Submitted:
18 April 2026
Posted:
20 April 2026
You are already at the latest version
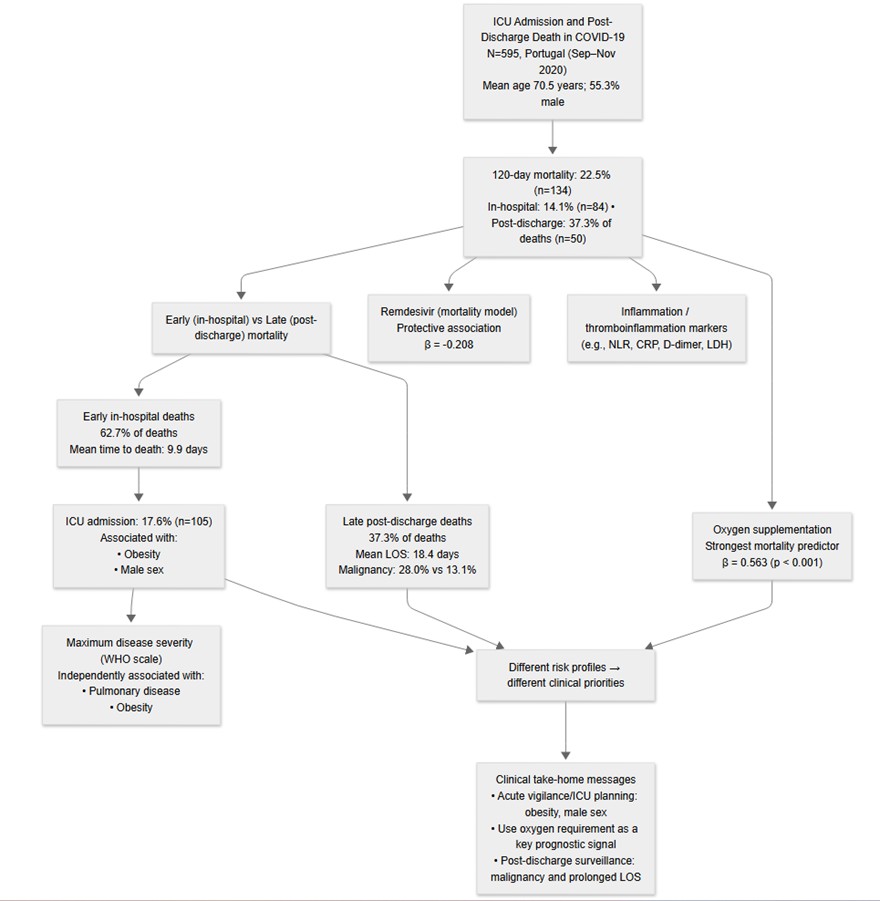
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Design and Patients
2.2. Baseline Demographic and Clinical Variables
2.3. WHO Ordinal Scale and Severity Definitions
2.4. Outcomes
2.5. Statistical Analysis
3. Results
3.1. Cohort Demographic and Clinical Characteristics
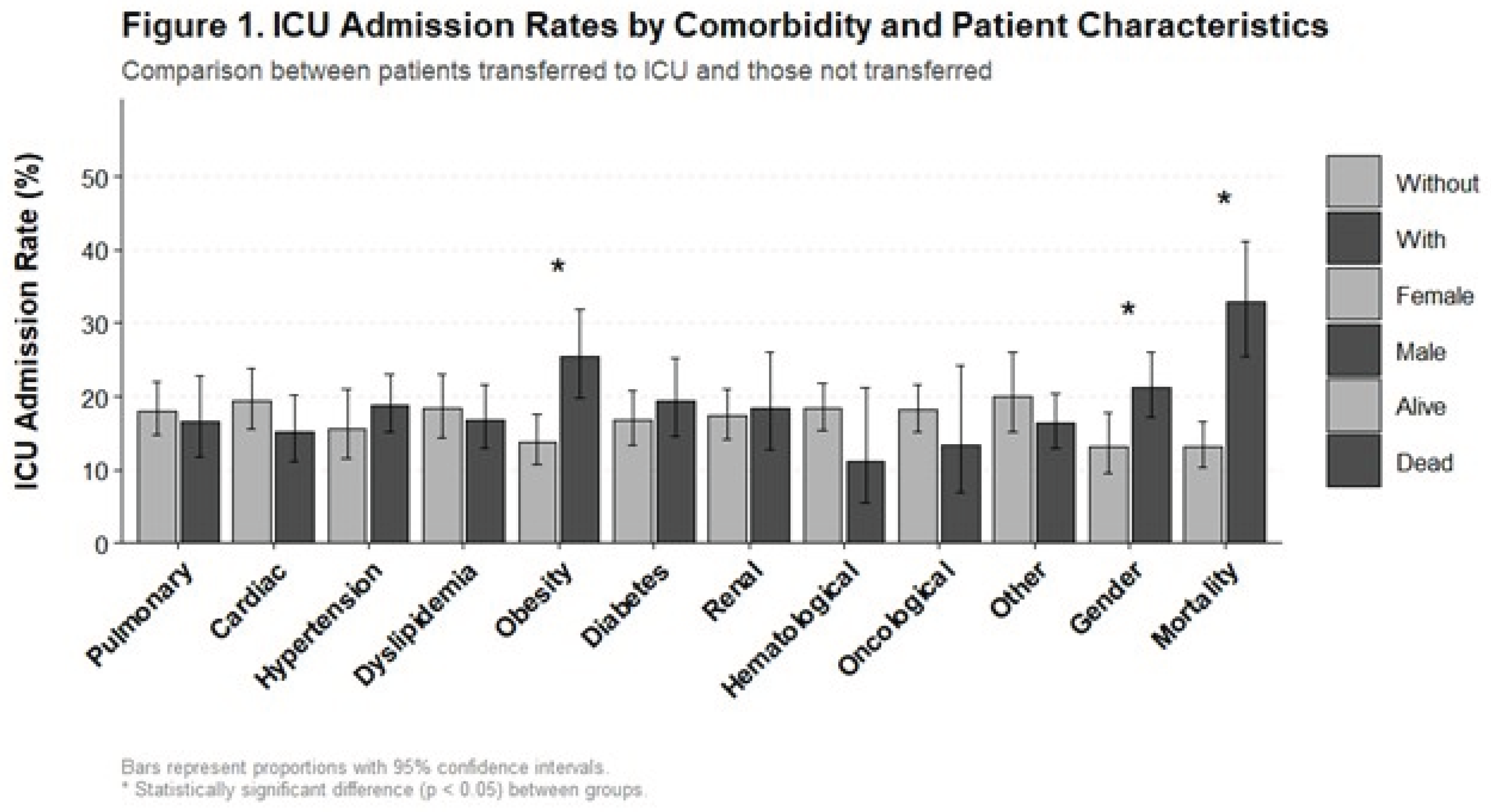
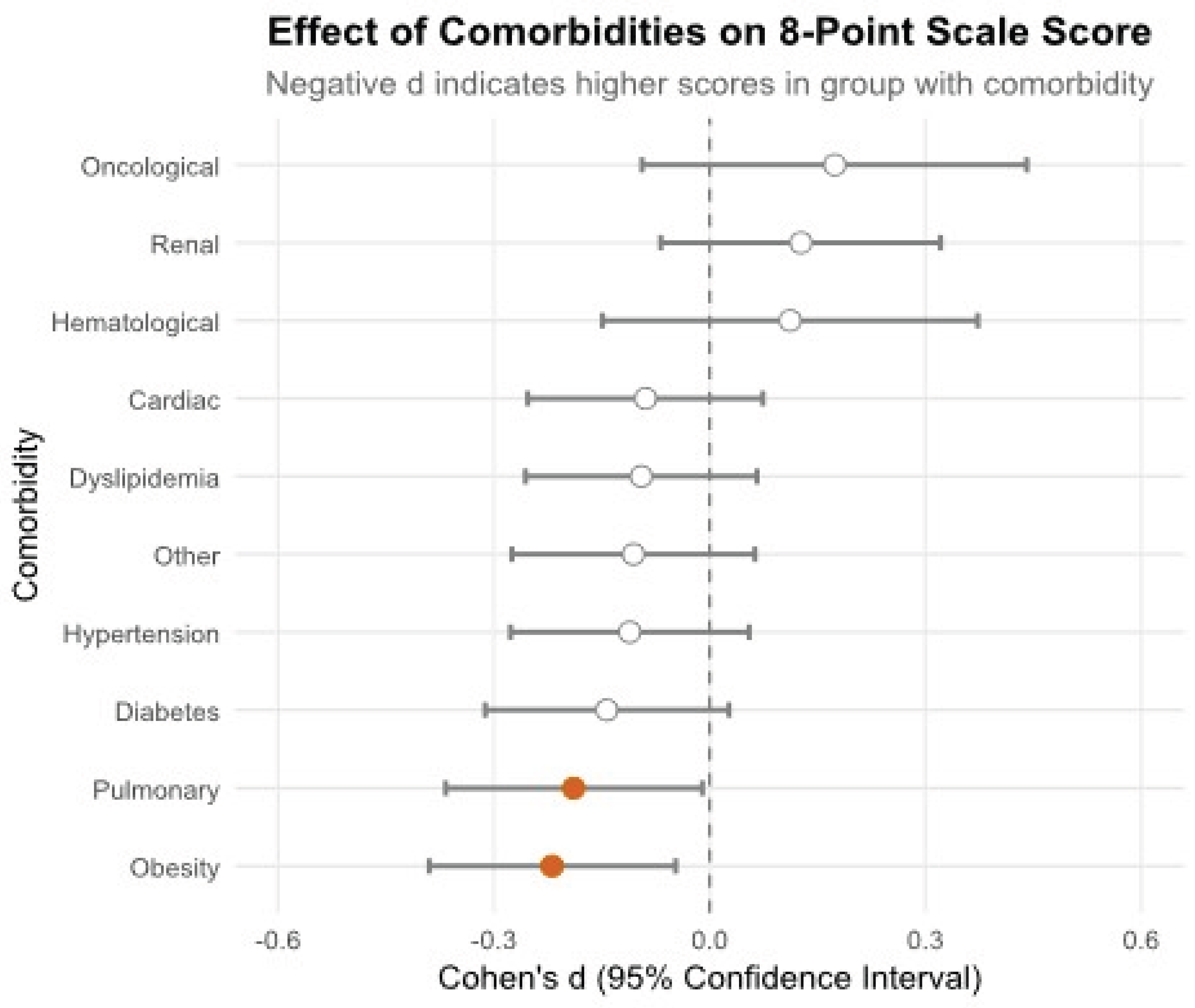
3.2. Group Comparisons
3.2.1. Mortality
| Comorbidity |
Without Comorbidity n (%) |
With Comorbidity n (%) |
N | X² | df | p-value | Φ | |
| Cardiac | Alive | 292 (63.3%) | 169 (36.7%) | 461 | 16.004 | 1 | <0.001 | 0.164 |
| Dead | 59 (44.0%) | 75 (56.0%) | 134 | |||||
| Renal | Alive | 372 (80.7%) | 89 (19.3%) | 461 | 7.752 | 1 | 0.009 | 0.114 |
| Dead | 93 (69.4%) | 41 (30.6%) | 134 | |||||
| Oncological | Alive | 426 (92.4%) | 35 (7.6%) | 461 | 14.018 | 1 | <0.001 | 0.153 |
| Dead | 109 (81.3%) | 25 (18.7%) | 134 | |||||
| Other | Alive | 169 (36.7%) | 292 (63.3%) | 461 | 4.410 | 1 | 0.036 | 0.086 |
| Dead | 36 (26.9%) | 98 (73.1%) | 134 | |||||
| Legend - N = frequencies; M = mean; SD = standard deviation; t = t-test; df = degrees of freedom; p = p-value; Cohen’s d = size effect; ALT = alanine aminotransferase; AST = aspartate transaminase; ALP = alkaline phosphatase; LDH = lactate dehydrogenase; CRP = C-reactive protein; NLR= neutrophil-to-lymphocyte ratio. X² = Chi-Squared test value; df = degrees of freedom; Φ = Phi coefficient (effect size). Significant p-values are in bold. | ||||||||
3.2.2. Oxygen Supplementation
| Comorbidity | O2 Suppl. |
Without Comorbidity n (%) |
With Comorbidity n (%) |
Total | X² | df | p-value | Φ |
| Pulmonary | No | 107 (82.3%) | 23 (17.7%) | 130 | 9.384 | 1 | 0.002 | 0.126 |
| Yes | 319 (68.6%) | 146 (31.4%) | 465 | |||||
| Obesity | No | 100 (76.9%) | 30 (23.1%) | 130 | 7.327 | 1 | 0.007 | 0.111 |
| Yes | 299 (64.3%) | 166 (35.7%) | 465 | |||||
| Diabetes | No | 95 (73.1%) | 35 (26.9%) | 130 | 4.002 | 1 | 0.045 | 0.082 |
| Yes | 296 (63.7%) | 169 (36.3%) | 465 | |||||
| Legend. N = frequencies; M = mean; SD = standard deviation; t = t-test; df = degrees of freedom; p = p-value; Cohen’s d = size effect; ALP = alkaline phosphatase; LDH = lactate dehydrogenase; CRP = C-reactive protein; NLR= neutrophil-to-lymphocyte ratio. X² = Chi-Squared test value; df = degrees of freedom; Φ = Phi coefficient (effect size). | ||||||||
3.2.3. Length of Stay (LOS)
3.2.4. Symptomatology
3.3. Correlations
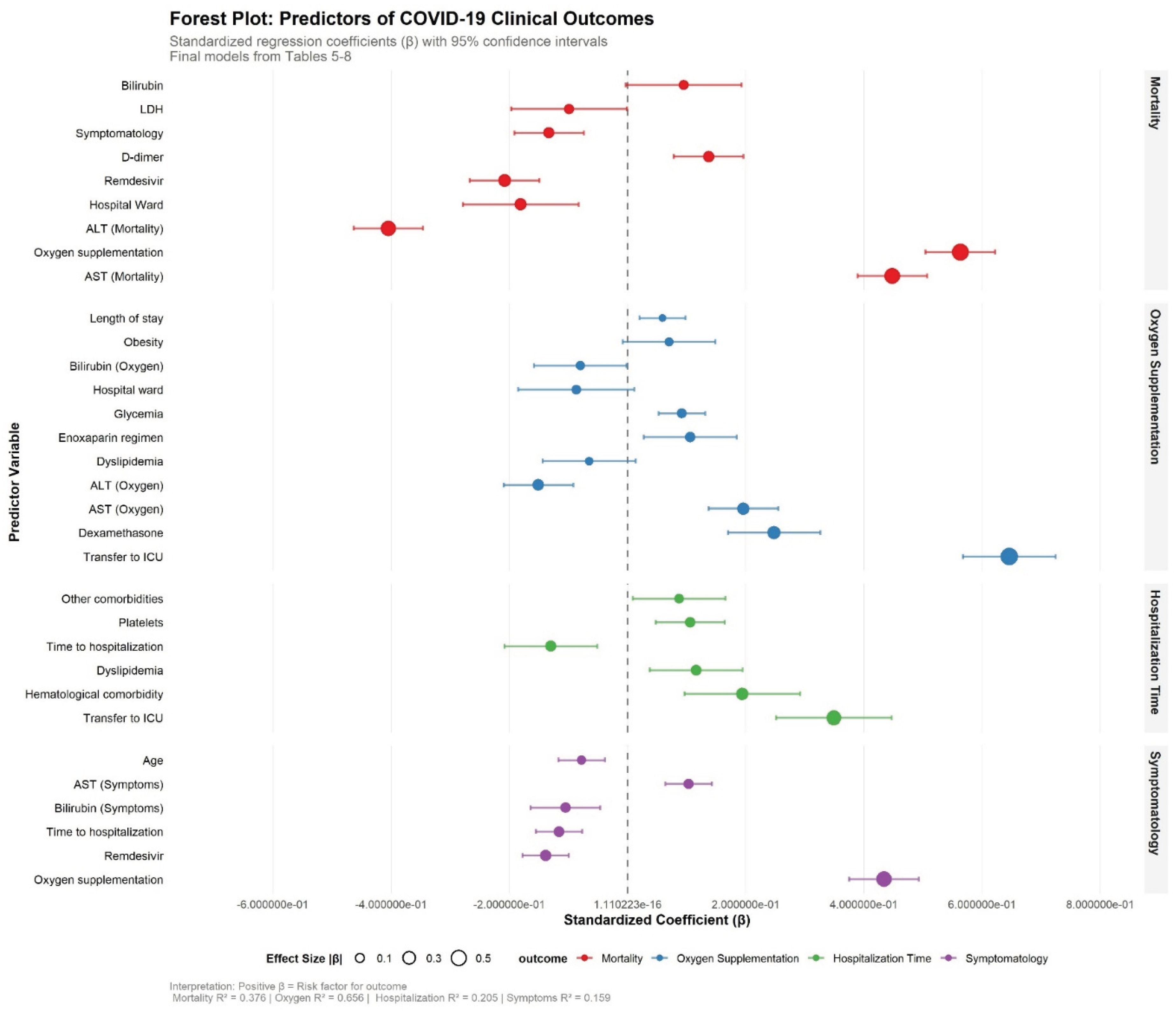
3.4. Regressions
3.5. Moderation Analysis
4. Discussion
Limitations and Future Directions
Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Competing Interests
Ethics Approval
References
- Zhu, N.; Zhang, D.; Wang, W.; et al. A novel coronavirus from patients with pneumonia in China, 2019. N. Engl. J. Med. 2020, 382, 727–733. [Google Scholar] [CrossRef]
- Huang, C.; Wang, Y.; Li, X.; et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 2020, 395, 497–506. [Google Scholar] [CrossRef]
- Wu, Z.; McGoogan, J.M. Characteristics of and important lessons from the coronavirus disease 2019 (COVID-19) outbreak in China. JAMA 2020, 323, 1239–1242. [Google Scholar] [CrossRef]
- World Health Organization. Global excess deaths associated with COVID-19, January 2020–December 2021; World Health Organization: Geneva, Switzerland, 2021; Available online: https://www.who.int/data/stories/global-excess-deaths-associated-with-covid-19-january-2020-december-2021 (accessed on 14 April 2026).
- Zhou, F.; Yu, T.; Du, R.; et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: A retrospective cohort study. Lancet 2020, 395, 1054–1062. [Google Scholar] [CrossRef] [PubMed]
- Docherty, A.B.; Harrison, E.M.; Green, C.A.; et al. Features of 20,133 UK patients in hospital with COVID-19. BMJ 2020, 369, m1985. [Google Scholar] [CrossRef]
- Williamson, E.J.; Walker, A.J.; Bhaskaran, K.; et al. Factors associated with COVID-19-related death using OpenSAFELY. Nature 2020, 584, 430–436. [Google Scholar] [CrossRef] [PubMed]
- Knights, H.; Mayor, N.; Millar, K.; et al. Characteristics and outcomes of patients with COVID-19 at a district general hospital in Surrey, UK. Clin. Med. 2020, 20, e148–e153. [Google Scholar] [CrossRef]
- Knight, S.R.; Ho, A.; Pius, R.; et al. Risk stratification of patients admitted to hospital with COVID-19 using the 4C Mortality Score. BMJ 2020, 370, m3339. [Google Scholar] [CrossRef]
- Liang, W.; Liang, H.; Ou, L.; et al. Development and validation of a clinical risk score to predict critical illness in hospitalized COVID-19 patients. JAMA Intern. Med. 2020, 180, 1081–1089. [Google Scholar] [CrossRef]
- Wynants, L.; Van Calster, B.; Collins, G.S.; et al. Prediction models for diagnosis and prognosis of COVID-19 infection. BMJ 2020, 369, m1328. [Google Scholar] [CrossRef] [PubMed]
- Wolkewitz, M.; Puljak, L. Methodological challenges of analysing COVID-19 data during the pandemic. BMC Med. Res. Methodol. 2020, 20, 81. [Google Scholar] [CrossRef]
- Nalbandian, A.; Sehgal, K.; Gupta, A.; et al. Post-acute COVID-19 syndrome. Nat. Med. 2021, 27, 601–615. [Google Scholar] [CrossRef]
- Xie, Y.; Xu, E.; Bowe, B.; Al-Aly, Z. Long-term cardiovascular outcomes of COVID-19. Nat. Med. 2022, 28, 583–590. [Google Scholar] [CrossRef] [PubMed]
- Bradley, B.T.; Maioli, H.; Johnston, R.; et al. Histopathology and ultrastructural findings of fatal COVID-19 infections. Lancet 2020, 396, 320–332. [Google Scholar] [CrossRef] [PubMed]
- Wichmann, D.; Sperhake, J.P.; Lütgehetmann, M.; et al. Autopsy findings and venous thromboembolism in patients with COVID-19. Ann. Intern. Med. 2020, 173, 268–277. [Google Scholar] [CrossRef]
- Tang, N.; Li, D.; Wang, X.; Sun, Z. Abnormal coagulation parameters and poor prognosis in COVID-19. J. Thromb. Haemost. 2020, 18, 844–847. [Google Scholar] [CrossRef]
- Smilowitz, N.R.; Kunichoff, D.; Garshick, M.; et al. C-reactive protein and clinical outcomes in COVID-19. Eur. Heart J. 2021, 42, 2270–2279. [Google Scholar] [CrossRef]
- Feikin, D.R.; Higdon, M.M.; Abu-Raddad, L.J.; et al. Duration of effectiveness of vaccines against SARS-CoV-2 infection and COVID-19 disease: Results of a systematic review and meta-regression. Lancet 2022, 399, 924–944. [Google Scholar] [CrossRef] [PubMed]
- Goldberg, Y.; Mandel, M.; Bar-On, Y.M.; et al. Waning immunity after the BNT162b2 vaccine in Israel. N. Engl. J. Med. 2021, 385, e85. [Google Scholar] [CrossRef]
- World Health Organization. From Emergency Response to Long-Term COVID-19 Disease Management: Sustaining Gains Made during the COVID-19 Pandemic; World Health Organization: Geneva, Switzerland, 2023; Available online: https://www.who.int/publications/i/item/WHO-WHE-SPP-2023.1 (accessed on 10 April 2026).
- von Elm, E.; Altman, D.G.; Egger, M.; Pocock, S.J.; Gøtzsche, P.C.; Vandenbroucke, J.P.; STROBE Initiative. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: Guidelines for reporting observational studies. BMJ 2007, 335, 806–808. [Google Scholar] [CrossRef]
- WHO Working Group on the Clinical Characterisation and Management of COVID-19 Infection. A minimal common outcome measure set for COVID-19 clinical research. Lancet Infect. Dis. 2020, 20, e192–e197. [CrossRef]
- Cai, M.; Xie, Y.; Topol, E.J.; Al-Aly, Z. Three-year outcomes of post-acute sequelae of COVID-19. Nat. Med. 2024, 30, 1564–1573. [Google Scholar] [CrossRef]
- Wiwattanakul, S.; Taweerutchana, R.; Khuancharee, K.; et al. Biochemical and hematological predictors of mortality in Thai patients with COVID-19. Med. Sci. 2025, 13, 281. [Google Scholar] [CrossRef] [PubMed]
- Rubio-Rivas, M.; Corbella, X.; Guillén, N.; et al. WHO ordinal scale and inflammation risk categories in COVID-19: Comparative study of the severity scales. J. Gen. Intern. Med. 2022, 37, 1980–1987. [Google Scholar] [CrossRef] [PubMed]
- Yang, O.O. The immunopathogenesis of SARS-CoV-2 infection: Overview of lessons learned in the first 5 years. J. Immunol. 2025, 214, 1095–1104. [Google Scholar] [CrossRef]
- Wang, D.; Hu, B.; Hu, C.; et al. Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus–infected pneumonia in Wuhan, China. JAMA 2020, 323, 1061–1069. [Google Scholar] [CrossRef]
- Wajdowicz, M.; Dobrowolska, K.; Brzdęk, K.; et al. Predictors of severe outcomes in COVID-19: Evidence from real-world multicenter retrospective study (2020–2024). J. Clin. Med. 2026, 15, 1207. [Google Scholar] [CrossRef]
- Leung, J.M.; Yang, C.X.; Tam, A.; et al. ACE-2 expression in the small airway epithelia of smokers and COPD patients: Implications for COVID-19. Eur. Respir. J. 2020, 55, 2000688. [Google Scholar] [CrossRef]
- Korakas, E.; Ikonomidis, I.; Kousathana, F.; et al. Obesity and COVID-19: Immune and metabolic derangement as a possible link to adverse clinical outcomes. Am. J. Physiol. Endocrinol. Metab. 2020, 319, E105–E109. [Google Scholar] [CrossRef] [PubMed]
- Leite, F.; Ribeiro, L. Dopaminergic pathways in obesity-associated inflammation. J. Neuroimmune Pharmacol. 2020, 15, 93–113. [Google Scholar] [CrossRef]
- Simonnet, A.; Chetboun, M.; Poissy, J.; et al. High prevalence of obesity in severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) requiring invasive mechanical ventilation. Obesity 2020, 28, 1195–1199. [Google Scholar] [CrossRef]
- Jackson, S.L.; Wood, J.; Dahl, A.; et al. Association between hypertension and diabetes control and COVID-19 severity: National Patient-Centered Clinical Research Network, United States, March 2020 to February 2022. J. Am. Heart Assoc. 2023, 12, e030240. [Google Scholar] [CrossRef] [PubMed]
- Taveira-Barbosa, J.; Pereira, E.; Lopes, J.; et al. Characteristics and mortality of patients with cancer diagnosed with SARS-CoV-2 in Northern Portugal. Anticancer Res. 2025, 45, 1749–1760. [Google Scholar] [CrossRef]
- Mendes, D.; Figueiredo, D.; Alves, C.; et al. Impact of the COVID-19 pandemic on cancer screenings in Portugal. Cancer Epidemiol. 2024, 88, 102496. [Google Scholar] [CrossRef]
- von Rekowski, C.P.; Pinto, I.; Fonseca, T.A.H.; et al. Analysis of six consecutive waves of ICU-admitted COVID-19 patients: Key findings and insights from a Portuguese population. Geroscience 2025, 47, 2399–2422. [Google Scholar] [CrossRef]
- Santos, M.M.S.; Pereira, I.J.; Cuboia, N.; et al. Predictors of early and long-term mortality after ICU discharge in critically ill COVID-19 patients: A prospective cohort study. PLOS ONE 2023, 18, e0293883. [Google Scholar] [CrossRef]
- Salas, M.; Hofman, A.; Stricker, B.H. Confounding by indication: An example of variation in the use of epidemiologic terminology. Am. J. Epidemiol. 1999, 149, 981–983. [Google Scholar] [CrossRef] [PubMed]
- White, E.; Baik, M.K.; Zahir, S.F.; et al. Management and outcomes of children hospitalised with COVID-19 including incidental and nosocomial infections in Australia 2020–2023: A national surveillance study. J. Clin. Virol. 2025, 179, 105824. [Google Scholar] [CrossRef] [PubMed]
- Instituto Nacional de Saúde Doutor Ricardo Jorge. Genomic epidemiology of novel coronavirus in Portugal from March 1st 2020 up to March 31st, 2021; INSA: Lisbon, Portugal, 2021; Available online: https://insaflu.insa.pt/ncov/F-Mar2020-Mar2021 (accessed on 17 April 2026).
| Group | N | M | SD | t | df | p | Cohen's d | |
|
Neutrophils x103/µL |
Alive | 461 | 5.39 | 3.08 | -2.189 | 178.301 | 0.030 | -0.249 |
| Dead | 132 | 6.21 | 3.98 | |||||
| NLR | Alive | 461 | 5.96 | 4.79 | -2.910 | 161.757 | 0.004 | -0.367 |
| Dead | 132 | 8.00 | 7.62 | |||||
|
Hemoglobin (g/dL) |
Alive | 461 | 12.92 | 1.90 | 2.294 | 187.495 | 0.023 | 0.249 |
| Dead | 132 | 12.42 | 2.26 | |||||
|
Urea (mg/dL) |
Alive | 461 | 51.10 | 38.18 | -4.139 | 180.664 | <0.001 | -0.249 |
| Dead | 132 | 69.95 | 48.18 | |||||
|
Glucose (mg/dL) |
Alive | 456 | 138.02 | 62.00 | -2.067 | 584 | 0.039 | -0.206 |
| Dead | 130 | 151.65 | 79.70 | |||||
| ALP (U/L) | Alive | 457 | 74.27 | 37.97 | -3.242 | 170.409 | 0.001 | -0.382 |
| Dead | 130 | 90.07 | 51.75 | |||||
| LDH (U/L) | Alive | 452 | 319.84 | 122.62 | -4.185 | 167.695 | <0.001 | -0490 |
| Dead | 126 | 401.75 | 172.40 | |||||
| CRP (mg/L) | Alive | 461 | 86.76 | 69.66 | -2.392 | 591 | 0.017 | -0.236 |
| Dead | 132 | 109.37 | 63.41 | |||||
|
D-dimer (ng/mL) |
Alive | 334 | 864.19 | 883.57 | -2.736 | 103.627 | 0.007 | -0.426 |
| Dead | 86 | 1292.03 | 1378.93 |
| O2 Suppl. | N | M | SD | t | df | p | Cohen's d | |
|
Hemoglobin (g/dL) |
No | 129 | 12.23 | 2.14 | -3.707 | 591 | <0.001 | -0.37 |
| Yes | 464 | 12.96 | 1.93 | |||||
| Neutrophilsx103/µL | No | 129 | 4.59 | 2.80 | -3.858 | 591 | <0.001 | -0.38 |
| Yes | 464 | 5.84 | 3.40 | |||||
| NLR | No | 129 | 3.94 | 2.79 | -8.534 | 456,985 | <0.001 | -0.58 |
| Yes | 464 | 7.10 | 5.98 | |||||
| Glucose (mg/dL) | No | 125 | 124.56 | 50.82 | -3.755 | 263,439 | <0.001 | -0.32 |
| Yes | 461 | 145.51 | 69.51 | |||||
| ALP (U/L) | No | 125 | 84.86 | 37.47 | 2.14 | 585 | 0.033 | 0.22 |
| Yes | 462 | 75.85 | 42.84 | |||||
| LDH (U/L) | No | 124 | 273.54 | 94.48 | -7.183 | 284,221 | <0.001 | -0.59 |
| Yes | 454 | 350.40 | 138.92 | |||||
| CRP (mg/L) | No | 129 | 46.99 | 50.99 | -10.048 | 273,598 | <0.001 | -0.84 |
| Yes | 464 | 102.51 | 69.38 | |||||
| Ferritin (ng/mL) | No | 58 | 972.12 | 1062.51 | -2.432 | 378 | 0.015 | -0.35 |
| Yes | 322 | 1392.73 | 1237.16 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




