Submitted:
20 April 2026
Posted:
21 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Experiments
2.1. Preparation of Porous Titanium Doped with Different Nickel Contents
2.2. Preparation of TiO2 Nanotube Thin Films
2.3. Investigation on the Effect of Heat Treatment Temperature on the Crystal Structure and Photocatalytic Performance of Catalysts
2.4. Characterization of Materials
3. Results
3.1. Characterization and Analysis of Titanium Dioxide Nanotubes on Porous Titanium with Different Nickel Contents
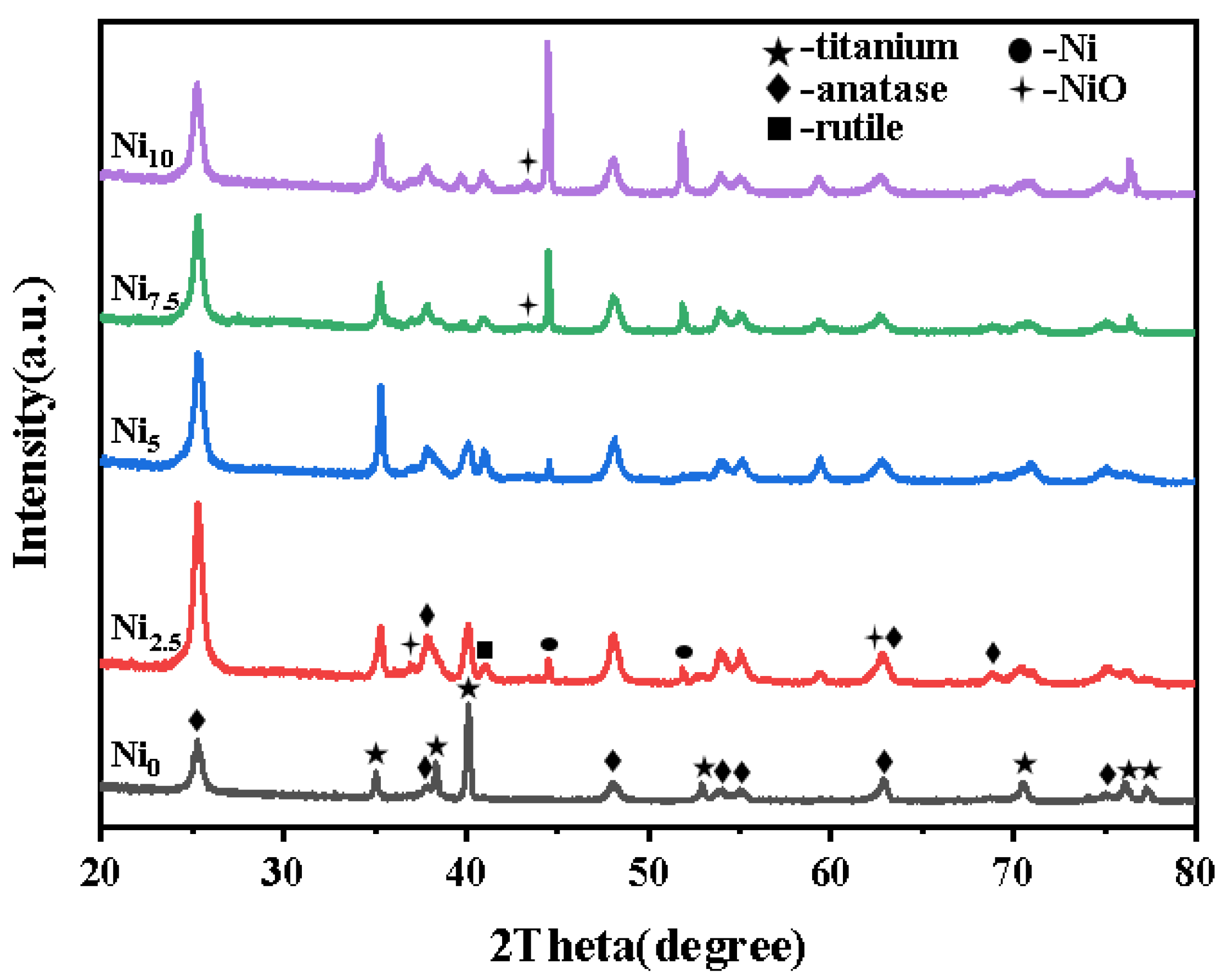
3.1.1. XRD Analysis
3.1.2. SEM Analysis
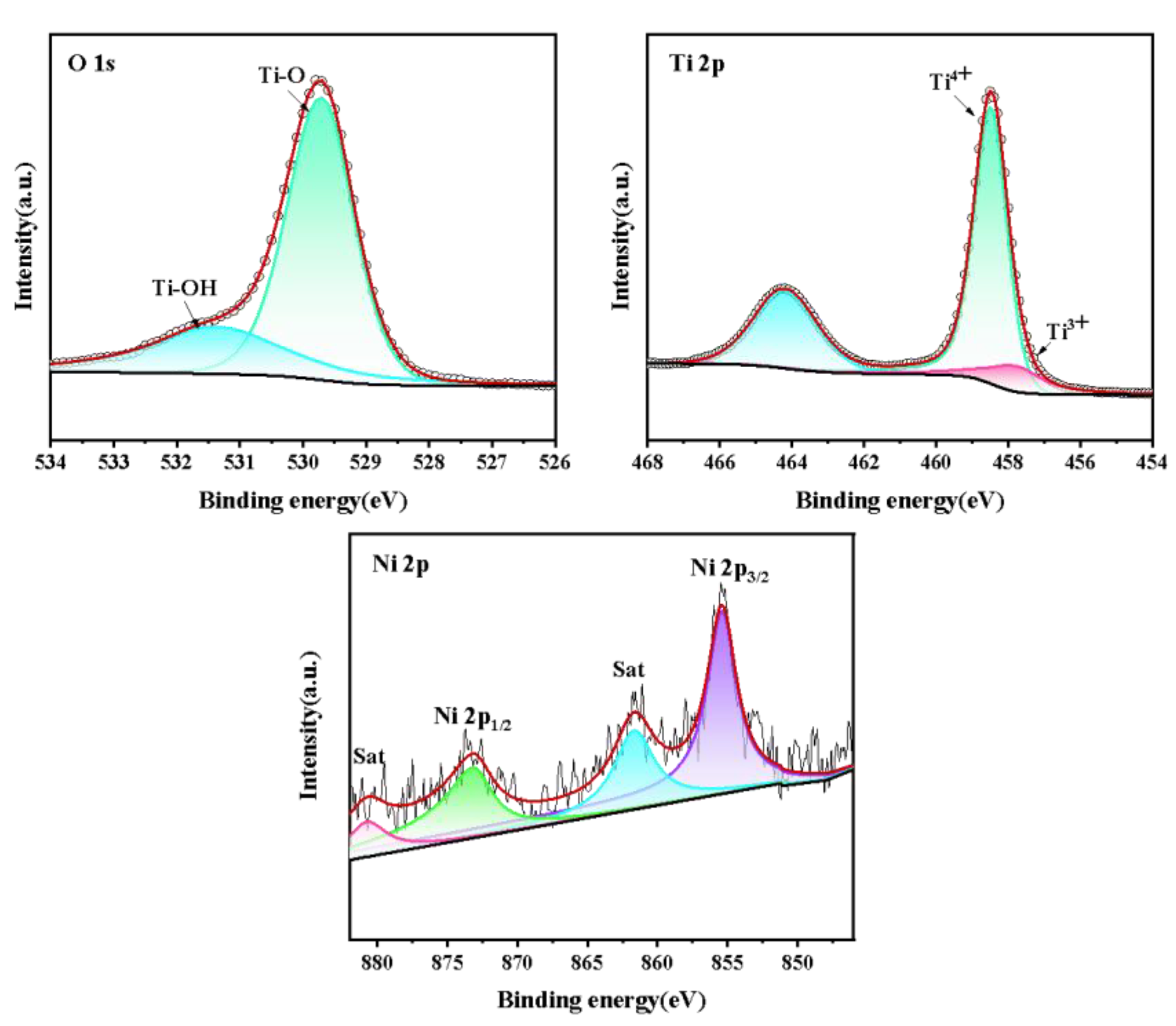
3.1.3. XPS Analysis
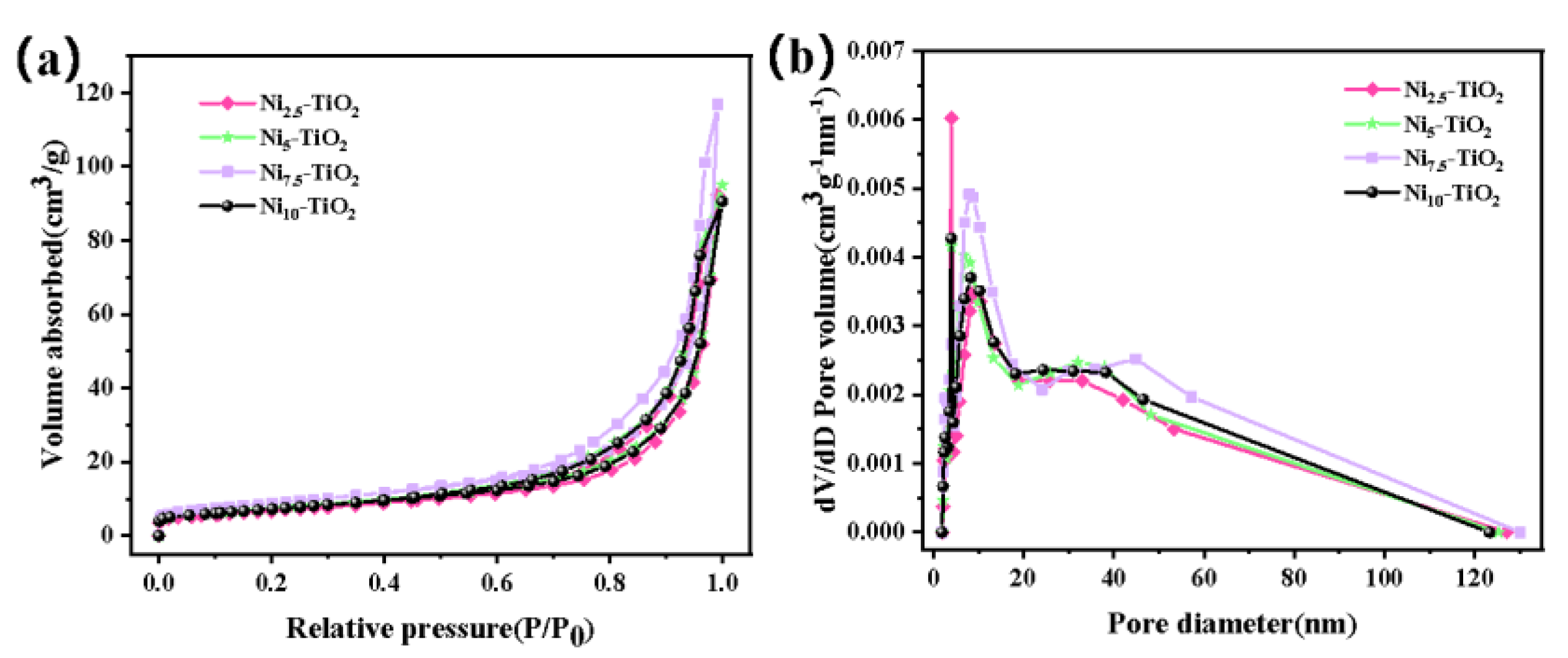
3.1.4. BET Analysis
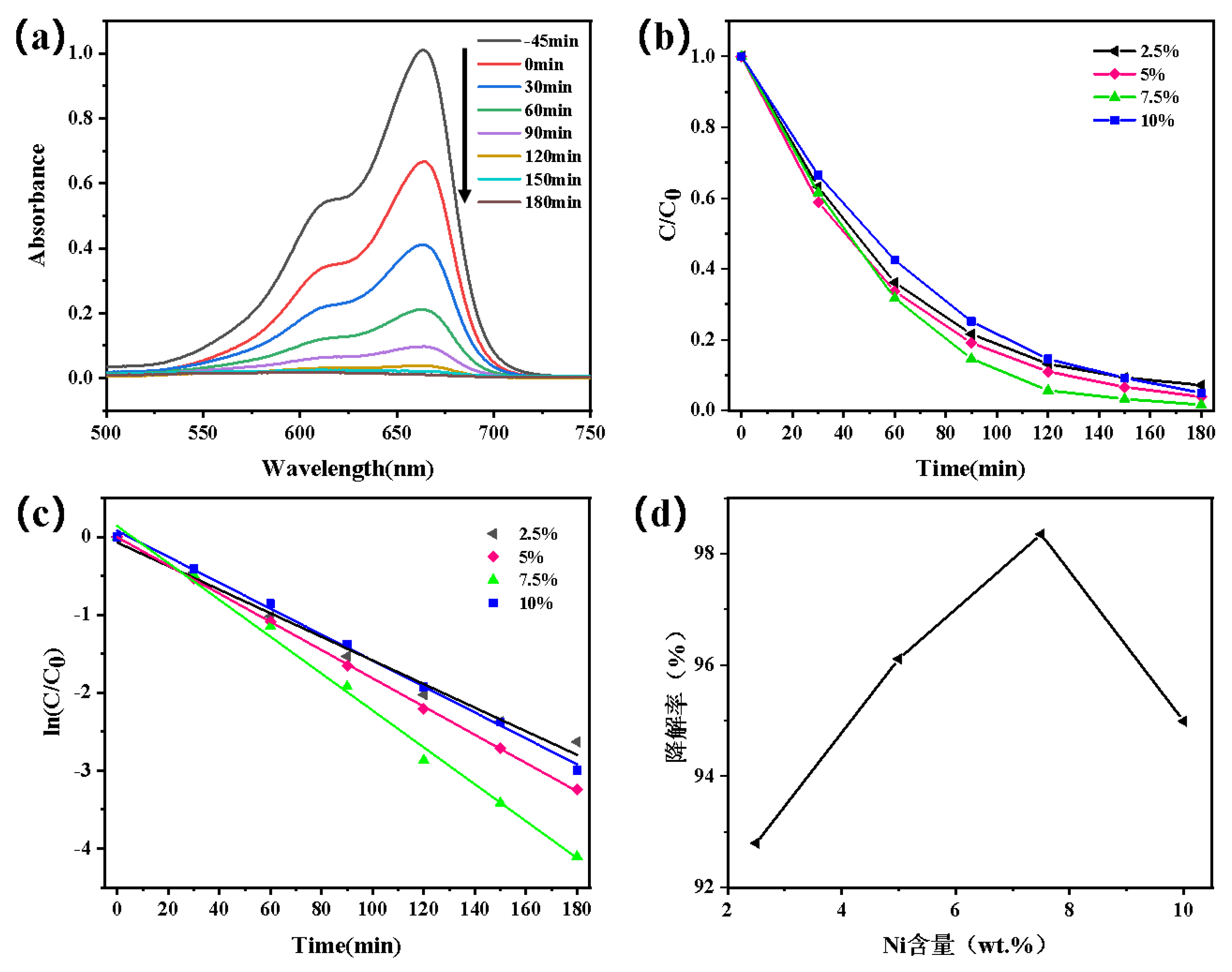
3.1.5. Study on the Performance of Photocatalytic Degradation of Dyes
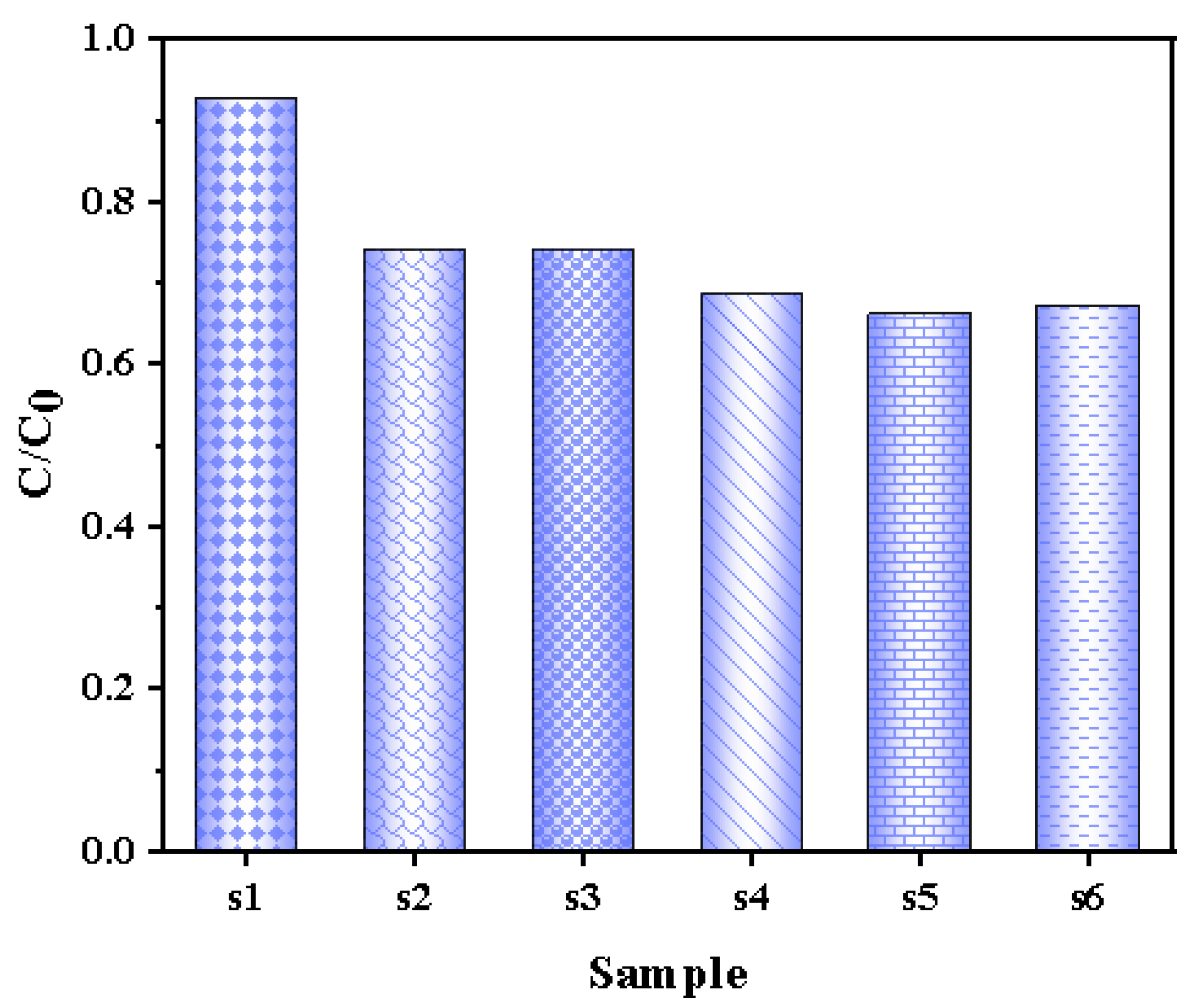
3.1.6. Adsorption Performance of Photocatalysts
3.2. Effect of Heat Treatment Temperature on the Crystal Phase and Photocatalytic Performance of Catalysts
3.2.1. XRD Analysis
3.2.2. SEM Analysis
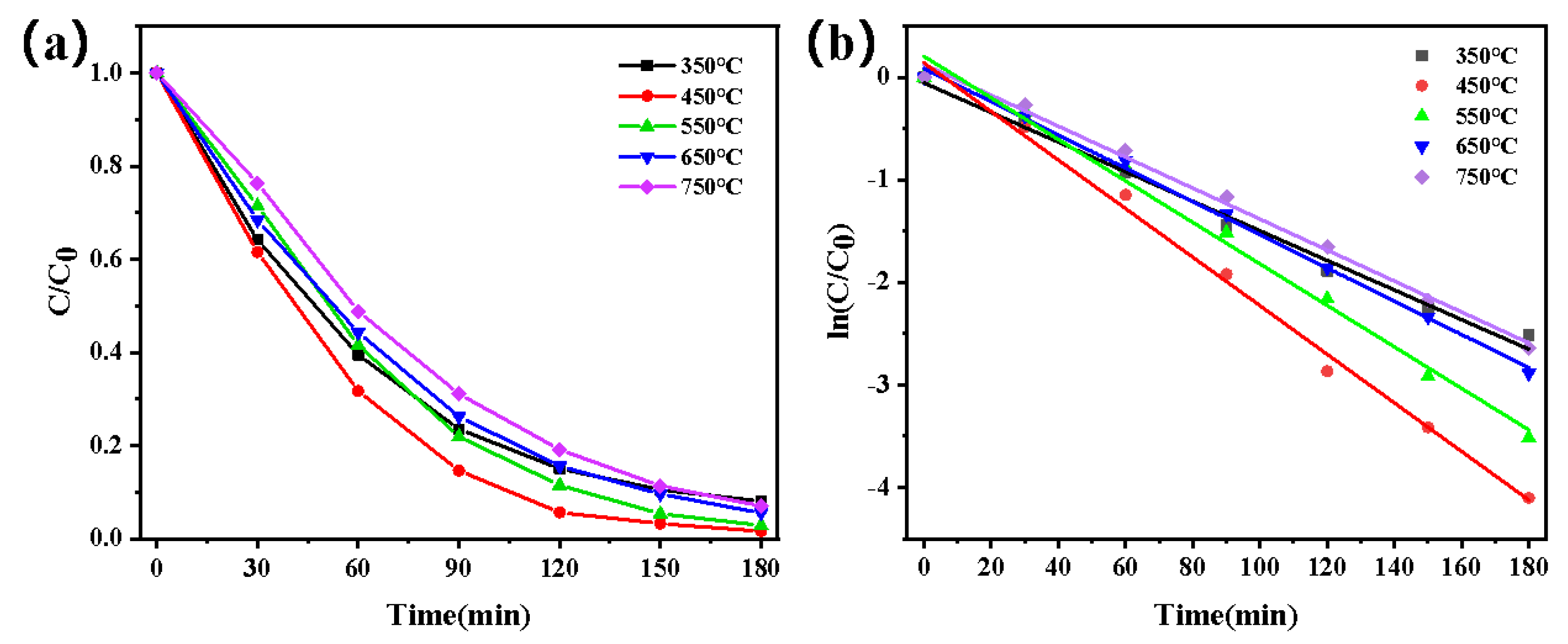
3.2.3. Study on Photocatalytic Degradation Performance of Dyes
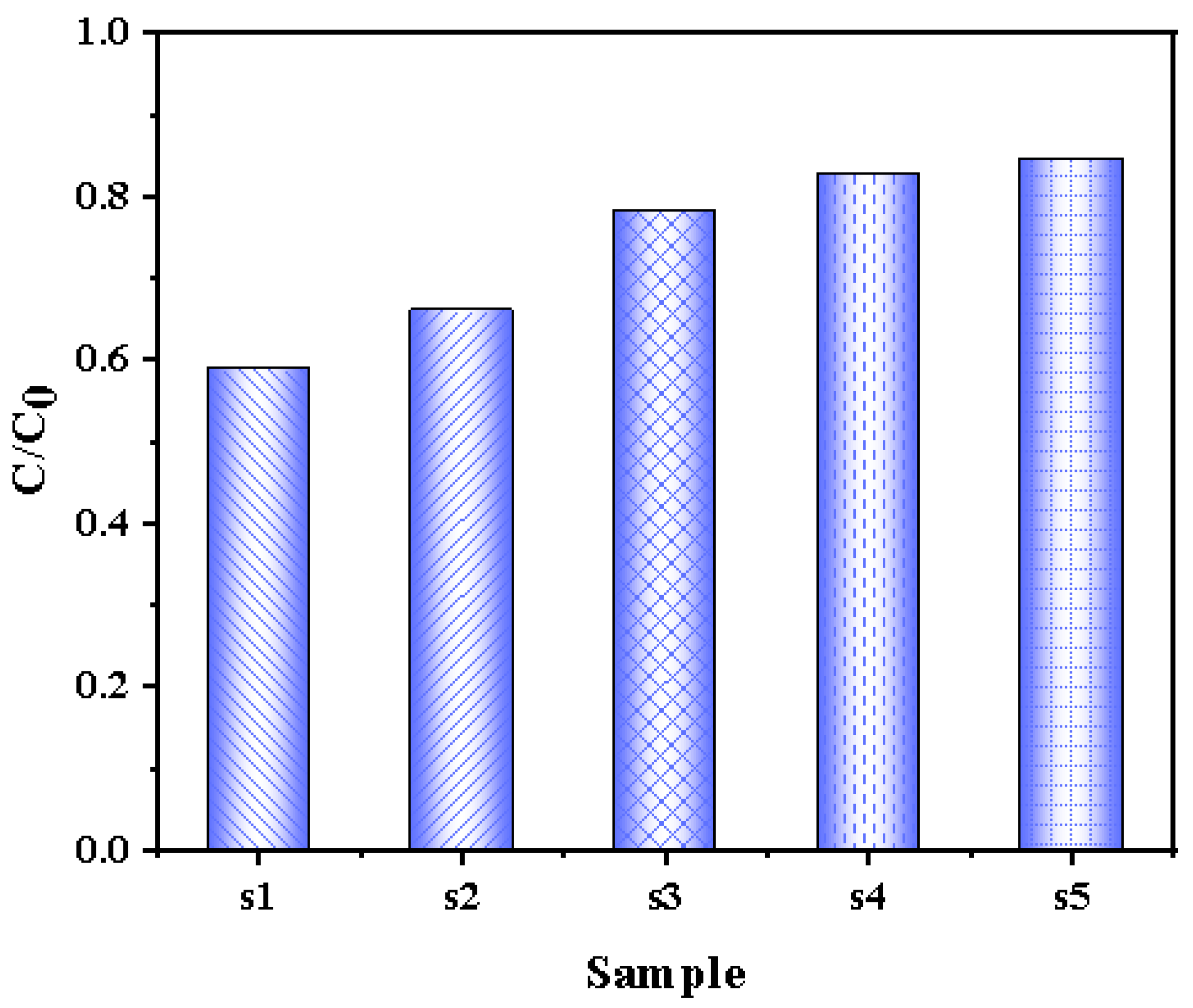
3.2.4. Adsorption Performance of Photocatalysts
4. Conclusions
Funding
Data Availability Statement
References
- Lu, E. J.; Zou, J. H. Research progress of metal/TiO₂ photocatalysts. Journal of Ceramics 2024, vol. 45(no. 06), 1083–1097. [Google Scholar]
- Sun, R. Y.; Zhao, X. Y. Preparation and application progress of titanium dioxide photocatalysts. Contemporary Chemical Industry 2023, vol. 52(no. 01), 202–208. [Google Scholar]
- Hu, H. S.; Zhang, P. H. Preparation of S-scheme TiO₂/C₃N₅ heterojunction photocatalyst and its performance in methylene blue degradation. China Surfactant Detergent & Cosmetics (in Chinese and English) 2025, vol. 55(no. 08), 980–988. [Google Scholar]
- Gao, Z. Y.; Cen, Y. H.; Zhang, C. Y. Preparation of titanium dioxide nanospheres and study on their resonance properties in visible light band. Journal of Guangzhou University (Natural Science Edition) 2025, vol. 24(no. 03), 1–9. [Google Scholar]
- Zhang, J.; M. R. Guo, S.; et al. Photocatalytic degradation of tetracycline hydrochloride by Fe/Ni-TiO₂ composite. In Environmental Protection of Chemical Industry; Online in Press, 2025; pp. 1–7. [Google Scholar]
- Zhang, H. J.; Zhang, G. D.; Wang, C. Preparation and photocatalytic performance of porous TiO₂/CdS nanomaterials. Transducer and Microsystem Technologies 2022, vol. 41(no. 12), 14–18. [Google Scholar]
- Wei, J. P.; Ge, Y. L.; et al. Preparation of two-dimensional anatase/rutile composite and its photocatalytic performance. Chemical Engineer 2025, vol. 39(no. 04), 14–18. [Google Scholar]
- Wang, D. Z.; F. T. Liu, L.; et al. Preparation of porous nano-titanium dioxide thin films and their photocatalytic activity. Journal of Ceramics 2006, no. 04, 397–401. [Google Scholar]
- Chen, X.; Wang, L. Y.; et al. Preparation of anatase-rutile mixed crystal TiO₂ nanofibers and their photocatalytic performance. Journal of Jilin Jianzhu University 2021, vol. 38(no. 03), 57–62. [Google Scholar]
- JIANG, B. F. Study on photocatalytic treatment of dye-containing wastewater by supported modified nano-TiO₂. Dissertation, Harbin Inst. Technol., Harbin, China, 2010. [Google Scholar]
- DAI, J. W.; YANG, J. Y.; GAO, X.; et al. Preparation and performance of zinc-doped titanium dioxide photocatalytic antibacterial films. Mod. Transp. Metall. Mater. vol. 5(no. 6), 12–18, 49, 2025.
- LOU, F. P. Preparation, characterization and photocatalytic performance of nano-titanium dioxide. Dissertation, Zhejiang Univ. Technol., Hangzhou, China, 2015. [Google Scholar]
- ZHANG, J.; GUO, M. R.; WANG, S. Q.; et al. Photocatalytic degradation of tetracycline hydrochloride by Fe/Ni-TiO₂ composites. Environ. Prot. Chem. Ind. 2025, vol. 45(no. 6), 876–881. [Google Scholar]
- LI, P. Y. Preparation and adsorption-degradation performance of carbon/titanium dioxide composites. Dissertation, Henan Normal Univ., Xinxiang, China, 2017. [Google Scholar]
- YU, C. Y.; WANG, B.; SONG, T. Research progress in photocatalytic treatment of organic wastewater. Liaoning Chem. Ind. 2025, vol. 54(no. 9), 1568–1571. [Google Scholar]
- LI, P. Y. Preparation and adsorption-degradation performance of carbon/titanium dioxide composites. Dissertation, Henan Normal Univ., Xinxiang, China, 2017. [Google Scholar]
- ZHENG, X. Research on the application of photocatalytic technology in sewage treatment. Chem. Eng. Equip. 2025, no. 7, 173–175. [Google Scholar]
- WU, Z. Preparation and performance study of titanium dioxide photocatalytic functional materials. Dissertation, Nanjing Univ. Sci. Technol., Nanjing, China, 2018. [Google Scholar]
- WEI, F.; DING, H.; LI, H.; et al. Preparation of TiO₂ self-cleaning antibacterial glaze by low-temperature secondary firing method. China Ceramics 2025, vol. 61(no. 7), 64–69. [Google Scholar]
- XIANG, W. J.; ZHU, C. J.; LIU, D.; et al. Photocatalytic degradation of nitrogen oxides by titanocene dichloride under visible light. Appl. Chem. Ind. 2019, vol. 48(no. 9), 2153–2155. [Google Scholar]
- HE, X. W.; ZHOU, S. R.; LI, G. Z.; et al. Preparation and photocatalytic study of rare earth praseodymium and gadolinium ions doped modified nano-TiO₂. J. Funct. Mater. vol. 56(no. 5), 5135–5142, 5166, 2025.
- WANG, Z. G.; WEI, J. J.; WANG, H. C. Application progress of titanium dioxide photocatalysis technology in water quality treatment. Guangzhou Chem. Ind. 2020, vol. 48(no. 6), 27–29, 32. [Google Scholar]
- SHANG, Z. H.; LYU, M. Z.; LI, X. Y.; et al. Preparation and application of different photothermal and photocatalytic nanofiber membranes. Knitting Ind. 2025, no. 2, 44–47. [Google Scholar]
- LIU, Q. H.; LI, K. J.; WU, S. H.; et al. Degradation of dye wastewater by nano-porous titanium dioxide microspheres photocatalyst. J. Qingdao Univ. Sci. Technol. (Nat. Sci. Ed.) 2021, vol. 42(no. 1), 72–77. [Google Scholar]
- HOU, B. S.; ZENG, J. T.; LU, L. Y.; et al. Research progress of modified TiO₂ photocatalytic degradation of organic dyes. Contemp. Chem. Res. 2024, no. 23, 16–18. [Google Scholar]
- LYU, M. Z.; LIU, L. W.; LIU, Z. Q.; et al. Preparation and photocatalytic performance of FePc/TiO₂ loaded nanofiber membranes. Knitting Ind. 2021, no. 3, 58–61. [Google Scholar]
- ZHANG, J. R.; MA, Z. S.; HE, S. H. Research progress on preparation and modification of nano-TiO₂ photocatalyst materials. Dyeing Finishing 2024, vol. 50(no. 11), 70–75, 79. [Google Scholar]
- ZHANG, Y. F. Preparation of TiO₂-based nano-composite photocatalysts and their photodegradation performance on dyes. Dissertation, Soochow Univ., Suzhou, China, 2021. [Google Scholar]
- PENG, H. K.; ZHANG, M.; YAO, N.; et al. Research progress of photocatalytic treatment of microplastics in water environment. Appl. Chem. Ind. 2024, vol. 53(no. 10), 2479–2483. [Google Scholar]
- LIU, Y.; XU, H. L.; WANG, X. Q. Research progress on preparation and application of TiO₂ photocatalysts. Contemp. Chem. Ind. 2021, vol. 50(no. 9), 2217–2220. [Google Scholar]
- YANG, J.; CHEN, X.; LIU, C. H.; et al. Development and application of photocatalytic technology in energy and environment. Solar Energy 2024, no. 7, 50–61. [Google Scholar]
- WANG, J. Z.; AO, Q. B.; JING, P.; et al. Preparation and application of porous titanium. Rare Met. Mater. Eng. 2022, vol. 51(no. 5), 1907–1918. [Google Scholar]
- WANG, H. O.; YE, F.; LIAO, C. Z.; et al. Open comprehensive teaching design and practice of titanium dioxide doping modification and its application in photocatalytic degradation of water pollution. Guangdong Chem. Ind. 2024, vol. 51(no. 8), 146–149, 145. [Google Scholar]
- LI, C. W.; YANG, Y. F.; TAO, S. P. Effect of sintering process on microstructure and properties of porous titanium-based alloys. World Nonferrous Met. 2023, no. 9, 1–3. [Google Scholar]
- LI, L. R.; YANG, X. Z.; DENG, Z. W.; et al. Rare earth modified photocatalytic hydrogen production pathway under the 'double carbon' goal. Oil Gas New Energy 2024, vol. 36(no. 1), 59–65. [Google Scholar]
- DU, R. C.; LI, Y.; WANG, T.; et al. Research progress on modification of nano-TiO₂ photocatalysts. Chem. Bull. 2023, vol. 86(no. 10), 1172–1180. [Google Scholar]
- ZHOU, Y.; YE, L.J. Preparation of Nd³⁺-doped TiO₂/rGO and its photocatalytic performance. Chem. Technol. Dev. 2024, vol. 53 Suppl. 1, 17–25. [Google Scholar]
- XIE, H. Y.; MA, C.W.; SUN, Z. G.; et al. Preparation of polycrystalline nano-titanium dioxide by flame chemical vapor deposition and its application in gas purification. J. Shanghai Polytech. Univ. 2014, vol. 31(no. 1), 18–31. [Google Scholar]
- WU, D. K.; LEI, K.; SUN, H. P. Characteristics of Pt-doped TiO₂ photocatalysts and their effects on photocatalytic hydrogen production performance. Adhesion 2025, vol. 52(no. 11), 18–21. [Google Scholar]
- HUANG, R. Y. Preparation and photocatalytic performance of titanium dioxide-based photocatalysts. Dissertation, Jiangxi Univ. Sci. Technol., Ganzhou, China, 2016. [Google Scholar]
- WANG, S. Z. Synthesis and performance study of titanium nanotubes. Environ. Sci. Guide (in Chinese). vol. 44(no. 5), 58–61, 2025. [CrossRef]
- JIANG, Z. Synthesis and activity study of titanium-containing photocatalysts. Dissertation, Wuhan Univ., Wuhan, China, 2017. [Google Scholar]
- HAN, L. H.; GUO, J. Y.; MA, Q. H.; et al. Research progress in modified nano-TiO₂ photocatalysts for dye degradation. Liaoning Chem. Ind. 2025, vol. 54(no. 8), 1412–1416. [Google Scholar]
- WANG, J. G. Construction and catalytic performance of high-efficiency photocatalysts. Proc. 11th Natl. Conf. Environ. Catal. Environ. Mater., Shanghai Univ. Eng. Sci., Shanghai, China, 2018; p. 130. [Google Scholar]
- LIN, Y. J.; QIAO, P.; LI, X. R.; et al. Heterostructure construction strategies and application research progress of TiO₂ nano-photocatalysts. Chem. Ind. Eng. Prog. 2025, vol. 44 Suppl. 1, 159–177. [Google Scholar]
- ZHANG, T.; YAN, X. Y.; YUAN, H. F.; et al. Research progress of titanium dioxide photocatalysts. Shanxi Chem. Ind. 2018, vol. 38(no. 5), 40–42. [Google Scholar]
- HUANG, R. F. Preparation of water-soluble titanium citrate-based photocatalysts and their quasi-homogeneous photocatalytic performance. Henan Univ. Technol., Zhengzhou, China, 2025. [Google Scholar]
- MA, Y.; ZHENG, Q. Research progress of metal-doped nano-titanium dioxide photocatalysts. J. Yulin Univ. 2020, vol. 30(no. 2), 50–55. [Google Scholar]
- QIAO, Y. J.; SUN, J.; LI, J.; et al. Preparation and characterization of nano-titanium dioxide photocatalysts. Henan Build. Mater. 2020, no. 5, 35–36, 39. [Google Scholar]

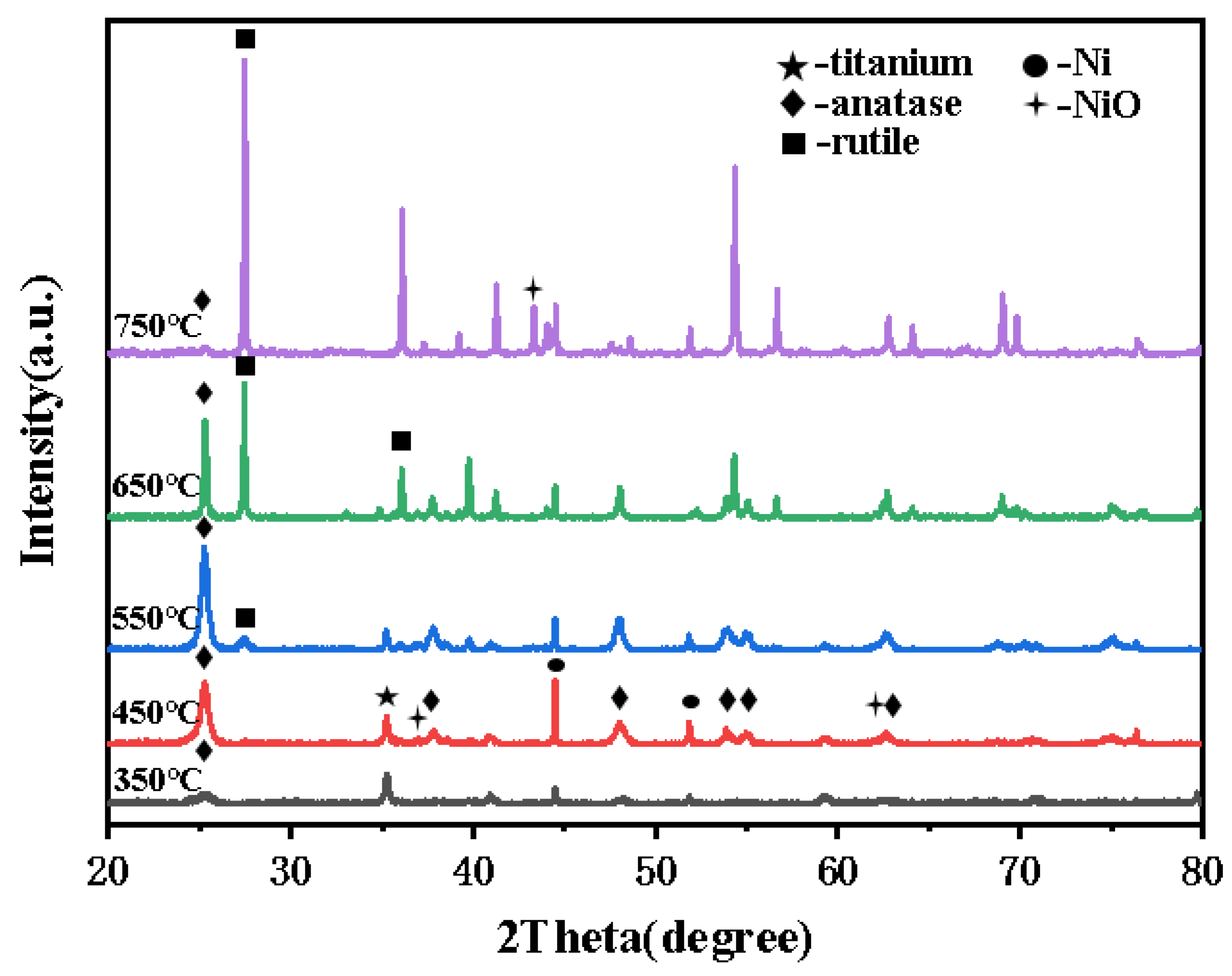

| calcination temperature (℃) | average grain size (nm) | mass fraction(%) | ||
| anatase(101) | Rutile (110) | anatase | Rutile | |
| 350 | 9.1 | —— | 100 | —— |
| 450 | 14.05 | —— | 100 | —— |
| 550 | 17.4 | 16.8 | 81.7 | 18.3 |
| 650 | 44.15 | 65.4 | 40.1 | 59.9 |
| 750 | 27.7 | 85.5 | 4.9 | 95.1 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




