Submitted:
20 April 2026
Posted:
22 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Methods
2.1. Research Questions
2.2. Eligibility Criteria
2.3. Information Sources and Search Strategy
2.4. Selection of Sources of Evidence
2.5. Data Charting
2.6. Synthesis and Algorithm Development
3. Results
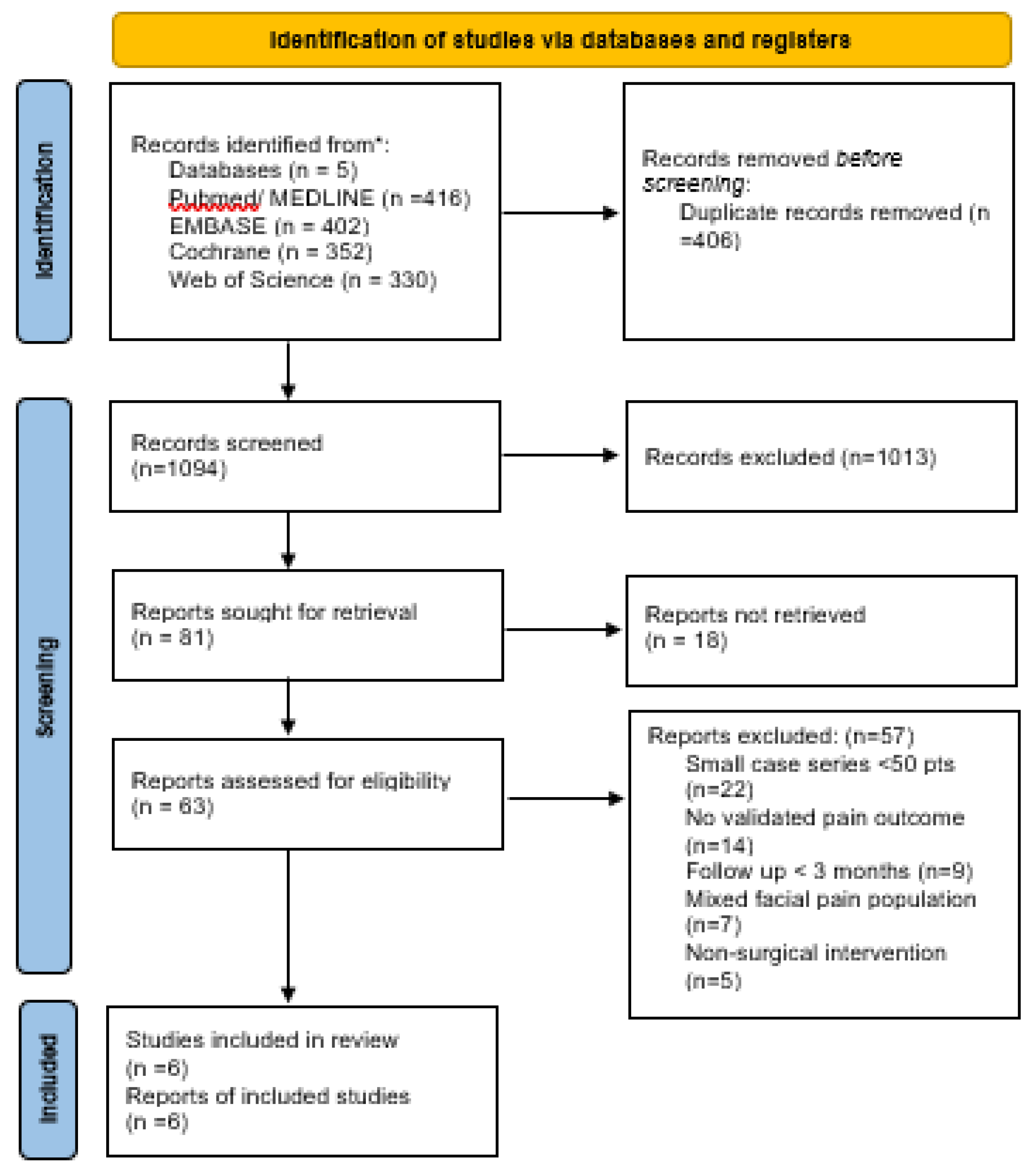
3.1. Study Selection
3.2. Characteristics of Included Studies
3.3. Synthesis of Evidence by Procedure
3.3.1. Microvascular Decompression
3.3.2. Stereotactic Radiosurgery
3.3.3. Percutaneous Ablative Techniques
3.3.4. Peripheral Nerve Stimulation and Neuromodulatory Options
4. Clinical Decision Algorithm
5. Discussion
5.1. Principal Findings
5.2. Evidence Gaps
5.3. Comparison with Existing Literature
5.4. Limitations
6. Conclusions
Supplementary Materials
Funding
References
- Headache Classification Committee of the International Headache Society (IHS). The International Classification of Headache Disorders, 3rd edition. Cephalalgia. 2018;38(1):1–211. [CrossRef]
- Cruccu G, Di Stefano G, Truini A. Trigeminal neuralgia. N Engl J Med. 2020;383(8):754–762. [CrossRef]
- Zakrzewska JM, Wu J, Mon-Williams M, Phillips N, Pavitt SH. Evaluating the impact of trigeminal neuralgia. Pain. 2017;158(6):1166–1174. [CrossRef]
- Trigeminal neuralgia. Nat Rev Dis Primers. 2024;10:40. [CrossRef]
- Bendtsen L, Zakrzewska JM, Abbott J, Braschinsky M, Di Stefano G, Donnet A, et al. European Academy of Neurology guideline on trigeminal neuralgia. Eur J Neurol. 2019;26(6):831–849. [CrossRef]
- Tricco AC, Lillie E, Zarin W, O’Brien KK, Colquhoun H, Levac D, et al. PRISMA Extension for Scoping Reviews (PRISMA-ScR): checklist and explanation. Ann Intern Med. 2018;169(7):467–473. [CrossRef]
- Peters MDJ, Marnie C, Tricco AC, Pollock D, Munn Z, Alexander L, et al. Updated methodological guidance for the conduct of scoping reviews. JBI Evid Synth. 2020;18(10):2119–2126. [CrossRef]
- Montano N, Papacci F, Cioni B, Di Bonaventura R, Meglio M. What is the best treatment of drug-resistant trigeminal neuralgia in patients affected by multiple sclerosis? A literature analysis of surgical procedures. Clin Neurol Neurosurg. 2013;115(5):567–572. [CrossRef]
- Liu H, Hou H, Li F, Zheng R, Zhang Y, Cheng J, Han S. Structural and functional brain changes in patients with classic trigeminal neuralgia: a combination of voxel-based morphometry and resting-state functional MRI study. Front Neurosci. 2022;16:930765. [CrossRef]
- Rapisarda A, Battistelli M, Izzo A, D’Ercole M, D’Alessandris QG, Polli FM, Santi S, Martinelli R, Montano N. Outcome comparison of drug-resistant trigeminal neuralgia surgical treatments — an umbrella review of meta-analyses and systematic reviews. Brain Sci. 2023;13(4):530. [CrossRef]
- Gomes-da Silva de Rosenzweig P, Pastrana-Brandes S, Merikansky-Gerson S, Victoria-Garcia LO, Curtius-Caruso MS, Carrillo-Ruiz JD. Factors associated with outcomes following microvascular decompression for the treatment of primary trigeminal neuralgia in adults: a systematic review and meta-analysis. J Dent Anesth Pain Med. 2024;24(4):227–243. [CrossRef]
- Régis J, Tuleasca C, Resseguier N, Carron R, Donnet A, Yomo S, Gaudart J, Levivier M. The very long-term outcome of radiosurgery for classical trigeminal neuralgia. Stereotact Funct Neurosurg. 2016;94(1):24–32. [CrossRef]
- Di Carlo DT, Benedetto N, Perrini P. Clinical outcome after microvascular decompression for trigeminal neuralgia: a systematic review and meta-analysis. Neurosurg Rev. 2022;46(1):8. [CrossRef]
- Chang KW, Jung HH, Chang JW. Percutaneous procedures for trigeminal neuralgia. J Korean Neurosurg Soc. 2022;65(5):622–632. [CrossRef]
- Latif U, Moghim R, Valimahomed A, Lam CM, Abd-Elsayed A, Gulati A, et al. Consensus guidelines for the use of peripheral nerve stimulation in the treatment of chronic pain and neurological diseases: a Neuron Project from the American Society of Pain and Neuroscience. J Pain Res. 2025;18:5949–5990. [CrossRef]

| PCC Element | Component | Inclusion / Exclusion Criteria |
| Population | Adults with ICHD-3 TN | Adults (≥18 years) with confirmed TN per ICHD-3 2018 criteria [1], encompassing classical, secondary, and idiopathic subtypes. Mixed facial pain populations eligible only where TN-specific data were reported separately. |
| Concept | Surgical or neuromodulatory intervention | Any surgical procedure (MVD; radiofrequency thermocoagulation; percutaneous balloon compression; glycerol rhizolysis; stereotactic radiosurgery) or neuromodulatory intervention (peripheral nerve stimulation; spinal cord stimulation; motor cortex stimulation; deep brain stimulation). Pharmacological-only studies excluded. |
| Context | Any clinical setting; January 2016 – January 2026 | No restriction on healthcare setting or country. English-language publications. Minimum follow-up ≥3 months. Minimum n ≥50 for primary studies; no minimum for systematic reviews and meta-analyses. |
| Study designs | Eligible designs | Systematic reviews and meta-analyses (OCEBM Level I–II); large prospective or retrospective series (n≥50; OCEBM Level III); evidence-based consensus guidelines. Editorials, case reports, conference abstracts, and non-English publications excluded. |
| First Author (Year) [Ref] | Country | Design | Population | Intervention | Outcome Measure | Inclusion Rationale |
| Rapisarda et al. (2023) [10] | Italy | Umbrella review of systematic reviews and meta-analyses | All surgical TN procedures; all ICHD-3 subtypes; 10 meta-analyses included | MVD, RF, PBC, PGR, SRS (all modalities compared) | BNI; VAS; procedure-specific pain-free rates | Sole study directly comparing all five surgical modalities within a single umbrella analysis. Highest-level synthesis of the available TN surgical evidence base. Provides cross-procedure outcome comparisons anchoring Steps 4–5 of the decision algorithm. OCEBM Level I–II. |
| Rosenzweig et al. (2024) [11] | Mexico / Multi-national | Systematic review and meta-analysis | MVD for primary TN; adults; multiple centres | Microvascular decompression — outcome predictors | BNI; multivariable predictor analysis | Systematic review and meta-analysis of factors associated with MVD outcomes. Identifies MRI-confirmed arterial neurovascular conflict as the principal independent predictor of surgical success. Directly anchors Step 3 (NVC assessment) of the decision algorithm. OCEBM Level I–II. |
| Régis et al. (2016) [12] | France | Prospective cohort series | n=497; classical TN; Gamma Knife SRS; up to 15-year follow-up | Gamma Knife stereotactic radiosurgery | BNI; 15-year outcomes | Largest and longest prospective SRS series in the literature. Provides unique 15-year outcome data and quantifies repeat SRS neuropathy risk. Anchors Step 5 (SRS pathway). OCEBM Level III. |
| Di Carlo et al. (2022) [13] | Italy | Systematic review and meta-analysis | Multiple series; classical TN; MVD; multi-centre | Microvascular decompression: clinical outcomes | BNI; VAS; complication rate; recurrence | Systematic review and meta-analysis providing pooled MVD outcome data: pain-free rates, recurrence, and complication profile. Provides the complementary MVD evidence base alongside Rosenzweig et al. [11], focused on clinical outcomes rather than predictors. OCEBM Level I–II. |
| Chang et al. (2022) [14] | South Korea | Narrative and evidence synthesis review | Adults with TN; RF, PBC, and PGR; multiple international series | Percutaneous procedures: RF thermocoagulation, balloon compression, glycerol rhizolysis | Pain-free rate; recurrence; complication profile | The only included source to provide a comprehensive comparative synthesis of all three percutaneous Gasserian ganglion procedures (RF, PBC, PGR), addressing candidate selection, technique, outcomes, and complications. Fills the critical evidence gap for percutaneous techniques. Anchors Step 5 (non-MVD sub-profile selection) of the decision algorithm. OCEBM Level III. |
| Latif et al. (2025) [15] | USA / Multi-national | Evidence-based consensus guidelines | Chronic pain and neurological conditions; PNS; multiple indications including TN | Peripheral nerve stimulation: indications, technique, and outcomes | Responder rate (≥50% pain reduction); complication profile | Neuron Project consensus guidelines (American Society of Pain and Neuroscience) on PNS use in chronic pain and neurological disease. Provides the evidence base and minimum dataset for PNS in TN, anchoring Step 7 (neuromodulation hierarchy) of the decision algorithm. OCEBM Level I–II (consensus guideline). |
| Procedure | Included Study | Initial Pain-Free / Responder Rate (%) | Recurrence | Principal Complication | Key Evidence Statement |
| Microvascular decompression | Di Carlo et al. [13]; Rosenzweig et al. [11]; Rapisarda et al. [10] | 85–96% [10,13] | 15–25% at 5 years [13] | Mortality 0.1–0.5%; hearing loss 1–3%; CSF leak 1–3% [13] | Preferred surgical option for classical TN with MRI-confirmed arterial NVC in surgically fit patients. Arterial NVC is the single strongest independent predictor of MVD success [11]. OCEBM Level I–II. |
| Gamma Knife SRS | Régis et al. [12]; Rapisarda et al. [10] | 65–85% [10,12] | 43% at 3 years; late recurrence common [12] | Delayed facial numbness 6–13%; onset 1–3 months [12] | Most favourable safety profile of any TN procedure [10,12]. Preferred for absent/venous NVC, MS-TN, high operative risk, anticoagulated patients. Repeat SRS: neuropathy risk ≥20% [12]. OCEBM Level I–II (Rapisarda et al. [[10]); Level III (Régis et al. [[12]). |
| RF thermocoagulation | Chang et al. [14]; Rapisarda et al. [10] | 85–95% [10,14] | 25–45% at 5 years [14] | Dysaesthesia 6–24%; anaesthesia dolorosa <2% [14] | Division-specific targeting; effective for V2–V3; re-intervention candidate after SRS failure. No head-to-head RCT data against MVD or SRS [14]. OCEBM Level III. |
| Percutaneous balloon compression | Chang et al. [14]; Rapisarda et al. [10] | 75–95% [10,14] | 25–50% at 5 years [14] | Transient masseter weakness 10–66%; corneal reflex preserved [14] | Preferred for V1 involvement where corneal reflex preservation is required. Requires brief general anaesthesia. Short-stay procedure [14]. OCEBM Level III. |
| Glycerol rhizolysis | Chang et al. [14]; Rapisarda et al. [10] | 70–90% [10,14] | 30–50% at 5 years [14] | Mild sensory loss; lowest deafferentation risk of percutaneous procedures [14] | Preferred in anticoagulated patients and MS-TN where sensory preservation is paramount. Lowest deafferentation risk among percutaneous options [14]. OCEBM Level III. |
| Peripheral nerve stimulation (PNS) | Latif et al. [15]; Rapisarda et al. [10] | ~60–70% responders (≥50% pain reduction) [15] | Variable; device-dependent [15] | Lead migration 8–15%; infection 2–4% [15] | Supported by ASPN consensus guidelines as a reversible neuromodulatory option after failure of surgical/radiosurgical procedures. Supraorbital and infraorbital targets for V1–V2 TN [15]. OCEBM Level I–II (guideline). |
| Step | Clinical Question | YES → Action | NO → Redirect |
| 1 | ICHD-3 criteria confirmed? [1] | Confirmed TN. Proceed to Step 2. | Exclude mimics: PIFP, dental pain, cluster headache, paroxysmal hemicrania, SUNCT. Do NOT proceed to surgical pathway. |
| 2 | Pharmacotherapy failed or not tolerated? (EAN guideline: CBZ ≥800 mg or OXC ≥1,200 mg, ≥3 months) [5] | Drug-refractory TN confirmed. Proceed to Step 3. | Optimise and titrate pharmacotherapy per EAN guideline [5]. Neurology review. Re-assess at 3 months. |
| 3 | 3T MRI (CISS/FIESTA): arterial NVC confirmed? [11] | Arterial NVC confirmed. Proceed to Step 4 — MVD pathway. NVC grade (Sindou I–III) informs prognosis. | Absent or venous NVC. MVD not indicated per [11]. Proceed to Step 5 — non-MVD pathway. |
| 4 | Fit for retrosigmoid craniotomy? (ASA I–III, age typically <75, no severe comorbidity) [13] | → OFFER MVD. Initial pain-free 85–96% [10,13]. 5-yr recurrence 15–25% [13]. Lowest long-term recurrence of any procedure. | High operative risk or patient declines craniotomy. Proceed to Step 5. |
| 5 | Clinical sub-profile for non-MVD selection? [10,12,14] | V1/corneal reflex concern → PBC [14]. Anticoagulated / MS-TN / no-NVC → PGR or Gamma Knife SRS [5,10,14]. Default non-MVD → Gamma Knife SRS [10,12]. | If sub-profile unclear, Gamma Knife SRS is the evidence-based default (most favourable safety profile) [10,12]. |
| 6 | Failed prior surgical procedure? [10,13,14] | Failed MVD → mandatory post-MVD MRI; then SRS or RF [10,13]. Failed SRS → RF thermocoagulation [14]. Failed percutaneous → repeat same or switch modality per clinical profile [14]. | Reassess from Step 3. Confirm technical adequacy of prior procedure and recurrence mechanism. |
| 7 | Multiple procedure failures or deafferentation pain? MDT review complete? | Neuromodulation hierarchy (MDT supervision): PNS (supraorbital/infraorbital) [15] → cervical SCS (C1–C2) → MCS/DBS at specialist centre. Avoid further ablative surgery. | Reassess diagnosis. Exclude central sensitisation and deafferentation pain. MDT review mandatory before any further intervention. |
| Evidence Gap | Description | Research Priority |
| Absence of RCT data comparing any surgical modalities | Across all six included studies, no RCT directly comparing two or more surgical TN procedures was identified within the 2016–2026 search window. The only published prospective comparative study (Pollock & Ecker, 2005; J Neurosurg) was excluded as it predates the search window, predates ICHD-3, and is underpowered (n=42), yet remains the highest-quality direct comparison available. | Multicentre RCT comparing MVD, Gamma Knife SRS, and at least one percutaneous technique in ICHD-3 classical TN with confirmed arterial NVC, stratified by Sindou grade, with co-primary endpoints of BNI pain-free rate and EQ-5D QoL at 1, 3, 5, and 10 years. |
| Outcome measurement heterogeneity | Three distinct pain scales (BNI, VAS, NRS) were used across the six included studies, preventing precise cross-procedure comparisons. Only Di Carlo et al. [13] and Rapisarda et al. [10] used the BNI consistently. | Adoption of BNI as the universal mandatory outcome measure in all future TN surgical trials, reported at standardised time points (6 months, 1, 3, 5, and 10 years), alongside validated QoL instruments (EQ-5D-5L or SF-36). |
| Absence of brain-level QoL and neuroplasticity outcome data | Liu et al. (2022) [9] demonstrated significant structural and functional brain changes in classical TN, yet none of the six included surgical studies reported neuroimaging or neuroplasticity outcomes post-intervention. Psychosocial burden and suicidality data were not captured in any included study [3]. | Mandatory co-primary QoL reporting (EQ-5D, PHQ-9 for depression, suicidality screening) in all future TN surgical trials. Prospective neuroimaging sub-studies to assess reversal of maladaptive cortical reorganisation following successful surgical decompression. |
| Limited long-term neuromodulation outcome data | Latif et al. [15] provides ASPN consensus guidance for PNS but the evidence base comprises predominantly short-to-medium-term series. No included study provided controlled neuromodulation data beyond 36 months, and MCS and DBS outcome data in TN were not represented in the included study set. | Prospective multicentre neuromodulation registry with a mandatory minimum dataset: device type, stimulation parameters, validated pain (BNI/NRS) and QoL outcomes, hardware complications, collected at 6, 12, 24, 36, and 60 months. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




