Submitted:
17 April 2026
Posted:
21 April 2026
You are already at the latest version
Abstract
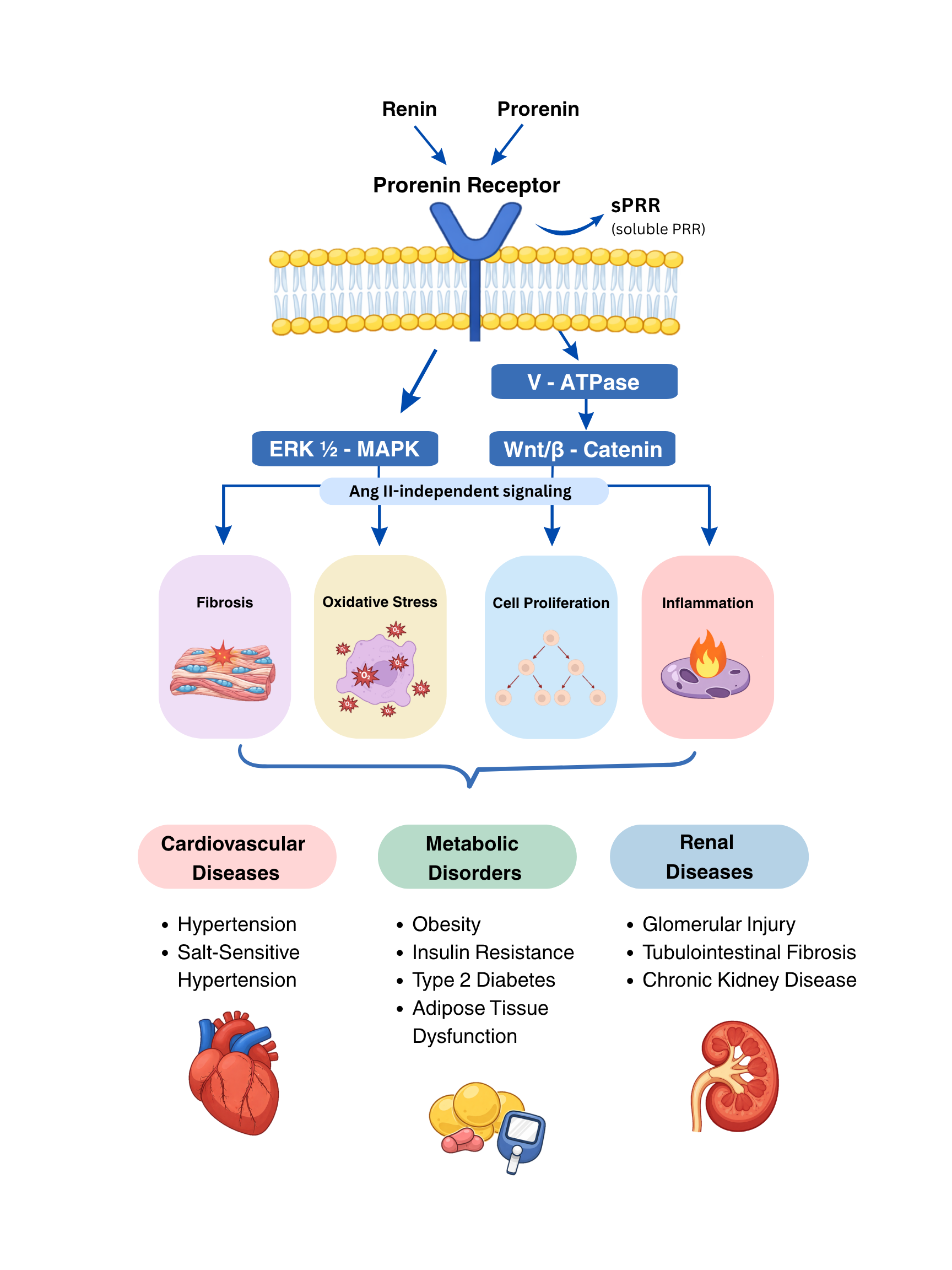
Keywords:
1. Introduction
2. Prorenin Receptor in Cardiovascular, Metabolic and Renal Diseases
2.1. Prorenin Receptor Signaling in Metabolic Homeostasis and Disease
2.2. Prorenin Receptor as a Regulator of Endothelial Signaling, Angiogenesis, and Vascular Remodeling
2.3. Prorenin Receptor Pathogenic Feedback Loops and Chronic Kidney Disease
2.4. Prorenin Receptor Contributes to Systemic and Renal Inflammation
3. The Soluble Prorenin Receptor: A Circulating Link Between the RAAS and Cardiorenal System
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ADAM19 | A Disintegrin And Metalloproteinase 19 |
| Akt | Protein kinase B |
| Ang I | Angiotensin I |
| Ang II | Angiotensin II |
| AP-1 | Activator protein-1 |
| ATP6AP2 | ATPase H+-transporting accessory protein 2 (gene encoding PRR) |
| BP | Blood pressure |
| CKD | Chronic kidney disease |
| COX-2 | Cyclooxygenase-2 |
| CREB | cAMP response element-binding protein |
| CVD | Cardiovascular disease |
| ECM | Extracellular matrix |
| ENaC | Epithelial sodium channel |
| ERK1/2 | Extracellular signal-regulated kinase 1/2 |
| HSP27 | Heat shock protein 27 |
| HRP | Handle region peptide |
| IL-1β | Interleukin-1 beta |
| IS | Indoxyl sulfate |
| MAPK | Mitogen-activated protein kinase |
| MCP-1 | Monocyte chemoattractant protein-1 |
| MMP9 | Matrix metalloproteinase-9 |
| NF-κB | Nuclear factor κB |
| NOX4 | NADPH oxidase 4 |
| p38 MAPK | p38 mitogen-activated protein kinase |
| PAI-1 | Plasminogen activator inhibitor-1 |
| PI3K | Phosphoinositide 3-kinase |
| PRR | Prorenin receptor |
| PRO20 | PRR decoy inhibitor derived from the first 20 amino acids of the prorenin pro-segment |
| RAAS | Renin–angiotensin–aldosterone system |
| ROS | Reactive oxygen species |
| sPRR | Soluble prorenin receptor |
| SP-1 | Site-1 protease |
| TGF-β | Transforming growth factor-β |
| TIMP1 | Tissue inhibitor of metalloproteinase-1 |
| TNF-α | Tumor necrosis factor-α |
| V-ATPase | Vacuolar H+-ATPase |
| VSMC | Vascular smooth muscle cell |
| Wnt/β-catenin | Wnt/β-catenin signaling pathway |
| 2K1C | Two-kidney, one-clip (Goldblatt hypertension model) |
References
- Crowley, S.D.; Navar, L.G.; Prieto, M.C.; Gurley, S.B.; Coffman, T.M. Kidney Renin–Angiotensin System: Lost in a RAS Cascade. Hypertension 2024, 81, 682–686. [Google Scholar] [CrossRef]
- Nguyen, G.; Delarue, F.; Burcklé, C.; Bouzhir, L.; Giller, T.; Sraer, J.-D. Pivotal Role of the Renin/Prorenin Receptor in Angiotensin II Production and Cellular Responses to Renin. J. Clin. Invest. 2002, 109, 1417–1427. [Google Scholar] [CrossRef] [PubMed]
- Advani, A.; Kelly, D.J.; Cox, A.J.; White, K.E.; Advani, S.L.; Thai, K.; Connelly, K.A.; Yuen, D.; Trogadis, J.; Herzenberg, A.M.; et al. The (Pro)Renin Receptor: Site-Specific and Functional Linkage to the Vacuolar H+ -ATPase in the Kidney. Hypertension 2009, 54, 261–269. [Google Scholar] [CrossRef]
- Gonzalez, A.A.; Lara, L.S.; Luffman, C.; Seth, D.M.; Prieto, M.C. Soluble Form of the (pro)Renin Receptor Is Augmented in the Collecting Duct and Urine of Chronic Angiotensin II-Dependent Hypertensive Rats. Hypertension 2011, 57, 859–864. [Google Scholar] [CrossRef] [PubMed]
- Ramkumar, N.; Stuart, D.; Peterson, C.S.; Hu, C.; Wheatley, W.; Min Cho, J.; Symons, J.D.; Kohan, D.E. Loss of Soluble (Pro)Renin Receptor Attenuates Angiotensin-II Induced Hypertension and Renal Injury. Circulation Research 2021, 129, 50–62. [Google Scholar] [CrossRef] [PubMed]
- Prieto, M.C.; Gonzalez, A.A.; Visniauskas, B.; Navar, L.G. The Evolving Complexity of the Collecting Duct Renin-Angiotensin System in Hypertension. Nat Rev Nephrol 2021, 17, 481–492. [Google Scholar] [CrossRef]
- Gogulamudi, V.R.; Arita, D.Y.; Bourgeois, C.R.T.; Jorgensen, J.; He, J.; Wimley, W.C.; Satou, R.; Gonzalez, A.A.; Prieto, M.C. High Glucose Induces Trafficking of Prorenin Receptor and Stimulates Profibrotic Factors in the Collecting Duct. Sci Rep 2021, 11, 13815. [Google Scholar] [CrossRef]
- Visniauskas, B.; Arita, D.Y.; Rosales, C.B.; Feroz, M.A.; Luffman, C.; Accavitti, M.J.; Dawkins, G.; Hong, J.; Curnow, A.C.; Thethi, T.K.; et al. Sex Differences in Soluble Prorenin Receptor in Patients with Type 2 Diabetes. Biol Sex Differ 2021, 12, 33. [Google Scholar] [CrossRef]
- Wang, B.; Jie, H.; Wang, S.; Dong, B.; Zou, Y. The Role of (pro)Renin Receptor and Its Soluble Form in Cardiovascular Diseases. Front. Cardiovasc. Med. 2023, 10, 1086603. [Google Scholar] [CrossRef]
- Ichihara, A.; Yatabe, M.S. The (pro)Renin Receptor in Health and Disease. Nat Rev Nephrol 2019, 15, 693–712. [Google Scholar] [CrossRef]
- Nakagawa, T.; Suzuki-Nakagawa, C.; Watanabe, A.; Asami, E.; Matsumoto, M.; Nakano, M.; Ebihara, A.; Uddin, M.N.; Suzuki, F. Site-1 Protease Is Required for the Generation of Soluble (pro)Renin Receptor. The Journal of Biochemistry 2017, 161, 369–379. [Google Scholar] [CrossRef]
- Figueiredo, M.; Daryadel, A.; Sihn, G.; Müller, D.N.; Popova, E.; Rouselle, A.; Nguyen, G.; Bader, M.; Wagner, C.A. The (pro)Renin Receptor (ATP6ap2) Facilitates Receptor-Mediated Endocytosis and Lysosomal Function in the Renal Proximal Tubule. Pflugers Arch 2021, 473, 1229–1246. [Google Scholar] [CrossRef] [PubMed]
- Kinouchi, K.; Ichihara, A.; Sano, M.; Sun-Wada, G.-H.; Wada, Y.; Kurauchi-Mito, A.; Bokuda, K.; Narita, T.; Oshima, Y.; Sakoda, M.; et al. The (Pro)Renin Receptor/ATP6AP2 Is Essential for Vacuolar H+ -ATPase Assembly in Murine Cardiomyocytes. Circulation Research 2010, 107, 30–34. [Google Scholar] [CrossRef]
- Li, L.; Cui, Y.; Liu, Y.; Li, H.; Su, Y.; Li, S.; Wang, L.; Zhao, Y.; Wang, S.; Yan, F.; et al. ATP6AP2 Knockdown in Cardiomyocyte Deteriorates Heart Function via Compromising Autophagic Flux and NLRP3 Inflammasome Activation. Cell Death Discov. 2022, 8, 161. [Google Scholar] [CrossRef]
- Hayakawa, Y.; Aoyama, T.; Yokoyama, C.; Okamoto, C.; Komaki, H.; Minatoguchi, S.; Iwasa, M.; Yamada, Y.; Kawamura, I.; Kawasaki, M.; et al. High Salt Intake Damages the Heart through Activation of Cardiac (Pro) Renin Receptors Even at an Early Stage of Hypertension. PLoS ONE 2015, 10, e0120453. [Google Scholar] [CrossRef] [PubMed]
- Okamoto, C.; Hayakawa, Y.; Aoyama, T.; Komaki, H.; Minatoguchi, S.; Iwasa, M.; Yamada, Y.; Kanamori, H.; Kawasaki, M.; Nishigaki, K.; et al. Excessively Low Salt Diet Damages the Heart through Activation of Cardiac (pro) Renin Receptor, Renin-Angiotensin-Aldosterone, and Sympatho-Adrenal Systems in Spontaneously Hypertensive Rats. PLoS ONE 2017, 12, e0189099. [Google Scholar] [CrossRef]
- Moilanen, A.-M.; Rysä, J.; Serpi, R.; Mustonen, E.; Szabò, Z.; Aro, J.; Näpänkangas, J.; Tenhunen, O.; Sutinen, M.; Salo, T.; et al. (Pro)Renin Receptor Triggers Distinct Angiotensin II-Independent Extracellular Matrix Remodeling and Deterioration of Cardiac Function. PLoS ONE 2012, 7, e41404. [Google Scholar] [CrossRef]
- Ma, J.; Li, Y.; Yang, X.; Liu, K.; Zhang, X.; Zuo, X.; Ye, R.; Wang, Z.; Shi, R.; Meng, Q.; et al. Signaling Pathways in Vascular Function and Hypertension: Molecular Mechanisms and Therapeutic Interventions. Sig Transduct Target Ther 2023, 8, 168. [Google Scholar] [CrossRef]
- Prieto, M.C.; Reverte, V.; Mamenko, M.; Kuczeriszka, M.; Veiras, L.C.; Rosales, C.B.; McLellan, M.; Gentile, O.; Jensen, V.B.; Ichihara, A.; et al. Collecting Duct Prorenin Receptor Knockout Reduces Renal Function, Increases Sodium Excretion, and Mitigates Renal Responses in ANG II-Induced Hypertensive Mice. American Journal of Physiology-Renal Physiology 2017, 313, F1243–F1253. [Google Scholar] [CrossRef] [PubMed]
- Peng, K.; Lu, X.; Wang, F.; Nau, A.; Chen, R.; Zhou, S.-F.; Yang, T. Collecting Duct (pro)Renin Receptor Targets ENaC to Mediate Angiotensin II-Induced Hypertension. American Journal of Physiology-Renal Physiology 2017, 312, F245–F253. [Google Scholar] [CrossRef]
- Yosypiv, I.V.; Sequeira-Lopez, M.L.S.; Song, R.; De Goes Martini, A. Stromal Prorenin Receptor Is Critical for Normal Kidney Development. Am J Physiol Regul Integr Comp Physiol 2019, 316, R640–R650. [Google Scholar] [CrossRef]
- Wang, F.; Lu, X.; Peng, K.; Fang, H.; Zhou, L.; Su, J.; Nau, A.; Yang, K.T.; Ichihara, A.; Lu, A.; et al. Antidiuretic Action of Collecting Duct (Pro)Renin Receptor Downstream of Vasopressin and PGE2 Receptor EP4. JASN 2016, 27, 3022–3034. [Google Scholar] [CrossRef]
- Gatineau, E.; Arthur, G.; Poupeau, A.; Nichols, K.; Spear, B.T.; Shelman, N.R.; Graf, G.A.; Temel, R.E.; Yiannikouris, F.B. The Prorenin Receptor and Its Soluble Form Contribute to Lipid Homeostasis. American Journal of Physiology-Endocrinology and Metabolism 2021, 320, E609–E618. [Google Scholar] [CrossRef]
- Nichols, K.; Yiannikouris, F. The Role of (Pro)Renin Receptor in the Metabolic Syndrome. CHYR 2022, 18, 117–124. [Google Scholar] [CrossRef]
- Hoffmann, N.; Peters, J. Functions of the (pro)Renin Receptor (Atp6ap2) at Molecular and System Levels: Pathological Implications in Hypertension, Renal and Brain Development, Inflammation, and Fibrosis. Pharmacological Research 2021, 173, 105922. [Google Scholar] [CrossRef] [PubMed]
- Cousin, C.; Bracquart, D.; Contrepas, A.; Corvol, P.; Muller, L.; Nguyen, G. Soluble Form of the (Pro)Renin Receptor Generated by Intracellular Cleavage by Furin Is Secreted in Plasma. Hypertension 2009, 53, 1077–1082. [Google Scholar] [CrossRef] [PubMed]
- Visniauskas, B.; Reverte, V.; Abshire, C.M.; Ogola, B.O.; Rosales, C.B.; Galeas-Pena, M.; Sure, V.N.; Sakamuri, S.S.V.P.; Harris, N.R.; Kilanowski-Doroh, I.; et al. High-Plasma Soluble Prorenin Receptor Is Associated with Vascular Damage in Male, but Not Female, Mice Fed a High-Fat Diet. American Journal of Physiology-Heart and Circulatory Physiology 2023, 324, H762–H775. [Google Scholar] [CrossRef] [PubMed]
- Patel, N.R.; K C, R.; Blanks, A.; Li, Y.; Prieto, M.C.; Meadows, S.M. Endothelial Cell Polarity and Extracellular Matrix Composition Require Functional ATP6AP2 during Developmental and Pathological Angiogenesis. JCI Insight 2022, 7, e154379. [Google Scholar] [CrossRef]
- Sakoda, M.; Ichihara, A.; Kaneshiro, Y.; Takemitsu, T.; Nakazato, Y.; Nabi, A.H.M.N.; Nakagawa, T.; Suzuki, F.; Inagami, T.; Itoh, H. (Pro)Renin Receptor-Mediated Activation of Mitogen-Activated Protein Kinases in Human Vascular Smooth Muscle Cells. Hypertens Res 2007, 30, 1139–1146. [Google Scholar] [CrossRef]
- Luo, R.; Yang, K.; Wang, F.; Xu, C.; Yang, T. (Pro)Renin Receptor Decoy Peptide PRO20 Protects against Adriamycin-Induced Nephropathy by Targeting the Intrarenal Renin-Angiotensin System. American Journal of Physiology-Renal Physiology 2020, 319, F930–F940. [Google Scholar] [CrossRef]
- Lara, L.S.; Gonzalez, A.A.; Hennrikus, M.T.; Prieto, M.C. Hormone-Dependent Regulation of Renin and Effects on ProreninReceptor Signaling in the Collecting Duct. CHYR 2022, 18, 91–100. [Google Scholar] [CrossRef]
- Fu, Z.; Kaewsaro, K.; Soodvilai, S.; Kimball, A.; Yang, T. Collecting Duct-Derived (pro)Renin Receptor Contributes to 2-Kidney, 1-Clip-Induced Ischemic Nephropathy and Renovascular Hypertension in Mice. American Journal of Physiology-Renal Physiology 2025, 329, F46–F58. [Google Scholar] [CrossRef] [PubMed]
- Reverte, V.; Gogulamudi, V.R.; Rosales, C.B.; Musial, D.C.; Gonsalez, S.R.; Parra-Vitela, A.J.; Galeas-Pena, M.; Sure, V.N.; Visniauskas, B.; Lindsey, S.H.; et al. Urinary Angiotensinogen Increases in the Absence of Overt Renal Injury in High Fat Diet-Induced Type 2 Diabetic Mice. J Diabetes Complications 2020, 34, 107448. [Google Scholar] [CrossRef] [PubMed]
- Narumi, K.; Sato, E.; Hirose, T.; Yamamoto, T.; Nakamichi, T.; Miyazaki, M.; Sato, H.; Ito, S. (Pro)Renin Receptor Is Involved in Mesangial Fibrosis and Matrix Expansion. Sci Rep 2018, 8, 16. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez, A.A.; Zamora, L.; Reyes-Martinez, C.; Salinas-Parra, N.; Roldan, N.; Cuevas, C.A.; Figueroa, S.; Gonzalez-Vergara, A.; Prieto, M.C. (Pro)Renin Receptor Activation Increases Profibrotic Markers and Fibroblast-like Phenotype through MAPK-Dependent ROS Formation in Mouse Renal Collecting Duct Cells. Clin Exp Pharmacol Physiol 2017, 44, 1134–1144. [Google Scholar] [CrossRef]
- Lin, M.; Wang, D.; Chen, Y.; Chen, G.; Zhou, Y.; Ou, J.; Xiao, L. PRR Promotes Hypertensive Renal Injury by Activating Wnt/β-Catenin Signaling and Inflammation Infiltration in Mice. Biochim Biophys Acta Mol Basis Dis 2025, 1871, 167517. [Google Scholar] [CrossRef]
- Feldt, S.; Maschke, U.; Dechend, R.; Luft, F.C.; Muller, D.N. The Putative (Pro)Renin Receptor Blocker HRP Fails to Prevent (Pro)Renin Signaling. Journal of the American Society of Nephrology 2008, 19, 743–748. [Google Scholar] [CrossRef]
- Li, W.; Sullivan, M.N.; Zhang, S.; Worker, C.J.; Xiong, Z.; Speth, R.C.; Feng, Y. Intracerebroventricular Infusion of the (Pro)Renin Receptor Antagonist PRO20 Attenuates Deoxycorticosterone Acetate-Salt–Induced Hypertension. Hypertension 2015, 65, 352–361. [Google Scholar] [CrossRef]
- Fang, H.; Yang, T.; Zhou, B.; Li, X. (Pro)Renin Receptor Decoy Peptide PRO20 Protects against Oxidative Renal Damage Induced by Advanced Oxidation Protein Products. Molecules 2023, 28, 3017. [Google Scholar] [CrossRef]
- Larenas, P.E.; Cárdenas, P.; Aguirre-Delgadillo, M.; Moris, C.; Casarini, D.E.; Vallotton, Z.; Prieto, M.C.; Gonzalez, A.A. GLUT1 and Prorenin Receptor Mediate Differential Regulation of TGF-β and CTGF in Renal Inner Medullary Collecting Duct Cells during High Glucose Conditions. Biol Res 2024, 57, 81. [Google Scholar] [CrossRef]
- Cárdenas, P.; Cid-Salinas, C.; León, A.C.; Castillo-Geraldo, J.; De Oliveira, L.C.G.; Yokota, R.; Vallotton, Z.; Casarini, D.E.; Prieto, M.C.; Lorca, R.A.; et al. (Pro)Renin Receptor Blockade Prevents Increases in Systolic Blood Pressure, Sodium Retention, and αENaC Protein Expression in the Kidney of 2K1C Goldblatt Mice. IJMS 2025, 26, 4177. [Google Scholar] [CrossRef]
- Ramkumar, N.; Kohan, D.E. The (pro)Renin Receptor: An Emerging Player in Hypertension and Metabolic Syndrome. Kidney Int 2019, 95, 1041–1052. [Google Scholar] [CrossRef]
- Gonzalez, A.A.; Luffman, C.; Bourgeois, C.R.T.; Vio, C.P.; Prieto, M.C. Angiotensin II–Independent Upregulation of Cyclooxygenase-2 by Activation of the (Pro)Renin Receptor in Rat Renal Inner Medullary Cells. Hypertension 2013, 61, 443–449. [Google Scholar] [CrossRef] [PubMed]
- Ellmers, L.J.; Rademaker, M.T.; Charles, C.J.; Yandle, T.G.; Richards, A.M. (Pro)Renin Receptor Blockade Ameliorates Cardiac Injury and Remodeling and Improves Function After Myocardial Infarction. J Card Fail 2016, 22, 64–72. [Google Scholar] [CrossRef] [PubMed]
- Feng, Y.; Peng, K.; Luo, R.; Wang, F.; Yang, T. Site-1 Protease-Derived Soluble (Pro)Renin Receptor Contributes to Angiotensin II–Induced Hypertension in Mice. Hypertension 2021, 77, 405–416. [Google Scholar] [CrossRef] [PubMed]
- Xu, C.; Liu, C.; Xiong, J.; Yu, J. Cardiovascular Aspects of the (pro)Renin Receptor: Function and Significance. The FASEB Journal 2022, 36, e22237. [Google Scholar] [CrossRef]
- Yang, T. Soluble (Pro)Renin Receptor in Hypertension. Nephron 2023, 147, 234–243. [Google Scholar] [CrossRef]
- Watanabe, N.; Bokuda, K.; Fujiwara, T.; Suzuki, T.; Mito, A.; Morimoto, S.; Jwa, S.C.; Egawa, M.; Arai, Y.; Suzuki, F.; et al. Soluble (Pro)Renin Receptor and Blood Pressure During Pregnancy: A Prospective Cohort Study. Hypertension 2012, 60, 1250–1256. [Google Scholar] [CrossRef]
- Yisireyili, M.; Saito, S.; Abudureyimu, S.; Adelibieke, Y.; Ng, H.-Y.; Nishijima, F.; Takeshita, K.; Murohara, T.; Niwa, T. Indoxyl Sulfate-Induced Activation of (pro)Renin Receptor Promotes Cell Proliferation and Tissue Factor Expression in Vascular Smooth Muscle Cells. PLoS One 2014, 9, e109268. [Google Scholar] [CrossRef]
- Li, Z.; Zhou, L.; Wang, Y.; Miao, J.; Hong, X.; Hou, F.F.; Liu, Y. (Pro)Renin Receptor Is an Amplifier of Wnt/β-Catenin Signaling in Kidney Injury and Fibrosis. J Am Soc Nephrol 2017, 28, 2393–2408. [Google Scholar] [CrossRef]
- Yang, T.; Gao, Z.-X.; Mao, Z.-H.; Wu, P. Soluble (pro)Renin Receptor as a Novel Regulator of Renal Medullary Na+ Reabsorption. American Journal of Physiology-Renal Physiology 2025, 328, F239–F247. [Google Scholar] [CrossRef] [PubMed]
- Visniauskas, B.; Ogola, B.O.; Kilanowski-Doroh, I.; Harris, N.R.; Diaz, Z.T.; Horton, A.C.; Blessinger, S.A.; McNally, A.B.; Zimmerman, M.A.; Arnold, A.C.; et al. Hypertension Disrupts the Vascular Clock in Both Sexes. American Journal of Physiology-Heart and Circulatory Physiology 2024, 327, H765–H777. [Google Scholar] [CrossRef] [PubMed]
- Yang, T. Potential of Soluble (pro)Renin Receptor in Kidney Disease: Can It Go beyond a Biomarker? American Journal of Physiology-Renal Physiology 2022, 323, F507–F514. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



