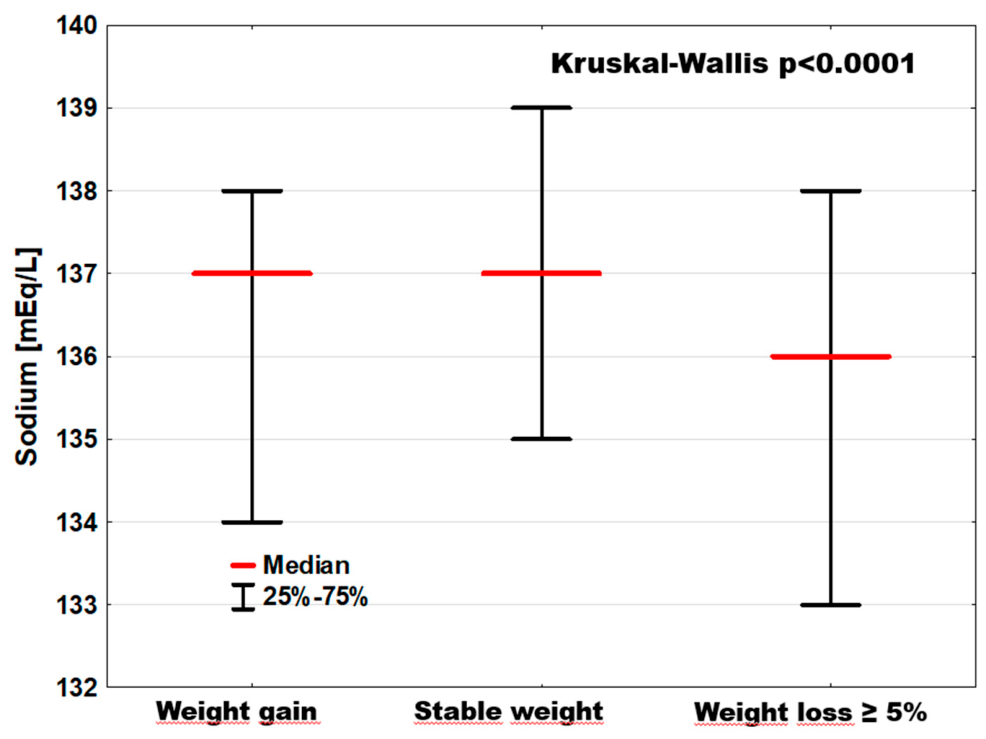
| Sodium [mEq/L] | 137; [134–138] | 137; [135–139] B | 136; [133–138] & | p = 0.0002 |
| Potassium [mEq/L] | 4.5; [4.3–4.8] | 4.5; [4.2–4.9] | 4.6; [4.3–5.0] & | p = 0.0126 |
| Glucose [mol/L] | 5.5; [5.0–6.4] | 5.6; [5.0–6.2] | 5.5; [4.9–6.4] | p = 0.95 |
| Total Cholesterol [mmol/L] | 4.3; [3.6–5.2] | 4.4; [3.7–5.3] | 4.3; [3.6–5.3] | p = 0.75 |
| LDL [mmol/L] | 2.5; [1.9–3.2] | 2.5; [2.0–3.4] | 2.5; [1.9–3.2] | p = 0.65 |
| HDL [mmol/L] | 1.1; [1.0–1.4] | 1.1; [0.9–1.4] | 1.2; [0.9–1.4] | p = 0.76 |
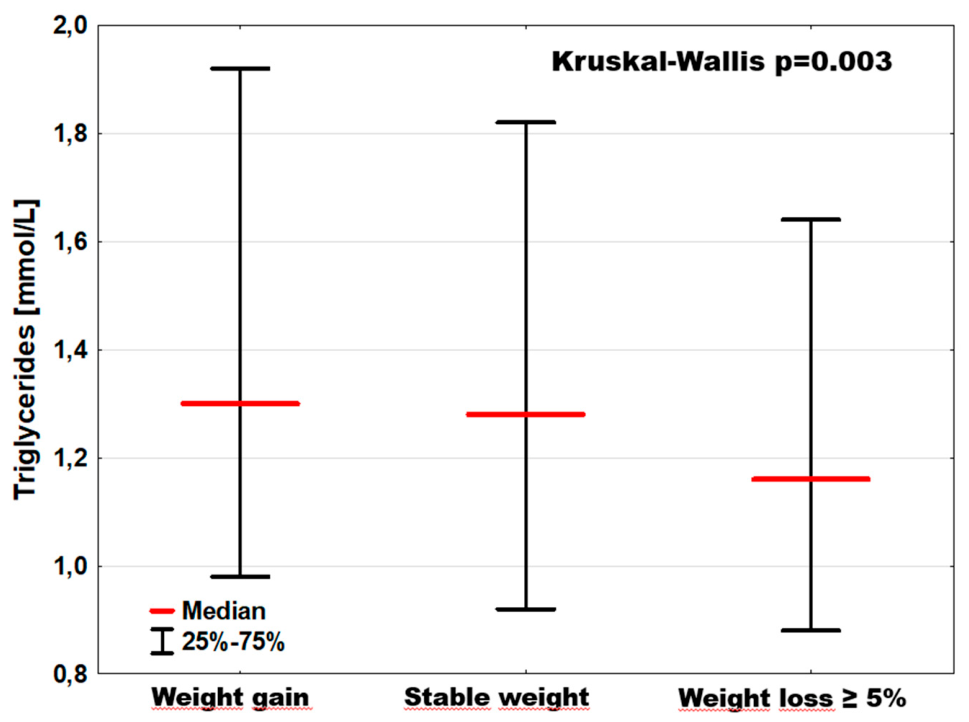
| Triglycerides [mmol/L] | 1.3; [1.0–1.9] | 1.3; [0.9–1.8] | 1.2; [0.9–1.6] | p = 0.0035 |
| Bilirubin [µmol//L] | 12.3; [9.5–18.1] | 12.9; [9.4–20.0] | 15.3; [10.5–22.8] % | p < 0.0001 |
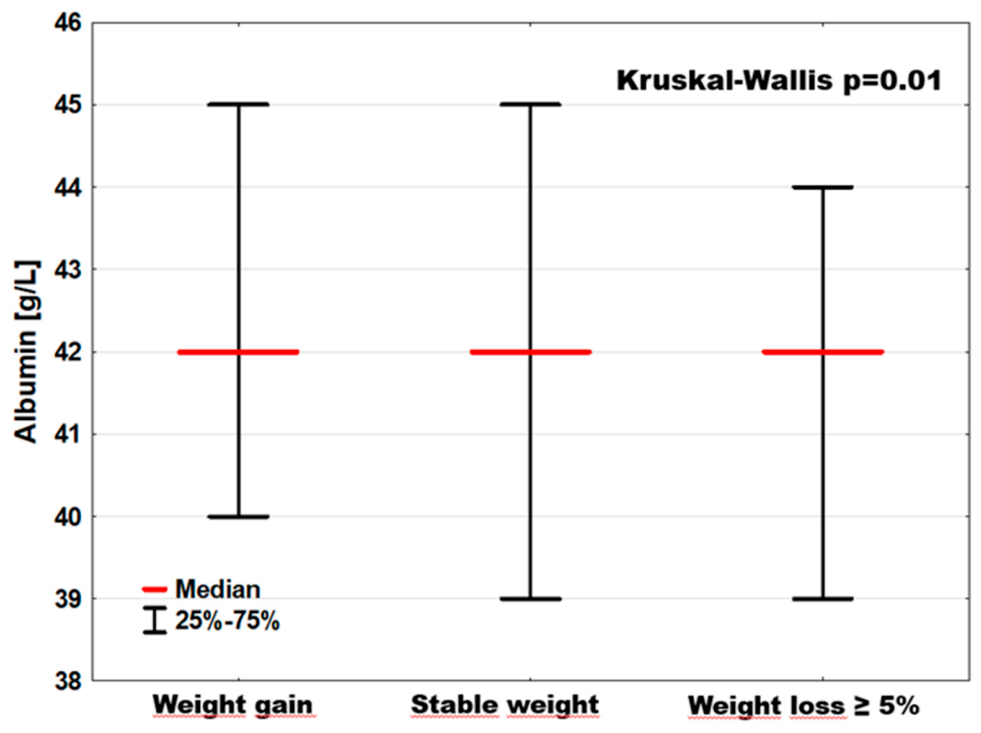
| Albumin [g/L] | 42.0; [40.0–45.0] | 42.0; [39.0–45.0] | 42.0; [39.0–44.0] & | p = 0.0134 |
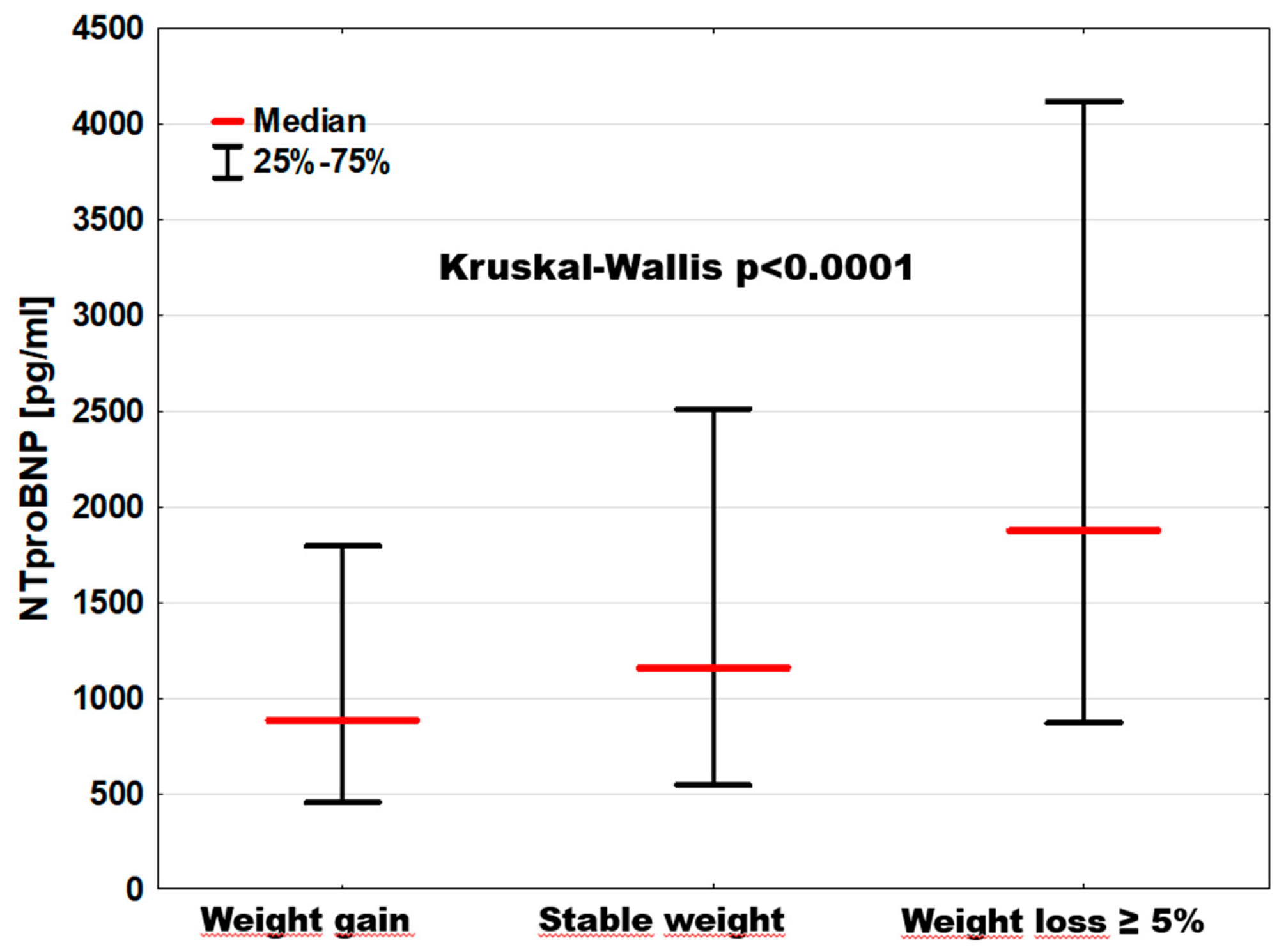
| NTproBNP [pg/mL] | 882; [454–1794] | 1157; [545–2511] C | 1874; [870–4114] $ | p < 0.0001 |
| Accompanying diseases [%] | ||||
| Hypertension | 56.7 | 59.9 | 53.0 | p = 0.20 |
| Hypercholesterolaemia | 64.0 | 64.3 | 58.1 | p = 0.15 |
| Hypertriglyceridaemia | 50.2 | 44.4 | 41.6 & | p = 0.08 |
| Diabetes melitus | 27.5 | 26.1 | 33.6 X | p = 0.06 |
| Smoking history | 6.4 | 10.6 | 12.1 & | p = 0.70 |
| Pharmacotherapy [%] | ||||
| ACEI | 98.7 | 95.2 | 91.0 % | p = 0.007 |
| BB | 98.7 | 98.1 | 96.9 | p = 0.29 |
| MRA | 84.3 | 89.9 | 90.8 % | p = 0.006 |
| Loop diuretics | 92.8 | 84.1 | 94.6 Y | p = 0.0002 |
| Prognosis | ||||
| Mortality at 3 years [%] | 16.7 | 17.9 | 25.5 Y,% | p = 0.0035 |
| Group 1 (Weight Gain) |
Group 2 (Stable Weight) |
Group 3 (Weight Loss) |
|
| Hazard ratio; (95% confidence intervals), p-value | |||
| Raw model | 1.03; (0.72–1.49), p = 0.24 | 1.0 | 1.51; (1.12–2.04), p = 0.0006 |
| Model 1 (adjusted for age and gender) | 1.06; (0.73–1.53), p = 0.28 | 1.0 | 1.55; (1.15–2.10), p = 0.0004 |
| Model 2—model 1 and BMI before HF, duration of HF, etiology of HF | 0.95; (0.66–1.38), p = 0.07 | 1.0 | 1.60; (1.18–2.17), p < 0.0001 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




