Submitted:
20 April 2026
Posted:
22 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Case Presentation
3. Discussion
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Zarate, Y.A.; Hopkin, R. J. Fabry's disease. Lancet 2008, 372, 1427–1435. [Google Scholar] [CrossRef] [PubMed]
- Germain, D.P. Fabry disease. Orphanet J Rare Dis. 2010, 5, 30. [Google Scholar] [CrossRef] [PubMed]
- Germain, D.P.; Moiseev, S.; Suárez-Obando, F.; Al Ismaili, F.; Al Khawaja, H.; Altarescu, G.; Barreto, F.C.; Haddoum, F.; Hadipour, F.; Maksimova, I.; Kramis, M.; Nampoothiri, S.; Nguyen, K.N.; Niu, D.M.; Politei, J.; Ro, L.S.; Vu Chi, D.; Chen, N.; Kutsev, S. The benefits and challenges of family genetic testing in rare genetic diseases-lessons from Fabry disease. Mol Genet Genomic Med. 2021, 9, e1666. [Google Scholar] [CrossRef] [PubMed]
- Schiffmann, R.; Warnock, D.G.; Banikazemi, M.; Bultas, J.; Linthorst, G.E.; Packman, S.; Sorensen, S.A.; Wilcox, W.R.; Desnick, R.J. Fabry disease: progression of nephropathy, and prevalence of cardiac and cerebrovascular events before enzyme replacement therapy. Nephrol Dial Transplant. 2009, 24, 2102–2111. [Google Scholar] [CrossRef]
- Waldek, S.; Feriozzi, S. Fabry nephropathy: a review - how can we optimize the management of Fabry nephropathy? BMC Nephrol 2014, 15, 72. [Google Scholar] [CrossRef]
- Mursă, A.; Militaru, S.; Rusu, E.; Onciul, S.; Neculae, G.; Adam, R.; Ciobotaru, L.; Stefănescu, V.; Dulămea, A.; Rădoi, V.; Popescu, B.A.; Ismail, G.; Jurcuţ, R. Fabry disease phenotyping in women from the complete Romanian cohort - time for early diagnostic awareness. Rom J Intern Med. 2024, 62, 414–429. [Google Scholar] [CrossRef]
- Wilcox, W.R.; Oliveira, J.P.; Hopkin, R.J.; Ortiz, A.; Banikazemi, M.; Feldt-Rasmussen, U.; Sims, K.; Waldek, S.; Pastores, G.M.; Lee, P.; Eng, C.M.; Marodi, L.; Stanford, K.E.; Breunig, F.; Wanner, C.; Warnock, D.G.; Lemay, R.M.; Germain, D.P. Fabry Registry. Females with Fabry disease frequently have major organ involvement: lessons from the Fabry Registry. Mol Genet Metab. 2008, 93, 112–128. [Google Scholar] [CrossRef]
- Najafian, B.; Svarstad, E.; Bostad, L.; Gubler, M.C.; Tøndel, C.; Whitley, C.; Mauer, M. Progressive podocyte injury and globotriaosylceramide (GL-3) accumulation in young patients with Fabry disease. Kidney Int. 2011, 79, 663–670. [Google Scholar] [CrossRef]
- Tøndel, C.; Bostad, L.; Hirth, A.; Svarstad, E. Renal biopsy findings in children and adolescents with Fabry disease and minimal albuminuria. Am J Kidney Dis. 2008, 51, 767–776. [Google Scholar] [CrossRef]
- Ramaswami, U.; Najafian, B.; Schieppati, A.; Mauer, M.; Bichet, D.G. Assessment of renal pathology and dysfunction in children with Fabry disease. Clin J Am Soc Nephrol. 2010, 5, 365–370. [Google Scholar] [CrossRef]
- Fogo, A.B.; Bostad, L.; Svarstad, E.; Cook, W.J.; Moll, S.; Barbey, F.; Geldenhuys, L.; West, M.; Ferluga, D.; Vujkovac, B.; Howie, A. J.; Burns, A.; Reeve, R.; Waldek, S.; Noël, L.H.; Grünfeld, J.P.; Valbuena, C.; Oliveira, J.P.; Müller, J.; Breunig, F.; Zhang, X.; Warnock, D.G. all members of the International Study Group of Fabry Nephropathy (ISGFN). Scoring system for renal pathology in Fabry disease: report of the International Study Group of Fabry Nephropathy (ISGFN). Nephrol Dial Transplant 2010, 25, 2168–2177. [Google Scholar] [CrossRef]
- Kim, I.Y.; Lee, H.J.; Cheon, C.K. Fabry nephropathy before and after enzyme replacement therapy: important role of renal biopsy in patients with Fabry disease. Kidney Res Clin Pract. 2021, 40, 611–619. [Google Scholar] [CrossRef]
- Rusu, E.E.; Zilisteanu, D.S.; Ciobotaru, L.M.; Gherghiceanu, M.; Procop, A.; Jurcut, R.O.; Dulamea, A.O.; Sorohan, B.M. The Impact of Kidney Biopsy for Fabry Nephropathy Evaluation on Patients’ Management and Long-Term Outcomes: Experience of a Single Center. Biomedicines 2022, 10, 1520. [Google Scholar] [CrossRef] [PubMed]
- Ortiz, A.; Germain, D.P.; Desnick, R.J.; Politei, J.; Mauer, M.; Burlina, A.; Eng, C.; Hopkin, R.J.; Laney, D.; Linhart, A.; Waldek, S.; Wallace, E.; Weidemann, F.; Wilcox, W.R. Fabry disease revisited: Management and treatment recommendations for adult patients. Mol. Genet. Metab. 2018, 123, 416–427. [Google Scholar] [CrossRef] [PubMed]
- Tøndel, C.; Kanai, T.; Larsen, K.K.; Ito, S.; Politei, J.M.; Warnock, D.G.; Svarstad, E. Foot process effacement is an early marker of nephropathy in young classic Fabry patients without albuminuria. Nephron. 2015, 129, 16–21. [Google Scholar] [CrossRef] [PubMed]
- Silva, C.A.B.; Moura-Neto, J.A.; Dos Reis, M.A.; Vieira Neto, O.M.; Barreto, F.C. Renal Manifestations of Fabry Disease: A Narrative Review. Can J Kidney Health Dis. 2021, 8, 2054358120985627. [Google Scholar] [CrossRef]
- Capelli, I.; Martano, L.; Berti, G.M.; Vischini, G.; Lerario, S.; Donadio, V.; Incensi, A.; Aiello, V.; Ciurli, F.; Fabbrizio, B.; Chilotti, S.; Mignani, R.; Pasquinelli, G.; La Manna, G. The Role of Kidney Biopsy in Fabry Disease. Biomedicines 2025, 13, 767. [Google Scholar] [CrossRef]
- Verovnik, F.; Benko, D.; Vujkovac, B.; Linthorst, G.E. Remarkable variability in renal disease in a large Slovenian family with Fabry disease. Eur J Hum Genet. 2004, 12, 678–681. [Google Scholar] [CrossRef]
- Tuttolomondo, A.; Simonetta, I.; Duro, G.; Pecoraro, R.; Miceli, S.; Colomba, P.; Zizzo, C.; Nucera, A.; Daidone, M.; Di Chiara, T.; Scaglione, R.; Della Corte, V.; Corpora, F.; Vogiatzis, D.; Pinto, A. Interfamilial and intrafamilial phenotypic variability in three Sicilian families with Anderson-Fabry disease. Oncotarget 2017, 8, 61415–61424. [Google Scholar] [CrossRef]
- Cammarata, G.; Fatuzzo, P.; Rodolico, M.S.; Colomba, P.; Sicurella, L.; Iemolo, F.; Zizzo, C.; Alessandro, R.; Bartolotta, C.; Duro, G.; Monte, I. High variability of Fabry disease manifestations in an extended Italian family. Biomed Res Int. 2015, 2015, 504784. [Google Scholar] [CrossRef]
- Rigoldi, M.; Concolino, D.; Morrone, A.; Pieruzzi, F.; Ravaglia, R.; Furlan, F.; Santus, F.; Strisciuglio, P.; Torti, G.; Parini, R. Intrafamilial phenotypic variability in four families with Anderson-Fabry disease. Clin Genet. 2014, 86, 258–263. [Google Scholar] [CrossRef]
- Li, J.; Wang, J.; Chen, Y. Phenotypic Diversity on Cardiac Magnetic Resonance in a Han Family With Fabry Disease. CJC Open 2025, 7, 736–739. [Google Scholar] [CrossRef]
- Altarescu, G.M.; Goldfarb, L.G.; Park, K.Y.; Kaneski, C.; Jeffries, N.; Litvak, S.; Nagle, J.W.; Schiffmann, R. Identification of fifteen novel mutations and genotype-phenotype relationship in Fabry disease. Clin Genet. 2001, 60, 46–51. [Google Scholar] [CrossRef]
- Mignani, R.; Moschella, M.; Cenacchi, G.; Donati, I.; Flachi, M.; Grimaldi, D.; Cerretani, D.; Giovanni, P.; Montevecchi, M.; Rigotti, A.; Ravasio, A. Different renal phenotypes in related adult males with Fabry disease with the same classic genotype. Mol Genet Genomic Med. 2017, 5, 438–442. [Google Scholar] [CrossRef]
- Militaru, S.; Adam, R.; Dorobantu, L.; Ferrazzi, P.; Iascone, M.; Radoi, V.; Ismail, G.; Popescu, B.A.; Jurcut, R. Rare presentation and wide intrafamilial variability of Fabry disease: A case report and review of the literature. Anatol J Cardiol. 2019, 22, 154–158. [Google Scholar] [CrossRef]
- Germain, D.P.; Charrow, J.; Desnick, R.J.; Guffon, N.; Kempf, J.; Lachmann, R.H.; Lemay, R.; Linthorst, G.E.; Packman, S.; Scott, C.R.; Waldek, S.; Warnock, D.G.; Weinreb, N.J.; Wilcox, W.R. Ten-year outcome of enzyme replacement therapy with agalsidase beta in patients with Fabry disease. J Med Genet. 2015, 52, 353–358. [Google Scholar] [CrossRef] [PubMed]
- Biegstraaten, M.; Arngrímsson, R.; Barbey, F.; Boks, L.; Cecchi, F.; Deegan, P.B.; Feldt-Rasmussen, U.; Geberhiwot, T.; Germain, D.P.; Hendriksz, C.; Hughes, D.A.; Kantola, I.; Karabul, N.; Lavery, C.; Linthorst, G. E.; Mehta, A.; van de Mheen, E.; Oliveira, J.P.; Parini, R.; Ramaswami, U.; Rudnicki, M.; Serra, A.; Sommer, C.; Sunder-Plassmann, G.; Svarstad, E.; Sweeb, A.; Terryn, W.; Tylki-Szymanska, A.; Tøndel, C.; Vujkovac, B.; Weidemann, F.; Wijburg, F.A.; Woolfson, P.; Hollak, C.E.M. Recommendations for initiation and cessation of enzyme replacement therapy in patients with Fabry disease: The European Fabry Working Group consensus document. Orphanet J. Rare Dis. 2015, 10, 36. [Google Scholar] [CrossRef] [PubMed]
- Singh, J.; Santosh, P.; Ramaswami, U. Epigenetic Mechanisms in Fabry Disease: A Thematic Analysis Linking Differential Methylation Profiles and Genetic Modifiers to Disease Phenotype. Curr. Issues Mol. Biol. 2025, 47, 855. [Google Scholar] [CrossRef] [PubMed]
- Altarescu, G.; Murik, O.; Jurcut, R.; Rusu, E.E.; Mursa, A.; Mann, T.; Eldar-Yedidia, Y.; Zeevi, D.A. Whole blood transcriptomic profiling in the interpretation of variable phenotype presentation in Fabry disease”. Mol Genet Metab. 2022, 138, 6–6. [Google Scholar] [CrossRef]
- Breznik, N.; Levstek, T.; Vujkovac, B.; Cokan Vujkovac, A.; Trebušak Podkrajšek, K. Transcriptomic Approach in Understanding Fabry Nephropathy: A Review of the Literature and Proof-of-Concept. Genes 2025, 16, 601. [Google Scholar] [CrossRef]
- Delaleu, N.; Marti, H.P.; Philipp, S.; Sekulic, M.; Osman, T.; Tøndel, C.; Skrunes, R.; Leh, S.; Svarstad, E.; Nowak, A.; Gaspert, A.; Rusu, E.; Kwee, I.; Rinaldi, A.; Flatberg, A.; Eikrem, O. Systems analyses of the Fabry kidney transcriptome and its response to enzyme replacement therapy identified and cross-validated enzyme replacement therapy-resistant targets amenable to drug repurposing. Kidney Int. 2023, 104, 803–819. [Google Scholar] [CrossRef]
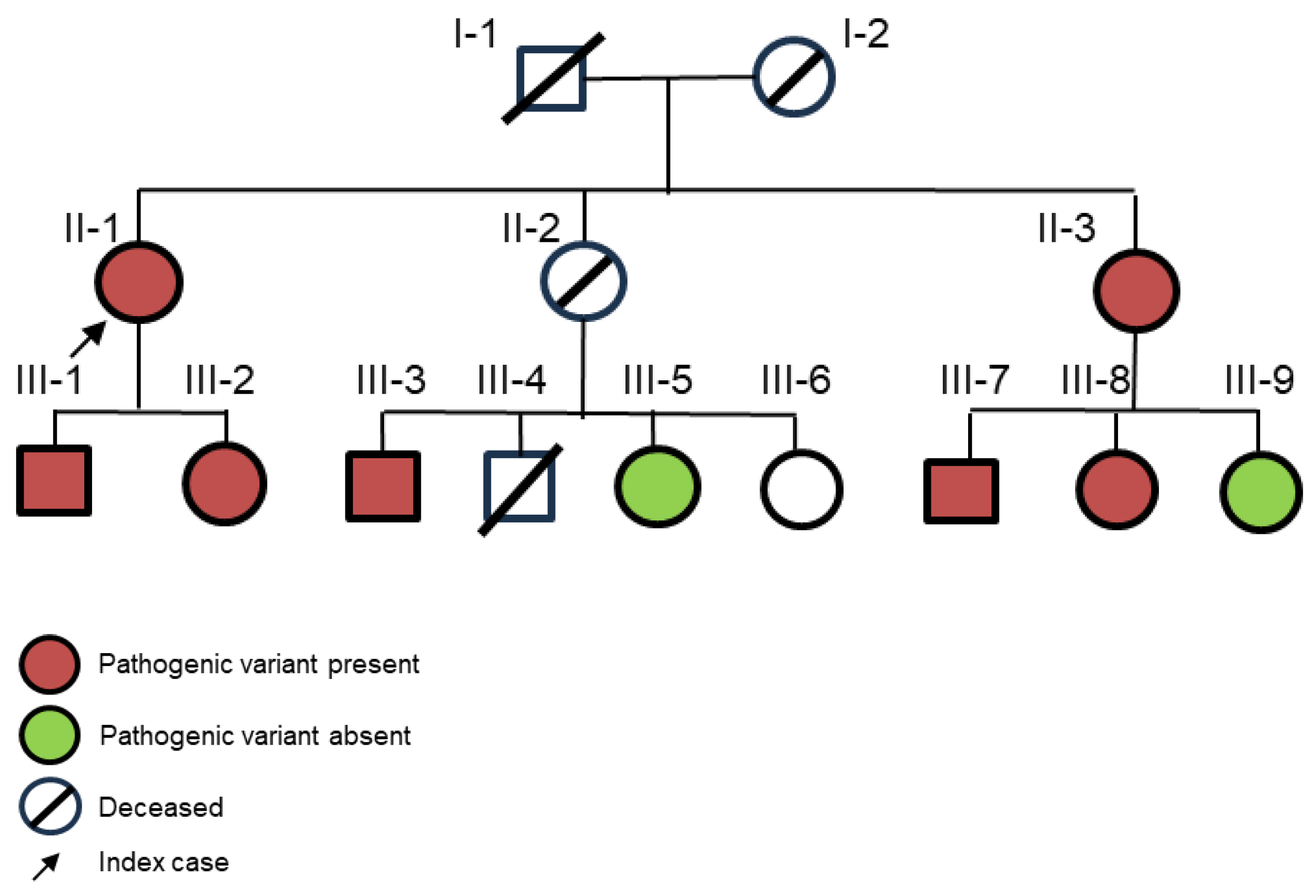

| N | II-1 | II-3 | III-1 | III-2 | III-3 | III-7 | III-8 | |
| Gender | Female | Female | Male | Female | Male | Male | Female | |
| Fabry features | Age at onset, years | 43 | 45 | 12 | 28 | 9 | 17 | |
| Age at genetic test, years | 49 | 45 | 29 | 30 | 29 | 9 | 17 | |
| α-GLA activity (µmol/l/h) | 1.2* | 0.4 | 0.0 | 0.7 | 0.0 | 0.0 | 0.5 | |
| Lyso-GL-3 (ng/ml) | 6.7 | 10.7 | 129.2 | 3.9 | 68.2 | 101.1 | 4.4 | |
| Total MSSI at baseline, points | 37 | 14 | 19 | 10 | 30 | 9 | 3 | |
| Renal manifestations at kidney biopsy |
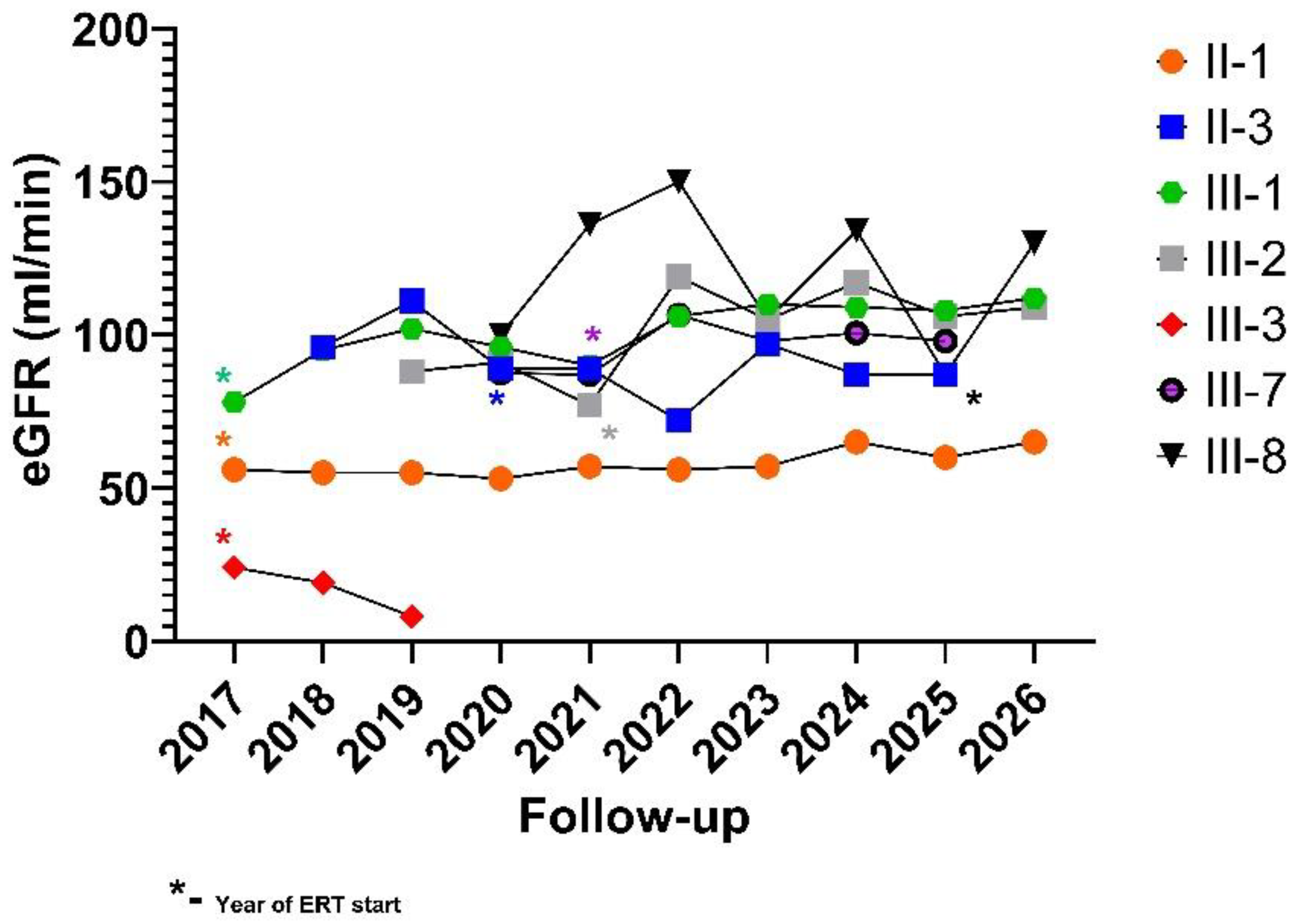
eGFR, ml/min/1.73m2 | 56 | 96 | 78 | 88 | 24 | 87.5 | 135 |
| CKD stage | 3 | 1 | 2 | 2 | 4 | 2 | 1 | |
| UACR (mg/g) | 100 | 10 | 30 | 10 | 300 | 10 | 10 | |
| Proteinuria (g/24h) | 0.4 | 0.1 | 0.2 | 0.2 | 1.7 | 0.08 | 0.08 | |
| Hypertension | Yes | No | No | No | Yes | No | No | |
| Kidney biopsy | Age at kidney biopsy | 50 | 46 | 29 | 30 | NA | 9 | 19 |
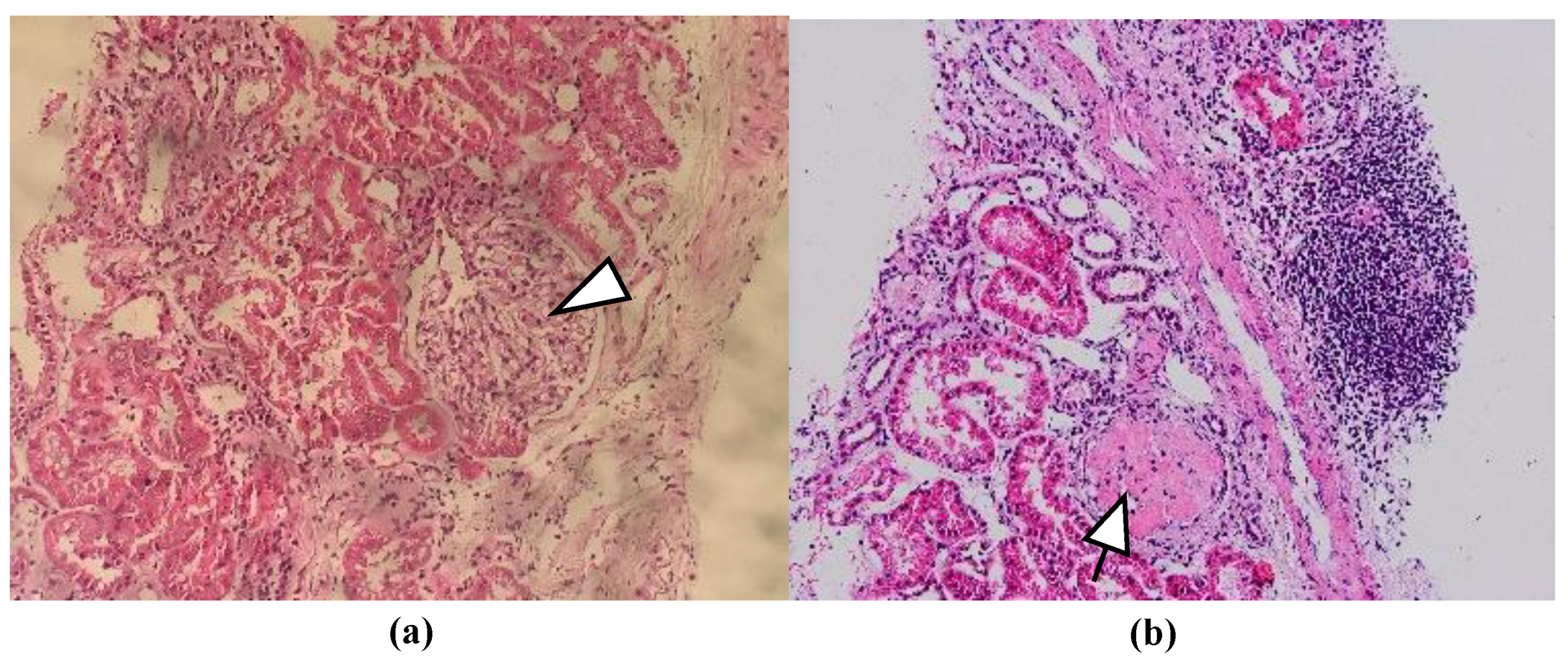
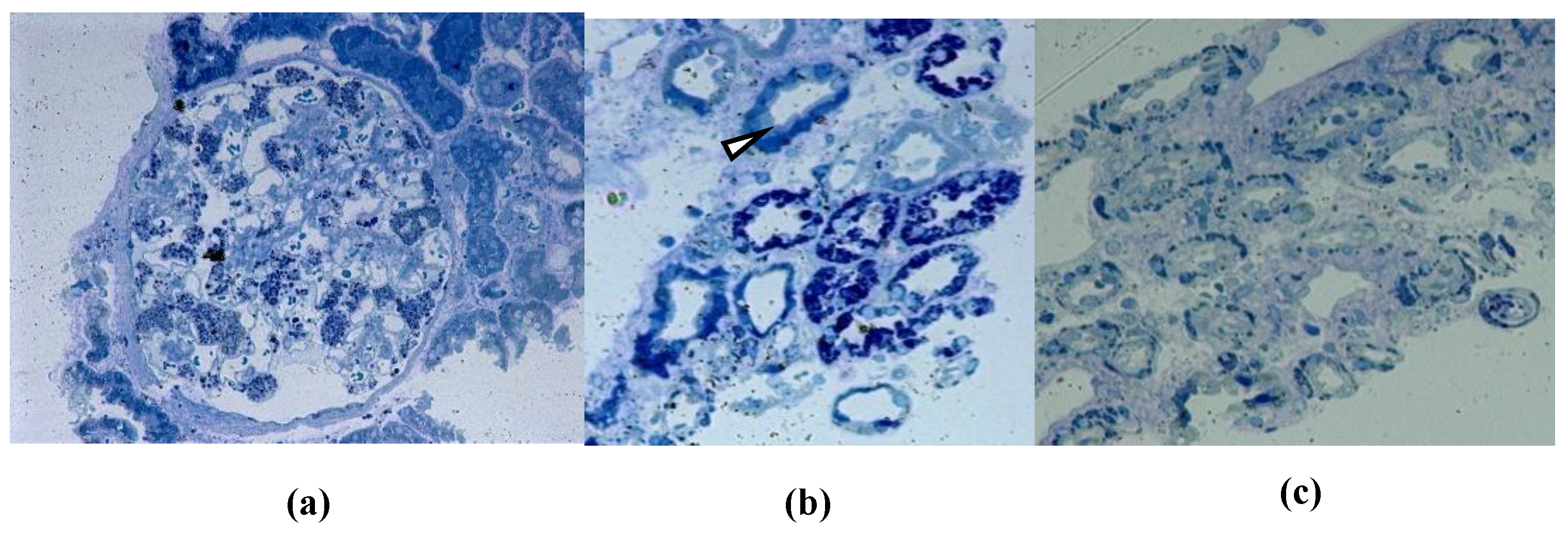
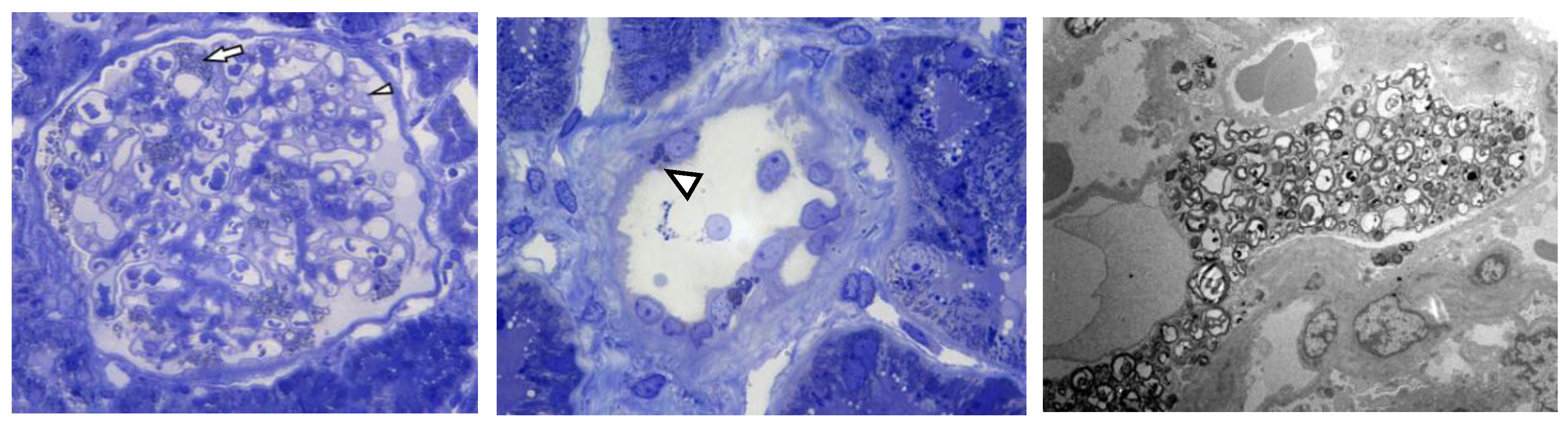
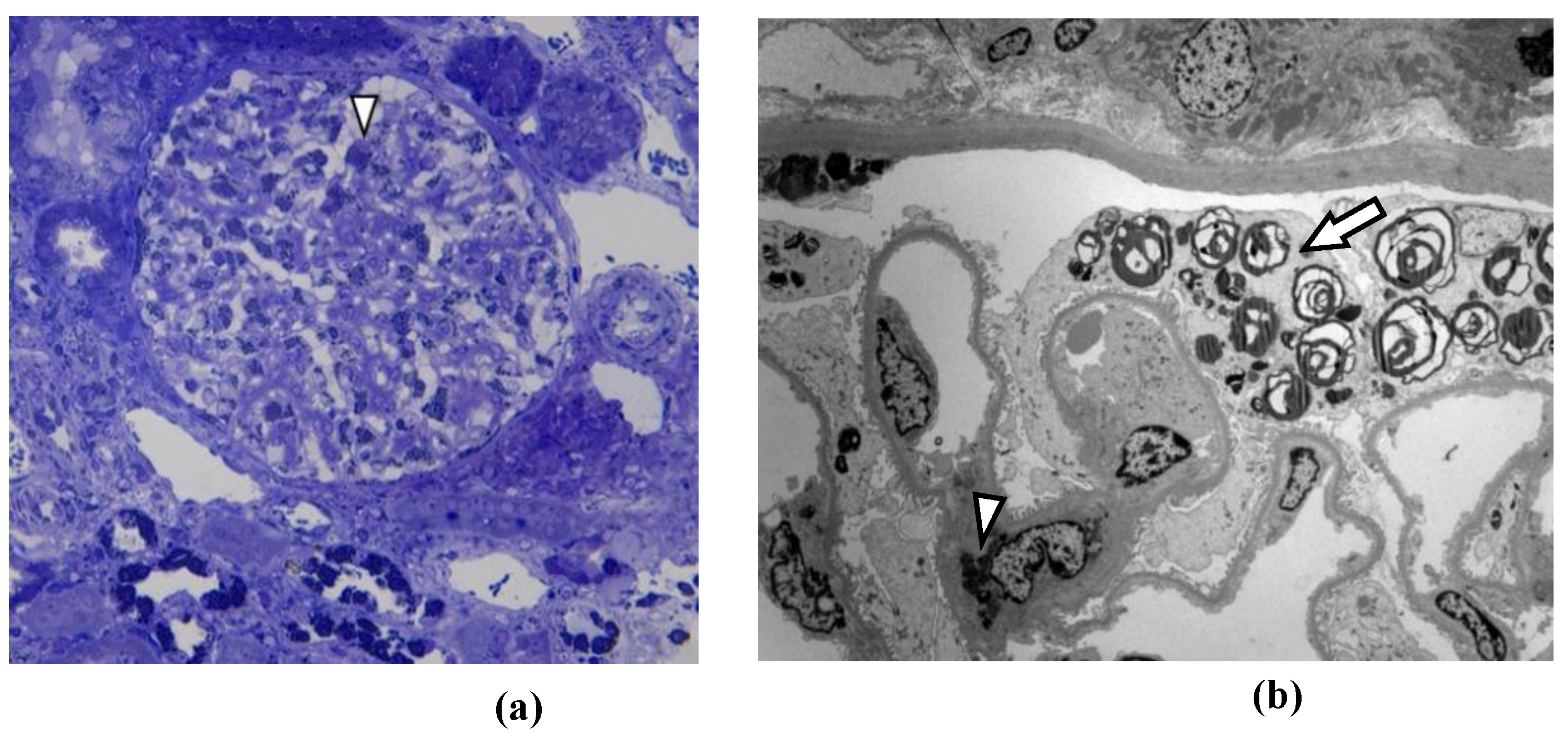
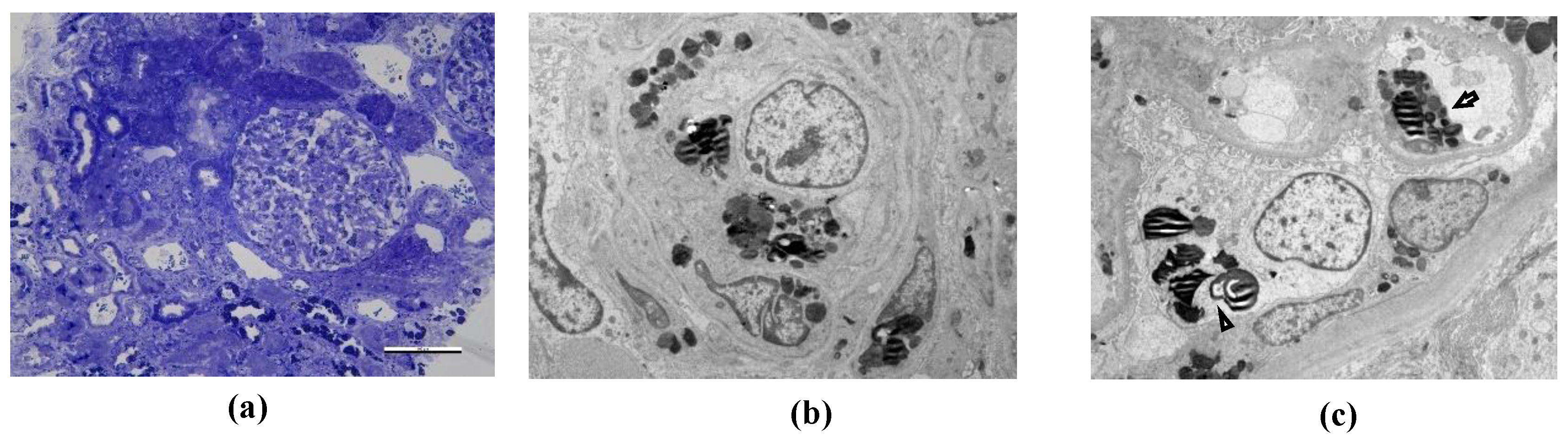
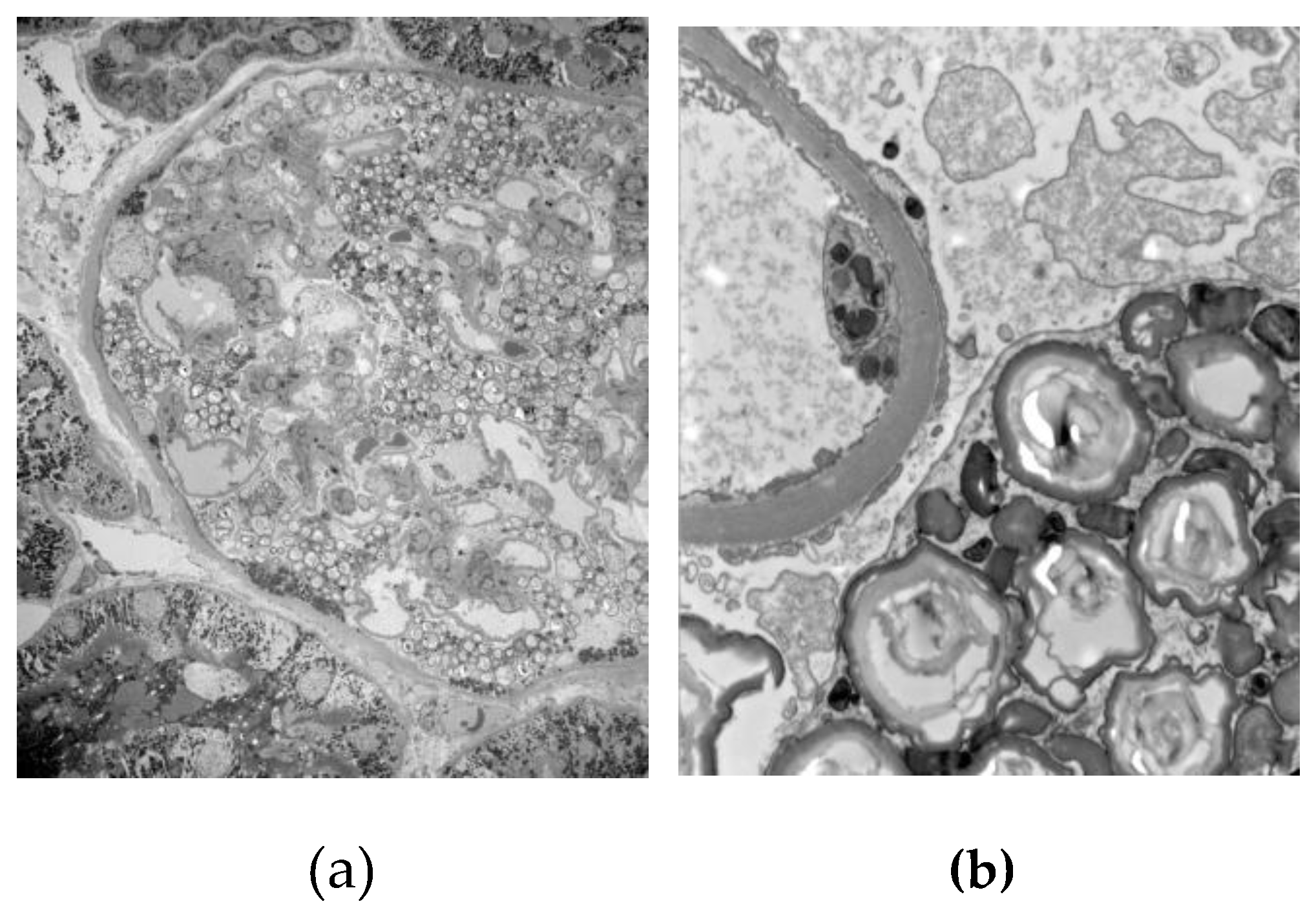
| Podocyte GL-3 deposits | + | + | + | + | + | + | ||
| Tubular GL-3 deposits | + | + | + | + | + | + | ||
| Glomerular endothelial cells GL-3 deposits | + | + | + | + | + | + | ||
| Podocyte foot effacement at EM | NA | No | Segmental | Segmental | Segmental | Segmental | ||
| Segmental sclerosis | + | - | + | - | - | - | ||
| Global sclerosis | + | - | - | - | - | - | ||
| Fibrosis | + | - | - | - | - | - | ||
| Tubular atrophy | + | - | - | - | - | - | ||
| Vasculopathy | + | - | - | - | - | - | ||
| Hypertrophic cardiomyopathy | Yes | No | No | No | Yes | Mild LVH | No | |
| Cardiac MRI changes | HOCM | No | No | No | NA | low T1 | No | |
| Extrarenal phenotype at baseline |
Hypohidrosis | Yes | Yes | Yes | Yes | Yes | No | No |
| Acroparesthesia | Yes | Yes | Yes | Yes | Yes | Yes | No | |
| Cornea verticillata | Yes | Yes | Yes | Yes | Yes | NA | Yes | |
| Cerebral MRI changes | Yes | Yes | No | No | No | No | No | |
| Angiokeratoma | Yes | Yes | Yes | No | Yes | Yes | No | |
| Concomitant disease | Type 1 DM |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




