Submitted:
21 April 2026
Posted:
22 April 2026
You are already at the latest version
Abstract
Keywords:
Introduction
Methods
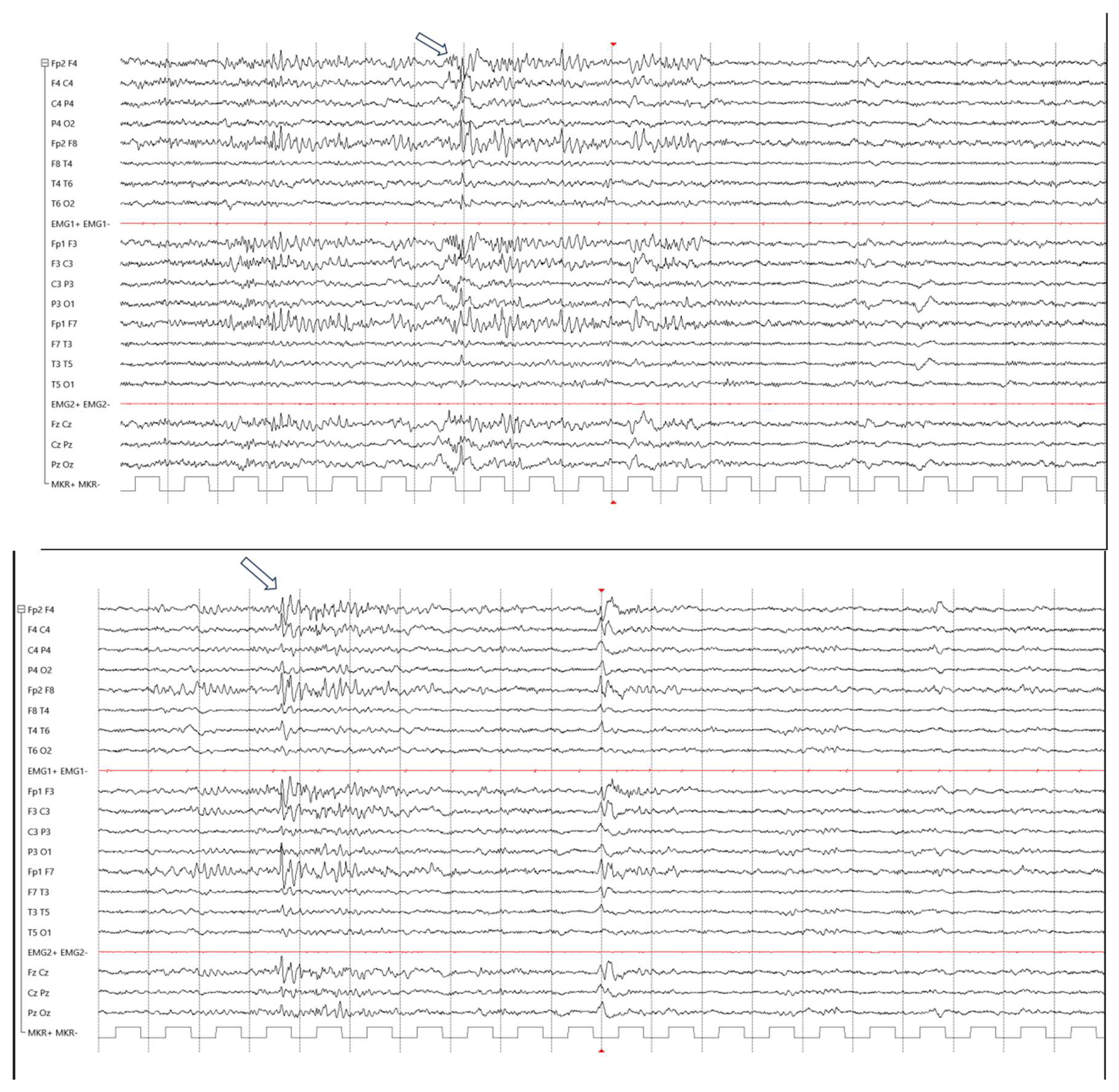
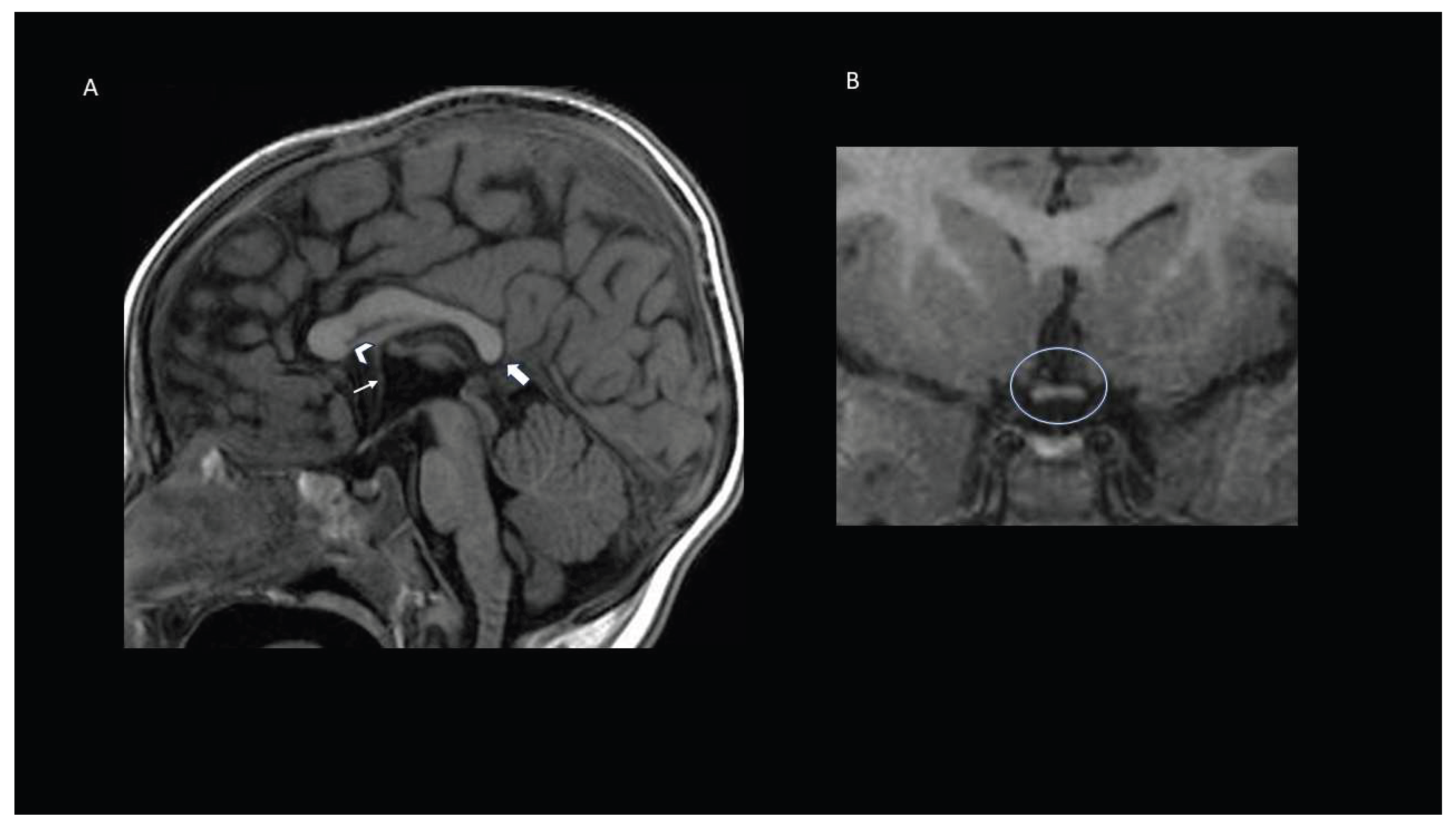
Results
Case Presentations
Patient 1
Patient 2
Discussion
Conclusions
Author Contributions
Funding
Informed Consent Statement
Conflicts of Interest
References
- Li, S.; Zhang, W.; Liang, P.; Zhu, M.; Zheng, B.; Zhou, W.; Wang, C.; Zhao, X. Novel variants in the CLCN4 gene associated with syndromic X-linked intellectual disability. Frontiers in Neurology 2023, 14, 1096969. [Google Scholar] [CrossRef] [PubMed]
- He, H.; Li, X.; Guzman, G.A.; Bungert-Plümke, S.; Franzen, A.; Lin, X.; Zhu, H.; Peng, G.; Zhang, H.; Yu, Y.; et al. Expanding the genetic and phenotypic relevance of CLCN4 variants in neurodevelopmental condition: 13 new patients. Journal of Neurology 2024, 271, 4933–4948. [Google Scholar] [CrossRef] [PubMed]
- Veeramah, K.R.; Johnstone, L.; Karafet, T.M.; Wolf, D.; Sprissler, R.; Salogiannis, J.; Barth-Maron, A.; Greenberg, M.E.; Stuhlmann, T.; Weinert, S.; et al. Exome sequencing reveals new causal mutations in children with epileptic encephalopathies. Epilepsia 2013, 54, 1270–1281. [Google Scholar] [CrossRef] [PubMed]
- Hu, H.; Haas, S.A.; Chelly, J.; Van Esch, H.; Raynaud, M.; de Brouwer, A.P.; Weinert, S.; Froyen, G.; Frints, S.G.; Laumonnier, F.; et al. X-exome sequencing of 405 unresolved families identifies seven novel intellectual disability genes. Molecular Psychiatry 2016, 21, 133–148. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Lu, F.; Zhang, L.; Li, H.; Du, S.; Tang, J. Novel CLCN4 variant associated with syndromic X-linked intellectual disability in a Chinese girl: A case report. BMC Pediatrics 2021, 21, 384. [Google Scholar] [CrossRef] [PubMed]
- Palmer, E.E.; Pusch, M.; Picollo, A.; Forwood, C.; Nguyen, M.H.; Suckow, V.; Gibbons, J.; Hoff, A.; Sigfrid, L.; Megarbane, A.; et al. Functional and clinical studies reveal pathophysiological complexity of CLCN4-related neurodevelopmental condition. Molecular Psychiatry 2023, 28, 668–697. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Luo, S.; Mei, S.; Shang, Q.; Zhang, W.; Zhang, X.; Liu, L.; Lei, Z.; Zhang, Y. Analysis of CLCN4 gene variant in a child with Raynaud-Claes syndrome. Zhonghua Yi Xue Yi Chuan Xue Za Zhi 2023, 40, 1280–1283. [Google Scholar] [PubMed]
- Sahly, A.N.; Sierra-Marquez, J.; Bungert-Plümke, S.; Franzen, A.; Mougharbel, L.; Berrahmoune, S.; Dassi, C.; Poulin, C.; Srour, M.; Guzman, R.E.; et al. Genotype–phenotype correlation in CLCN4-related developmental and epileptic encephalopathy. Human Genetics 2024, 143, 667–681. [Google Scholar] [CrossRef] [PubMed]
- Tettey-Matey, A.; Picollo, A.; Sbrana, F.; Coppola, M.A.; Rubino, E.; Giusto, A.; Festa, M.; Angeli, E.; Picco, C.; Barbieri, R.; et al. Dominant action of CLCN4 neurodevelopmental disease variants in heteromeric endosomal ClC-3/ClC-4 transporters. Cells 2025, 14, 1973. [Google Scholar] [CrossRef] [PubMed]
- He, H.; Guzman, R.E.; Cao, D.; Sierra-Marquez, J.; Yin, F.; Fahlke, C.; Peng, J.; Stauber, T. The molecular and phenotypic spectrum of CLCN4-related epilepsy. Epilepsia 2021, 62, 1401–1415. [Google Scholar] [CrossRef]
- Palmer, E.E.; Stuhlmann, T.; Weinert, S.; Haan, E.; Van Esch, H.; Holvoet, M.; Boyle, J.; Leffler, M.; Raynaud, M.; Moraine, C.; et al. De novo and inherited mutations in the X-linked gene CLCN4 are associated with syndromic intellectual disability and behavior and seizure disorders in males and females. Molecular Psychiatry 2018, 23(2), 222–230. [Google Scholar] [CrossRef]
- Raynaud, M.; Gendrot, C.; Dessay, B.; Moncla, A.; Ayrault, A.D.; Moizard, M.P.; Toutain, A.; Briault, S.; Villard, L.; Ronce, N.; et al. X-linked mental retardation with neonatal hypotonia in a French family (MRX15): Gene assignment to Xp11.22–Xp21.1. American Journal of Medical Genetics 1996, 64, 97–106. [Google Scholar] [CrossRef]
- Claes, S.; Gu, X.X.; Legius, E.; Lorenzetti, E.; Marynen, P.; Fryns, J.P.; Cassiman, J.J.; Raeymaekers, P. Linkage analysis in three families with nonspecific X-linked mental retardation. American Journal of Medical Genetics 1996, 64, 137–146. [Google Scholar] [CrossRef]
- Richards, S.; Aziz, N.; Bale, S.; Bick, D.; Das, S.; Gastier-Foster, J.; Grody, W.W.; Hegde, M.; Lyon, E.; ACMG Laboratory Quality Assurance Committee; et al. Standards and guidelines for the interpretation of sequence variants: A joint consensus recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genet Med 2015, 17, 405–424. [Google Scholar] [CrossRef] [PubMed]
- Green, E.; Stroud, L.; O’Connell, R.; Bloomfield, S.; Cronje, J.; Foxcroft, C.; Venter, D. Griffiths Scales of Child Development, 3rd ed.; Hogrefe, 2016. [Google Scholar]
- Bayley, N. Bayley Scales of Infant and Toddler Development, 3rd ed.; 2005. [Google Scholar]
- Sparrow, S.S.; Cicchetti, D.V.; Balla, D.A. Vineland Adaptive Behavior Scales, 2nd ed.; 2016. [Google Scholar]
- Zhou, P.; He, N.; Zhang, J.W.; Lin, Z.J.; Wang, J.; Yan, L.M.; Meng, H.; Tang, B.; Li, B.M.; Liu, X.R.; et al. Novel mutations and phenotypes of epilepsy-associated genes in epileptic encephalopathies. Genes, Brain and Behavior 2018, 17(8), e12456. [Google Scholar] [CrossRef] [PubMed]
- Garrett, A.; Kalscheuer, V.M.; Figueroa, R.R.; Palmer, E.E.; Morgan, A.T. CLCN4-related neurodevelopmental condition: Characterization of speech and language abilities. American Journal of Medical Genetics Part A 2025, 197(12), e64192. [Google Scholar] [CrossRef] [PubMed]
- Rossi, J.; Russo, M.; Gobbi, G.; Terracciano, A.; Zuntini, R.; Caraffi, S.G.; Novelli, A.; Garavelli, L.; Valzania, F.; Rizzi, R. Developmental and epileptic encephalopathy in a young Italian woman with a de novo missense variant in the CLCN4 gene: A case report. Brain and Development 2023, 45(8), 445–450. [Google Scholar] [CrossRef] [PubMed]
- Spagnoli, C.; Fusco, C.; Pisani, F. Rett Syndrome Spectrum in Monogenic Developmental-Epileptic Encephalopathies and Epilepsies: A Review. Genes 2021, 12(8), 1157. [Google Scholar] [CrossRef] [PubMed]
| Genetic Variant | Type of Variant | Gender | Age (years if not Specified) | Epilepsy | Neurologic Signs | Intellectual Disability | Language | ASD/Autistic Traits | Dysmorphisms | Brain MRI | Reference |
|---|---|---|---|---|---|---|---|---|---|---|---|
| c.1390-12(IVS9) T>G | Splicing | M | 6 | No | No | Yes (not specified the level) | Non-verbal | Yes | No | Right temporal arachnoid cyst and slightly wide left temporal subcranial plate gap | Li et al., 2023 |
| c.1576+5G>A | Splicing | M | 19 | Yes | walking | Borderline | Sentences (regression) | Yes | NR | NA | He et al., 2024 |
| p.(Ala448Val) | Missense LoF | F | 3 | No | Hypotonia, walking at 28 months | Severe | Non-verbal | Yes | Bushy eyebrows, downslanted palpebral fissures, esotropia, depressed nasal bridge, sparse teeth | Thin corpus callosum, mega cisterna magna, ventriculomegaly | Xu et al., 2021 |
| p.(Ala555Val) | Missense GoF | F | 3 | No | Hypertonia, walking at the age of 2 | Moderate | Severe delay | No | Microcephaly | Widened bilateral frontal-temporal extra brain space and enlarged the left ventricle | Li et al.,2023 |
| p.(Ala555Val) | Missense GoF | F | 5 | No | Hypertonia, non-ambulatory, swallowing difficulties | Severe | Non-verbal | No | Microcephaly | Small cranial volume, slightly thin the posterior of the corpus callosum, and widened ventricles | Li et al., 2023 |
| p.(Ala555Val) | Missense GoF | F | 9 | No | Non-ambulatory, swallowing difficulties with gastrostomy feeding | Yes, not specified the level | Non-verbal | No | Microcephaly | Diffuse cortical volume loss with mild lateral and third ventricular enlargement | Palmer et al., 2023 |
| p.(Ala555Val) | Missense GoF | F | 4 | No | Non-ambulatory, swallowing difficulties with gastrostomy feeding | Moderate | Non-verbal | Stereotypical hand movements | Microcephaly, positional plagiocephaly, deep-set and wide-spaced eyes, broad bulbous nose, large ears, small jaw, and high palate | Agenesis of the corpus callosum and anterior commissure (complete commissural agenesis) and abnormal orientation of the hippocampi | Palmer et al., 2023 |
| p.(Ala555Val) | Missense GoF | F | 4 | No | Non-ambulatory feeding difficulties | Yes, not specified the level | Non-verbal | No | Microcephaly | Small pons, immature myelination | Palmer et al., 2023 |
| p.(Ala555Val) | Missense GoF | F | 10 | No | Hypotonia, walking at 18 months | Yes, not specified the level | Two-to-three-word phrases | No | Microcephaly, posteriorly rotated ears, slightly arched eyebrows, slightly depressed nasal bridge, decreased muscle bulk, prominent columella, fifth finger clinodactyly | Normal | Palmer et al., 2023 |
| p.(Ala555Val) | Missense GoF | F | 18 months | No | Hypertonia, Non-ambulatory | Severe | Non-verbal | NR | Microcephaly, depressed nasal bridge | Dilation of the cerebral ventricles, thin corpus callosum, delayed myelination | He et al., 2024 |
| p.(Ala555Val) | Missense GoF | F | 5 | No | Walking | Severe | Few words | NR | NR | NA | He et al., 2024 |
| p.(Ala555Val) | Missense GoF | F | 28 | No | Walking | Severe | Simple sentences | NR | NR | NA | He et al., 2024 |
| p.(Arg41Trp) | Missense LoF | M | 4 | No | Non-ambulatory, autonomic, paroxysmal involuntary eye movements, tic disorder | Severe | Single words | NR | NR | NA | He et al., 2024 |
| p.(Arg360Ser) | Missense LoF | M | 18 | No | No | Severe (regression) | Non-verbal (regression) | Yes | Elongated face, long nose, mildly anteverted ears, prominent chin, and a pre-auricular pit at base of left helix. 5th finger proximal interphalangeal joint camptodactyly, mild pectus excavatum, thoracolumbar kyphosis and mild scoliosis with leg length discrepancy | Non-specific bilateral small punctate frontal white matter hyperintensities, mildly prominent Virchow-Robin spaces. Slightly bulky corpus callosum | Palmer et al., 2023 |
| p.(Arg603Trp) | Missense LoF | M | 2 | Infantile spasms, focal seizures | Non-ambulatory | Severe | Non-verbal | No | Depressed nasal bridge, small chin | Enlargement of subarachnoid spaces, dilation of the cerebral ventricles | He et al., 2024 |
| p.(Arg694Gln) | Missense LoF | M | 5 | No | Feeding difficulties, poor vision | Yes (not specified the level) | Non-verbal | Traits | No | Normal | Li et al., 2023 |
| p.(Arg718Trp) |
Missense LoF | F |
6 | Yes, blank stares, cyanosis, and emesis | No | Yes (not specified the level, regression) | Very limited expressive language | Yes, echolalia, stereotyped or repetitive motor movements, | No | NA | Palmer et al., 2023 |
| p.(Arg718Trp) |
Missense LoF | F |
9 | Hypertonia and staring during hyperpyrexia, erratic clonic movements and leftward head deviation followed by diffuse hypotonia, occurring during febrile and afebrile states | Hypotonia, walking at 24 months, gait instability, pyramidal signs, feeding difficulties | Profound (regression) | Non-verbal | Yes, midline stereotypies, hand-to-mouth movements, bruxism | Microcephaly | Bilateral frontal-parietal and periventricular white matter hyperintensities and corpus callosum thinning | This study |
| p.(Arg718Trp) |
Missense LoF | F |
8 | Absence seizures | Hypotonia | Severe, regression | NR | NR | NR | NR | Palmer et al., 2018 |
| p.(Arg718Trp) |
Missense LoF | F |
NR | NR | NR | NR | NR | NR | NR | NR | Palmer et al., 2023 |
| p.(Arg718Trp) |
Missense LoF | M | 1 month | Intractable epilepsy | NR | Severe | NR | NR | NR | NR | He et al., 2021 |
| p.(Arg718Trp) |
Missense LoF | M | 3 | Intractable epilepsy | NR | NR | NR | NR | NR | NR | Zhou et al., 2018 |
| p.(Arg718Trp) |
Missense LoF | NR (3) | NR | NR | NR | NR | NR | NR | NR | NR | Palmer et al., 2023 |
| p.(Arg718Trp) |
Missense LoF | F | 4 | Yes | Hypotonia, feeding difficulties | NR | First words at 12-15 months, CAS, dysarthria | Yes | NR | Mega cisterna magna | Garrett et al., 2025 |
| p.(Asn141Ser) | Missense LoF | M | 3 | No | NR | Moderate | Non-verbal | Yes | NR | NA | Li et al., 2023 |
| p.(Asn309Ser) | Missense LoF | M | 13 | Possible absence seizures | Hypotonia, walking at the age of 23 months, ataxia | Moderate-severe | Sentences | Yes | Long face with square chin and upslanting palpebral fissures, long fingers, pes planus, and joint hyperextensibility | Complete agenesis of the corpus callosum | Palmer et al., 2023 |
| p.(Asn309Ser) | Missense LoF | M | 21 | Focal seizures with secondary generalization | Hypotonia, ataxia | Moderate-severe | NR | No | Long face with square chin, distinct philtrum and tented upper lip, bilateral strabismus, long fingers, camptodactyly pes planus, and joint hyperextensibility | Complete agenesis of the corpus callosum | Palmer et al., 2023 |
| p.(Asp15Serfs*18) | Frameshift | M (5) | NR | NR | NA | Borderline (1/5), Mild (3/5), Moderate (1/5) | NR | No | No | NA | Hu et al. 2016 |
| p.(Gln392Ter) | Nonsense | M | 4 | No | NR | Global developmental delay | Speech disorder | NR | Yes | NA | Yi et al. 2023 |
| p.(Gln489Lys) | Missense LoF | F | 32 | No | Migraines, tremors, poor balance | No, IQ 131 (WAIS-IV) | Normal | Traits | No | Rathke cleft cyst | Palmer et al., 2023 |
| p.(Glu280Asp) | Missense LoF | M | 8 | Tonic-clonic and focal seizures, associated with fever or not | Hypotonia | Moderate | Sentences | Yes | No | NA | Palmer et al., 2023 |
| p.(Gly269Asp) | Missense LoF | F | 15 | No | Hypotonia, swallowing difficulties requiring gastrostomy | Mild | First words at 30 months | No | Microcephaly, elongated and narrow face, sloping forehead, prominent nose with high nasal bridge, long philtrum, thin upper lip vermillion, micrognathia, high arched palate, dental crowding, and bilateral 5th finger clinodactyly, spina bifida occulta | Mild prominence of the lateral ventricles with septation through the right lateral ventricle at the base of the frontal horn | Palmer et al., 2023 |
| p.(Gly342Arg) | Missense LoF | M | 5 | Focal epilepsy with recurrent status epilepticus | Walking with support | Moderate | 8 words | Yes | No | Small foci of T2-FLAIR hyperintensity, mostly within the bifrontal white matter (prominent perivascular spaces) | Sahly et al. 2024 |
| p.(Gly342Arg) | Missense LoF | M | 2 | Tonic and versive seizures with loss of consciousness, easily triggered by fever, often presenting in clusters | No | Mild | Sentences | No | No | Slightly widening of the left ventricle | Mao et al., 2025 |
| p.(Gly342Glu) | Missense LoF | M | 3 | One focal seizure | Walking at the age of 2 years | Yes, not specified the level | 3 inconstant words | No | No | Normal | Palmer et al., 2023 |
| p.Gly480Arg | Missense LoF | M | 2 | Febrile seizures | Walking | Moderate | Non-verbal (regression) | No | No | Normal | He et al., 2024 |
| p.(Gly484Arg) | Missense LoF | NR | NR | NR | NR | NR | NR | NR | NR | NR | Palmer et al., 2023 |
| p.(Gly526Ser) | Missense LoF | M | 17 | Focal onset seizures with secondary generalized seizures and myoclonic seizures | Severe feeding difficulties | Mild | NR | NR | Microcephaly, hypoplastic upper maxilla and dental malocclusion | NA | Palmer et al., 2023 |
| p.(Gly544Arg) | Missense LoF | F | 24 | Myoclonic, tonic-clonic seizure, atypical absences, and focal impaired awareness seizures | Hypotonia, walking at 18 months | Yes, not specified the level | Non-verbal | Autistic regression | Obesity, short stature, round face, round and flat nasal tip, fleshy lips, and posteriorly rotated ears, small hands and feet | Asymmetry of the mesial temporal lobes and lateral ventricles due to a smaller size of the left hippocampus | Rossi et al., 2023 |
| p.(Gly544Arg) | Missense LoF | M | NR | Early onset epileptic encephalopathy | NR | Severe | NR | NR | NR | NR | Veeramah et al., 2013 |
| p.(Gly544Arg) (mosaic) | Missense LoF | M | NR | Early onset epileptic encephalopathy | NR | Severe | NR | NR | NR | NR | Palmer et al., 2023 |
| p.(Gly545Asp) | Missense LoF | NR | NR | NR | NR | NR | NR | NR | NR | NR | Palmer et al., 2023 |
| p.(Gly545Ser) | Missense LoF | NR | NR | NR | NR | NR | NR | NR | NR | NR | Palmer et al., 2023 |
| p.(Gly731Arg) | Missense LoF | M (3) | NR | No | Hypotonia (3/3) | Severe(1/3) profound (2/3) | NR | No | Strabismus (2/3), scoliosis (3/3) | Cortical atrophy (1/2 studied) | Hu et al. 2016 |
| p.(Gly731Val) | Missense LoF | M | 11 | No | No | No | Mildly delayed | No | Synophrys, straight eyebrows, a high nasal bridge and enophthalmia | Normal | Palmer et al., 2023 |
| p.(Gly78Ser) | Missense LoF |
M (3) | NR | Yes | NA | Moderate | NR | No | No | NA | Hu et al. 2016; Palmer et al., 2023 |
| p.(Ile272Val) | Missense LoF | NR | NR | NR | NR | NR | NR | NR | NR | NR | Palmer et al., 2023 |
| p.(Ile374Thr) | Missense LoF | NR (2) | NR | NR | NR | NR | NR | NR | NR | NR | Palmer et al., 2023 |
| p.(Ile549Leu) | Missense LoF | M | 20 | Myoclonic, atonic epilepsy which evolved to Lennox-Gastaut syndrome | Wheel-chaired | Severe | Non-verbal | No | NR | Normal | Sahly et al., 2024 |
| p.(Ile549Leu) | Missense LoF | NR | NR | NR | NR | NR | NR | NR | NR | NR | Palmer et al., 2023 |
| p.(Leu221Val) | Missense LoF | M (4) | Yes (1/4) | Hypotonia (1/4 studied) | Mild (1/4), profound (2/4), not known (1/4) | No | No | Normal (1 studied) | Hu et al. 2016 | ||
| p.(Leu276Phe) | Missense LoF | M | 3 | Generalized tonic-clonic seizures | central hypotonia and peripheral spasticity, swallowing difficulties with gastrostomy | Profound | Non-verbal | No | Microcephaly, hyperpigmented lesions on the trunk | Severe cerebral and cerebellar atrophy with thinning of the corpus callosum, mild atrophy of bilateral thalami, abnormal cerebral white matter signal and mild bifrontal cerebral collections | Palmer et al., 2023 |
| p.(Leu279Val) | Missense LoF | F | 39 | Atypical and recurrent tonic- clonic convulsions associated with fevers | Generally increased tone and brisk reflexes | Moderate | Regression | Yes | Broad mouth and short philtrum with minimal micrognathia | NA | Palmer et al., 2023 |
| p.(Leu348Val) | Missense LoF | M | 5 | No | Walking, auditory nerve injury | Moderate | Non-verbal (regression) | Yes | Right groin hernia | Normal | He et al., 2024 |
| p.(Lys560Glu) | Missense LoF | NR | NR | NR | NR | NR | NR | NR | NR | NR | Palmer et al., 2023 |
| p.(Lys62Arg) | Missense LoF | M |
14 | Mixed seizure semiology including absences, eye blinking, tonic-clonic seizures, and episodes can be associated with nausea, vomiting and tremor | Balance difficulties | Mild | Delayed | Yes | High palate, macrodontia of the central incisors and restricted extension at the elbows | NA | Palmer et al., 2023 |
| p.(Phe319Ser) | Missense LoF | M | 20 | Drug-resistant epilepsy (tonic-clonic, absence seizures, sometimes provoked by fevers | Hypotonia, walking at 40 months | Moderate | First words at 54 months | Yes | No | Normal | Palmer et al., 2023 |
| p.(Phe319Ser) | Missense LoF | M | 12 | Tonic-clonic seizures often associated with febrile illnesses, absence seizures associated with eyelid myoclonus | Hypotonia, unstable gait | Mild, mainly non-verbal, verbal IQ in the normal range | Speech articulation difficulties | No | No | Normal | Palmer et al., 2023 |
| p.(Pro226Leu) | Missense LoF | M | 20 | Generalized tonic-clonic, absences, sometimes provoked by fevers | Walking at 3.5 years | Moderate | Firs words at 54 months | Yes | No | Normal | Palmer et al., 2023 |
| p.(Pro635Arg) | Missense LoF | F | 8 | Focal and generalized tonic clonic seizures | NA | Moderate | NA | Yes | Hypertelorism with epicanthal folds and full cheeks | Underdevelopment of the sulci in the left frontal region | Palmer et al., 2023 |
| p.(Ser278Arg) | Missense LoF | NR (2) | NR | NR | NR | NR | NR | NR | NR | NR | Palmer et al., 2023 |
| p.(Ser283Asn) | Missense LoF | F | 37 | No | Hypotonia, walking at the age of 2, Progressive spastic diplegia from this age | Moderate | Sentences | Yes | Cushingoid features | NA | Palmer et al., 2023 |
| p.(Thr203Ile) | Missense LoF | M | 3 | No | Hypotonia, walking at 26 months, ataxia | Mild | Some words after the age of 4 | Yes | Microcephaly, mild malar flatness, long philtrum, left-sided single palmar crease | Corpus callosum hypoplasia | Palmer et al., 2023 |
| p.(Thr203Ile) | Missense LoF | F | 11 months | Infantile spasms | Not walking | Yes, not specified the level | Non-verbal | No | No | Delayed myelination | He et al., 2024 |
| p.(Val212Gly) | Missense LoF | M (2) | NR | NR | NR | NR | NR | NR | NR | NR | Hu et al. 2016; Palmer et al., 2023 |
| p.(Val275Leu) | Missense LoF | M | 4 | Absences, infantile spasms, focal seizures | Hypotonia, walking at 4.5 years | Severe | Non-verbal | No | Elongated face, facial hypotonia with an open mouth, full cheeks and micrognathia | Dysgenesis of the corpus callosum | Palmer et al., 2023 |
| p.(Val275Met) | Missense LoF | F | NR | NR | NR | NR | NR | NR | NR | NR | Palmer et al., 2023 |
| p.(Val275Met) | Missense LoF | M | 2 | Infantile spasms, tonic– clonic seizures | No ambulation, hypertonia, left-sided unilateral positive ankle clonus and babinski sig | Moderate | Single words | NR | No | Increased T2 signal in white matter, thin corpus callosum | He et al., 2024 |
| p.(Val275Met) | Missense LoF | M | 6 | Myoclonic, tonic, focal seizures | No ambulation | Severe (regression) | Non-verbal | NR | NA | Enlargement of subarachnoid spaces, dilation of bilateral lateral and third ventricle | He et al., 2024 |
| p.(Val275Met) | Missense LoF | M | 21 months | Tonic, focal, status epilepticus | No ambulation | Severe | Non-verbal | NR | Microcephaly | Dilation of the cerebral ventricles, thin corpus callosum, small hippocampi | He et al., 2024 |
| p.(Val275Met) | Missense LoF | M | 8 | Tonic–clonic, tonic, absence seizures | No ambulation | Profound (regression) | Non-verbal | NR | NA | N | He et al., 2024 |
| p.(Val533Met) | Missense LoF | M | 15 | Focal | NA | Moderate | NA | Traits | No | NA | Palmer et al., 2023 |
| p.(Val536Met) | Missense LoF | M (7), F (2) | NR | Yes (8/8, 1 NR) | Progressive spasticity (2/8, 1 NR) | Mild (4), moderate (2), profound (2) 1 NR |
NR | No | Coarse facial features, broad nasal tip, flat midface, prominent ears (4/4 studied) | NA | Hu et al. 2016, Palmer 2018 |
| p.(Val636Met) | Missense LoF | F | 4 | No | Walking at 23 months | Moderate | Few words | Yes | Microcephaly, down slanting palpebral fissures, telecanthus, small and widely spaced teeth, bilateral clinodactyly of the 5th fingers and slight oedema of the dorsum of the feet | Palmer et al., 2023 | |
| p.(Val92Met) | Missense LoF | F | 9 | No | No | Yes, not specified the level | Delayed | Synophrys and posteriorly rotated ears | No | Palmer et al., 2023 | |
| p.(Arg315His) | Missense GoF | F | 3 | One brief febrile seizure at 34 months | Hypotonia, walking at 21 months | Moderate | Delayed | Yes | No | NA | Palmer et al., 2023 |
| p.(Arg315His) | Missense GoF | NR (2) | NR | NR | NR | NR | NR | NR | NR | NR | Palmer et al., 2023 |
| p.(Arg364Gly) | Missense GoF | M | 27 | Absence seizures, focal seizures, and occasionally secondary generalized seizures | normal | Borderline to mild | Normal | No | No | NA | Palmer et al., 2023 |
| p.(Arg432Gln) | Missense GoF | NR | NR | NR | NR | NR | NR | NR | NR | NR | Palmer et al., 2023 |
| p.(Arg652Thr) | Missense GoF | NR | NR | NR | NR | NR | NR | NR | NR | NR | Palmer et al., 2023 |
| p.(Arg718Gln) | Missense GoF | M (1) NR (2) |
NR | NR | NR | NR | NR | NR | NR | NR | Palmer et al., 2023 |
| p.(Asp29Glu) | Missense GoF | M |
10 | No | Hypotonia, standing with support, few steps with waking frame | Severe | 4 words | Mannerisms/involuntary movements: hand regard, hand, and arm flapping, clapping, smiling, and laughing most of the time | Triangular/trapezoidal face shape, high anterior hairline, prominent forehead, deep-set eyes with straight eyebrows, long/large mouth with thin lips, square, widely spaced teeth | Multiple dilated perivascular spaces, delayed myelination, grey matter heterotopia | Palmer et al., 2023 |
| p.(Asp29Glu) | Missense GoF | M |
2 | Infantile spasms, myoclonic/tonic seizures | Hypotonia, not sitting up unaided, convergent strabismus, nystagmus, | Moderate | Babbling | Mannerisms/involuntary movements: hand regard, hand and arm flapping, clapping, smiling, and laughing most of the time | Triangular/trapezoidal face shape, high anterior hairline, prominent forehead, deep-set eyes with straight eyebrows, long/large mouth with thin lips, square, widely spaced teeth | Normal | Palmer et al., 2023 |
| p.(Asp29Glu) | Missense GoF | F |
NR | NR | NR | Mild | NR | NR | NR | NR | Palmer et al., 2023 |
| p.(Asp34Asn) | Missense GoF (VOUS) | M | 9 | Focal, atonic, and febrile seizures | Walking at 18 months | Yes, not specified the level | Sentences | Yes | Almond shaped eyes, pointed teeth and a short, upturned nose, rhizomelic limb shortening consistent with Desbuquois Dysplasia | Normal | Palmer et al., 2023 |
| p.(Asp621Gly) | Missense GoF | NR | NR | NR | NR | NR | NR | NR | NR | NR | Palmer et al., 2023 |
| p.(Asp89Asn) | Missense GoF | M | 5 | Tonic-clonic seizures | Not sitting independently, no purposeful movements | Profound (regression) | Non-verbal | Yes | Microcephaly and microsomia | complete agenesis of the corpus callosum and cerebellar and brainstem hypoplasia | Sahly et al., 2024 |
| p.(Asp89Asn) | Missense GoF | F | 13 | No | Ataxia | Mild | Delayed | No | Microcephaly, broad nasal root, high palate, fifth finger clinodactyly, pectus carinatum | N | Palmer et al., 2023 |
| p.(Asp89Asn) | Missense GoF | F | 3 | No | Ataxia, hypertonia, not walking until the age of 2 | Mild | Delayed | Microcephaly, right eye internal strabismus and low hairline | Delayed myelination | Li et al., 2023 | |
| p.(Asp89Asn) | Missense GoF | M | Fetus | - | - | - | - | - | Bilateral talipes equino-varus | Agenesis of the corpus callosum, hypoplastic pons, absent septum pellucidum, colpocephaly | Lam et al., 2023 |
| p.(Asp89Asn) | Missense GoF | M | Fetus | - | - | - | - | - | - | Agenesis of the corpus callosum, hypoplastic pons, absent septum pellucidum, colpocephaly | Lam et al., 2023 |
| p.(Ile549Asn) | Missense GoF | F | 7 | No | Walking at the age of 5 | Regression | Regression, single words | Yes | Microcephaly, short palpebral fissures, and a prominent lower lip | Delayed myelination and considerable white matter deficiency with enlarged lateral ventricles and a thin corpus callosum | Palmer et al., 2023 |
| p.(Ile646Thr) | Missense GoF | NR (2) | NR | NR | NR | NR | NR | NR | NR | NR | Palmer et al., 2023 |
| p.(Ile655Val) | Missense GoF | NR | NR | NR | NR | NR | NR | NR | NR | NR | Palmer et al., 2023 |
| p.(Phe238Leu) | Missense GoF | NR | NR | NR | NR | NR | NR | NR | NR | NR | Palmer et al., 2023 |
| p.(Phe268Leu) | Missense GoF | F | 8 | No | Hypotonia, walking at the age of 2, swallowing difficulties | Mild-moderate | Delayed | Yes | Microcephaly | Lipoma, dysgenesis of the corpus callosum | Palmer et al., 2023 |
| p.(Pro310Ser) | Missense GoF | F | 7 | Yes | Hypotonia, walking at the age of 2 | Yes, not specified the level | 20 words | No | Microcephaly with ridged metopic suture, strabismus, left-sided ptosis, slight ocular telecanthus, lateral fullness of the nose, mild bilateral limited elbow extension, transverse palmer crease | Normal | Palmer et al., 2023 |
| p.(Pro369Leu) | Missense GoF | M | 13 | Yes | Walking at 48 months, wide-based gait, sensorineural hearing loss, optic nerve hypoplasia, swallowing difficulties with gastrostomy feeding | Moderate | Sentences | Yes | Diaphragmatic hernia, left lung hypoplasia, bilateral cryptorchidism, full cheeks, long philtrum, micrognathia, wide nasal bridge, anteverted nares, frontal bossing, exaggerated cupid’s bow, downturned corners of mouth, high anterior hairline, prominent forehead, lagophthalmos, hypertelorism, preauricular pit, posteriorly rotated ears | NA | Palmer et al., 2023 |
| p.(Ser105Cys) | Missense GoF | NR (2) | NR | NR | NR | NR | NR | NR | NR | NR | Palmer et al., 2023 |
| p.(Ser395Arg) | Missense GoF | F | 13 | No | Walking at 24 months | Mild | Sentences | No | No | Arachnoid cyst | Palmer et al., 2023 |
| p.(Ser69Leu) | Missense GoF (VOUS) | M | 5 | No | 7th cranial nerve palsy, severe myopia, mild ataxia | Severe | Delayed | No | Hypospadias, long eyelashes, synophrys, thick eyebrows, low set ears, short but normal fifth digit phalanges in hands and feet, second toe longer than the hallux on the left side | Normal | Palmer et al., 2023 |
| p.(Thr629Ile) | Missense GoF | F | 14 | No | Walking at the age of 3 | Moderate-severe | Delayed, short sentences | No | Long face and arched eyebrows, 2-vessel umbilical cord at prenatal ultrasound | Small remote lacunar infarct and dilated perivascular space, mild prominence of fourth ventricle | Palmer et al., 2023 |
| p.(Val317Ile) | Missense GoF | M | 5 | No | Hypotonia, walking at 5 years | Severe | Non-verbal | Yes | Mildly flat face, everted lower lip, anteverted nares | Partial corpus callosum agenesis (posterior part of the corpus and splenium) with colpocephaly, and mild third ventricle dilation | Palmer et al., 2023 |
| p.(Val317Ile) | Missense GoF | M | 13 | Focal onset frontal lobe hypermotoric epilepsy | Hypotonia, walking at 3.5 years, bilateral optic nerve hypoplasia, strabismus | Moderate | Delayed, first words at the age of 2 | No | Elevated finger pads, fifth finger clinodactyly, right preauricular skin tag | Mildly small optic chiasm and optic nerves bilaterally suggestive of optic hypoplasia as well as a dysplastic corpus callosum | Palmer et al., 2023 |
| p.(Val317Ile) | Missense GoF | M | 18 | No | Hypotonia, walking at the age of 3 years, bilateral optic atrophy, swallowing difficulties, gastrostomy feeding | Severe | Delayed, first words at the age of 4 | No | Bilateral ptosis, widely spaced teeth, slightly simple ears, malar flatness with long face and pointed chin | Bilateral optic atrophy, hypoplasia of the corpus callosum, prominent subarachnoid spaces | Palmer et al., 2023 |
| p.(Val317Ile) | Missense GoF | M | 6 | No | Hypotonia, feeding difficulties | Severe | First words at 15-18 months, unclear and slow rate speech, limited vocabulary | Traits | NR | Agenesis corpus callosum and hypoplasia of cerebellar vermis | Garrett et al., 2025 |
| p.(Val317Ile) | Missense GoF | M | 6 | Generalized/myoclonic mainly during febrile episodes or intercurrent illnesses | Hypotonia, walking at the age of 20 months, swallowing difficulties for liquids | Moderate-to-severe | Delayed, first word wat the age of 2; At the age of 6 years two-words associations | No | No | White matter hyperintensities, temporal horns dysmorphism with hippocampal eversion, dysmorphic corpus callosum and thin optic chiasma. | This study |
| p.(Val317Phe) | Missense GoF | NR | NR | NR | NR | NR | NR | NR | NR | NR | Palmer et al., 2023 |
| p.(Val550Leu) | Missense GoF | F | 20 | No | Walking at 18 months | Moderate | Mixed receptive-expressive language disorder | Traits | Round face, deep-set eyes, bulbous nose with small nares, partial cutaneous syndactyly of the 2nd and 3rd toes, joint hypermobility | NA | Palmer et al., 2023 |
| p.(Asn309Profs) | Truncating | M | 52 | No | No | Mild-moderate | Delayed | Yes | Prominent ears with simple helix, prominent nose |
NA | Palmer et al., 2023 |
| p.(Gln663Glyfs) | Truncating | M | 18 | Generalized seizures | No | Mild-moderate | Delayed | No | Microcephaly, long face, straight eyebrow, thin lips |
NA | Palmer et al., 2023 |
| p.(Tyr675Ter) | Truncating | M | 13 | Febrile seizure, tonic-clonic seizures | Hypotonia, walking at 18 months |
Severe | Severely delayed, few words | No | No | Bilateral mesial temporal sclerosis, asymmetric, right side more affected |
Palmer et al., 2023 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




