Submitted:
20 April 2026
Posted:
22 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. New Combinations: Promise and Disappointment in the Adjuvant Setting
2.1. ICIs Combinations
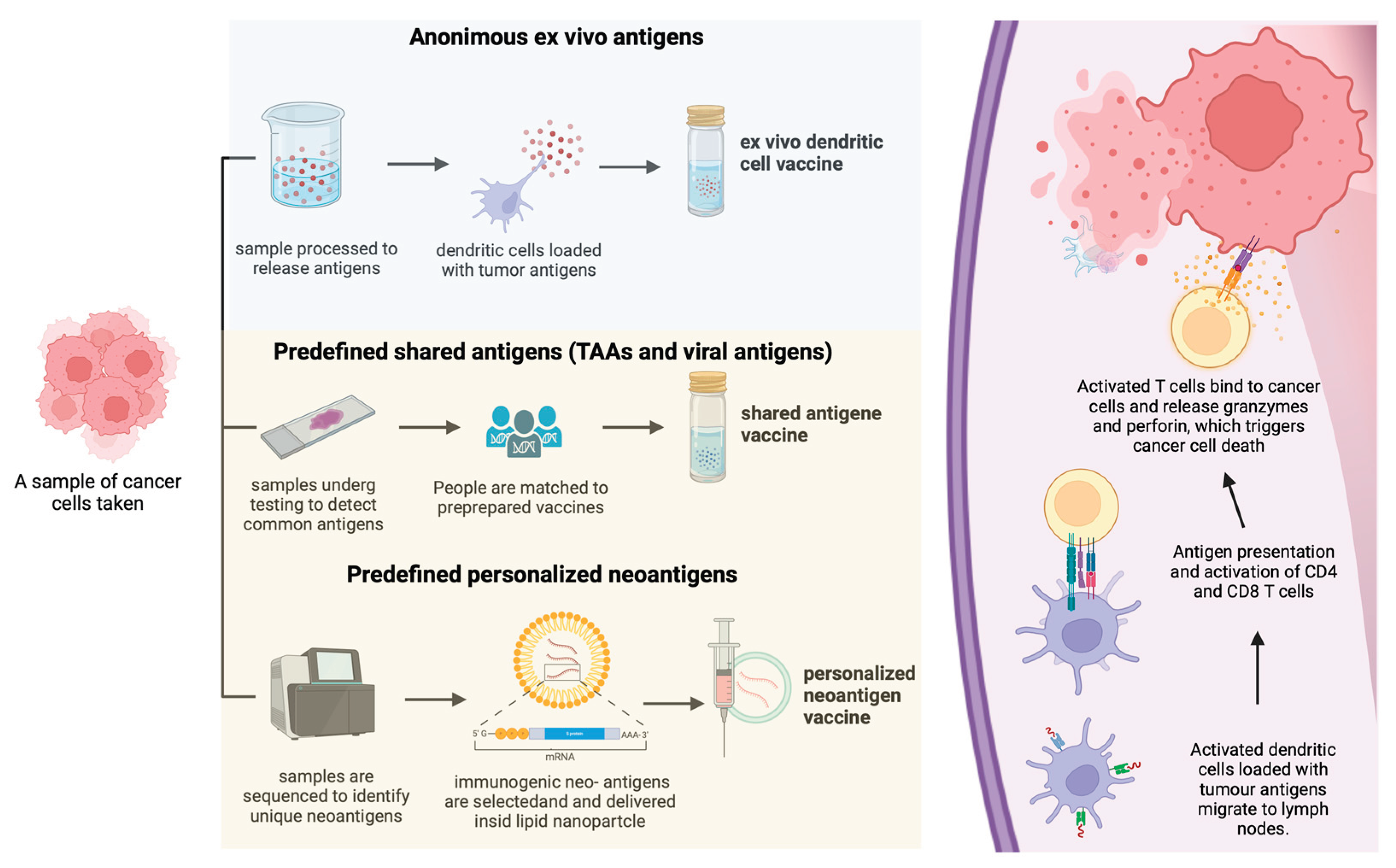
2.2. Cancer Vaccines: From Concept to Clinic
3. Open Issues in Neoadjuvant Therapy
4. Open Issues and Controversies
4.1. Adjuvant Treatment of Stage IIIA Melanoma
4.2. Overall Survival and Recurrence-Free Survival in Adjuvant Trials
4.3. Retreatment at Relapse After Adjuvant Therapy
5. Translational Data
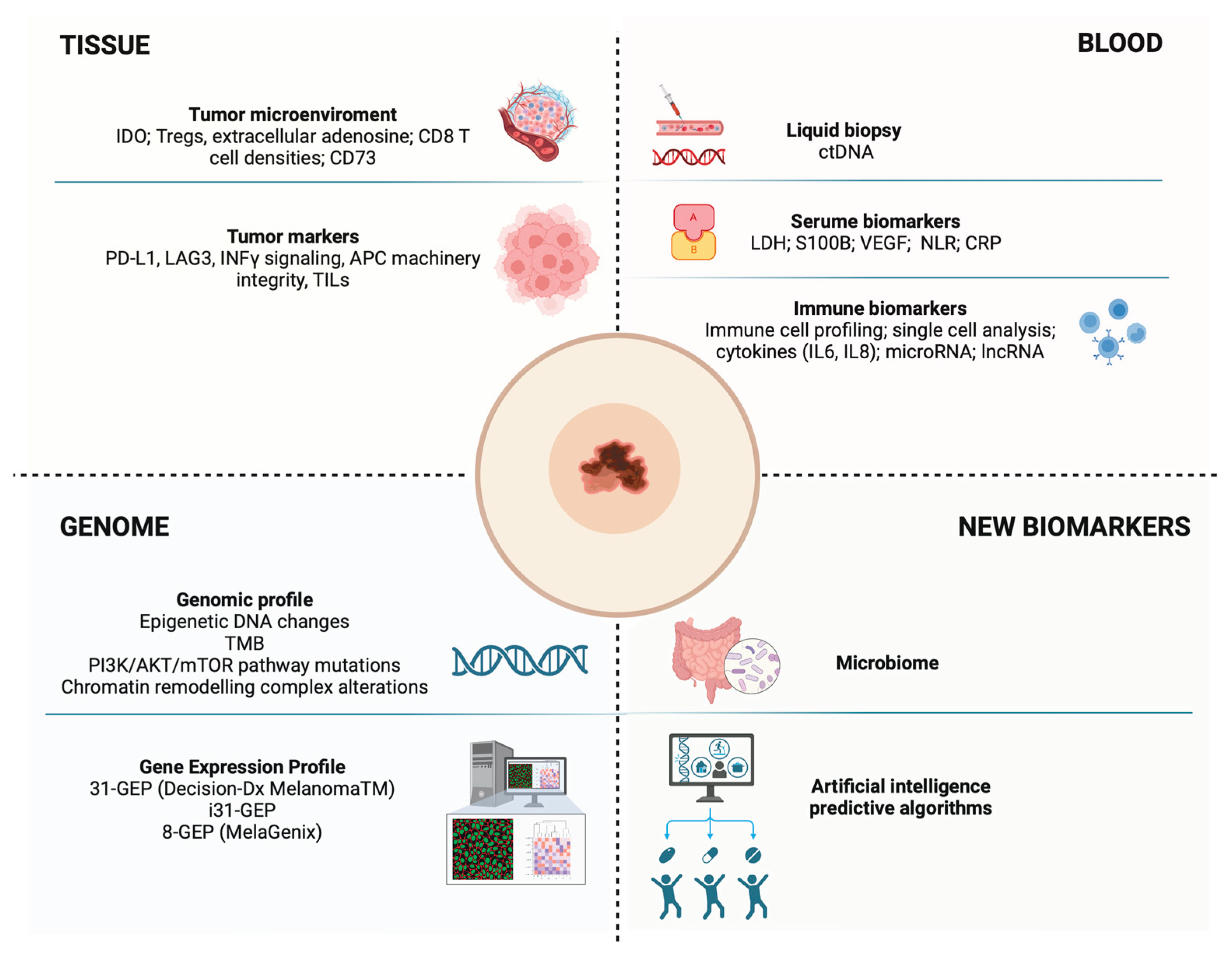
5.1. Biomarkers in Adjuvant Therapy
5.2. Improve Prognostication of Stage II Melanoma: Focus on Gene Expression Profile (GEP)
6. Conclusions
Funding
Acknowledgments
References
- Hodi, F. S.; et al. ‘Long-term survival in advanced melanoma for patients treated with nivolumab plus ipilimumab in CheckMate 067.’. https://doi.org/10.1200/JCO.2022.40.16_suppl.9522. 2022, vol. 40(no. 16_) suppl, pp. 9522. [Google Scholar] [CrossRef]
- Weber, J. S.; et al. ‘Adjuvant Therapy of Nivolumab Combined With Ipilimumab Versus Nivolumab Alone in Patients With Resected Stage IIIB-D or Stage IV Melanoma (CheckMate 915)’. J. Clin. Oncol. 2023, vol. 41(no. 3), 517–527. [Google Scholar] [CrossRef]
- Livingstone, E.; et al. ‘Adjuvant nivolumab plus ipilimumab or nivolumab alone versus placebo in patients with resected stage IV melanoma with no evidence of disease (IMMUNED): Final results of a randomised, double-blind, phase 2 trial’. The Lancet 2022, vol. 400(no. 10358), 1117–1129. [Google Scholar] [CrossRef]
- Long, G. V.; et al. ‘Adjuvant nivolumab and relatlimab in stage III/IV melanoma: The randomized phase 3 RELATIVITY-098 trial’. Nature Medicine 2025, 2025 31:12, vol. 31(no. 12), 4301–4309. [Google Scholar] [CrossRef]
- Dummer, R.; et al. ‘Vibostolimab coformulated with pembrolizumab versus pembrolizumab alone as adjuvant therapy for high-risk stage IIB–IV melanoma (KEYVIBE-010): A randomised, double-blind, phase 3 study’. Lancet Oncol. 2026, vol. 27(no. 3), 327–339. [Google Scholar] [CrossRef]
- Peng, M.; et al. ‘Neoantigen vaccine: An emerging tumor immunotherapy’. Mol. Cancer 2019, vol. 18(no. 1). [Google Scholar] [CrossRef] [PubMed]
- Kruit, W. H. J.; et al. ‘Selection of immunostimulant AS15 for active immunization with MAGE-A3 protein: Results of a randomized phase II study of the European Organisation for Research and Treatment of Cancer Melanoma Group in Metastatic Melanoma’. J. Clin. Oncol. 2013, vol. 31(no. 19), 2413–2420. [Google Scholar] [CrossRef] [PubMed]
- Dreno, B.; et al. ‘MAGE-A3 immunotherapeutic as adjuvant therapy for patients with resected, MAGE-A3-positive, stage III melanoma (DERMA): A double-blind, randomised, placebo-controlled, phase 3 trial’. Lancet Oncol. 2018, vol. 19(no. 7), 916–929. [Google Scholar] [CrossRef] [PubMed]
- Schwartzentruber, D. J.; et al. ‘gp100 Peptide Vaccine and Interleukin-2 in Patients with Advanced Melanoma’. New England Journal of Medicine 2011, vol. 364(no. 22), 2119–2127. [Google Scholar] [CrossRef]
- Hu, Z.; et al. ‘Personal neoantigen vaccines induce persistent memory T cell responses and epitope spreading in patients with melanoma’. Nature Medicine 2021, 2021 27:3, vol. 27(no. 3), 515–525. [Google Scholar] [CrossRef]
- Sahin, U.; et al. ‘Personalized RNA mutanome vaccines mobilize poly-specific therapeutic immunity against cancer’. Nature 2017, 2017 547:7662, vol. 547(no. 7662), 222–226. [Google Scholar] [CrossRef] [PubMed]
- Weber, J. S.; et al. ‘Individualized neoantigen therapy mRNA-4157 (V940) plus pembrolizumab in resected melanoma: 3-year update from the mRNA-4157-P201 (KEYNOTE-942) trial.’. Journal of Clinical Oncology 2024, vol. 42(no. 17_) suppl, LBA9512–LBA9512. [Google Scholar] [CrossRef]
- Therien, A. D.; Chime-Eze, C. M.; Rhodin, K. E.; Beasley, G. M. ‘Neoadjuvant therapy for melanoma: Past, present, and future’. Surg. Oncol. 2024, vol. 56, 102127. [Google Scholar] [CrossRef] [PubMed]
- Topalian, S. L.; Taube, J. M.; Pardoll, D. M. ‘Neoadjuvant checkpoint blockade for cancer immunotherapy’, Science (1979). 2020, vol. 367(no. 6477). [Google Scholar] [CrossRef]
- Blank, C. U.; et al. ‘Neoadjuvant Nivolumab and Ipilimumab in Resectable Stage III Melanoma’. New England Journal of Medicine 2024, vol. 391(no. 18), 1696–1708. [Google Scholar] [CrossRef] [PubMed]
- Hoeijmakers, L. L.; et al. ‘Neoadjuvant ipilimumab plus nivolumab in melanoma: 5-year survival and biomarker analysis from the phase 2 PRADO-trial’. Nature Medicine 2026, 2026 32:3, vol. 32(no. 3), 952–963. [Google Scholar] [CrossRef]
- Lucas, M. W.; et al. ‘LBA57 Two-year clinical update and first biomarker analyses of the phase III NADINA trial comparing neoadjuvant nivolumab plus ipilimumab versus adjuvant nivolumab in resectable stage III melanoma’. Annals of Oncology 2025, vol. 36, S1600–S1601. [Google Scholar] [CrossRef]
- Rozeman, E. A.; et al. ‘Identification of the optimal combination dosing schedule of neoadjuvant ipilimumab plus nivolumab in macroscopic stage III melanoma (OpACIN-neo): A multicentre, phase 2, randomised, controlled trial’. Lancet Oncol. 2019, vol. 20(no. 7), 948–960. [Google Scholar] [CrossRef]
- Reijers, I. L. M.; et al. ‘Personalized response-directed surgery and adjuvant therapy after neoadjuvant ipilimumab and nivolumab in high-risk stage III melanoma: The PRADO trial’. Nature Medicine 2022, 2022 28:6, vol. 28(no. 6), 1178–1188. [Google Scholar] [CrossRef] [PubMed]
- Robert, C.; et al. ‘Open-label, phase II study of talimogene laherparepvec plus pembrolizumab for the treatment of advanced melanoma that progressed on prior anti–PD-1 therapy: MASTERKEY-115’. Eur. J. Cancer 2024, vol. 207. [Google Scholar] [CrossRef]
- Kähler, K. C.; et al. ‘Neoadjuvant intralesional targeted immunocytokines (daromun) in stage III melanoma’. Annals of Oncology 2025, vol. 36(no. 10), 1166–1177. [Google Scholar] [CrossRef] [PubMed]
- Gershenwald, J.E.; Scolyer, R.A.; Hess, K.R.; Sondak, V.K.; Long, G.V.; Ross, M.I.; Lazar, A.J.; Faries, M.B.; Kirkwood, J.M.; McArthur, G.A.; Haydu, L.E.; Eggermont, A.M.M.; Flaherty, K.T.; Balch, C.M.; Thompson, J.F. Melanoma staging: Evidence-based changes in the American Joint Committee on Cancer eighth edition cancer staging manual. CA: A Cancer Journal for Clinicians 2017, 67, 472–492. [Google Scholar] [CrossRef]
- Dewar, D. J.; Newell, B.; Green, M. A.; Topping, A. P.; Powell, B. W. E. M.; Cook, M. G. ‘The microanatomic location of metastatic melanoma in sentinel lymph nodes predicts nonsentinel lymph node involvement’. J. Clin. Oncol. 2004, vol. 22(no. 16), 3345–3349. [Google Scholar] [CrossRef]
- Kretschmer, L.; et al. ‘The sentinel node invasion level (SNIL) as a prognostic parameter in melanoma’. Mod. Pathol. 2021, vol. 34(no. 10), 1839–1849. [Google Scholar] [CrossRef] [PubMed]
- Satzger, I.; Leiter, U.; Gräger, N.; Keim, U.; Garbe, C.; Gutzmer, R. ‘Melanoma-specific survival in patients with positive sentinel lymph nodes: Relevance of sentinel tumor burden’. Eur. J. Cancer 2019, vol. 123, 83–91. [Google Scholar] [CrossRef]
- Amaral, T.; et al. ‘Isolated melanoma cells in sentinel lymph node in stage IIIA melanoma correlate with a favorable prognosis similar to stage IB’. Eur. J. Cancer 2024, vol. 201, 113912. [Google Scholar] [CrossRef]
- Moncrieff, M. D.; et al. ‘Clinical Outcomes and Risk Stratification of Early-Stage Melanoma Micrometastases from an International Multicenter Study: Implications for the Management of American Joint Committee on Cancer IIIA Disease’. Journal of Clinical Oncology 2022, vol. 40(no. 34), 3940–3951. [Google Scholar] [CrossRef]
- Grover, P.; et al. ‘Efficacy of adjuvant therapy in patients (pts) with AJCC v8 stage IIIA cutaneous melanoma.’. https://doi.org/10.1200/JCO.2023.41.16_suppl.9518. 2023, vol. 41(no. 16_) suppl, 9518–9518. [Google Scholar] [CrossRef]
- Stassen, R. C.; et al. ‘Development and validation of a novel model to predict recurrence-free survival and melanoma-specific survival after sentinel lymph node biopsy in patients with melanoma: An international, retrospective, multicentre analysis’. Lancet Oncol. 2024, vol. 25(no. 4), 509–517. [Google Scholar] [CrossRef]
- Suciu, S.; et al. ‘Relapse-Free Survival as a Surrogate for Overall Survival in the Evaluation of Stage II-III Melanoma Adjuvant Therapy’. J. Natl. Cancer Inst. 2018, vol. 110(no. 1), 87–96. [Google Scholar] [CrossRef] [PubMed]
- Eggermont, M. M.; et al. ‘Prolonged Survival in Stage III Melanoma with Ipilimumab Adjuvant Therapy’. N. Engl. J. Med. 2016, vol. 375(no. 19), 1845–1855. [Google Scholar] [CrossRef] [PubMed]
- Ascierto, P. A.; et al. ‘Adjuvant nivolumab versus ipilimumab in resected stage IIIB-C and stage IV melanoma (CheckMate 238): 4-year results from a multicentre, double-blind, randomised, controlled, phase 3 trial’. Lancet Oncol. 2020, vol. 21(no. 11), 1465–1477. [Google Scholar] [CrossRef]
- Luke, J. J.; et al. ‘Pembrolizumab versus placebo as adjuvant therapy in stage IIB or IIC melanoma: Final analysis of distant metastasis-free survival in the phase 3 KEYNOTE-716 study.’. https://doi.org/10.1200/JCO.2023.41.17_suppl.LBA9505. 2023, vol. 41(no. 17_) suppl, LBA9505–LBA9505. [Google Scholar] [CrossRef]
- van Akkooi, A. C. J.; et al. ‘Adjuvant systemic therapy in melanoma: Relative versus absolute benefit; the number needed to treat (NNT) versus the number needed to harm (NNH)?’. EJC Skin Cancer 2024, vol. 2, 100021. [Google Scholar] [CrossRef]
- Eggermont, A. M. M.; et al. ‘Five-Year Analysis of Adjuvant Pembrolizumab or Placebo in Stage III Melanoma’. NEJM Evidence 2022, vol. 1(no. 11). [Google Scholar] [CrossRef]
- Owen, C. N.; et al. ‘Management of early melanoma recurrence despite adjuvant anti-PD-1 antibody therapy☆’. Ann. Oncol. 2020, vol. 31(no. 8), 1075–1082. [Google Scholar] [CrossRef]
- VanderWalde, A.; et al. ‘Ipilimumab with or without nivolumab in PD1/PDL1 blockade refractory metastatic melanoma: A randomized phase 2 trial’. Nat. Med. 2023, vol. 29(no. 9), 2278. [Google Scholar] [CrossRef]
- Dima, D.; et al. ‘Real-world outcomes of patients with resected stage III melanoma treated with adjuvant therapies’. Cancer Med. 2024, vol. 13(no. 12), e7257. [Google Scholar] [CrossRef]
- Kluger, H. M.; et al. ‘Defining tumor resistance to PD-1 pathway blockade: Recommendations from the first meeting of the SITC Immunotherapy Resistance Taskforce’. J. Immunother. Cancer 2020, vol. 8(no. 1). [Google Scholar] [CrossRef]
- Taylor, A. M.; et al. ‘Efficacy and safety of “Second Adjuvant” therapy with BRAF/MEK inhibitors after local therapy for recurrent melanoma following adjuvant PD-1 based immunotherapy’. Eur. J. Cancer 2024, vol. 199. [Google Scholar] [CrossRef] [PubMed]
- Indini, A.; et al. ‘Developing a score system to predict therapeutic outcomes to anti-PD-1 immunotherapy in metastatic melanoma’. Tumori 2019, vol. 105(no. 6), 465–473. [Google Scholar] [CrossRef]
- Hauschild, A.; et al. ‘S100B protein detection in serum is a significant prognostic factor in metastatic melanoma’. Oncology 1999, vol. 56(no. 4), 338–344. [Google Scholar] [CrossRef]
- Tan, L.; et al. ‘Prediction and monitoring of relapse in stage III melanoma using circulating tumor DNA’. Annals of Oncology 2019, vol. 30(no. 5), 804–814. [Google Scholar] [CrossRef]
- Holmgaard, R. B.; et al. ‘Tumor-expressed IDO recruits and activates MDSCs in a Treg-dependent manner’. Cell Rep. 2015, vol. 13(no. 2), 412. [Google Scholar] [CrossRef] [PubMed]
- Kunte, C.; et al. ‘Prognostic factors associated with sentinel lymph node positivity and effect of sentinel status on survival: An analysis of 1049 patients with cutaneous melanoma’. Melanoma Res. 2010, vol. 20(no. 4), 330–337. [Google Scholar] [CrossRef] [PubMed]
- Chen, P. L.; et al. ‘Analysis of immune signatures in longitudinal tumor samples yields insight into biomarkers of response and mechanisms of resistance to immune checkpoint blockade’. Cancer Discov. 2016, vol. 6(no. 8), 827. [Google Scholar] [CrossRef] [PubMed]
- Snyder, A.; et al. ‘Genetic basis for clinical response to CTLA-4 blockade in melanoma’. N. Engl. J. Med. 2014, vol. 371(no. 23), 2189–2199. [Google Scholar] [CrossRef]
- Hugo, W.; et al. ‘Genomic and Transcriptomic Features of Response to Anti-PD-1 Therapy in Metastatic Melanoma’. Cell 2016, vol. 165(no. 1), 35–44. [Google Scholar] [CrossRef]
- Long, G. V.; et al. ‘Association of biomarkers (BMs) with efficacy of adjuvant nivolumab (NIVO) vs placebo (PBO) in patients with resected stage IIB/C melanoma (CA209-76K).’. https://doi.org/10.1200/JCO.2023.41.16_suppl.9504. 2023, vol. 41(no. 16_) suppl, pp. 9504. [Google Scholar] [CrossRef]
- Dummer, R.; et al. ‘Adjuvant dabrafenib plus trametinib versus placebo in patients with resected, BRAFV600-mutant, stage III melanoma (COMBI-AD): Exploratory biomarker analyses from a randomised, phase 3 trial’. Lancet Oncol. 2020, vol. 21(no. 3), 358–372. [Google Scholar] [CrossRef]
- Danilova, L.; et al. ‘Association of PD-1/PD-L axis expression with cytolytic activity, mutational load, and prognosis in melanoma and other solid tumors’. Proc. Natl. Acad. Sci. U. S. A 2016, vol. 113(no. 48), E7769–E7777. [Google Scholar] [CrossRef] [PubMed]
- Rozeman, E. A.; et al. ‘Survival and biomarker analyses from the OpACIN-neo and OpACIN neoadjuvant immunotherapy trials in stage III melanoma’. Nature Medicine 2021, 2021 27:2, vol. 27(no. 2), 256–263. [Google Scholar] [CrossRef] [PubMed]
- Wheeler, C. E.; et al. ‘The tumor microbiome as a predictor of outcomes in patients with metastatic melanoma treated with immune checkpoint inhibitors’. bioRxiv 2023. [Google Scholar] [CrossRef]
- Cozzolino, C.; et al. ‘Machine learning to predict overall short-term mortality in cutaneous melanoma’. Discover. Oncology 2023, vol. 14(no. 1), 13. [Google Scholar] [CrossRef] [PubMed]
- Cohen, P. R.; Kurzrock, R. ‘Dermatologic Disease-Directed Targeted Therapy (D3T2): The Application of Biomarker-Based Precision Medicine for the Personalized Treatment of Skin Conditions-Precision Dermatology’. Dermatol. Ther. (Heidelb) 2022, vol. 12(no. 10), 2249–2271. [Google Scholar] [CrossRef]
- Harbour, J. W. ‘A Prognostic Test to Predict the Risk of Metastasis in Uveal Melanoma Based on a 15-Gene Expression Profile’. Methods Mol. Biol. 2014, vol. 1102, 427. [Google Scholar] [CrossRef]
- Gerami, P.; et al. ‘Development of a Prognostic Genetic Signature to Predict the Metastatic Risk Associated with Cutaneous Melanoma’. Clinical Cancer Research 2015, vol. 21(no. 1), 175–183. [Google Scholar] [CrossRef]
- Greenhaw, B. N.; et al. ‘Molecular risk prediction in cutaneous melanoma: A meta-analysis of the 31-gene expression profile prognostic test in 1,479 patients’. J. Am. Acad. Dermatol. 2020, vol. 83(no. 3), 745–753. [Google Scholar] [CrossRef]
- Brunner, G.; et al. ‘A Prognostic Gene Signature Expressed in Primary Cutaneous Melanoma: Synergism With Conventional Staging’. JNCI Cancer Spectr. 2018, vol. 2(no. 3). [Google Scholar] [CrossRef]
- Dhillon, S.; et al. ‘Routine imaging guided by a 31-gene expression profile assay results in earlier detection of melanoma with decreased metastatic tumor burden compared to patients without surveillance imaging studies’. Arch. Dermatol. Res. 2023, vol. 315(no. 8), 2295–2302. [Google Scholar] [CrossRef]
- Jarell, A.; et al. ‘Optimizing treatment approaches for patients with cutaneous melanoma by integrating clinical and pathologic features with the 31-gene expression profile test’. J. Am. Acad. Dermatol. 2022, vol. 87(no. 6), 1312–1320. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



