Submitted:
21 April 2026
Posted:
22 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Bioactive Components of Food and Medicine Continuum Herbs
2.1. Flavonoid Compounds
2.2. Terpenoid Compounds
2.3. Polysaccharide Compounds
2.4. Phenolic Acid and Gingerol Compounds
2.5. Other Bioactive Components
3. Anti-Atherosclerotic Molecular Mechanisms
3.1. Improving Endothelial Dysfunction
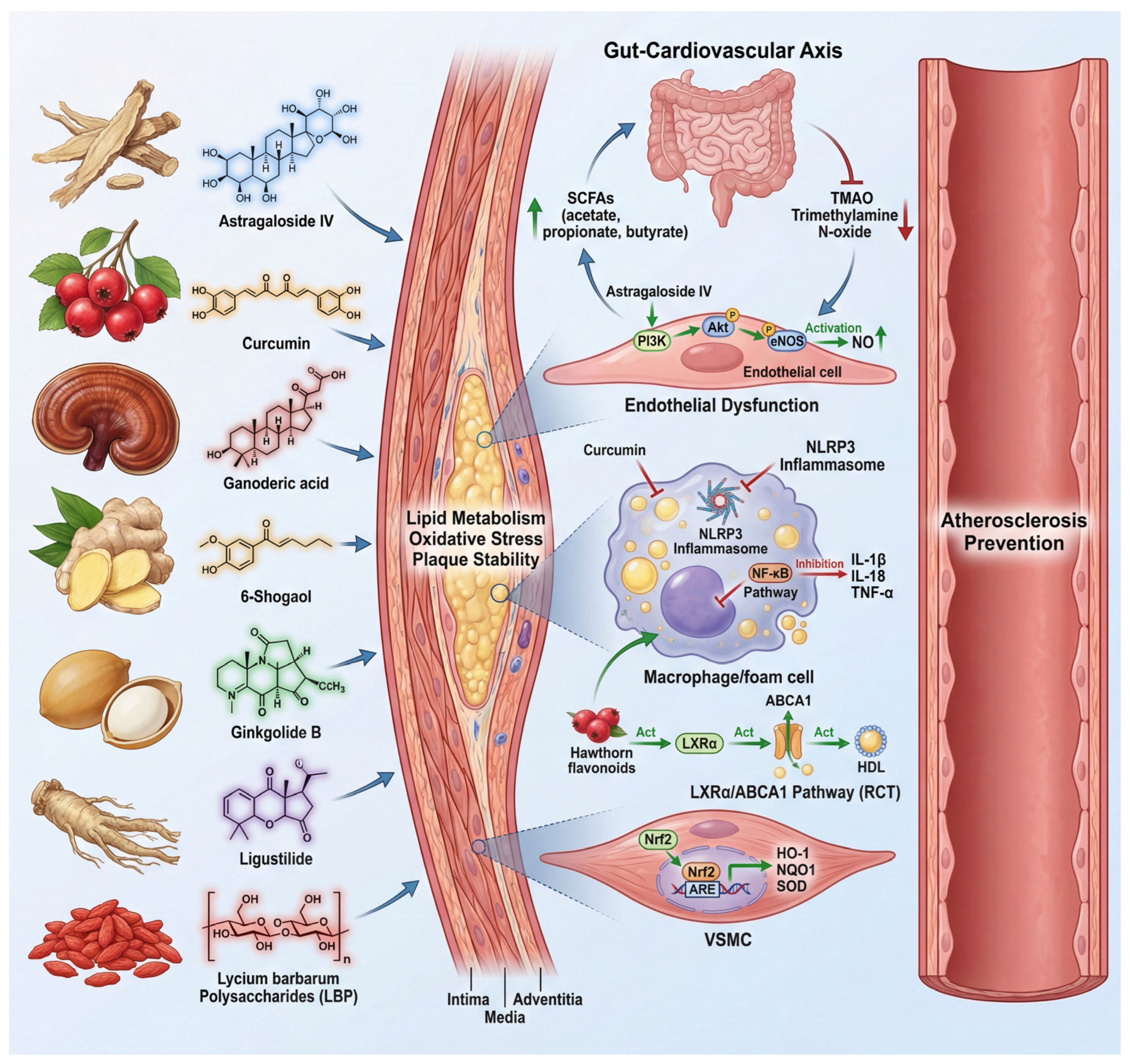
3.2. Regulating Lipid Metabolism
3.3. Inhibiting Vascular Inflammation
3.4. Anti-Oxidative Stress
3.5. Inhibiting Foam Cell Formation
3.6. Maintaining Plaque Stability
3.7. Regulating the Gut Microbiota-Metabolite-Cardiovascular Axis
3.8. Network Regulatory Mechanisms of Multi-Component Synergy
3.8.1. Component Synergy Within the Same Herb
3.8.2. Comparison with Single-Target Western Medicine Treatment
4. Preventive Applications: From Traditional Medicinal Cuisine to Functional Foods
4.1. Modernization of Traditional Medicinal Cuisine
4.2. Clinical Evidence for Standardized Extracts
4.3. Functional Food Development
4.4. Safety Management and Drug Interactions
| Herb | Risk Mechanism | Specific Manifestations | High-Risk Populations/Drugs |
|---|---|---|---|
| Ginkgo semen | Ginkgotoxin, anti-PAF, antiplatelet aggregation | Tonic-Clonic Seizure, prolonged bleeding time | Patients with epilepsy; heavy consumers; warfarin, aspirin, clopidogrel users; |
| Ginger | High dose (>4g/day) inhibits platelets | Increased bleeding risk | Perioperative period, anticoagulation therapy patients |
| Glossy Ganoderma | Immunomodulatory effects | Adverse event risk increased 1.67-fold (non-serious) | Post-organ transplant (immunosuppressant users); active autoimmune disease |
| Hawthorn Fruit | Positive inotropic effect, vasodilation | May enhance vasodilator and digoxin effects | Heart failure patients with LVEF≤35%; digoxin users |
| Turmeric | High dose inhibits platelet aggregation; inhibits CYP3A4/2C9 | Theoretically increases bleeding risk; affects drug metabolism | Anticoagulation therapy patients, perioperative period; statin users |
| Barbary Wolfberry Fruit | Immune activation; may enhance warfarin effects | Active autoimmune disease risk; elevated INR | Active systemic lupus erythematosus, rheumatoid arthritis; warfarin users |
5. Summary and Outlook
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| CVD | Cardiovascular disease |
| AS | Atherosclerosis |
| FMC | Food and medicine continuum |
| TCM | Traditional Chinese medicine |
| AMPK | Adenosine 5’-monophosphate-activated protein kinase |
| SREBP-1c | Sterol regulatory element-binding protein-1c |
| PPARα | Peroxisome proliferator-activated receptor alpha |
| PAF | Platelet-activating factor |
| Nrf2 | Nuclear factor erythroid 2-related factor 2 |
| PI3K | Phosphatidylinositol 3-kinase |
| Akt | Protein kinase B |
| eNOS | Endothelial nitric oxide synthase |
| HMG-CoA | 3-hydroxy-3-methylglutaryl-coenzyme A |
| NF-κB | Nuclear factor kappa-light-chain-enhancer of activated B cells |
| MAPK | Mitogen-activated protein kinase |
| HO-1 | Heme oxygenase-1 |
| NQO1 | NAD(P)H quinone oxidoreductase 1 |
| RG-I | Rhamnogalacturonan-I |
| COX | Cyclooxygenase |
| TXA2 | Thromboxane A2 |
| NO | Nitric oxide |
| iNOS | Inducible nitric oxide synthase |
| ARE | Antioxidant response element |
| LDL | Low-density lipoprotein |
| TLR4 | Toll-like receptor 4 |
| TNF-α | Tumor necrosis factor-alpha |
| ICAM-1 | Intercellular adhesion molecule-1 |
| VCAM-1 | Vascular cell adhesion molecule-1 |
| NADPH | Nicotinamide adenine dinucleotide phosphate |
| ROS | Reactive oxygen species |
| LOX-1 | Lectin-like oxidized low-density lipoprotein receptor-1 |
| LDLR | Low-density lipoprotein receptor |
| LXRα | Liver X receptor alpha |
| CYP7A1 | Cytochrome P450 family 7 subfamily A member 1 |
| SCAP | SREBP cleavage-activating protein |
| RCT | Reverse cholesterol transport |
| ABCA1 | ATP-binding cassette transporter A1 |
| HDL | High-density lipoprotein |
| IL-1β | Interleukin-1 beta |
| NLRP3 | NLR family pyrin domain containing 3 |
| MMP | Matrix metalloproteinase |
| TMAO | Trimethylamine N-oxide |
References
- Global, Regional, and National Burden of Cardiovascular Diseases and Risk Factors in 204 Countries and Territories, 1990-2023. J Am Coll Cardiol 2025, 86, 2167–2243. [CrossRef]
- Libby, P.; Buring, J.E.; Badimon, L.; Hansson, G.K.; Deanfield, J.; Bittencourt, M.S.; Tokgözoğlu, L.; Lewis, E.F. Atherosclerosis. Nat Rev Dis Primers 2019, 5, 56. [CrossRef]
- Banach, M.; Patti, A.M.; Giglio, R.V.; Cicero, A.F.G.; Atanasov, A.G.; Bajraktari, G.; Bruckert, E.; Descamps, O.; Djuric, D.M.; Ezhov, M.; et al. The Role of Nutraceuticals in Statin Intolerant Patients. J Am Coll Cardiol 2018, 72, 96–118. [CrossRef]
- Collins, R.; Reith, C.; Emberson, J.; Armitage, J.; Baigent, C.; Blackwell, L.; Blumenthal, R.; Danesh, J.; Smith, G.D.; DeMets, D.; et al. Interpretation of the evidence for the efficacy and safety of statin therapy. Lancet 2016, 388, 2532–2561. [CrossRef]
- Riccardi, G.; Giosuè, A.; Calabrese, I.; Vaccaro, O. Dietary recommendations for prevention of atherosclerosis. Cardiovasc Res 2022, 118, 1188–1204. [CrossRef]
- Marques-Vidal, P.; Tsampasian, V.; Cassidy, A.; Biondi-Zoccai, G.; Chrysohoou, C.; Koskinas, K.; Verschuren, W.M.M.; Czapla, M.; Kavousi, M.; Kouvari, M.; et al. Diet and nutrition in cardiovascular disease prevention: a scientific statement of the European Association of Preventive Cardiology and the Association of Cardiovascular Nursing & Allied Professions of the European Society of Cardiology. Eur J Prev Cardiol 2025, 32, 1540–1552. [CrossRef]
- Law, S.K.; Au, D.C.T. A review of medicine and food homology on traditional Chinese medicine as functional food. Food & Medicine Homology 2026, 3, 9420091. [CrossRef]
- Qu, S.; Yu, S.; Ma, X.; Wang, R. “Medicine food homology” plants promote periodontal health: antimicrobial, anti-inflammatory, and inhibition of bone resorption. Front Nutr 2023, 10, 1193289. [CrossRef]
- Hou, Y.; Jiang, J.G. Origin and concept of medicine food homology and its application in modern functional foods. Food Funct 2013, 4, 1727–1741. [CrossRef]
- Lyu, C.G.; Kang, C.Z.; Yang, J.; Wang, S.; Wang, R.S.; Wan, X.F.; Guo, L.P. [Problems and strategy of further development of Chinese medicinal materials with edible values]. Zhongguo Zhong Yao Za Zhi 2022, 47, 6810–6816. [CrossRef]
- Downer, S.; Berkowitz, S.A.; Harlan, T.S.; Olstad, D.L.; Mozaffarian, D. Food is medicine: actions to integrate food and nutrition into healthcare. Bmj 2020, 369, m2482. [CrossRef]
- Zhou, D.D.; Luo, M.; Shang, A.; Mao, Q.Q.; Li, B.Y.; Gan, R.Y.; Li, H.B. Antioxidant Food Components for the Prevention and Treatment of Cardiovascular Diseases: Effects, Mechanisms, and Clinical Studies. Oxid Med Cell Longev 2021, 2021, 6627355. [CrossRef]
- Zarenezhad, E.; Hadi, A.T.; Nournia, E.; Rostamnia, S.; Ghasemian, A. A Comprehensive Review on Potential In Silico Screened Herbal Bioactive Compounds and Host Targets in the Cardiovascular Disease Therapy. Biomed Res Int 2024, 2024, 2023620. [CrossRef]
- Yamagata, K. Polyphenols Regulate Endothelial Functions and Reduce the Risk of Cardiovascular Disease. Curr Pharm Des 2019, 25, 2443–2458. [CrossRef]
- Sánchez, M.; Romero, M.; Gómez-Guzmán, M.; Tamargo, J.; Pérez-Vizcaino, F.; Duarte, J. Cardiovascular Effects of Flavonoids. Curr Med Chem 2019, 26, 6991–7034. [CrossRef]
- Garcia, C.; Blesso, C.N. Antioxidant properties of anthocyanins and their mechanism of action in atherosclerosis. Free Radic Biol Med 2021, 172, 152–166. [CrossRef]
- Rodriguez-Mateos, A.; Le Sayec, M.; Cheok, A. Dietary (poly)phenols and cardiometabolic health: from antioxidants to modulators of the gut microbiota. Proc Nutr Soc 2025, 84, 279–289. [CrossRef]
- Oak, M.H.; Auger, C.; Belcastro, E.; Park, S.H.; Lee, H.H.; Schini-Kerth, V.B. Potential mechanisms underlying cardiovascular protection by polyphenols: Role of the endothelium. Free Radic Biol Med 2018, 122, 161–170. [CrossRef]
- Guo, S.; Qiu, S.; Cai, Y.; Wang, Z.; Yang, Q.; Tang, S.; Xie, Y.; Zhang, A. Mass spectrometry-based metabolomics for discovering active ingredients and exploring action mechanism of herbal medicine. Front Chem 2023, 11, 1142287. [CrossRef]
- Sharma, B.; Yadav, D.K. Metabolomics and Network Pharmacology in the Exploration of the Multi-Targeted Therapeutic Approach of Traditional Medicinal Plants. Plants (Basel) 2022, 11. [CrossRef]
- Li, X.; Li, X.; Wang, L.; Hou, Y.; Liu, Y.; Mao, J.; Zhang, L.; Li, X. Advancing Traditional Chinese Medicine Research through Network Pharmacology: Strategies for Target Identification, Mechanism Elucidation and Innovative Therapeutic Applications. Am J Chin Med 2025, 53, 2021–2042. [CrossRef]
- Wang, Z.; Li, Y.; Yu, P.; Guo, Z.; Huang, X.; Ma, C.; Dong, L.; Luo, P.; Wang, G.; Hu, X.; et al. Research Progress on The Chemical Composition And Pharmacological Effects of Hawthron (Crataegus spp.): A Review. J Agric Food Chem 2026, 74, 145–170. [CrossRef]
- Zhang, J.; Liang, R.; Wang, L.; Yan, R.; Hou, R.; Gao, S.; Yang, B. Effects of an aqueous extract of Crataegus pinnatifida Bge. var. major N.E.Br. fruit on experimental atherosclerosis in rats. J Ethnopharmacol 2013, 148, 563–569. [CrossRef]
- Zheng, L.; Lu, Z.; Ma, Y.; Cui, P.; Zhang, X.; Gan, J.; Li, G. Hawthorn total flavonoids ameliorate hyperlipidemia through AMPK/SREBP1-c and PPARα/PGC-1α/CPT-1A pathway activation and gut microbiota modulation. J Sci Food Agric 2025, 105, 4326–4337. [CrossRef]
- Li, M.; Si, J.Y.; Xie, P.F.; Feng, Z.J.; Li, S.J.; Zhou, F.; Li, J.M. Procyanidin C1 from hawthorn modulates metabolic-inflammatory axis to combat vascular senescence in atherosclerosis. Food Res Int 2026, 227, 118224. [CrossRef]
- Jia, H.; Wei, W.X.; Jiang, P.P.; Yu, W.W. Variation in seed quality among Ginkgo biloba L. individuals and selection of superior nut-producing tree. Plant Science Journal 2025, 43, 770–780.
- Shan, S.J.; Shi, L.L.; Liu, C.C.; Jiang, Y.R.; Chen, G.R. Study on Chemical Constituents of n-Butanol Extract of Ginkgo Biloba Seeds. Liaoning Chemical Industry 2023, 52, 798–800+804. [CrossRef]
- Zhou, G.S. Study on Resources Chemistry of Ginkgo Biloba Seeds. Master’s degree, Nanjing University of Chinese Medicine, 2013.
- Tomova, T.; Doncheva, N.; Mihaylova, A.; Kostadinov, I.; Peychev, L.; Argirova, M. An experimental study on phytochemical composition and memory enhancing effect of Ginkgo biloba seed extract. Folia Med (Plovdiv) 2021, 63, 203–212. [CrossRef]
- Chen, Q.; Wang, J.; Sun, L.; Ba, B.; Shen, D. Mechanism of Astragalus membranaceus (Huangqi, HQ) for treatment of heart failure based on network pharmacology and molecular docking. J Cell Mol Med 2024, 28, e18331. [CrossRef]
- Li, J.; Shao, N.; Gao, Y.; Li, B.; Liang, Y.; Yang, Y.; Li, J. Mechanistic Investigation of Astragalus Root in the Management of T2DM-NAFLD Comorbidity: An Integrated Network Pharmacology, Molecular Docking, Molecular Dynamics Simulation, and In Vitro Study. Pharmaceuticals (Basel) 2026, 19. [CrossRef]
- Ma, C.; Wu, H.; Yang, G.; Xiang, J.; Feng, K.; Zhang, J.; Hua, Y.; Kang, L.; Fan, G.; Yang, S. Calycosin ameliorates atherosclerosis by enhancing autophagy via regulating the interaction between KLF2 and MLKL in apolipoprotein E gene-deleted mice. Br J Pharmacol 2022, 179, 252–269. [CrossRef]
- Li, C.; He, Y.; Yang, Y.; Gou, Y.; Li, S.; Wang, R.; Zeng, S.; Zhao, X. Antioxidant and Inflammatory Effects of Nelumbo nucifera Gaertn. Leaves. Oxid Med Cell Longev 2021, 2021, 8375961. [CrossRef]
- Moore, G.; Brooks, P.; Pappalardo, L.; Boufridi, A. Phenolic profiles of Australian monofloral Eucalyptus, Corymbia, Macadamia and Lophostemon honeys via HPLC-DAD analysis. Food Chem 2025, 462, 140900. [CrossRef]
- Gośliński, M.; Nowak, D.; Szwengiel, A. Multidimensional Comparative Analysis of Bioactive Phenolic Compounds of Honeys of Various Origin. Antioxidants (Basel) 2021, 10. [CrossRef]
- Guo, J.; Tang, W.; Tang, W.; Gao, T.; Yuan, M.; Wu, Y.; Wang, G. Research progress on the types, functions, biosynthesis, and metabolic regulation of ginkgo terpenoids. Plant Physiol Biochem 2024, 212, 108754. [CrossRef]
- Forman, V.; Luo, D.; Geu-Flores, F.; Lemcke, R.; Nelson, D.R.; Kampranis, S.C.; Staerk, D.; Møller, B.L.; Pateraki, I. A gene cluster in Ginkgo biloba encodes unique multifunctional cytochrome P450s that initiate ginkgolide biosynthesis. Nat Commun 2022, 13, 5143. [CrossRef]
- Rather, L.J.; Mir, S.S.; Ganie, S.A.; Assiri, M.A.; Li, Q. Ginkgo biloba: The Traditional Use, Phytochemistry, Pharmacology, and Therapeutic Applications. Am J Chin Med 2025, 53, 2403–2434. [CrossRef]
- Liu, X.W.; Yang, J.L.; Niu, W.; Jia, W.W.; Olaleye, O.E.; Wen, Q.; Duan, X.N.; Huang, Y.H.; Wang, F.Q.; Du, F.F.; et al. Human pharmacokinetics of ginkgo terpene lactones and impact of carboxylation in blood on their platelet-activating factor antagonistic activity. Acta Pharmacol Sin 2018, 39, 1935–1946. [CrossRef]
- Li, W.Z.; Wu, W.Y.; Huang, H.; Wu, Y.Y.; Yin, Y.Y. Protective effect of bilobalide on learning and memory impairment in rats with vascular dementia. Mol Med Rep 2013, 8, 935–941. [CrossRef]
- Salehi, B.; Carneiro, J.N.P.; Rocha, J.E.; Coutinho, H.D.M.; Morais Braga, M.F.B.; Sharifi-Rad, J.; Semwal, P.; Painuli, S.; Moujir, L.M.; de Zarate Machado, V.; et al. Astragalus species: Insights on its chemical composition toward pharmacological applications. Phytother Res 2021, 35, 2445–2476. [CrossRef]
- Li, S.; Hu, X.; Liu, F.; Hu, W. Bioactive components and clinical potential of Astragalus species. Front Pharmacol 2025, 16, 1585697. [CrossRef]
- Yang, C.; Pan, Q.; Ji, K.; Tian, Z.; Zhou, H.; Li, S.; Luo, C.; Li, J. Review on the protective mechanism of astragaloside IV against cardiovascular diseases. Front Pharmacol 2023, 14, 1187910. [CrossRef]
- Lin, X.P.; Cui, H.J.; Yang, A.L.; Luo, J.K.; Tang, T. Astragaloside IV Improves Vasodilatation Function by Regulating the PI3K/Akt/eNOS Signaling Pathway in Rat Aorta Endothelial Cells. J Vasc Res 2018, 55, 169–176. [CrossRef]
- Meng, P.; Yang, R.; Jiang, F.; Guo, J.; Lu, X.; Yang, T.; He, Q. Molecular Mechanism of Astragaloside IV in Improving Endothelial Dysfunction of Cardiovascular Diseases Mediated by Oxidative Stress. Oxid Med Cell Longev 2021, 2021, 1481236. [CrossRef]
- Yao, J.; Liu, J.; He, Y.; Liu, L.; Xu, Z.; Lin, X.; Liu, N.; Kai, G. Systems pharmacology reveals the mechanism of Astragaloside IV in improving immune activity on cyclophosphamide-induced immunosuppressed mice. J Ethnopharmacol 2023, 313, 116533. [CrossRef]
- Pozzobon, R.G.; Rutckeviski, R.; de Lima, L.S.; Oliveira, C.S.; Smiderle, F.R. Anti-Inflammatory Potential of Ganoderma lucidum Triterpenes: A Systematic Review and Meta-Analysis of Preclinical Evidence. Pharmaceuticals (Basel) 2026, 19. [CrossRef]
- Hu, Z.; Du, R.; Xiu, L.; Bian, Z.; Ma, C.; Sato, N.; Hattori, M.; Zhang, H.; Liang, Y.; Yu, S.; et al. Protective effect of triterpenes of Ganoderma lucidum on lipopolysaccharide-induced inflammatory responses and acute liver injury. Cytokine 2020, 127, 154917. [CrossRef]
- Kim, H.; Ramirez, C.N.; Su, Z.Y.; Kong, A.N. Epigenetic modifications of triterpenoid ursolic acid in activating Nrf2 and blocking cellular transformation of mouse epidermal cells. J Nutr Biochem 2016, 33, 54–62. [CrossRef]
- Krajka-Kuźniak, V.; Paluszczak, J.; Oszmiański, J.; Baer-Dubowska, W. Hawthorn (Crataegus oxyacantha L.) bark extract regulates antioxidant response element (ARE)-mediated enzyme expression via Nrf2 pathway activation in normal hepatocyte cell line. Phytother Res 2014, 28, 593–602. [CrossRef]
- Reisman, S.A.; Aleksunes, L.M.; Klaassen, C.D. Oleanolic acid activates Nrf2 and protects from acetaminophen hepatotoxicity via Nrf2-dependent and Nrf2-independent processes. Biochem Pharmacol 2009, 77, 1273–1282. [CrossRef]
- Xu, L.; Xiao, S.; Sun, R.; Li, Z.; Feng, Z.; Cheng, J.; Yang, L.; Ma, C.; Fan, H.; Chai, Z. Assessment of the structural characterization and anti-inflammatory activities of various parts of Astragalus (root, stem, leaf, flower) from a polysaccharide perspective. Int J Biol Macromol 2026, 339, 149904. [CrossRef]
- Li, C.X.; Liu, Y.; Zhang, Y.Z.; Li, J.C.; Lai, J. Astragalus polysaccharide: a review of its immunomodulatory effect. Arch Pharm Res 2022, 45, 367–389. [CrossRef]
- Sha, W.; Zhao, B.; Wei, H.; Yang, Y.; Yin, H.; Gao, J.; Zhao, W.; Kong, W.; Ge, G.; Lei, T. Astragalus polysaccharide ameliorates vascular endothelial dysfunction by stimulating macrophage M2 polarization via potentiating Nrf2/HO-1 signaling pathway. Phytomedicine 2023, 112, 154667. [CrossRef]
- Chen, G.; Jiang, N.; Zheng, J.; Hu, H.; Yang, H.; Lin, A.; Hu, B.; Liu, H. Structural characterization and anti-inflammatory activity of polysaccharides from Astragalus membranaceus. Int J Biol Macromol 2023, 241, 124386. [CrossRef]
- Yang, M.; Qin, X.; Liu, X. A review of polysaccharides from Ganoderma lucidum: Preparation methods, structural characteristics, bioactivities, structure-activity relationships and potential applications. Int J Biol Macromol 2025, 303, 140645. [CrossRef]
- Ren, L.; Zhang, J.; Zhang, T. Immunomodulatory activities of polysaccharides from Ganoderma on immune effector cells. Food Chem 2021, 340, 127933. [CrossRef]
- Li, J.; Gu, F.; Cai, C.; Hu, M.; Fan, L.; Hao, J.; Yu, G. Purification, structural characterization, and immunomodulatory activity of the polysaccharides from Ganoderma lucidum. Int J Biol Macromol 2020, 143, 806–813. [CrossRef]
- Wang, Y.; Zuo, Y.; Weng, J.; Peng, X. Health benefits of Ganoderma lucidum polysaccharides: A review of potential cardiovascular protective effects. Int J Biol Macromol 2025, 330, 148001. [CrossRef]
- Li, L.F.; Liu, H.B.; Zhang, Q.W.; Li, Z.P.; Wong, T.L.; Fung, H.Y.; Zhang, J.X.; Bai, S.P.; Lu, A.P.; Han, Q.B. Comprehensive comparison of polysaccharides from Ganoderma lucidum and G. sinense: chemical, antitumor, immunomodulating and gut-microbiota modulatory properties. Sci Rep 2018, 8, 6172. [CrossRef]
- Wang, B.; Yang, J.; Tao, L.; Zhou, X.; Ding, X. Structural-activity relationship of Lycium barbarum polysaccharides in immunomodulation: integrating molecular insights with target identification for therapeutic development. Front Immunol 2026, 17, 1730418. [CrossRef]
- Li, Y.; Yang, B.; Zhang, X.; Shen, X.; Ma, Y.; Jing, L. Lycium barbarum polysaccharide antagonizes cardiomyocyte apoptosis by inhibiting the upregulation of GRK2 induced by I/R injury, and salvage mitochondrial fission/fusion imbalance and AKT/eNOS signaling. Cell Signal 2022, 92, 110252. [CrossRef]
- Chen, M.M.; Dong, S.; Wang, K.W.; Zhou, T. Physicochemical and functional properties of octenyl succinic anhydride-modified polysaccharides from fermented lotus leaves. Int J Biol Macromol 2026, 344, 150509. [CrossRef]
- Ke, Y.; Lin, L.; Zhao, M. Rhamnogalacturonan I-Enriched Pectin, Flavonoids, and Alkaloids from Lotus Leaf Infusion in Regulating Glycolipid Absorption and Metabolism: Isolation, In Vitro Bioactivity Verification, and Structural Characterization. J Agric Food Chem 2023, 71, 8969–8980. [CrossRef]
- Hong, Q.; Ma, Z.C.; Huang, H.; Wang, Y.G.; Tan, H.L.; Xiao, C.R.; Liang, Q.D.; Zhang, H.T.; Gao, Y. Antithrombotic activities of ferulic acid via intracellular cyclic nucleotide signaling. Eur J Pharmacol 2016, 777, 1–8. [CrossRef]
- Li, W.J.; Cai, Y.F.; Ouyang, Y.; Li, X.Y.; Shi, X.L.; Cao, S.X.; Huang, Y.; Wu, H.W.; Yang, H.J. Quality evaluation of Angelica Sinensis Radix dispensing granules by integrating microvascular activity and chemical analysis. J Ethnopharmacol 2024, 319, 117236. [CrossRef]
- An, W.; Tian, Q.; Guo, P.; Chen, M.; Zhang, T.; Yang, P.; Zhang, S. Danggui Buxue Decoction and its components dilate coronary artery through activating the inward rectification K(+) channels pathway. J Ethnopharmacol 2025, 338, 119064. [CrossRef]
- Zhang, K.; Shen, X.; Yang, L.; Chen, Q.; Wang, N.; Li, Y.; Song, P.; Jiang, M.; Bai, G.; Yang, P.; et al. Exploring the Q-markers of Angelica sinensis (Oliv.) Diels of anti-platelet aggregation activity based on spectrum-effect relationships. Biomed Chromatogr 2022, 36, e5422. [CrossRef]
- Singh, A.K.; Singla, R.K.; Pandey, A.K. Chlorogenic Acid: A Dietary Phenolic Acid with Promising Pharmacotherapeutic Potential. Curr Med Chem 2023, 30, 3905–3926. [CrossRef]
- Vardhini, N.M.; Punia, J.; Jat, S.; Pawar, S.D.; Devi, N.; Radhakrishnanand, P.; Murty, U.S.; Saini, A.; Sethi, K.K.; Kumar, P. Purification and characterization of pure curcumin, desmethoxycurcumin, and bisdemethoxycurcumin from North-East India Lakadong turmeric (Curcuma longa). J Chromatogr A 2023, 1708, 464358. [CrossRef]
- Praveen, A.; Prasad, D.; Mishra, S.; Nagarajan, S.; Chaudhari, S.R. Facile NMR approach for profiling curcuminoids present in turmeric. Food Chem 2021, 341, 128646. [CrossRef]
- Younis, H.M.; Mohamed, A.A. A Review on Curcumin: Pharmacological Promises and Biomedical Activities. Arch Pharm (Weinheim) 2025, 358, e70129. [CrossRef]
- El-Saadony, M.T.; Saad, A.M.; Mohammed, D.M.; Alkafaas, S.S.; Ghosh, S.; Negm, S.H.; Salem, H.M.; Fahmy, M.A.; Mosa, W.F.A.; Ibrahim, E.H.; et al. Curcumin, an active component of turmeric: biological activities, nutritional aspects, immunological, bioavailability, and human health benefits - a comprehensive review. Front Immunol 2025, 16, 1603018. [CrossRef]
- Pan-On, S.; Dilokthornsakul, P.; Tiyaboonchai, W. Trends in advanced oral drug delivery system for curcumin: A systematic review. J Control Release 2022, 348, 335–345. [CrossRef]
- Saavedra, D.I.; Carter, D.T.; Dawson, J.T.; Shah, S.A.; Stevens, N.; Poudel, A.; Satyal, P.; Bascoul, C. Turmeric (curcuma longa) rhizome essential oil: analytical profile of authenticated and commercial samples, safety and pharmacology review. Pharm Biol 2026, 64, 379–397. [CrossRef]
- Gao, Y.; Lu, Y.; Zhang, N.; Udenigwe, C.C.; Zhang, Y.; Fu, Y. Preparation, pungency and bioactivity of gingerols from ginger (Zingiber officinale Roscoe): a review. Crit Rev Food Sci Nutr 2024, 64, 2708–2733. [CrossRef]
- Garza-Cadena, C.; Ortega-Rivera, D.M.; Machorro-García, G.; Gonzalez-Zermeño, E.M.; Homma-Dueñas, D.; Plata-Gryl, M.; Castro-Muñoz, R. A comprehensive review on Ginger (Zingiber officinale) as a potential source of nutraceuticals for food formulations: Towards the polishing of gingerol and other present biomolecules. Food Chem 2023, 413, 135629. [CrossRef]
- Krüger, S.; Bergin, A.; Morlock, G.E. Effect-directed analysis of ginger (Zingiber officinale) and its food products, and quantification of bioactive compounds via high-performance thin-layer chromatography and mass spectrometry. Food Chem 2018, 243, 258–268. [CrossRef]
- He, X.; Shang, Y.; Liao, X.; Liu, G.; Yang, Q.; Liu, J.; Xu, X.; Liu, X. 6-Gingerol ameliorates endothelial injury in hyperlipidemia mice via Nrf2 activation and inflammation modulation. Food Funct 2026, 17, 1007–1017. [CrossRef]
- Ahmed, S.H.H.; Gonda, T.; Agbadua, O.G.; Girst, G.; Berkecz, R.; Kúsz, N.; Tsai, M.C.; Wu, C.C.; Balogh, G.T.; Hunyadi, A. Preparation and Evaluation of 6-Gingerol Derivatives as Novel Antioxidants and Antiplatelet Agents. Antioxidants (Basel) 2023, 12. [CrossRef]
- Sang, S.; Snook, H.D.; Tareq, F.S.; Fasina, Y. Precision Research on Ginger: The Type of Ginger Matters. J Agric Food Chem 2020, 68, 8517–8523. [CrossRef]
- Bischoff-Kont, I.; Fürst, R. Benefits of Ginger and Its Constituent 6-Shogaol in Inhibiting Inflammatory Processes. Pharmaceuticals (Basel) 2021, 14. [CrossRef]
- Du, Y.T.; Zheng, Y.L.; Ji, Y.; Dai, F.; Hu, Y.J.; Zhou, B. Applying an Electrophilicity-Based Strategy to Develop a Novel Nrf2 Activator Inspired from Dietary [6]-Shogaol. J Agric Food Chem 2018, 66, 7983–7994. [CrossRef]
- Bratosin, E.D.; Tit, D.M.; Purza, A.L.; Pasca, M.B.; Bungau, G.S.; Marin, R.C.; Radu, A.F.; Gitea, D. Exploratory Analysis of Phenolic Profiles and Antioxidant Capacity in Selected Romanian Monofloral Honeys: Influence of Botanical Origin and Acquisition Source. Antioxidants (Basel) 2025, 14. [CrossRef]
- Combarros-Fuertes, P.; Estevinho, L.M.; Dias, L.G.; Castro, J.M.; Tomás-Barberán, F.A.; Tornadijo, M.E.; Fresno-Baro, J.M. Bioactive Components and Antioxidant and Antibacterial Activities of Different Varieties of Honey: A Screening Prior to Clinical Application. J Agric Food Chem 2019, 67, 688–698. [CrossRef]
- Li, H.; Li, W.; Wu, Y.; Wu, H.; Cai, X. Integrating network pharmacology and animal experimental validation to investigate the mechanism of lotus leaf in obesity. Int Immunopharmacol 2025, 145, 113719. [CrossRef]
- Inchan, A.; Bualeong, T.; Kaewkong, W.; Nuengchamnong, N.; Apaikawee, P.; Sa-Nguanpong, P.; Sumsakul, W.; Charoenphon, N.; Chatturong, U.; Deetud, W.; et al. Antihypertensive Effects of Lotus Seed (Nelumbo nucifera Gaertn.) Extract via eNOS Upregulation and Oxidative Stress Reduction in L-NAME-Induced Hypertensive Rats. Pharmaceuticals (Basel) 2025, 18. [CrossRef]
- Wicha, P.; Onsa-Ard, A.; Chaichompoo, W.; Suksamrarn, A.; Tocharus, C. Vasorelaxant and Antihypertensive Effects of Neferine in Rats: An In Vitro and In Vivo Study. Planta Med 2020, 86, 496–504. [CrossRef]
- Ma, R.H.; Zhang, X.X.; Ni, Z.J.; Thakur, K.; Wang, W.; Yan, Y.M.; Cao, Y.L.; Zhang, J.G.; Rengasamy, K.R.R.; Wei, Z.J. Lycium barbarum (Goji) as functional food: a review of its nutrition, phytochemical structure, biological features, and food industry prospects. Crit Rev Food Sci Nutr 2023, 63, 10621–10635. [CrossRef]
- Zhu, X.; Cheang, I.; Tang, Y.; Shi, M.; Zhu, Q.; Gao, R.; Liao, S.; Yao, W.; Zhou, Y.; Zhang, H.; et al. Associations of Serum Carotenoids With Risk of All-Cause and Cardiovascular Mortality in Hypertensive Adults. J Am Heart Assoc 2023, 12, e027568. [CrossRef]
- Parichatikanond, W.; Duangrat, R.; Nuamnaichati, N.; Mangmool, S. Role of A(1) adenosine receptor in cardiovascular diseases: Bridging molecular mechanisms with therapeutic opportunities. Exp Mol Pathol 2025, 141, 104952. [CrossRef]
- Borea, P.A.; Gessi, S.; Merighi, S.; Varani, K. Adenosine as a Multi-Signalling Guardian Angel in Human Diseases: When, Where and How Does it Exert its Protective Effects? Trends Pharmacol Sci 2016, 37, 419–434. [CrossRef]
- Girma, A.; Seo, W.; She, R.C. Antibacterial activity of varying UMF-graded Manuka honeys. PLoS One 2019, 14, e0224495. [CrossRef]
- Leng, B.; Tang, F.; Lu, M.; Zhang, Z.; Wang, H.; Zhang, Y. Astragaloside IV improves vascular endothelial dysfunction by inhibiting the TLR4/NF-κB signaling pathway. Life Sci 2018, 209, 111–121. [CrossRef]
- Choi, E.S.; Yoon, J.J.; Han, B.H.; Jeong, D.H.; Lee, Y.J.; Kang, D.G.; Lee, H.S. Ligustilide attenuates vascular inflammation and activates Nrf2/HO-1 induction and, NO synthesis in HUVECs. Phytomedicine 2018, 38, 12–23. [CrossRef]
- Moore, K.J.; Koplev, S.; Fisher, E.A.; Tabas, I.; Björkegren, J.L.M.; Doran, A.C.; Kovacic, J.C. Macrophage Trafficking, Inflammatory Resolution, and Genomics in Atherosclerosis: JACC Macrophage in CVD Series (Part 2). J Am Coll Cardiol 2018, 72, 2181–2197. [CrossRef]
- Bischoff-Kont, I.; Primke, T.; Niebergall, L.S.; Zech, T.; Fürst, R. Ginger Constituent 6-Shogaol Inhibits Inflammation- and Angiogenesis-Related Cell Functions in Primary Human Endothelial Cells. Front Pharmacol 2022, 13, 844767. [CrossRef]
- Zhang, S.; Chen, B.; Wu, W.; Bao, L.; Qi, R. Ginkgolide B reduces inflammatory protein expression in oxidized low-density lipoprotein-stimulated human vascular endothelial cells. J Cardiovasc Pharmacol 2011, 57, 721–727. [CrossRef]
- Feng, Z.; Yang, X.; Zhang, L.; Ansari, I.A.; Khan, M.S.; Han, S.; Feng, Y. Ginkgolide B ameliorates oxidized low-density lipoprotein-induced endothelial dysfunction via modulating Lectin-like ox-LDL-receptor-1 and NADPH oxidase 4 expression and inflammatory cascades. Phytother Res 2018, 32, 2417–2427. [CrossRef]
- Hu, H.; Weng, J.; Cui, C.; Tang, F.; Yu, M.; Zhou, Y.; Shao, F.; Zhu, Y. The Hypolipidemic Effect of Hawthorn Leaf Flavonoids through Modulating Lipid Metabolism and Gut Microbiota in Hyperlipidemic Rats. Evid Based Complement Alternat Med 2022, 2022, 3033311. [CrossRef]
- Chen, B.; Tian, J.; Zhang, J.; Wang, K.; Liu, L.; Yang, B.; Bao, L.; Liu, H. Triterpenes and meroterpenes from Ganoderma lucidum with inhibitory activity against HMGs reductase, aldose reductase and α-glucosidase. Fitoterapia 2017, 120, 6–16. [CrossRef]
- Zhu, J.; Jin, J.; Ding, J.; Li, S.; Cen, P.; Wang, K.; Wang, H.; Xia, J. Ganoderic Acid A improves high fat diet-induced obesity, lipid accumulation and insulin sensitivity through regulating SREBP pathway. Chem Biol Interact 2018, 290, 77–87. [CrossRef]
- Yu, X.H.; Tang, C.K. ABCA1, ABCG1, and Cholesterol Homeostasis. Adv Exp Med Biol 2022, 1377, 95–107. [CrossRef]
- Dergunov, A.D.; Baserova, V.B. Different Pathways of Cellular Cholesterol Efflux. Cell Biochem Biophys 2022, 80, 471–481. [CrossRef]
- Lee, S.M.; Moon, J.; Cho, Y.; Chung, J.H.; Shin, M.J. Quercetin up-regulates expressions of peroxisome proliferator-activated receptor γ, liver X receptor α, and ATP binding cassette transporter A1 genes and increases cholesterol efflux in human macrophage cell line. Nutr Res 2013, 33, 136–143. [CrossRef]
- Dong, P.; Pan, L.; Zhang, X.; Zhang, W.; Wang, X.; Jiang, M.; Chen, Y.; Duan, Y.; Wu, H.; Xu, Y.; et al. Hawthorn (Crataegus pinnatifida Bunge) leave flavonoids attenuate atherosclerosis development in apoE knock-out mice. J Ethnopharmacol 2017, 198, 479–488. [CrossRef]
- Wang, W.; Sun, R.; Zhang, J.; Jia, L.; Dong, Y. Study on the Mechanism of Ganoderma lucidum Polysaccharides for Ameliorating Dyslipidemia via Regulating Gut Microbiota and Fecal Metabolites. Biomolecules 2026, 16. [CrossRef]
- Zhang, S.; Zou, J.; Li, P.; Zheng, X.; Feng, D. Curcumin Protects against Atherosclerosis in Apolipoprotein E-Knockout Mice by Inhibiting Toll-like Receptor 4 Expression. J Agric Food Chem 2018, 66, 449–456. [CrossRef]
- Nievergelt, A.; Marazzi, J.; Schoop, R.; Altmann, K.H.; Gertsch, J. Ginger phenylpropanoids inhibit IL-1beta and prostanoid secretion and disrupt arachidonate-phospholipid remodeling by targeting phospholipases A2. J Immunol 2011, 187, 4140–4150. [CrossRef]
- Liu, X.; Zhao, G.; Yan, Y.; Bao, L.; Chen, B.; Qi, R. Ginkgolide B reduces atherogenesis and vascular inflammation in ApoE(-/-) mice. PLoS One 2012, 7, e36237. [CrossRef]
- Yin, H.; Guo, Q.; Li, X.; Tang, T.; Li, C.; Wang, H.; Sun, Y.; Feng, Q.; Ma, C.; Gao, C.; et al. Curcumin Suppresses IL-1β Secretion and Prevents Inflammation through Inhibition of the NLRP3 Inflammasome. J Immunol 2018, 200, 2835–2846. [CrossRef]
- Wu, J.; He, S.; Song, Z.; Chen, S.; Lin, X.; Sun, H.; Zhou, P.; Peng, Q.; Du, S.; Zheng, S.; et al. Macrophage polarization states in atherosclerosis. Front Immunol 2023, 14, 1185587. [CrossRef]
- Zhang, X.; Liu, S.; Kong, F.; Shu, L.; Li, Y.; Wang, D.; Li, L. Acidic polysaccharide from Ganoderma tsugae: Structural characterization and antiatherosclerotic related to macrophage polarization. Food Res Int 2025, 203, 115913. [CrossRef]
- Quan, Y.Z.; Ma, A.; Ren, C.Q.; An, Y.P.; Qiao, P.S.; Gao, C.; Zhang, Y.K.; Li, X.W.; Lin, S.M.; Li, N.N.; et al. Ganoderic acids alleviate atherosclerosis by inhibiting macrophage M1 polarization via TLR4/MyD88/NF-κB signaling pathway. Atherosclerosis 2024, 391, 117478. [CrossRef]
- Zhou, L.; Yang, J.; Liu, H.; Rang, Y.; Xu, L.; Wang, X.; Li, Y.; Liu, C. Lycium barbarum polysaccharides attenuate oxidative stress and mitochondrial toxicity induced by mixed plasticizers in HepG2 cells through activation of Nrf2. Life Sci 2024, 336, 122346. [CrossRef]
- Yang, Y.; Yu, L.; Zhu, T.; Xu, S.; He, J.; Mao, N.; Liu, Z.; Wang, D. Neuroprotective effects of Lycium barbarum polysaccharide on light-induced oxidative stress and mitochondrial damage via the Nrf2/HO-1 pathway in mouse hippocampal neurons. Int J Biol Macromol 2023, 251, 126315. [CrossRef]
- Liu, Y.; Wang, F.; Xu, H.; Wang, H.; Lu, M.; Cheng, L. Ginkgolide B attenuates hyperlipidemia by restoring sphingolipid homeostasis and activating PPARα and Nrf2 pathways. Sci Rep 2025, 15, 28774. [CrossRef]
- Zhou, L.; Wang, X.; Meng, C.; Li, T.; Wang, Q.; Zhang, M.; Wu, M. Exploring the role of curcumin in anti-atherosclerosis: Mechanisms and pathways of action. Phytomedicine 2026, 152, 157809. [CrossRef]
- Wu, S.; Liao, X.; Zhu, Z.; Huang, R.; Chen, M.; Huang, A.; Zhang, J.; Wu, Q.; Wang, J.; Ding, Y. Antioxidant and anti-inflammation effects of dietary phytochemicals: The Nrf2/NF-κB signalling pathway and upstream factors of Nrf2. Phytochemistry 2022, 204, 113429. [CrossRef]
- Battino, M.; Giampieri, F.; Cianciosi, D.; Ansary, J.; Chen, X.; Zhang, D.; Gil, E.; Forbes-Hernández, T. The roles of strawberry and honey phytochemicals on human health: A possible clue on the molecular mechanisms involved in the prevention of oxidative stress and inflammation. Phytomedicine 2021, 86, 153170. [CrossRef]
- Wu, J.; Han, B.; Zhao, S.; Zhong, Y.; Han, W.; Gao, J.; Wang, S. Bioactive characterization of multifloral honeys from Apis cerana cerana, Apis dorsata, and Lepidotrigona flavibasis. Food Res Int 2022, 161, 111808. [CrossRef]
- Ma, L.; Liu, X.; Zhao, Y.; Chen, B.; Li, X.; Qi, R. Ginkgolide B reduces LOX-1 expression by inhibiting Akt phosphorylation and increasing Sirt1 expression in oxidized LDL-stimulated human umbilical vein endothelial cells. PLoS One 2013, 8, e74769. [CrossRef]
- Bai, X.; Wang, S.; Shu, L.; Cao, Q.; Hu, H.; Zhu, Y.; Chen, C. Hawthorn leaf flavonoids alleviate the deterioration of atherosclerosis by inhibiting SCAP-SREBP2-LDLR pathway through sPLA2-ⅡA signaling in macrophages in mice. J Ethnopharmacol 2024, 327, 118006. [CrossRef]
- Kremastiotis, G.; Handa, I.; Jackson, C.; George, S.; Johnson, J. Disparate effects of MMP and TIMP modulation on coronary atherosclerosis and associated myocardial fibrosis. Sci Rep 2021, 11, 23081. [CrossRef]
- Liu, Y.; Lai, G.; Guo, Y.; Tang, X.; Shuai, O.; Xie, Y.; Wu, Q.; Chen, D.; Yuan, X. Protective effect of Ganoderma lucidum spore extract in trimethylamine-N-oxide-induced cardiac dysfunction in rats. J Food Sci 2021, 86, 546–562. [CrossRef]
- Sang, T.; Guo, C.; Guo, D.; Wu, J.; Wang, Y.; Wang, Y.; Chen, J.; Chen, C.; Wu, K.; Na, K.; et al. Suppression of obesity and inflammation by polysaccharide from sporoderm-broken spore of Ganoderma lucidum via gut microbiota regulation. Carbohydr Polym 2021, 256, 117594. [CrossRef]
- Jiang, T.; Zhu, R.; Guo, X.; Li, J.; Zhu, X.; Bao, R.; Chen, J. Comprehensive evaluation of Ganoderma lucidum extracts: digestion kinetics, gut microbiota modulation, and immunoregulatory mechanisms. Food Res Int 2026, 230, 118664. [CrossRef]
- Wang, Z.; Ren, X.; Peng, Z.; Zeng, M.; Wang, Z.; Chen, Q.; Chen, J.; Dai, X.; Christian, M.; Qie, X.; et al. Flavonoid-rich extracts of Nelumbo nucifera leaves alleviate obesity in HFD-fed mice via microbiota-dependent modulation of brown fat thermogenesis. J Ethnopharmacol 2026, 354, 120513. [CrossRef]
- Shi, D.; Xu, X.; Wang, J.; Bu, T.; Sun, P.; Yang, K.; Cai, M. Synergistic anti-inflammatory effects of Ganoderma lucidum polysaccharide and ganoderic acid A on LPS-induced RAW264.7 cells by inhibition of TLR4/NF-κB activation. Int J Biol Macromol 2025, 309, 143074. [CrossRef]
- Li, Y.; Tang, J.; Gao, H.; Xu, Y.; Han, Y.; Shang, H.; Lu, Y.; Qin, C. Ganoderma lucidum triterpenoids and polysaccharides attenuate atherosclerotic plaque in high-fat diet rabbits. Nutr Metab Cardiovasc Dis 2021, 31, 1929–1938. [CrossRef]
- Caruso, L.; Nadur, N.F.; da Fonseca, M.B.; Peixoto Ferreira, L.A.; Lacerda, R.B.; Graebin, C.S.; Kümmerle, A.E. The Design of Multi-target Drugs to Treat Cardiovascular Diseases: Two (or more) Birds on One Stone. Curr Top Med Chem 2022, 22, 366–394. [CrossRef]
- Peters, J.U. Polypharmacology - foe or friend? J Med Chem 2013, 56, 8955–8971. [CrossRef]
- Yang, S.; Yang, H.; Zhang, Y. Yao-Shan of traditional Chinese medicine: an old story for metabolic health. Front Pharmacol 2023, 14, 1194026. [CrossRef]
- Li, Y.; Xu, Z.; Du, P.; Gao, J.; Wang, S.; Pang, X.; Ren, C.; Liu, Y.; Zhang, C. Methodological challenges in pilot trials of herbal medicine: barriers to evidence-based practice. J Clin Epidemiol 2025, 182, 111754. [CrossRef]
- Crichton, M.; Davidson, A.R.; Innerarity, C.; Marx, W.; Lohning, A.; Isenring, E.; Marshall, S. Orally consumed ginger and human health: an umbrella review. Am J Clin Nutr 2022, 115, 1511–1527. [CrossRef]
- Hasani, H.; Arab, A.; Hadi, A.; Pourmasoumi, M.; Ghavami, A.; Miraghajani, M. Does ginger supplementation lower blood pressure? A systematic review and meta-analysis of clinical trials. Phytother Res 2019, 33, 1639–1647. [CrossRef]
- Pourmasoumi, M.; Hadi, A.; Rafie, N.; Najafgholizadeh, A.; Mohammadi, H.; Rouhani, M.H. The effect of ginger supplementation on lipid profile: A systematic review and meta-analysis of clinical trials. Phytomedicine 2018, 43, 28–36. [CrossRef]
- Ahmed, A.; Tul-Noor, Z.; Lee, D.; Bajwah, S.; Ahmed, Z.; Zafar, S.; Syeda, M.; Jamil, F.; Qureshi, F.; Zia, F.; et al. Effect of honey on cardiometabolic risk factors: a systematic review and meta-analysis. Nutr Rev 2023, 81, 758–774. [CrossRef]
- Gholami, Z.; Sohrabi, Z.; Zare, M.; Pourrajab, B.; Nasimi, N. The effect of honey on lipid profiles: a systematic review and meta-analysis of controlled clinical trials. Br J Nutr 2022, 127, 1482–1496. [CrossRef]
- Ma, Z.; Wang, N.; He, H.; Tang, X. Pharmaceutical strategies of improving oral systemic bioavailability of curcumin for clinical application. J Control Release 2019, 316, 359–380. [CrossRef]
- Tan, O.J.; Loo, H.L.; Thiagarajah, G.; Palanisamy, U.D.; Sundralingam, U. Improving oral bioavailability of medicinal herbal compounds through lipid-based formulations - A Scoping Review. Phytomedicine 2021, 90, 153651. [CrossRef]
- Flory, S.; Sus, N.; Haas, K.; Jehle, S.; Kienhöfer, E.; Waehler, R.; Adler, G.; Venturelli, S.; Frank, J. Increasing Post-Digestive Solubility of Curcumin Is the Most Successful Strategy to Improve its Oral Bioavailability: A Randomized Cross-Over Trial in Healthy Adults and In Vitro Bioaccessibility Experiments. Mol Nutr Food Res 2021, 65, e2100613. [CrossRef]
- Preciado Iñiga, G.; Martínez-Carrera, D.; Meneses, M.E.; Sánchez, M.; Argumedo, A.; Bonilla, M.; Castillo, I.; Petlacalco, B.; Morales, A.; Fernández, N.; et al. Characterisation and Hypolipidaemic Effects of Tlayudas, Widely Consumed Tortillas, Containing Ganoderma lucidum Extracts on an In Vivo Model of Hypercholesterolaemia. Int J Food Sci 2025, 2025, 8096060. [CrossRef]
- Chow, S.L.; Bozkurt, B.; Baker, W.L.; Bleske, B.E.; Breathett, K.; Fonarow, G.C.; Greenberg, B.; Khazanie, P.; Leclerc, J.; Morris, A.A.; et al. Complementary and Alternative Medicines in the Management of Heart Failure: A Scientific Statement From the American Heart Association. Circulation 2023, 147, e4–e30. [CrossRef]
- Boateng, I.D. A critical review of current technologies used to reduce ginkgotoxin, ginkgotoxin-5’-glucoside, ginkgolic acid, allergic glycoprotein, and cyanide in Ginkgo biloba L. seed. Food Chem 2022, 382, 132408. [CrossRef]
- Azuma, F.; Nokura, K.; Kako, T.; Kobayashi, D.; Yoshimura, T.; Wada, K. An Adult Case of Generalized Convulsions Caused by the Ingestion of Ginkgo biloba Seeds with Alcohol. Intern Med 2020, 59, 1555–1558. [CrossRef]
- Kajiyama, Y.; Fujii, K.; Takeuchi, H.; Manabe, Y. Ginkgo seed poisoning. Pediatrics 2002, 109, 325–327. [CrossRef]
- Liperoti, R.; Vetrano, D.L.; Bernabei, R.; Onder, G. Herbal Medications in Cardiovascular Medicine. J Am Coll Cardiol 2017, 69, 1188–1199. [CrossRef]
- Bone, K.M. Potential interaction of Ginkgo biloba leaf with antiplatelet or anticoagulant drugs: what is the evidence? Mol Nutr Food Res 2008, 52, 764–771. [CrossRef]
- Burkina, V.; Zamaratskaia, G.; Rasmussen, M.K. Curcumin and quercetin modify warfarin-induced regulation of porcine CYP1A2 and CYP3A expression and activity in vitro. Xenobiotica 2022, 52, 435–441. [CrossRef]
- Leite, P.M.; Martins, M.A.P.; Carvalho, M.D.G.; Castilho, R.O. Mechanisms and interactions in concomitant use of herbs and warfarin therapy: An updated review. Biomed Pharmacother 2021, 143, 112103. [CrossRef]
- Vogel, J.H.; Bolling, S.F.; Costello, R.B.; Guarneri, E.M.; Krucoff, M.W.; Longhurst, J.C.; Olshansky, B.; Pelletier, K.R.; Tracy, C.M.; Vogel, R.A.; et al. Integrating complementary medicine into cardiovascular medicine. A report of the American College of Cardiology Foundation Task Force on Clinical Expert Consensus Documents (Writing Committee to Develop an Expert Consensus Document on Complementary and Integrative Medicine). J Am Coll Cardiol 2005, 46, 184–221. [CrossRef]
- Villaescusa, L.; Zaragozá, C.; Zaragozá, F.; Tamargo, J. Herbal medicines for the treatment of cardiovascular diseases: Benefits and risks - A narrative review. Int J Cardiol 2023, 385, 44–52. [CrossRef]
- Min, D.E.; Han, D.E.; Kang, K.; Ding, K.; Xu, G.; Lee, M.Y.; An, D.S.; Hur, N.; Kim, J.M.; Hong, J.Y.; et al. A comparative analysis of regulatory frameworks and their market impacts on health functional foods: perspectives from South Korea, Canada, the United States, and China. Crit Rev Food Sci Nutr 2026, 1–24. [CrossRef]
- Shan, F.; Liu, L.; Li, L.; Wang, W.; Bi, Y.; Li, M. Management, Safety, and Efficacy Evaluation of Nutraceutical and Functional Food: A Global Perspective. Compr Rev Food Sci Food Saf 2025, 24, e70222. [CrossRef]
- Chen, F.; Wen, Q.; Jiang, J.; Li, H.L.; Tan, Y.F.; Li, Y.H.; Zeng, N.K. Could the gut microbiota reconcile the oral bioavailability conundrum of traditional herbs? J Ethnopharmacol 2016, 179, 253–264. [CrossRef]

| Herb | Major Bioactive Components | Main Chemical Structure | Primary Cardiovascular Activities |
|---|---|---|---|
| Hawthorn Fruit | Hyperoside, Ursolic acid | 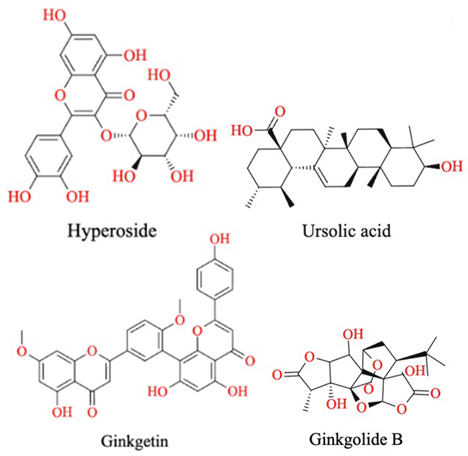 |
Antioxidant, lipid regulation, inhibit LDL oxidation, inhibit HMG-CoA reductase, activate Nrf2 |
| Ginkgo Seed | Ginkgetin, ginkgolide B | Antioxidant, anti-inflammatory, anti-PAF, neuroprotection | |
| Milkvetch Root | Astragaloside IV | 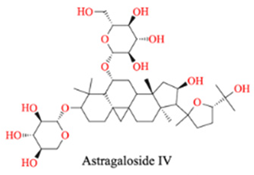 |
Improve endothelial function, anti-fibrosis |
| Turmeric | Curcumin | 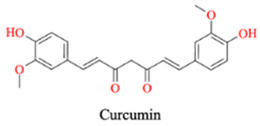 |
Anti-inflammatory, antioxidant |
| Ginger | 6-Gingerol | 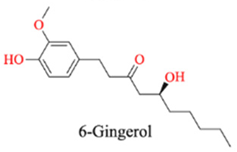 |
Antioxidant, anti-inflammatory, antiplatelet |
| Glossy Ganoderma | Ganoderic acid A | 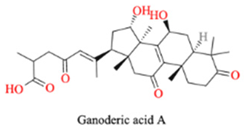 |
Lipid regulation, anti-inflammatory |
| Angelica sinensis |
Ferulic acid, ligustilide | 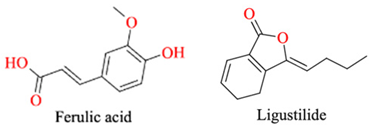 |
Antithrombotic, vasodilation, antiplatelet |
| Barbary Wolfberry Fruit | Zeaxanthin | 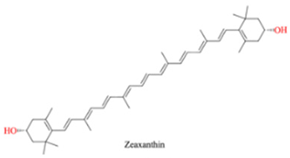 |
Antioxidant, anti-LDL oxidation |
| Lotus leaf | Nuciferine |  |
Lipid regulation and weight loss, antiarrhythmic |
| Honey | Quercetin |  |
Antioxidant, anti-LDL oxidation |
| Medicinal Cuisine Name | Competition and Preparation |
Traditional Functions | Target Population |
Modern Mechanistic Research |
|---|---|---|---|---|
| Hawthorn Fruit-Lotus Leaf Lipid-Regulating Tea | Dried hawthorn fruit fruit 10g, dried lotus leaf 6g; steep in boiling water or decoct for 15 minutes | Promote digestion and resolve accumulation, raise the clear and lower the turbid | Hyperlipidemia, pre-obesity | Hawthorn fruit flavonoids inhibit cholesterol synthesis; nuciferine activates AMPK |
| Milkvetch Root-Angelica sinensis Stewed Chicken | Milkvetch root 30g, Angelica sinensis 10g, chicken 500g; stew for 1.5 hours | Tonify qi and nourish blood, invigorate blood and unblock collaterals | Qi-blood deficiency type, postoperative recovery | Astragaloside IV improves endothelial function; ferulic acid is antithrombotic |
| Glossy Ganoderma-Poria Spirit-Calming Congee | Glossy ganoderma powder 3g, Poria 15g, polished rice 100g; cook as congee | Calm the heart and spirit, strengthen spleen and promote diuresis | Insomnia, anxiety, low immunity | Glossy ganoderma polysaccharides provide immunomodulation; triterpenes are anti-inflammatory |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




