Submitted:
21 April 2026
Posted:
22 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Cultivation of Euglena Gracilis and PFOS Exposure
2.1.1. PFOS Quantification
2.1.2. Visualization of PFOS Accumulation in the Eyespot
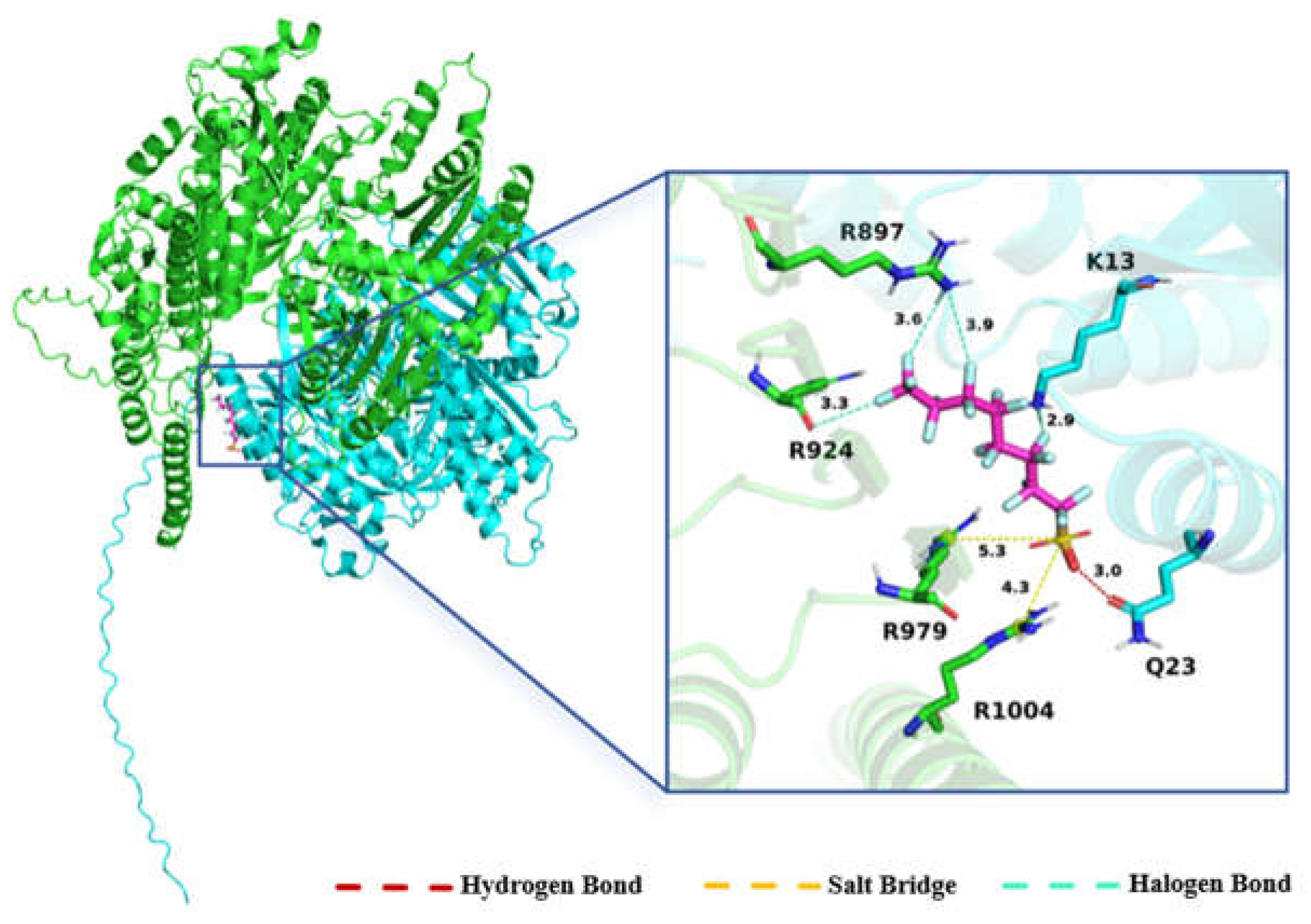
2.1.3. Molecular Docking Analysis
2.2. Phototactic Behavior Assays
2.2.1. Determination of Vertical and Horizontal Swimming Speed

2.2.2. Phototactic Movement Assay
2.2.3. Determination of Flagellar Shedding Rate
2.3. Growth Curve and Photosynthetic Activity Measurement
2.4. Chlorophyll Extraction and Quantification
2.5. Intracellular ATP Level Measurement
2.6. Intracellular Reactive Oxygen Species (ROS) Level Measurement
2.7. Assays of Isolated Chloroplast Function
2.7.1. Photophosphorylation Assay
2.7.2. Mg²⁺-ATPase Activity Assay
2.7.3. Ca²⁺-ATPase Activity Assay
2.7.4. Cytochrome b₆f Complex Activity Assay
2.7.5. Electron Transport Rate Assay
2.7.6. Cyclic Voltammetry (CV) Measurements
2.7.7. Electrochemical Impedance Spectroscopy (EIS) Measurements
2.8. Transcriptomic and Proteomic Analyses
2.8.1. Transcriptome Analysis
2.8.2. Quantitative Proteome Analysis
2.9. Statistical Analysis
3. Results and Discussion
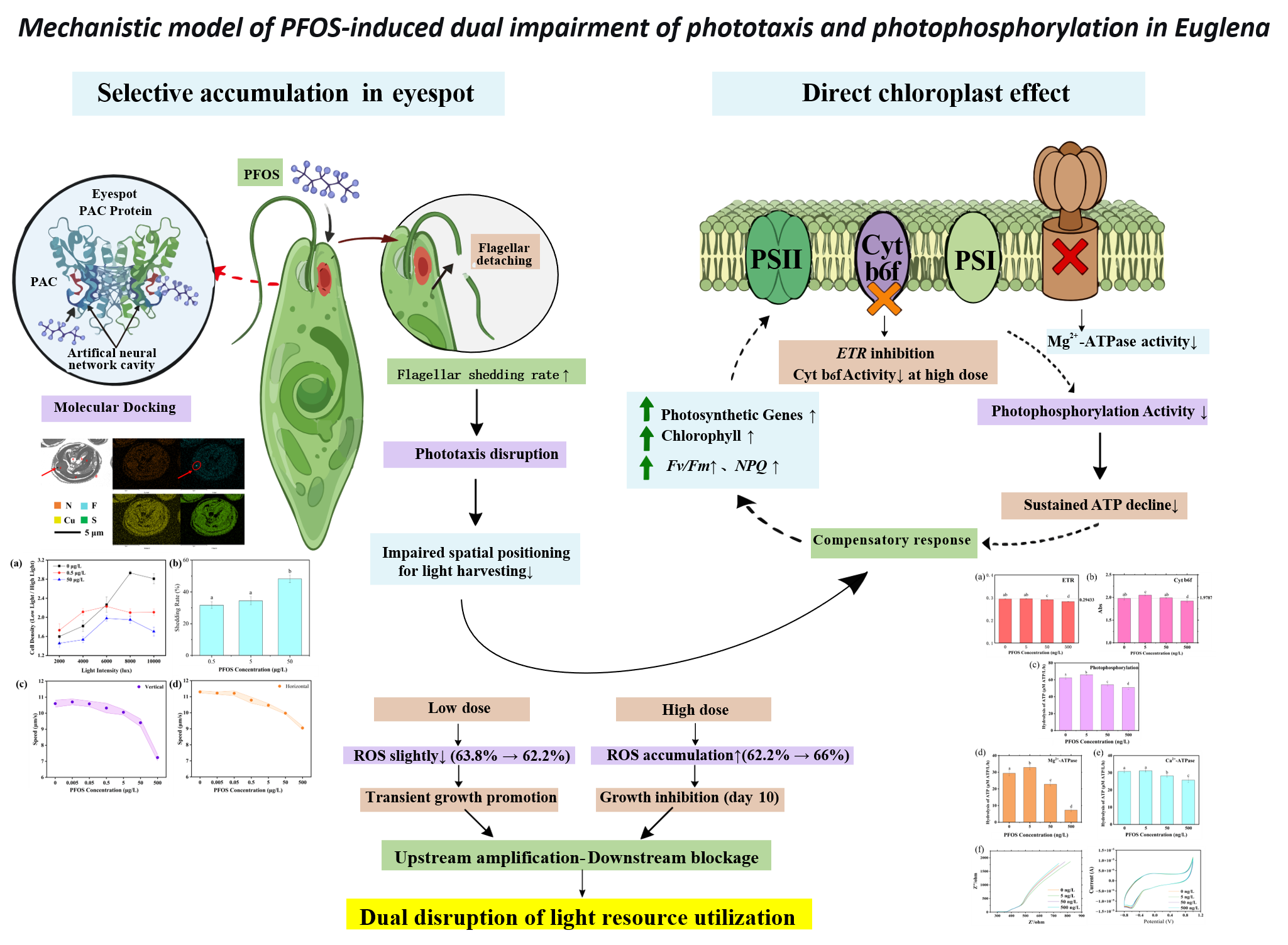
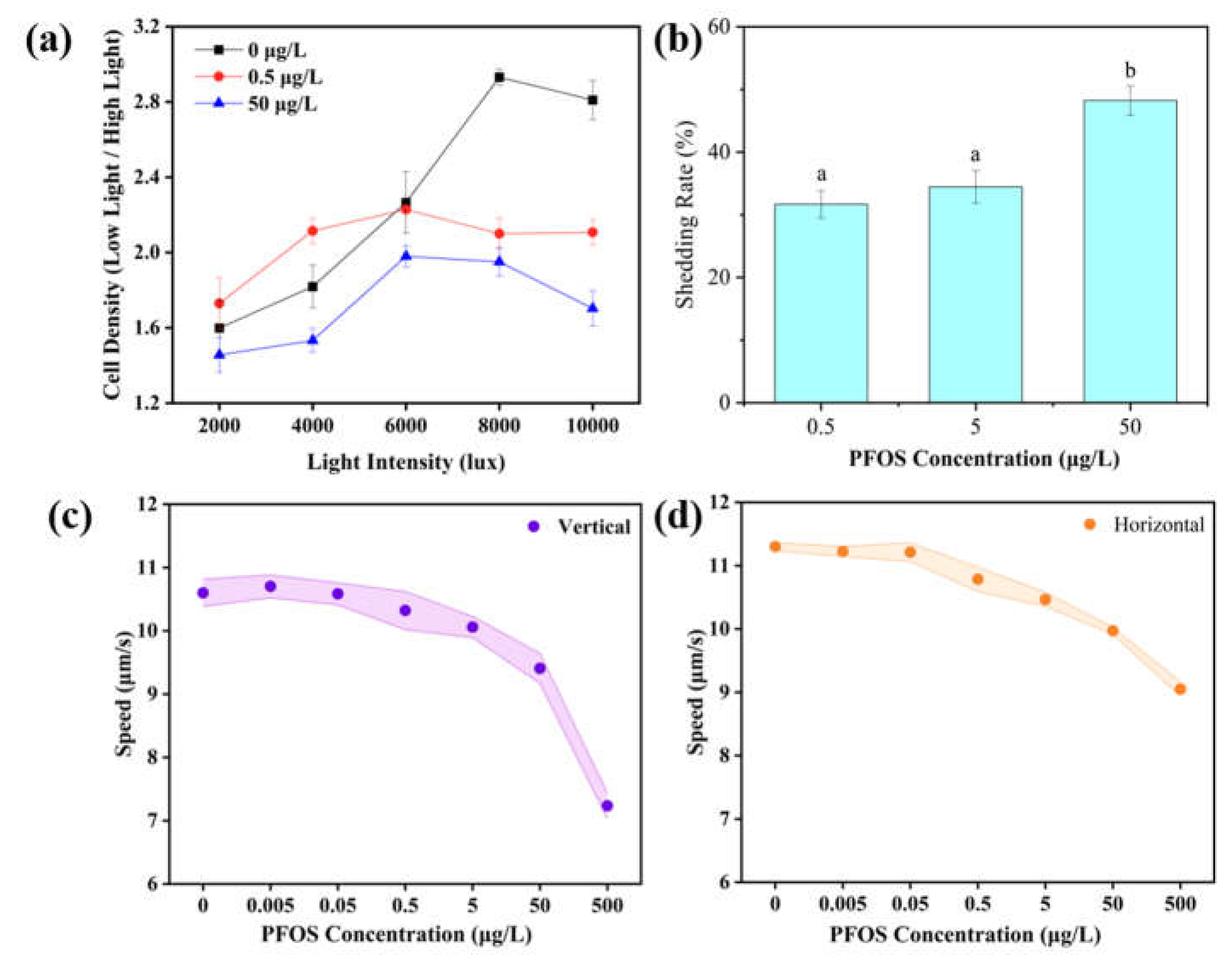
3.1. Accumulation of PFOS in the Eyespot and Its Effects on Phototaxis and Motility
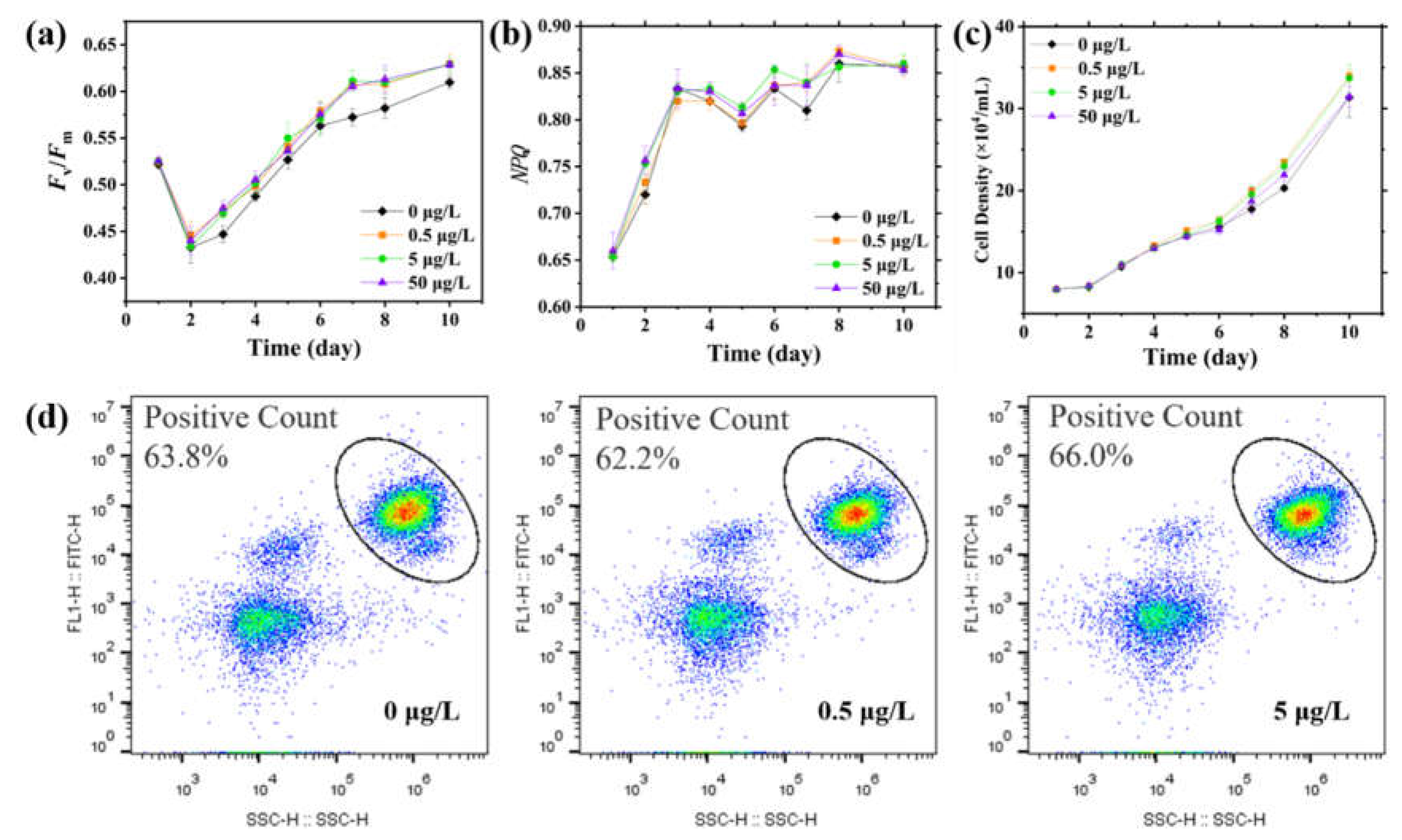
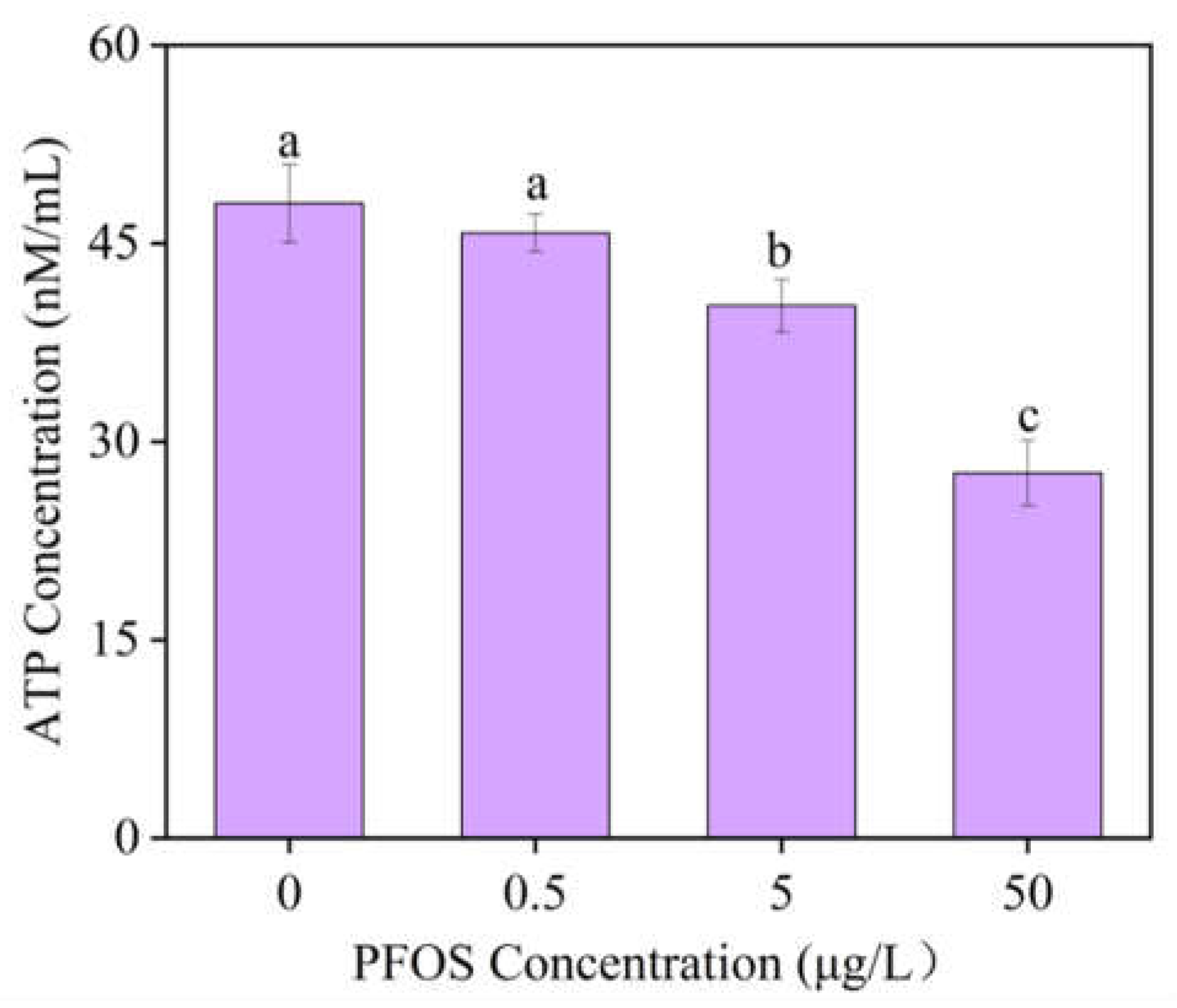
3.2. Changes in Photosynthetic Parameters and Their Decoupling from Energy Metabolism
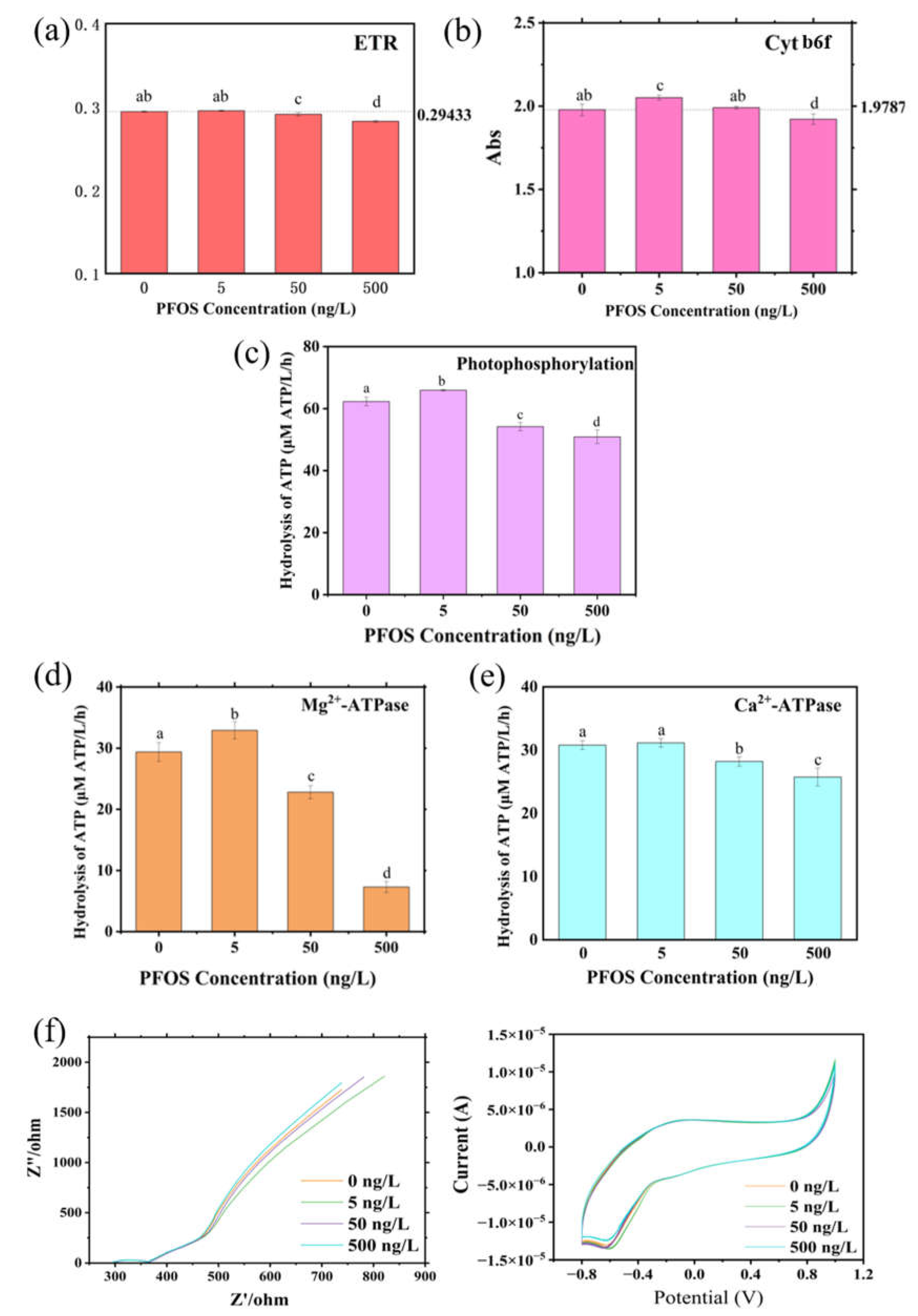
3.3. Isolated Chloroplast Assays Reveal Direct Inhibition of Mg²⁺-ATP Synthase and Secondary Impairment of Electron Transport by PFOS
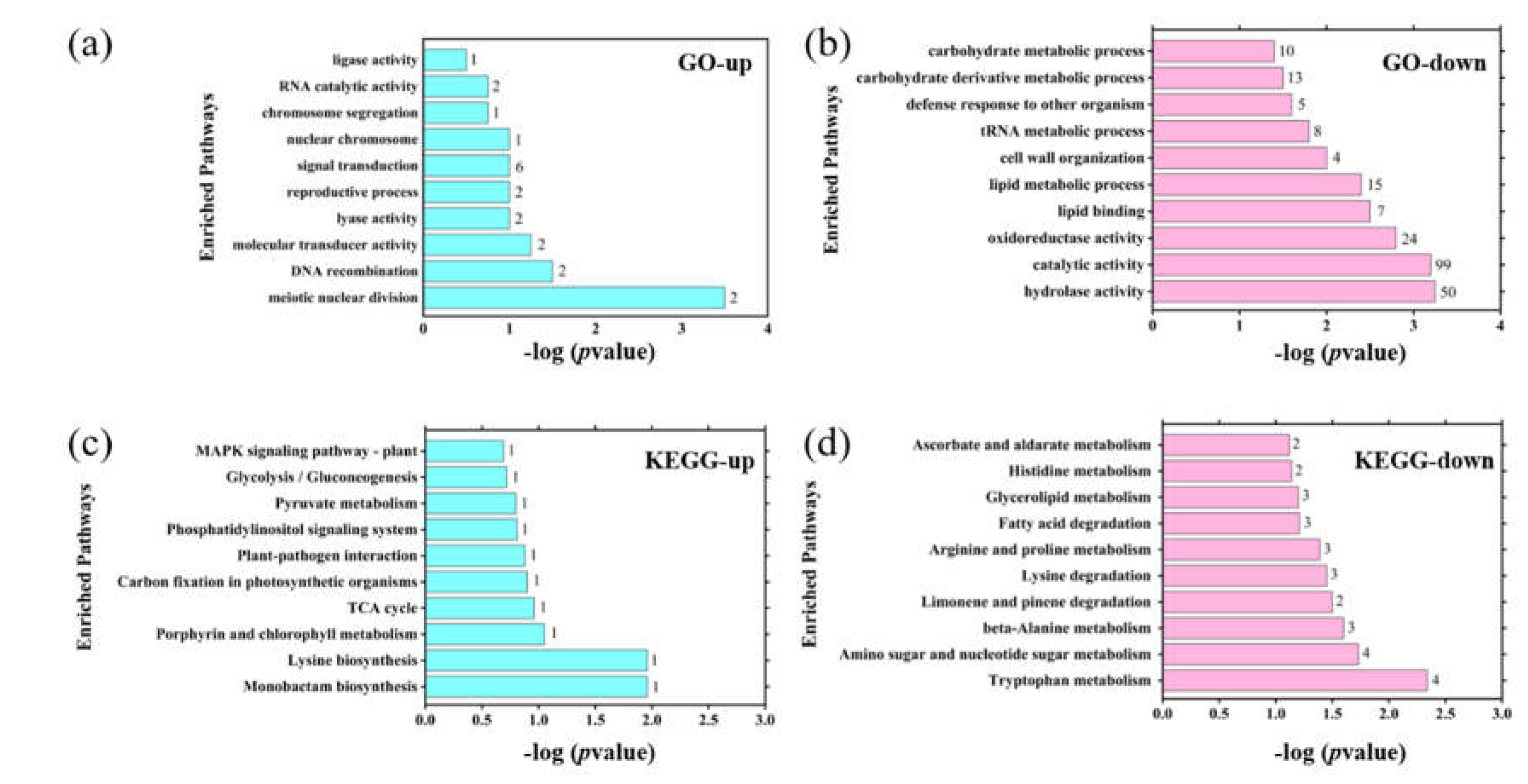
3.4. Integrative Transcriptomic and Proteomic Analyses Reveal Compensatory Responses and Energy Metabolism Imbalance Induced by PFOS
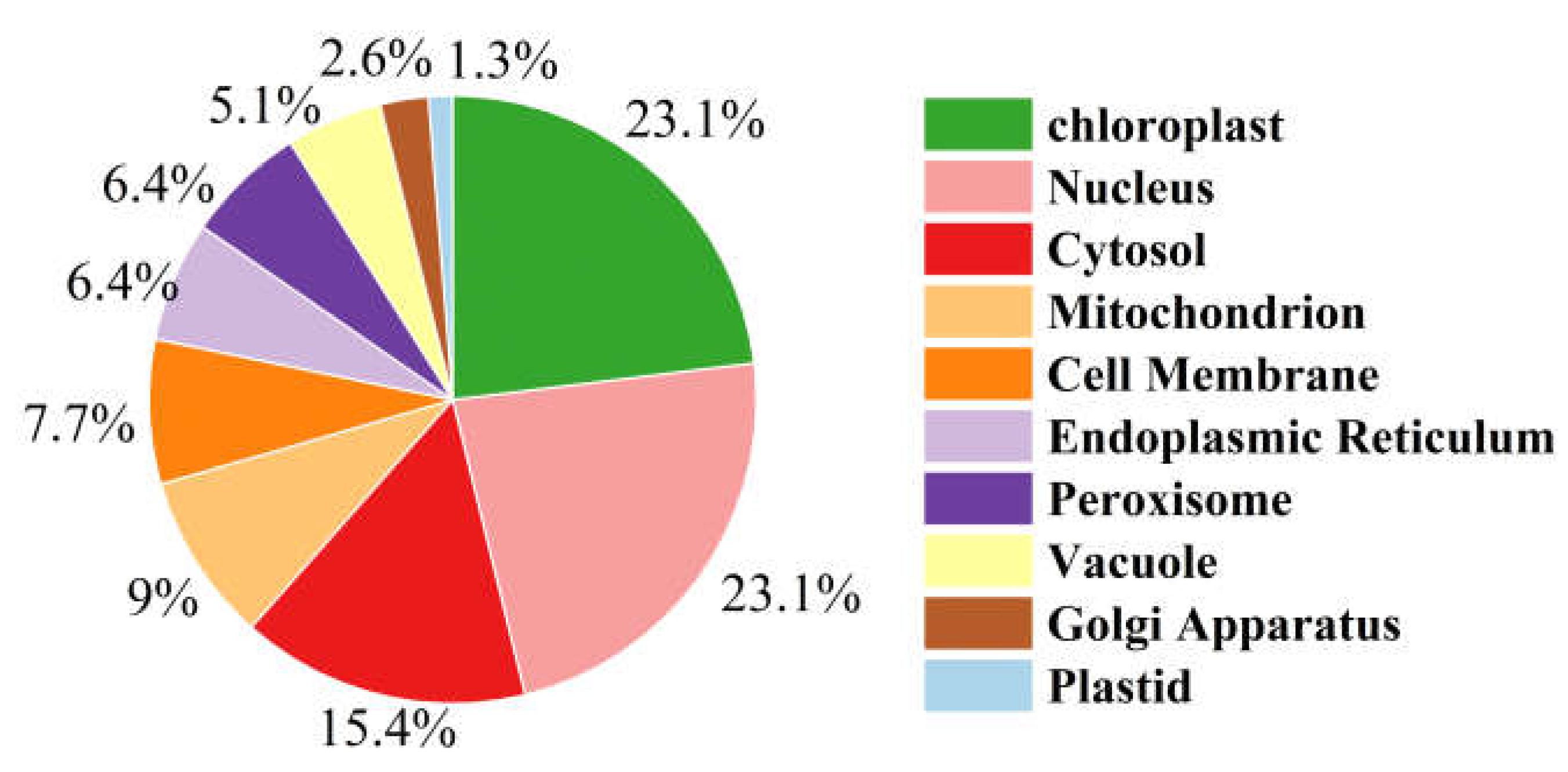
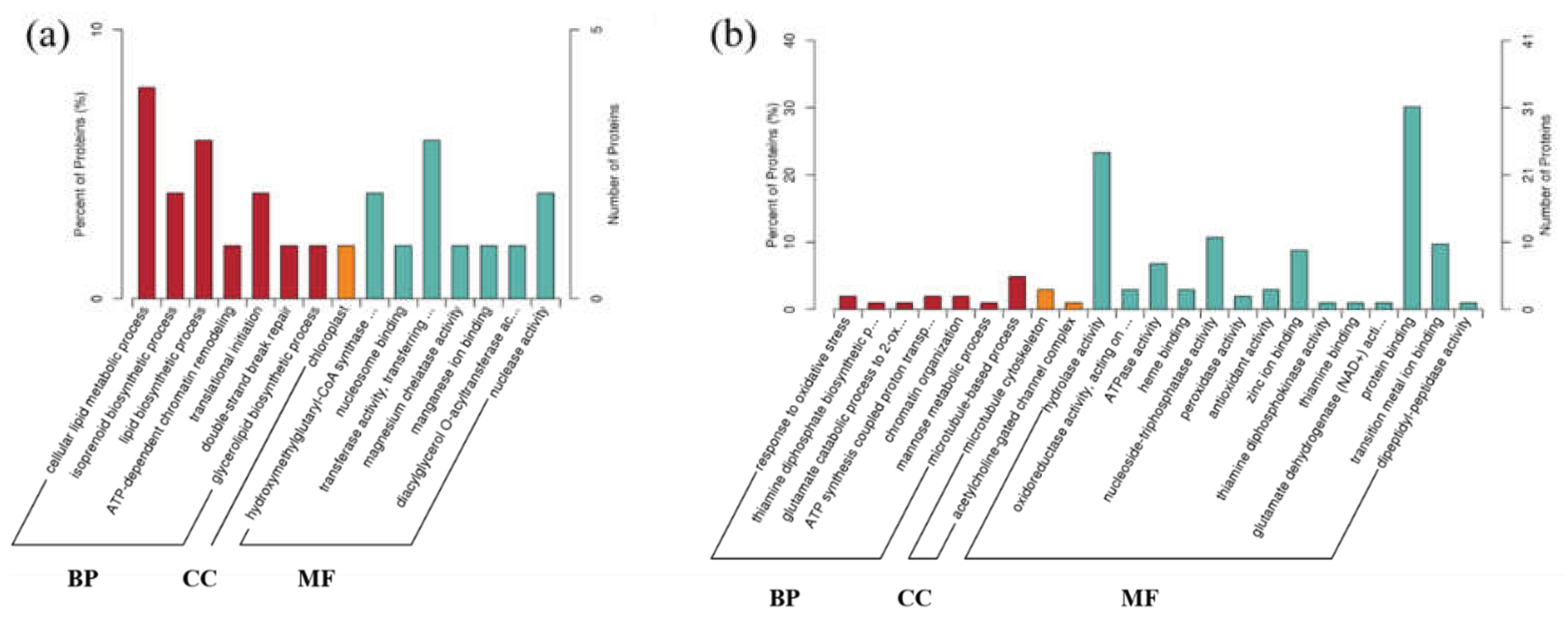
4. Conclusions
Supplementary Materials
Acknowledgments
References
- Ge, X.F.; Wang, K.; Xiao, X.; Chu, C.H.; Zhu, X.Y.; Chen, B.L. Beyond molecules: In situ imaging unveils dom-driven pfas nanoclusters with mitigated phytotoxicity. Environmental Science & Technology 2026, 60, 7393–7404. [Google Scholar] [CrossRef]
- Liao, J.; Sun, B.; Wang, C.; Cao, Z.; Wu, Z.; An, X.; Liang, Z.a.; Huang, X.; Lu, Y. Uptake and cellular responses of microcystis aeruginosa to pfos in various environmental conditions. Ecotoxicology and Environmental Safety 2024. [Google Scholar] [CrossRef]
- Xu, C.; Xu, C.; Zhou, Q.; Shen, C.; Peng, L.; Liu, S.; Yin, S.; Li, F. Spatial distribution, isomer signature and air-soil exchange of legacy and emerging poly- and perfluoroalkyl substances. Environmental Pollution 2024, 343, 123222. [Google Scholar] [CrossRef]
- Chen, Y.; Wei, L.J.; Luo, W.; Jiang, N.; Shi, Y.L.; Zhao, P.; Ga, B.L.; Pei, Z.G.; Li, Y.M.; Yang, R.Q.; et al. Occurrence, spatial distribution, and sources of pfass in the water and sediment from lakes in the tibetan plateau. Journal of Hazardous Materials 2023, 443, 130170. [Google Scholar] [CrossRef]
- Liao, J.M.; Sun, B.; Wang, C.; Cao, Z.W.; Wu, Z.Y.; An, X.P.; Liang, Z.A.; Huang, X.; Lu, Y. Uptake and cellular responses of microcystis aeruginosa to pfos in various environmental conditions. Ecotoxicology and Environmental Safety 2024, 272, 116041. [Google Scholar] [CrossRef]
- Li, W.Z.; Li, H.Z.; Zhang, D.N.; Tong, Y.J.; Li, F.X.; Cheng, F.; Huang, Z.B.; You, J. Legacy and emerging per- and polyfluoroalkyl substances behave distinctly in spatial distribution and multimedia partitioning: A case study in the pearl river, china. Environmental Science & Technology 2022, 56, 3492–3502. [Google Scholar] [CrossRef]
- Meng, L.Y.; Song, B.Y.; Zhong, H.F.; Ma, X.D.; Wang, Y.J.; Ma, D.H.; Lu, Y.; Gao, W.; Wang, Y.W.; Jiang, G.B. Legacy and emerging per- and polyfluoroalkyl substances (pfas) in the bohai sea and its inflow rivers. Environment International 2021, 156, 106735. [Google Scholar] [CrossRef]
- Xiao, S.K.; Wu, Q.; Pan, C.G.; Yin, C.; Wang, Y.H.; Yu, K.F. Distribution, partitioning behavior and potential source of legacy and alternative per- and polyfluoroalkyl substances (pfass) in water and sediments from a subtropical gulf, south china sea. Environmental Research 2021, 201, 111485. [Google Scholar] [CrossRef] [PubMed]
- Jones, P.D.; Hu, W.; De Coen, W.; Newsted, J.L.; Giesy, J.P. Binding of perfluorinated fatty acids to serum proteins. Environmental Toxicology and Chemistry 2003. [Google Scholar] [CrossRef] [PubMed]
- Iseki, M.; Matsunaga, S.; Murakami, A.; Ohno, K.; Shiga, K.; Yoshida, K.; Sugai, M.; Takahashi, T.; Hori, T.; Watanabe, M. A blue-light-activated adenylyl cyclase mediates photoavoidance in euglena gracilis. Nature 2002. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Tan, X.; Hafid, H.S.; Wakisaka, M. Enhancement of biomass yield and lipid accumulation of freshwater microalga euglena gracilis by phenolic compounds from basic structures of lignin. Bioresource Technology 2020. [Google Scholar] [CrossRef] [PubMed]
- Häder, D.-P.; Lebert, M. Photoorientation in photosynthetic flagellates. Methods in Molecular Biology 2009. [Google Scholar]
- Xu, D.; Chen, X.; Shao, B. Oxidative damage and cytotoxicity of perfluorooctane sulfonate on chlorella vulgaris. Bulletin of Environmental Contamination and Toxicology 2016. [Google Scholar] [CrossRef] [PubMed]
- Poothong, S.; Boontanon, S.K.; Boontanon, N. Determination of perfluorooctane sulfonate and perfluorooctanoic acid in food packaging using liquid chromatography coupled with tandem mass spectrometry. Journal of Hazardous Materials 2012. [Google Scholar] [CrossRef]
- Kanna, S.D.; Domonkos, I.; Kobori, T.O.; Dergez, A.; Bode, K.; Nagyapati, S.; Zsiros, O.; Unnep, R.; Nagy, G.; Garab, G.; et al. Salt stress induces paramylon accumulation and fine-tuning of the macro-organization of thylakoid membranes in euglena gracilis cells. Frontiers in Plant Science 2021, 12, 725699. [Google Scholar] [CrossRef]
- Liu, Y.; Yang, X.; Gan, J.; Chen, S.; Xiao, Z.-X.; Cao, Y. Cb-dock2: Improved protein-ligand blind docking by integrating cavity detection, docking and homologous template fitting. Nucleic Acids Research 2022. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Fan, X.; Gao, G.; Beardall, J.; Inaba, K.; Hall-Spencer, J.M.; Xu, D.; Zhang, X.; Han, W.; McMinn, A.; et al. Decreased motility of flagellated microalgae long-term acclimated to co2-induced acidified waters. Nature Climate Change 2020, 10, 561–567. [Google Scholar] [CrossRef]
- Zhu, J.; Tan, X.; Hafid, H.S.; Wakisaka, M. Enhancement of biomass yield and lipid accumulation of freshwater microalga euglena gracilis by phenolic compounds from basic structures of lignin. Bioresource Technology 2021, 321. [Google Scholar] [CrossRef]
- Kim, H.; Van Duong, H.; Kim, E.; Lee, B.G.; Han, S. Effects of phytoplankton cell size and chloride concentration on the bioaccumulation of methylmercury in marine phytoplankton. Environmental Toxicology 2014, 29, 936–941. [Google Scholar] [CrossRef]
- Zheng, S.; Zhang, C.; Shi, K.; Wang, J.; Sun, G.; Hu, Q.; Zhao, F.; Wang, X. Bioaccumulation, subcellular distribution and chemical forms of yttrium in rice seedling. Journal of Rare Earths 2018, 36, 331–336. [Google Scholar] [CrossRef]
- Cara, B.; Lies, T.; Thimo, G.; Robin, L.; Lieven, B. Bioaccumulation and trophic transfer of perfluorinated alkyl substances (pfas) in marine biota from the belgian north sea: Distribution and human health risk implications. Environmental Pollution 2022, 311, 119907. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Sun, J.; Li, P. Exposure routes, bioaccumulation and toxic effects of per- and polyfluoroalkyl substances (pfass) on plants: A critical review. Environment International 2022, 158, 106891. [Google Scholar] [CrossRef] [PubMed]
- Heyno, E.; Ermakova, M.; Lopez-Calcagno, P.E.; Woodford, R.; Brown, K.L.; Matthews, J.S.A.; Osmond, B.; Raines, C.A.; von Caemmerer, S. Rieske fes overexpression in tobacco provides increased abundance and activity of cytochrome b6f. Physiol Plant 2022, 174, e13803. [Google Scholar] [CrossRef]
- U DWIVEDI, R.B.; SHARMA, M. Alteration in the acceptor side of photosystem ii of chloroplast by high light. Journal of Biosciences 1996, 21, 527–533. [Google Scholar] [CrossRef]
- Richter, L.V.; Mansfeldt, C.B.; Kuan, M.M.; Cesare, A.E.; Menefee, S.T.; Richardson, R.E.; Ahner, B.A. Altered microbiome leads to significant phenotypic and transcriptomic differences in a lipid accumulating chlorophyte. Environmental Science & Technology 2018, 52, 6854–6863. [Google Scholar] [CrossRef]
- Gschloessl, B.; Dorkeld, F.; Berges, H.; Beydon, G.; Bouchez, O.; Branco, M.; Bretaudeau, A.; Burban, C.; Dubois, E.; Gauthier, P.; et al. Draft genome and reference transcriptomic resources for the urticating pine defoliator thaumetopoea pityocampa (lepidoptera: Notodontidae). Mol Ecol Resour 2018, 18, 602–619. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Zhang, Z.; Li, M.; Luo, J.; Chen, F.; Gong, Y.; Li, Y.; Wei, Y.; Su, Y.; Kong, L. Transcriptomic profiles of 33 opium poppy samples in different tissues, growth phases, and cultivars. Scientific Data 2019, 6, 66. [Google Scholar] [CrossRef]
- Muntel, J.; Xuan, Y.; Berger, S.T.; Reiter, L.; Bachur, R.; Kentsis, A.; Steen, H. Advancing urinary protein biomarker discovery by data-independent acquisition on a quadrupole-orbitrap mass spectrometer. Journal of Proteome Research 2015, 14, 4752–4762. [Google Scholar] [CrossRef]
- Chiva, C.; Mendes Maia, T.; Panse, C.; Stejskal, K.; Douche, T.; Matondo, M.; Loew, D.; Helm, D.; Rettel, M.; Mechtler, K.; et al. Quality standards in proteomics research facilities: Common standards and quality procedures are essential for proteomics facilities and their users. Science & Society 2021, 22, e52626. [Google Scholar]
- Bischel, H.N.; MacManus-Spencer, L.A.; Luthy, R.G. Noncovalent interactions of long-chain perfluoroalkyl acids with serum albumin. Environmental Science & Technology 2010. [Google Scholar]
- Mao, W.; Li, M.; Xue, X.; Cao, W.; Wang, X.; Xu, F.; Jiang, W. Bioaccumulation and toxicity of perfluorooctanoic acid and perfluorooctane sulfonate in marine algae chlorella sp. Science of the Total Environment 2023. [Google Scholar] [CrossRef] [PubMed]
- Jahanban-Esfahlan, A.; Ostadrahimi, A.; Jahanban-Esfahlan, R.; Roufegarinejad, L.; Tabibiazar, M.; Amarowicz, R. Recent developments in the detection of bovine serum albumin. International Journal of Biological Macromolecules 2019. [Google Scholar] [CrossRef]
- Ozasa, K.; Won, J.; Song, S.; Shinomura, T.; Maeda, M. Phototaxis and photo-shock responses of euglena gracilis under gravitaxis. Algal Research 2019. [Google Scholar] [CrossRef]
- Hu, W.y.; Jones, P.D.; DeCoen, W.; King, L.; Fraker, P.; Newsted, J.; Giesy, J.P. Alterations in cell membrane properties caused by perfluorinated compounds. In Comparative Biochemistry and Physiology C: Toxicology & Pharmacology; 2003. [Google Scholar]
- Scheuerlein, R.; Treml, S.; Thar, B.; Tirlapur, U.K.; Häder, D.-P. Evidence for uv-b-induced DNA degradation in euglena gracilis mediated by activation of metal-dependent nucleases. Journal of Photochemistry and Photobiology B 1995, Biology. [Google Scholar] [CrossRef]
- Davis, S.N.; Klumker, S.M.; Mitchell, A.A.; Coppage, M.A.; Labonté, J.M.; Quigg, A. Life in the pfas lane: The impact of perfluoroalkyl substances on photosynthesis, cellular exudates, nutrient cycling, and composition of a marine microbial community. Science of the Total Environment 2024. [Google Scholar] [CrossRef]
- Homburg, S.V.; Kruse, O.; Patel, A.V. Growth and photosynthetic activity of chlamydomonas reinhardtii entrapped in lens-shaped silica hydrogels. Journal of Biotechnology 2019. [Google Scholar] [CrossRef]
- Calabrese, E.J.; Baldwin, L.A. Hormesis: The dose-response revolution. In Annual Review of Pharmacology and Toxicology; 2002. [Google Scholar]
- Mitchell, P. Coupling of phosphorylation to electron and hydrogen transfer by a chemi-osmotic type of mechanism. Nature 1961. [Google Scholar] [CrossRef] [PubMed]
- Heidarian, F.; Fallah, S.; Pokhrel, L.R.; Rostamnejadi, A. Magnetite nanoparticles (Fe3O4NPs) promote drought tolerance and improve plant health, grain quality and yield in kidney bean (phaseolus vulgaris l.). Science of the Total Environment 2025. [Google Scholar] [CrossRef] [PubMed]
- Johnson, M.P. Structure, regulation and assembly of the photosynthetic electron transport chain. Nature Reviews Molecular Cell Biology 2025. [Google Scholar] [CrossRef]



Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.



