Submitted:
17 April 2026
Posted:
20 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Conceptual and Mechanistic Foundations of Hormesis
2.1. Beyond Biphasic Curves: Rethinking Hormesis
2.2. Mitohormesis and Redox Signaling
2.3. Network-Level Integration
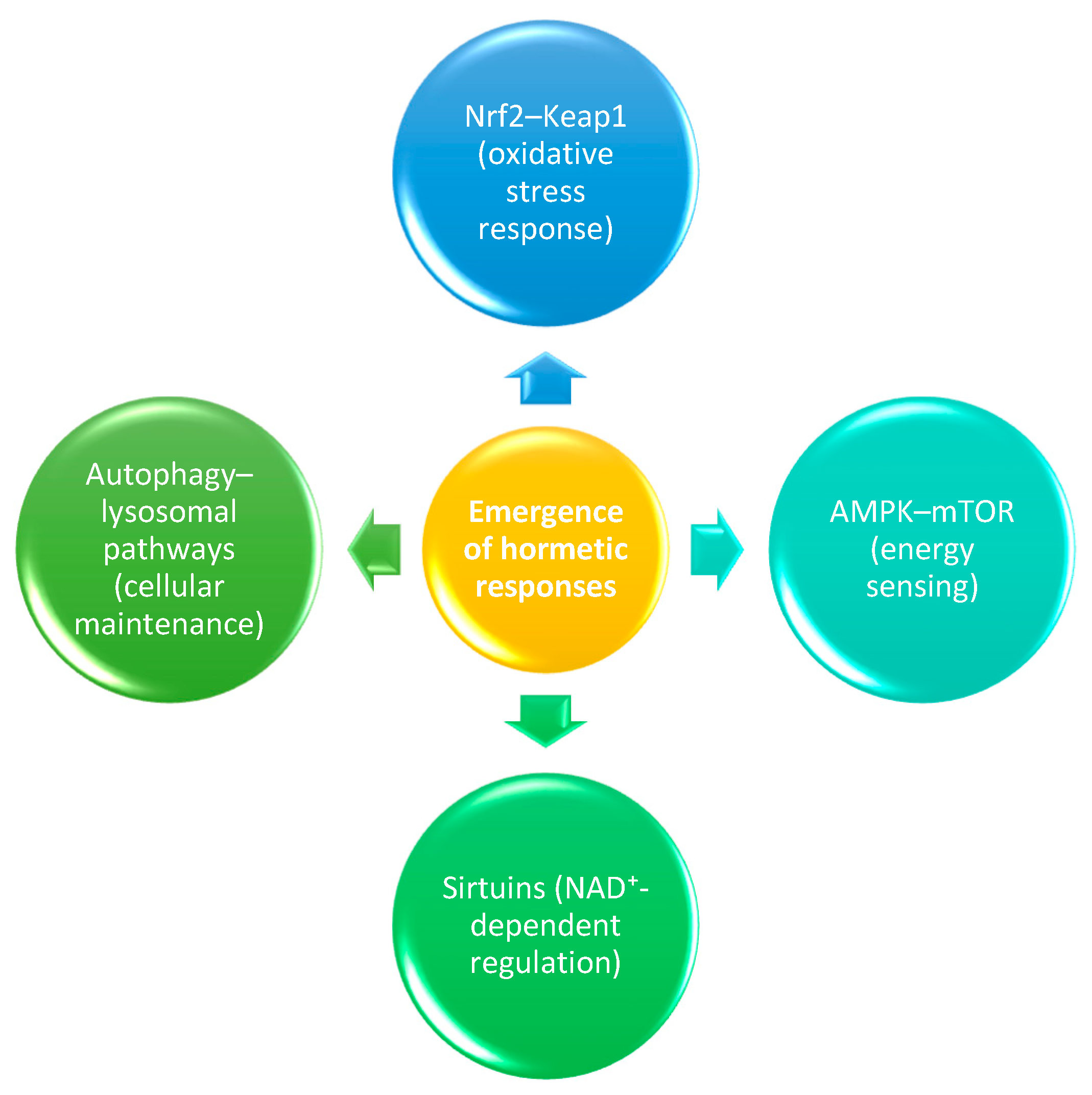
3. Molecular Mechanisms
3.1. Nrf2 Signaling: Central but Context-Dependent
3.2. Sirtuins: Metabolic Sensors or Longevity Regulators?
3.3. Autophagy: A Convergent Mechanism
4. Key Hormetic Interventions in Aging: Mechanistic Depth and Critical Integration
4.1. Physical Exercise: A Prototypical Hormetic Stimulus
4.2. Caloric Restriction and Dietary Stress: Robust but Context-Dependent
4.3. Thermal Stress: Emerging but Heterogeneous Evidence
4.3.1. Heat Exposure
4.3.2. Cold Exposure
4.3.3. Critical Perspective
4.4. Pharmacological Hormetic Interventions
4.5. Clinical Outcomes of Hormetic Interventions
5. Comparative and Integrative Perspective
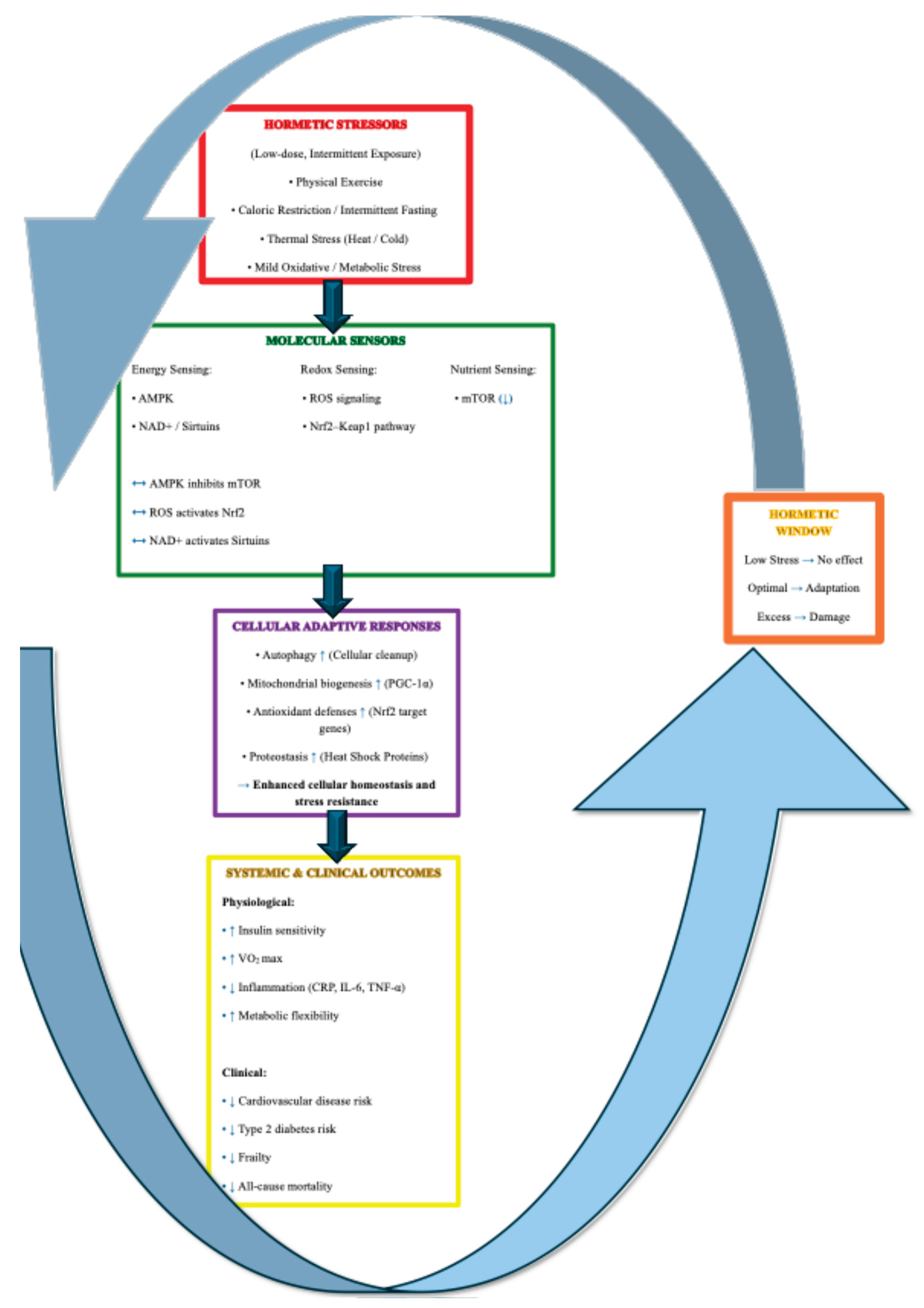
5.1. A Multilevel Model of Hormetic Adaptation in Aging
6. Discussion: Toward a Critical and Integrative Framework of Hormesis in Aging
6.1. Hormesis as a Unifying but Overextended Concept
6.2. The Antioxidant Paradox and Redox Complexity
6.3. Sirtuins and the Longevity Debate
6.4. Caloric Restriction vs. Intermittent Fasting: Mechanistic Overlap or Distinction?
6.5. Decline of Hormetic Capacity with Age
6.6. Toward Precision Hormesis
6.7. Hormesis in Neurodegenerative Diseases
6.8. Hormesis in Dementia
6.9. Hormesis in Cancer
6.10. Hormesis in Alzheimer’s Disease
6.11. Hormesis in Parkinson’s Disease
6.12. Hormesis in Cardiovascular Diseases
7. Future Directions and Open Questions
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Aman, Y.; Schmauck-Medina, T.; Hansen, M.; Morimoto, R.I.; Simon, A.K.; Bjedov, I.; Palikaras, K.; Simonsen, A.; Johansen, T.; Tavernarakis, N.; Rubinsztein, D.C.; Partridge, L.; Kroemer, G.; Labbadia, J.; Fang, E.F. Autophagy in healthy aging and disease. Nature Aging 2021, 1(8), 634–650. [Google Scholar] [CrossRef]
- Anton, S.D.; Moehl, K.; Donahoo, W.T.; Marosi, K.; Lee, S.A.; Mainous, A.G., 3rd; Leeuwenburgh, C.; Mattson, M.P. Flipping the Metabolic Switch: Understanding and Applying the Health Benefits of Fasting. Obesity (Silver Spring, Md.) 2018, 26(2), 254–268. [Google Scholar] [CrossRef]
- Bjelakovic, G.; Nikolova, D.; Gluud, L.L.; Simonetti, R.G.; Gluud, C. Antioxidant supplements for prevention of mortality in healthy participants and patients with various diseases. The Cochrane Database of Systematic Reviews 2012, 2012(3), CD007176. [Google Scholar] [CrossRef]
- Blair, S.N.; Kampert, J.B.; Kohl, H.W., 3rd; Barlow, C.E.; Macera, C.A.; Paffenbarger, R.S., Jr.; Gibbons, L.W. Influences of cardiorespiratory fitness and other precursors on cardiovascular disease and all-cause mortality in men and women. JAMA 1996, 276(3), 205–210. [Google Scholar] [CrossRef]
- Blondin, D.P.; Labbé, S.M.; Tingelstad, H.C.; Noll, C.; Kunach, M.; Phoenix, S.; Guérin, B.; Turcotte, E.E.; Carpentier, A.C.; Richard, D.; Haman, F. Increased brown adipose tissue oxidative capacity in cold-acclimated humans. The Journal of Clinical Endocrinology & Metabolism 2014, 99(3), E438–E446. [Google Scholar] [CrossRef] [PubMed]
- Booth, F.W.; Roberts, C.K.; Laye, M.J. Lack of exercise is a major cause of chronic diseases. Comprehensive Physiology 2012, 2(2), 1143–1211. [Google Scholar] [CrossRef]
- Bruno, G.; La Monica, M.; Ziegenfuss, T.N. Effects of Spermidine-Rich Rice Germ Extract Supplement on Biomarkers of Healthy Aging and Autophagy-Proof-of-Concept Pilot Study. Alternative Therapies in Health and Medicine 2025, 31(6), 9–13. [Google Scholar] [PubMed]
- Bruns, D.R.; Drake, J.C.; Biela, L.M.; Peelor, F.F., 3rd; Miller, B.F.; Hamilton, K.L. Nrf2 Signaling and the Slowed Aging Phenotype: Evidence from Long-Lived Models. Oxidative Medicine and Cellular Longevity 2015, 2015, 732596. [Google Scholar] [CrossRef]
- Burnett, C.; Valentini, S.; Cabreiro, F.; Goss, M.; Somogyvári, M.; Piper, M.D.; Hoddinott, M.; Sutphin, G.L.; Leko, V.; McElwee, J.J.; Vazquez-Manrique, R.P.; Orfila, A.M.; Ackerman, D.; Au, C.; Vinti, G.; Riesen, M.; Howard, K.; Neri, C.; Bedalov, A.; Kaeberlein, M.; Gems, D. Absence of effects of Sir2 overexpression on lifespan in C. elegans and Drosophila. Nature 2011, 477(7365), 482–485. [Google Scholar] [CrossRef] [PubMed]
- Burtscher, J.; Samaja, M. Healthy Aging at Moderate Altitudes: Hypoxia and Hormesis. Gerontology 2024, 70(11), 1152–1160. [Google Scholar] [CrossRef]
- Butterfield, D.A.; Boyd-Kimball, D.; Reed, T.T. Cellular Stress Response (Hormesis) in Response to Bioactive Nutraceuticals with Relevance to Alzheimer Disease. Antioxidants & Redox Signaling 2023, 38(7-9), 643–669. [Google Scholar] [CrossRef]
- Calabrese, E.J.; Baldwin, L.A. Defining hormesis. Human & Experimental Toxicology 21 2002, 91–97. [Google Scholar] [CrossRef]
- Calabrese, E.J.; Mattson, M.P. How does hormesis impact biology, toxicology, and medicine? NPJ Aging and Mechanisms of Disease 3 2017, 13. [Google Scholar] [CrossRef] [PubMed]
- Calabrese, E.J.; Nascarella, M.; Pressman, P.; Hayes, A.W.; Dhawan, G.; Kapoor, R.; Calabrese, V.; Agathokleous, E. Hormesis determines lifespan. Ageing Research Reviews 2024, 94, 102181. [Google Scholar] [CrossRef] [PubMed]
- Cannon, B.; Nedergaard, J. Brown adipose tissue: function and physiological significance. Physiological Reviews 2004, 84(1), 277–359. [Google Scholar] [CrossRef]
- Cantó, C.; Gerhart-Hines, Z.; Feige, J.N.; Lagouge, M.; Noriega, L.; Milne, J.C.; Elliott, P.J.; Puigserver, P.; Auwerx, J. AMPK regulates energy expenditure by modulating NAD+ metabolism and SIRT1 activity. Nature 2009, 458(7241), 1056–1060. [Google Scholar] [CrossRef] [PubMed]
- Chang, X.; Tamauchi, S.; Nakagawa, A.; Xinyuan, W.; Yoshida, K.; Yokoi, A.; Yoshikawa, N.; Kajiyama, H. Chemotherapeutic hormesis induced by the tumor microenvironment in refractory ovarian cancer. Scientific Reports 2025, 15(1), 596. [Google Scholar] [CrossRef]
- Colman, R.J.; Anderson, R.M.; Johnson, S.C.; Kastman, E.K.; Kosmatka, K.J.; Beasley, T.M.; Allison, D.B.; Cruzen, C.; Simmons, H.A.; Kemnitz, J.W.; Weindruch, R. Caloric restriction delays disease onset and mortality in rhesus monkeys. Science 2009, 325(5937), 201–204. [Google Scholar] [CrossRef]
- Da, W.; Chen, Q.; Shen, B. The current insights of mitochondrial hormesis in the occurrence and treatment of bone and cartilage degeneration. Biological Research 2024, 57(1), 37. [Google Scholar] [CrossRef]
- Davies, K.J.A. Adaptive homeostasis. Molecular Aspects of Medicine 49 2016, 1–7. [Google Scholar] [CrossRef]
- DeNicola, G.M.; Karreth, F.A.; Humpton, T.J.; Gopinathan, A.; Wei, C.; Frese, K.; Mangal, D.; Yu, K.H.; Yeo, C.J.; Calhoun, E.S.; Scrimieri, F.; Winter, J.M.; Hruban, R.H.; Iacobuzio-Donahue, C.; Kern, S.E.; Blair, I.A.; Tuveson, D.A. Oncogene-induced Nrf2 transcription promotes ROS detoxification and tumorigenesis. Nature 2011, 475(7354), 106–109. [Google Scholar] [CrossRef]
- Di Francesco, A.; Di Germanio, C.; Bernier, M.; de Cabo, R. A time to fast. Science 2018, 362(6416), 770–775. [Google Scholar] [CrossRef]
- Ebata, H.; Hansen, M. Links between autophagy and healthy aging. Journal of Molecular Biology 2026, 438(6), 169656. [Google Scholar] [CrossRef]
- Egan, B.; Zierath, J.R. Exercise metabolism and the molecular regulation of skeletal muscle adaptation. Cell Metabolism 17 2013, 162–184. [Google Scholar] [CrossRef]
- Ekelund, U.; Tarp, J.; Steene-Johannessen, J.; Hansen, B.H.; Jefferis, B.; Fagerland, M.W.; Whincup, P.; Diaz, K.M.; Hooker, S.P.; Chernofsky, A.; Larson, M.G.; Spartano, N.; Vasan, R.S.; Dohrn, I.M.; Hagströmer, M.; Edwardson, C.; Yates, T.; Shiroma, E.; Anderssen, S.A.; Lee, I.M. Dose-response associations between accelerometry measured physical activity and sedentary time and all cause mortality: systematic review and harmonised meta-analysis. BMJ (Clinical research ed.) 2019, 366, l4570. [Google Scholar] [CrossRef] [PubMed]
- Epstein, E.; Xu, I.; Patel, N.; Taub, P.R. Time-restricted eating: A tool for hormesis in cardiovascular risk reduction. Conditioning Medicine 2025, 7(2), 48–61. Available online: https://www.conditionmed.org/Data/View/16927.
- Fontana, L.; Partridge, L.; Longo, V.D. Extending healthy life span--from yeast to humans. Science 2010, 328(5976), 321–326. [Google Scholar] [CrossRef] [PubMed]
- Franceschi, C.; Garagnani, P.; Parini, P.; Giuliani, C.; Santoro, A. Inflammaging: A new immune–metabolic viewpoint for age-related diseases. Nature Reviews Endocrinology 2018, 14(10), 576–590. [Google Scholar] [CrossRef]
- Gibala, M.J.; Little, J.P.; Macdonald, M.J.; Hawley, J.A. Physiological adaptations to low-volume, high-intensity interval training in health and disease. The Journal of Physiology 2012, 590(5), 1077–1084. [Google Scholar] [CrossRef]
- Gleeson, M.; Bishop, N.C.; Stensel, D.J.; Lindley, M.R.; Mastana, S.S.; Nimmo, M.A. The anti-inflammatory effects of exercise: Mechanisms and implications for the prevention and treatment of disease. Nature Reviews Immunology 2011, 11(9), 607–615. [Google Scholar] [CrossRef]
- Grenier, S.F.; Commisso, C. A hormetic response model for glutamine stress in cancer. Trends in Cancer 2025, 11(3), 196–203. [Google Scholar] [CrossRef]
- Guarente, L. Calorie restriction and sirtuins revisited. Genes & Development 2013, 27(19), 2072–2085. [Google Scholar] [CrossRef] [PubMed]
- Haigis, M.C.; Sinclair, D.A. Mammalian sirtuins: biological insights and disease relevance. Annual Review of Pathology 2010, 5, 253–295. [Google Scholar] [CrossRef] [PubMed]
- Harman, D. Aging: A theory based on free radical and radiation chemistry. Journal of Gerontology 1956, 11(3), 298–300. [Google Scholar] [CrossRef]
- Hood, D.A.; Memme, J.M.; Oliveira, A.N.; Triolo, M. Maintenance of Skeletal Muscle Mitochondria in Health, Exercise, and Aging. Annual review of physiology 2019, 81, 19–41. [Google Scholar] [CrossRef]
- Imai, S.; Guarente, L. NAD+ and sirtuins in aging and disease. Trends in Cell Biology 2014, 24(8), 464–471. [Google Scholar] [CrossRef]
- Kalyakulina, A.; Yusipov, I.; Trukhanov, A.; Franceschi, C.; Moskalev, A.; Ivanchenko, M. EpInflammAge: Epigenetic-Inflammatory Clock for Disease-Associated Biological Aging Based on Deep Learning. International Journal of Molecular Sciences 2025, 26(13), 6284. [Google Scholar] [CrossRef]
- Kampinga, H.H.; Bergink, S. Heat shock proteins as potential targets for protective strategies in neurodegeneration. The Lancet Neurology 2016, 15(7), 748–759. [Google Scholar] [CrossRef] [PubMed]
- Kensler, T.W.; Wakabayashi, N.; Biswal, S. Cell survival responses to environmental stresses via the Keap1-Nrf2-ARE pathway. Annual Review of Pharmacology and Toxicology 2007, 47, 89–116. [Google Scholar] [CrossRef]
- Kirkwood, T. B. Understanding the odd science of aging. Cell 2005, 120(4), 437–447. [Google Scholar] [CrossRef]
- Kodama, S.; Saito, K.; Tanaka, S.; Maki, M.; Yachi, Y.; Asumi, M.; Sugawara, A.; Totsuka, K.; Shimano, H.; Ohashi, Y.; Yamada, N.; Sone, H. Cardiorespiratory fitness as a quantitative predictor of all-cause mortality and cardiovascular events in healthy men and women: A meta-analysis. JAMA 2009, 301(19), 2024–2035. [Google Scholar] [CrossRef]
- Komatsu, M.; Kurokawa, H.; Waguri, S.; Taguchi, K.; Kobayashi, A.; Ichimura, Y.; Sou, Y.S.; Ueno, I.; Sakamoto, A.; Tong, K.I.; Kim, M.; Nishito, Y.; Iemura, S.; Natsume, T.; Ueno, T.; Kominami, E.; Motohashi, H.; Tanaka, K.; Yamamoto, M. The selective autophagy substrate p62 activates the stress responsive transcription factor Nrf2 through inactivation of Keap1. Nature Cell Biology 2010, 12(3), 213–223. [Google Scholar] [CrossRef]
- Kunutsor, S.K.; Lehoczki, A.; Laukkanen, J.A. The untapped potential of cold water therapy as part of a lifestyle intervention for promoting healthy aging. GeroScience 2025, 47(1), 387–407. [Google Scholar] [CrossRef] [PubMed]
- Laukkanen, T.; Khan, H.; Zaccardi, F.; Laukkanen, J.A. Association between sauna bathing and fatal cardiovascular and all-cause mortality events. JAMA Internal Medicine 2015, 175(4), 542–548. [Google Scholar] [CrossRef]
- Levine, B.; Kroemer, G. Biological Functions of Autophagy Genes: A Disease Perspective. Cell 2019, 176(1-2), 11–42. [Google Scholar] [CrossRef]
- Lim, S.H.Y.; Hansen, M.; Kumsta, C. Molecular Mechanisms of Autophagy Decline during Aging. Cells 2024, 13(16), 1364. [Google Scholar] [CrossRef]
- Lim, V.J.T.; Tabassum, N.I.; Orian, J.M.; Arumugam, T.V.; Weng, E.F. Hormesis and brain diseases. Progress in Brain Research 2025, 295, 229–258. [Google Scholar] [CrossRef]
- Liu, D.; Huang, Y.; Huang, C.; Yang, S.; Wei, X.; Zhang, P.; Guo, D.; Lin, J.; Xu, B.; Li, C.; He, H.; He, J.; Liu, S.; Shi, L.; Xue, Y.; Zhang, H. Calorie Restriction with or without Time-Restricted Eating in Weight Loss. The New England Journal of Medicine 2022, 386(16), 1495–1504. [Google Scholar] [CrossRef]
- Longo, V.D.; Panda, S. Fasting, Circadian Rhythms, and Time-Restricted Feeding in Healthy Lifespan. Cell Metabolism 2016, 23(6), 1048–1059. [Google Scholar] [CrossRef] [PubMed]
- López-Otín, C.; Blasco, M.A.; Partridge, L.; Serrano, M.; Kroemer, G. The hallmarks of aging. Cell 2013, 153(6), 1194–1217. [Google Scholar] [CrossRef] [PubMed]
- López-Otín, C.; Blasco, M.A.; Partridge, L.; Serrano, M.; Kroemer, G. Hallmarks of aging: An expanding universe. Cell 2023, 186(2), 243–278. [Google Scholar] [CrossRef] [PubMed]
- Ma, Q. Role of nrf2 in oxidative stress and toxicity. Annual Review of Pharmacology and Toxicology 2013, 53, 401–426. [Google Scholar] [CrossRef]
- Madeo, F.; Carmona-Gutierrez, D.; Hofer, S.J.; Kroemer, G. Caloric Restriction Mimetics against Age-Associated Disease: Targets, Mechanisms, and Therapeutic Potential. Cell Metabolism 2019, 29(3), 592–610. [Google Scholar] [CrossRef]
- Martins, R.; Lithgow, G.J.; Link, W. Long live FOXO: unraveling the role of FOXO proteins in aging and longevity. Aging cell 2016, 15(2), 196–207. [Google Scholar] [CrossRef]
- Mattson, M.P. Hormesis defined. Ageing Research Reviews 2008, 7(1), 1–7. [Google Scholar] [CrossRef]
- Mattson, M.P.; Leak, R.K. The hormesis principle of neuroplasticity and neuroprotection. Cell Metabolism 2024, 36(2), 315–337. [Google Scholar] [CrossRef] [PubMed]
- McEwen, B.S. Physiology and neurobiology of stress and adaptation: Central role of the brain. Physiological Reviews 2007, 87(3), 873–904. [Google Scholar] [CrossRef] [PubMed]
- McEwen, B.S.; Wingfield, J.C. The concept of allostasis in biology and biomedicine. Hormones and Behavior 2003, 43(1), 2–15. [Google Scholar] [CrossRef]
- Meier, H.C.S.; Mitchell, C.; Karadimas, T.; Faul, J.D. Systemic inflammation and biological aging in the Health and Retirement Study. GeroScience 2023, 45(6), 3257–3265. [Google Scholar] [CrossRef]
- Merry, T.L.; Ristow, M. Mitohormesis in exercise training. Free radical biology & medicine 2016, 98, 123–130. [Google Scholar] [CrossRef]
- Moreno, T.M.; Nieto-Torres, J.L.; Kumsta, C. Monitoring Autophagy in Human Aging: Key Cell Models and Insights. Frontiers in Bioscience (Landmark edition) 2025, 30(3), 27091. [Google Scholar] [CrossRef]
- Mundo Rivera, V.M.; Tlacuahuac Juárez, J.R.; Murillo Melo, N.M.; Leyva Garcia, N.; Magaña, J.J.; Cordero Martínez, J.; Jiménez Gutierrez, G.E. Natural Autophagy Activators to Fight Age-Related Diseases. Cells 2024, 13(19), 1611. [Google Scholar] [CrossRef]
- Palacios-Ramírez, R.; Francés, D.E.; Motiño, O. Autophagy in age-related liver disease. Geromedicine 2025, 1, 202509. [Google Scholar] [CrossRef]
- Pedersen, B.K.; Saltin, B. Exercise as medicine—evidence for prescribing exercise as therapy in 26 different chronic diseases. Scandinavian Journal of Medicine & Science in Sports 2015, 25 Suppl 3, 1–72. [Google Scholar] [CrossRef]
- Phillips, M.C.L.; Picard, M. Neurodegenerative disorders, metabolic icebergs, and mitohormesis. Translational Neurodegeneration 2024, 13(1), 46. [Google Scholar] [CrossRef]
- Pomatto, L.C.D.; Davies, K.J.A. The role of declining adaptive homeostasis in ageing. The Journal of Physiology 2017, 595(24), 7275–7309. [Google Scholar] [CrossRef]
- Pomatto, L.C.D.; Davies, K.J.A. Adaptive homeostasis and the free radical theory of ageing. Free Radical Biology & Medicine 2018, 124, 420–430. [Google Scholar] [CrossRef]
- Powers, S.K.; Nelson, W.B.; Hudson, M.B. Exercise-induced oxidative stress in humans: cause and consequences. Free Radical Biology & Medicine 2011, 51(5), 942–950. [Google Scholar] [CrossRef]
- Radak, Z.; Chung, H.Y.; Koltai, E.; Taylor, A.W.; Goto, S. Exercise, oxidative stress and hormesis. Ageing Research Reviews 2008, 7(1), 34–42. [Google Scholar] [CrossRef] [PubMed]
- Redding, A.; Grabocka, E. Stress granules and hormetic adaptation of cancer. Trends in Cancer 2023, 9(12), 995–1005. [Google Scholar] [CrossRef]
- Redman, L.M.; Smith, S.R.; Burton, J.H.; Martin, C.K.; Il’yasova, D.; Ravussin, E. Metabolic Slowing and Reduced Oxidative Damage with Sustained Caloric Restriction Support the Rate of Living and Oxidative Damage Theories of Aging. Cell Metabolism 2018, 27(4), 805–815.e4. [Google Scholar] [CrossRef]
- Ricciardi, N.R. Hormesis as a Therapeutic Mechanism in Alzheimer’s Disease and Memory Formation University of Miami). University of Miami eScholarship Repository. Doctoral dissertation, 2024. Available online: https://scholarship.miami.edu/esploro/outputs/doctoral/Hormesis-as-a-Therapeutic-Mechanism-in/991032075814602976.
- Ristow, M.; Zarse, K. How increased oxidative stress promotes longevity and metabolic health: The concept of mitochondrial hormesis (mitohormesis). Experimental Gerontology 2010, 45(6), 410–418. [Google Scholar] [CrossRef]
- Ristow, M.; Zarse, K.; Oberbach, A.; Klöting, N.; Birringer, M.; Kiehntopf, M.; Stumvoll, M.; Kahn, C.R.; Blüher, M. Antioxidants prevent health-promoting effects of physical exercise in humans. Proceedings of the National Academy of Sciences of the United States of America 2009, 106(21), 8665–8670. [Google Scholar] [CrossRef] [PubMed]
- Rubinsztein, D.C.; Mariño, G.; Kroemer, G. Autophagy and aging. Cell 2011, 146(5), 682–695. [Google Scholar] [CrossRef]
- Satoh, A.; Brace, C.S.; Rensing, N.; Cliften, P.; Wozniak, D.F.; Herzog, E.D.; Yamada, K.A.; Imai, S. Sirt1 extends life span and delays aging in mice through the regulation of Nk2 homeobox 1 in the DMH and LH. Cell Metabolism 2013, 18(3), 416–430. [Google Scholar] [CrossRef] [PubMed]
- Sena, L.A.; Chandel, N.S. Physiological roles of mitochondrial reactive oxygen species. Molecular Cell 2012, 48(2), 158–167. [Google Scholar] [CrossRef]
- Sohal, R.S.; Orr, W.C. The redox stress hypothesis of aging. Free Radical Biology & Medicine 2012, 52(3), 539–555. [Google Scholar] [CrossRef]
- Sterling, P.; Eyer, J. Fisher, S., Reason, J., Eds.; Allostasis: A new paradigm to explain arousal pathology. In Handbook of life stress, cognition and health; John Wiley & Sons, 1988; pp. 629–649. [Google Scholar]
- Subramanian, V.; Tucker, W.J.; Peters, A.E.; Upadhya, B.; Kitzman, D.W.; Pandey, A. Cardiovascular Aging and Exercise: Implications for Heart Failure Prevention and Management. Circulation Research 2025, 137(2), 205–230. [Google Scholar] [CrossRef]
- Sutton, E.F.; Beyl, R.; Early, K.S.; Cefalu, W.T.; Ravussin, E.; Peterson, C.M. Early Time-Restricted Feeding Improves Insulin Sensitivity, Blood Pressure, and Oxidative Stress Even without Weight Loss in Men with Prediabetes. Cell Metabolism 2018, 27(6), 1212–1221.e3. [Google Scholar] [CrossRef] [PubMed]
- Ungvari, Z.; Bailey-Downs, L.; Sosnowska, D.; Gautam, T.; Koncz, P.; Losonczy, G.; Ballabh, P.; de Cabo, R.; Sonntag, W.E.; Csiszar, A. Vascular oxidative stress in aging: a homeostatic failure due to dysregulation of NRF2-mediated antioxidant response. American Journal of Physiology. Heart and Circulatory Physiology 2011, 301(2), H363–H372. [Google Scholar] [CrossRef]
- van der Lans, A.A.; Hoeks, J.; Brans, B.; Vijgen, G.H.; Visser, M.G.; Vosselman, M.J.; Hansen, J.; Jörgensen, J.A.; Wu, J.; Mottaghy, F.M.; Schrauwen, P.; van Marken Lichtenbelt, W.D. Cold acclimation recruits human brown fat and increases nonshivering thermogenesis. The Journal of Clinical Investigation 2013, 123(8), 3395–3403. [Google Scholar] [CrossRef]
- Villareal, D.T.; Chode, S.; Parimi, N.; Sinacore, D.R.; Hilton, T.; Armamento-Villareal, R.; Napoli, N.; Qualls, C.; Shah, K. Weight loss, exercise, or both and physical function in obese older adults. The New England Journal of Medicine 2011, 364(13), 1218–1229. [Google Scholar] [CrossRef] [PubMed]
- Wan, Y.; Liu, J.; Mai, Y.; Hong, Y.; Jia, Z.; Tian, G.; Liu, Y.; Liang, H.; Liu, J. Current advances and future trends of hormesis in disease. NPJ Aging 2024, 10(1), 26. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Davies, K.J.A.; Forman, H.J. Oxidative stress response and Nrf2 signaling in aging. Free Radical Biology & Medicine 2015, 88 Pt B, 314–336. [Google Scholar] [CrossRef]
- Zhang, Z.; Zhang, L.; Zhang, Y.; Zhao, Y.; Gao, Y.; Zhang, C.; Li, D.; Zhang, X.; Yang, G.; Zhang, J. Inhibition of Sirtuin 2 enhances autophagy and restores neuronal function in aged hippocampal neurons. Brain Research Bulletin 2025, 230, 111501. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



