Submitted:
17 April 2026
Posted:
20 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AIH | Artificial Insemination with Husband’s Semen |
| AGA | Appropriate for Gestational Age |
| ART | Assisted Reproductive Technology |
| AGA-NWGI | Age–Neonatal Weight Gain Insufficiency |
| CV | Central Vein |
| c/s | Cesarean Section |
| DD | Dichorionic Diamniotic |
| EGG | Energetics of Gestation and Growth |
| EUGR | Extrauterine Growth Restriction |
| ET | Early Term |
| FET | Frozen-Thawed Embryo Transfer |
| FY | Fiscal Year |
| IV | Intravenous |
| IVF-ET | in vitro fertilization–embryo transfer |
| LGA | Large for Gestational Age |
| LP | Late Preterm |
| MD | Monochorionic Diamniotic |
| ND | No Data |
| NICU | Neonatal Intensive Care Unit |
| SDS | Standard Deviation Score |
| SGA | Small for Gestational Age |
| TTN | Transient Tachypnea of the Newborn |
| WISC-IV | Wechsler Intelligence Scale for Children–Fourth Edition |
References
- WHO: recommended definitions, terminology, and format for statistical tables related to the perinatal period and use of a new certificate for cause of perinatal deaths. Modifications recommended by FIGO as amended October 14, 1976. Acta Obstet Gynecol Scand 1977, 56, 247–253.
- Goldenberg, R.L.; Culhane, J.F.; Iams, J.D.; Romero, R. Epidemiology and causes of preterm birth. Lancet 2008, 371, 75–84. [Google Scholar] [CrossRef]
- Raju, T.N.; Higgins, R.D.; Stark, A.R.; Leveno, K.J. Optimizing care and outcome for late-preterm (near-term) infants: a summary of the workshop sponsored by the National Institute of Child Health and Human Development. Pediatrics 2006, 118, 1207–1214. [Google Scholar] [CrossRef]
- Spong, C.Y. Defining “term” pregnancy: recommendations from the Defining “Term” Pregnancy Workgroup. JAMA 2013, 309, 2445–2446. [Google Scholar] [CrossRef]
- Office for Vital and Health Statistics; Ministry of Health; Labor and Welfare. Annual Report Section II. “Response regarding statistical information on gestational weeks at birth.”. October 2023. [Google Scholar]
- Engle, W.A.; Tomashek, K.M.; Wallman, C. “Late-preterm” infants: a population at risk. Pediatrics 2007, 120, 1390–1401. [Google Scholar] [CrossRef]
- Petrini, J.R.; Dias, T.; McCormick, M.C.; Massolo, M.L.; Green, N.S.; Escobar, G.J. Increased risk of adverse neurological development for late preterm infants. J Pediatr 2009, 154, 169–176. [Google Scholar] [CrossRef] [PubMed]
- Morse, S.B.; Zheng, H.; Tang, Y.; Roth, J. Early school-age outcomes of late preterm infants. Pediatrics 2009, 123, e622–e629. [Google Scholar] [CrossRef]
- McGowan, J.E.; Alderdice, F.A.; Doran, J.; Holmes, V.A.; Jenkins, J.; Craig, S.; Johnston, L. Impact of neonatal intensive care on late preterm infants: developmental outcomes at 3 years. Pediatrics 2012, 130, e1105–e1112. [Google Scholar] [CrossRef] [PubMed]
- Chan, E.; Quigley, M.A. School performance at age 7 years in late preterm and early term birth: a cohort study. Arch Dis Child Fetal Neonatal Ed 2014, 99, F451–F457. [Google Scholar] [CrossRef]
- Delnord, M.; Zeitlin, J. Epidemiology of late preterm and early term births - An international perspective. Semin Fetal Neonatal Med 2019, 24, 3–10. [Google Scholar] [CrossRef] [PubMed]
- Stewart, D.L.; Barfield, W.D. Committee on Fetus and Newborn Updates on an at-risk population: late-preterm and early-term infants. In Pediatrics; Cummings, J.J., Adams-Chapman, I.S., Aucott, S.W., Goldsmith, J.P., Hand, I.L., Juul, S.E., Poindexter, B.B., Puopolo, K.M., Eds.; 2019; Volume 144, p. e20192760. [Google Scholar]
- Yang, S.; Platt, R.W.; Kramer, M.S. Variation in child cognitive ability by week of gestation among healthy term births. Am J Epidemiol 2010, 171, 399–406. [Google Scholar] [CrossRef] [PubMed]
- Stene-Larsen, K.; Lang, A.M.; Landolt, M.A.; Latal, B.; Vollrath, M.E. Emotional and behavioral problems in late preterm and early term births: outcomes at child age 36 months. BMC Pediatr 2016, 16, 196. [Google Scholar] [CrossRef]
- Neonatal Committee of the Japan Pediatric Society. Revision of the new standard values for physical measurements at birth by gestational age. J Jpn Pediatr Soc 2010, 114, 1771–1806. [Google Scholar]
- Gyamfi-Bannerman, C.; Gantt, A.B.; Miller, R.S. Medically indicated late-preterm and early-term deliveries: ACOG Committee Opinion, Number 831. Obstet Gynecol 2021, 138, e35–e39. [Google Scholar]
- e-Stat. Vital statistics survey. Vital statistics. Confirmed figures. Births. Supplementary statistical tables (tables not included in reports) > Births > Annual (2015). Available online: https://www.e-stat.go.jp/dbview?sid=0003411912 (accessed on 13 September 2025).
- National Institute of Population and Social Security Research. Population Statistics Handbook (2025 Edition). IV Births and stillbirths. Table 4-1 Number of births by gender and birth sex ratio: 1873-2023. Available online: https://www.ipss.go.jp/syoushika/tohkei/Popular/P_Detail2025.asp?fname=T04-01.htm (accessed on 11 September 2025).
- e-Stat. Vital statistics survey. Vital statistics. Confirmed figures. Births. Available online: https://www.e-stat.go.jp/dbview?sid=0003411616 (accessed on 12 September 2025).
- Japan Society of Obstetrics and Gynecology Registration and Survey Subcommittee. 2022 Clinical performance results of in vitro fertilization and embryo transfer. Available online: https://www.jsog.or.jp/activity/art/2022_JSOG-ART.pdf (accessed on 12 September 2025).
- Ministry of Health; Labor and Welfare. Overview of vital statistics [confirmed figures] for Reiwa 1 [2019]. 2020. Available online: https://www.mhlw.go.jp/toukei/saikin/hw/jinkou/kakutei19/ (accessed on 12 September 2025).
- Ministry of Health; Labour and Welfare. Overview of the 2023 medical facilities (static/dynamic) survey and hospital report. Available online: https://www.mhlw.go.jp/toukei/saikin/hw/iryosd/23/ (accessed on 12 September 2025).
- Ishitsuka, K.; Piedvache, A.; Kobayashi, S.; Iwama, N.; Nishimura, T.; Watanabe, M.; Metoki, H.; Iwata, H.; Miyashita, C.; Ishikuro, M.; Obara, T. The population-attributable fractions of small-for-gestational-age births: results from the Japan Birth Cohort Consortium. Nutrients 2024, 16, 186. [Google Scholar] [CrossRef]
- Iizuka, Y. The experience of mother-infant separation and reunification for mothers of low birth weight infants admitted to the NICU (translated by the author). J Dev Psychol Res (In Japanese) 2013, 24, 263–272. [Google Scholar]
- Mizuno, K.; Ueda, A. The maturation and coordination of sucking, swallowing, and respiration in preterm infants. J Pediatr 2003, 142, 36–40. [Google Scholar] [CrossRef]
- Dunsworth, H.M.; Warrener, A.G.; Deacon, T.; Ellison, P.T.; Pontzer, H. Metabolic hypothesis for human altriciality. Proc Natl Acad Sci U S A 2012, 109, 15212–15216. [Google Scholar] [CrossRef]
- Christiansen, F.; Uhart, M.M.; Bejder, L.; Clapham, P.; Ivashchenko, Y.; Tormosov, D.; Lewin, N.; Sironi, M. Fetal growth, birth size and energetic cost of gestation in southern right whales. J Physiol 2022, 600, 2245–2266. [Google Scholar] [CrossRef]
- Bhatia, J.; Mena, P.; Denne, S.; García, C. Evaluation of adequacy of protein and energy. J Pediatr 2013, 162, S31–S36. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, K.; Sakurai, M.; Itabashi, K.; et al. Study on the transition of SD scores of anthropometric measurements during NICU hospitalization in preterm low birth weight infants (translated by the author). J Jpn Soc Premature Newborn Med (In Japanese) 2015, 27, 77–83. [Google Scholar]
- Egashira, T.; Tomino, H.; Ogiwara, S.; et al. Influence of enteral nutrition content during NICU hospitalization on the incidence of extrauterine growth restriction (translated by the author). Jpn J Perinat Neonat Med (In Japanese) 2022, 58, 516–522. [Google Scholar]
- Embleton, N.E.; Pang, N.; Cooke, R.J. Postnatal malnutrition and growth retardation: an inevitable consequence of current recommendations in preterm infants. Pediatrics 2001, 107, 270–273. [Google Scholar] [CrossRef] [PubMed]
- Ehrenkranz, R.A.; Dusick, A.M.; Vohr, B.R.; Wright, L.L.; Wrage, L.A.; Poole, W.K. Growth in the neonatal intensive care unit influences neurodevelopmental and growth outcomes of extremely low birth weight infants. Pediatrics 2006, 117, 1253–1261. [Google Scholar] [CrossRef] [PubMed]
- Guellec, I.; Lapillonne, A.; Marret, S.; Picaud, J.C.; Mitanchez, D.; Charkaluk, M.L.; Fresson, J.; Arnaud, C.; Flamand, C.; Cambonie, G.; Kaminski, M. Effect of intra- and extrauterine growth on long-term neurologic outcomes of very preterm infants. J Pediatr 2016, 175, 93–9.e1. [Google Scholar] [CrossRef]
- González-López, C.; Solís-Sánchez, G.; Lareu-Vidal; Mantecón-Fernández, S.; Ibáñez-Fernández, L.; Rubio-Granda, A.; Suárez-Rodríguez, A.M. Variability in definitions and criteria of extrauterine growth restriction and its association with neurodevelopmental outcomes in preterm infants: a narrative review. Nutrients 2024, 16, 968. [Google Scholar] [CrossRef]
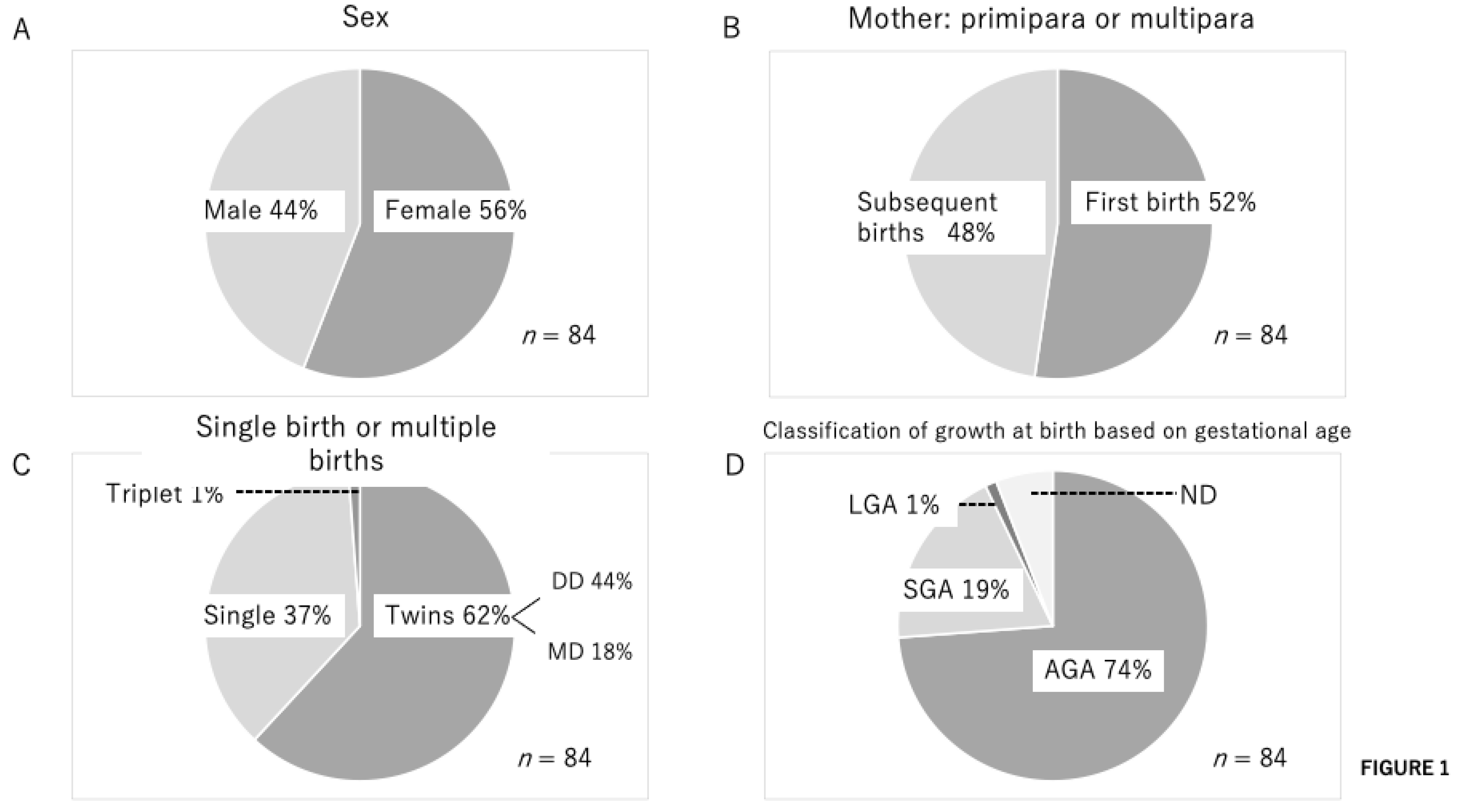
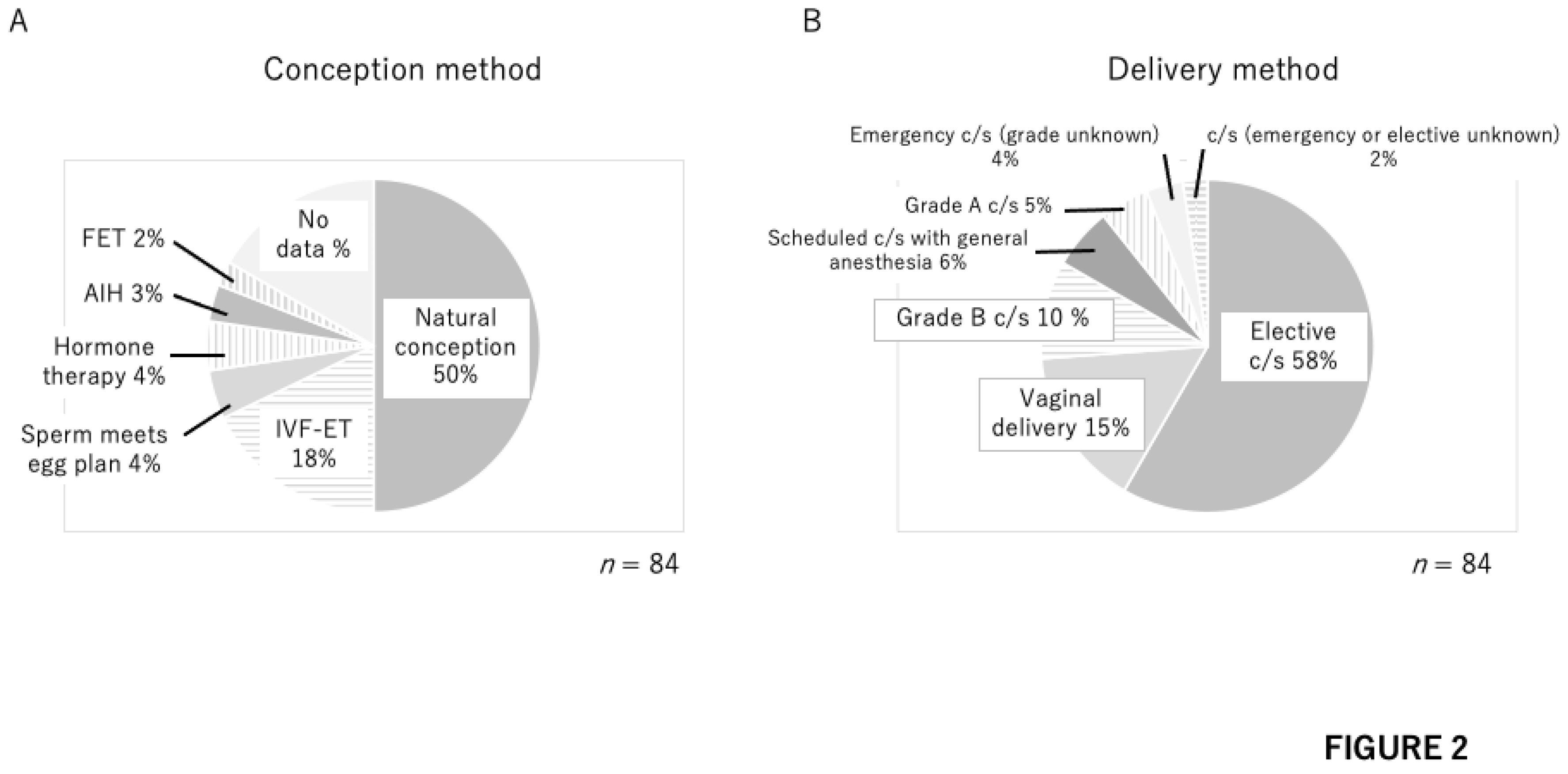
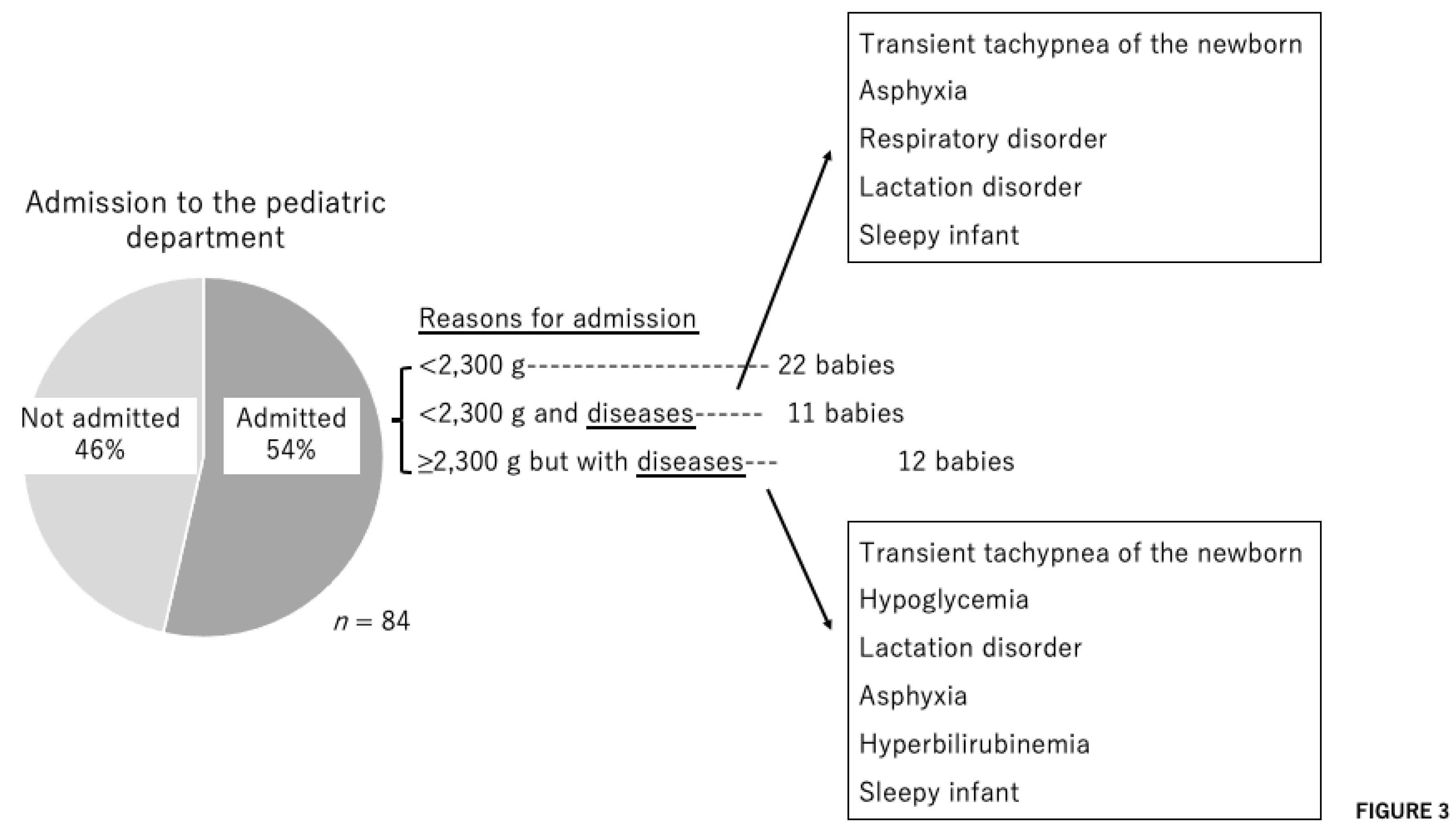
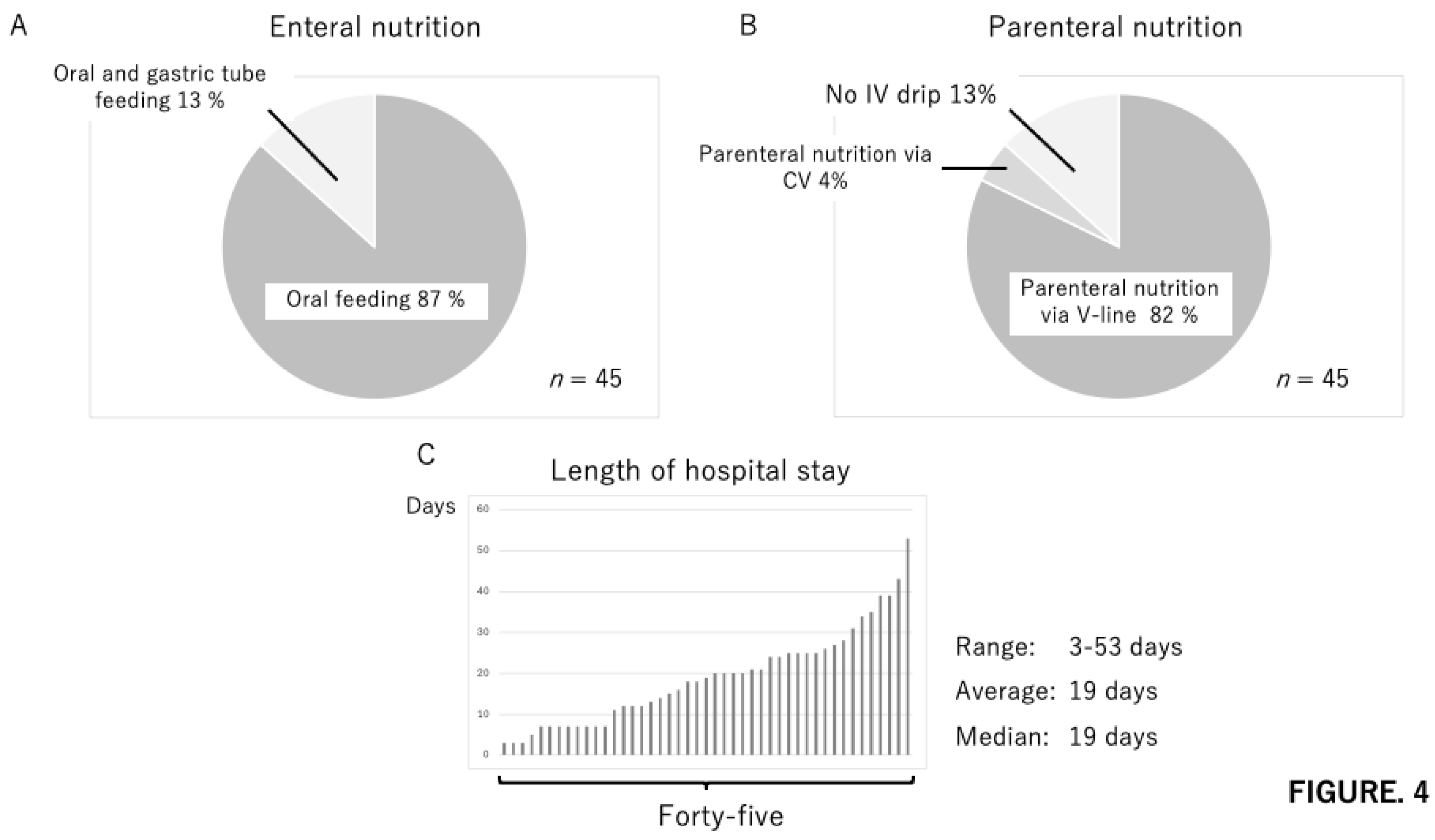
| Baby | Baby A | Baby B | Baby C |
| Sex | Female | Female | Female |
| Mother | Multipara | Multipara | Primipara |
| Gestational age | 36w5d | 36w5d | 36w6d |
| Birth weight | 2250 g | 2198 g | 2222 g |
| Birth height | 45.6 cm | 45.4 cm | 46.0 cm |
| Birth weight percentile | 16.4 | 12.6 | 17.4 |
| Birth height percentile | 27.1 | 24.3 | 29.8 |
| Growth class at birth | AGA | AGA | AGA |
| Weight measurement at 39 corrected weeks | Corrected 39w3d | Corrected 39w3d | Corrected 39w2d |
| Age | Day 18 | Day 18 | Day 18 |
| Birth weight at 39 corrected weeks | 2537 g | 2589 g | 2494 g |
| Birth weight percentile at 39 corrected weeks | 7.2 | 9.7 | 9.9 |
| Conception method | Natural conception | Natural conception | ND |
| Maternal complications | Graves’ disease | Graves’ disease | Uterine fibroid |
| Obstetric complications | None | None | Preterm labor, umbilical cord prolapse |
| Delivery method | Selective C/S | Selective C/S | GradeA C/S |
| Single or multiple birth | DD twin | DD twin | Single |
| Baby’s diagnosis | LBWI, TTN | LBWI | LBWI |
| Enteral nutrition | Oral feeding | Oral feeding | Oral feeding |
| Parenteral nutrition | None | None | None |
| Length of hospitalization | 20 days | 20 days | 20 days |
| Follow-up | Up to 1 year and 7 months old | Up to 1 year and 7 months old | Up to 1 month old |
| Child | Gestational age | Birth weight | Single or multiple births | Age: developmental assessment (results) |
| Child A | 36w4d | 2316 g | DD twin | 2 yrs and 2 mos: New Kyoto Scale (appropriate for age) |
| 2 yrs and 11 mos: Enjohji Test (appropriate for age) | ||||
| Child B | 36w4d | 2048 g | DD twin | 2 yrs and 2 mos: New Kyoto Scale (appropriate for age) |
| 2 yrs and 11 mos: Enjohji Test (appropriate for age) | ||||
| Child C | 36w5d | 1944 g | DD twin | 3 yrs and 0 mo: Enjohji Test (appropriate for age) |
| Child D | 36w0d | 2132 g | DD twin | 2 yrs and 4 mos: New Kyoto Scale (appropriate for age) |
| 3 yrs and 7 mos: Enjohji Test (appropriate for age) | ||||
| Child E | 36w0d | 1676 g | DD twin | 2 yrs and 4 mos: New Kyoto Scale (slightly below age-appropriate level) |
| Child F | 36w0d | 1502 g | Single | 1 yr and 6 mos: New Kyoto Scale (slightly below age-appropriate level) |
| 2 yrs and 11 mos: New Kyoto Scale (appropriate for age) | ||||
| 3 yrs and 6 mos: New Kyoto Scale (appropriate for age) | ||||
| Child G | 36w0d | 1588 g | MD twin | 1 yr and 7 mos: New Kyoto Scale (appropriate for age) |
| 3 yrs and 0 mo: Enjohji Test (appropriate for age) | ||||
| Child H | 36w0d | 1821 g | MD twin | 1 yr and 7 mos: New Kyoto Scale (appropriate for age) |
| 3 yrs and 0 mo: Enjohji Test (appropriate for age) | ||||
| Child I | 36w5d | 2080 g | DD twin | 1 yr and 6 mos: Enjohji Test (appropriate for age) |
| 2 yrs and 0 mo: Enjohji Test (appropriate for age), | ||||
| 9 yrs and 0 mo: WISC-4 (slightly below age-appropriate level) | ||||
| Child J | 36w4d | 1841 g | DD twin | 3 yrs and 0 mo: Enjohji Test (appropriate for age) |
| Child K | 36w0d | 2130 g | Single | 1 yr and 11 mos: Enjohji Test (appropriate for age) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




