Submitted:
16 April 2026
Posted:
20 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Participants
2.3. Ultrasound Protocol
2.4. Image Acquisition and Interpretation
2.5. Subgroup Analysis
2.6. Statistical Analysis
3. Results
3.1. Patient Characteristics
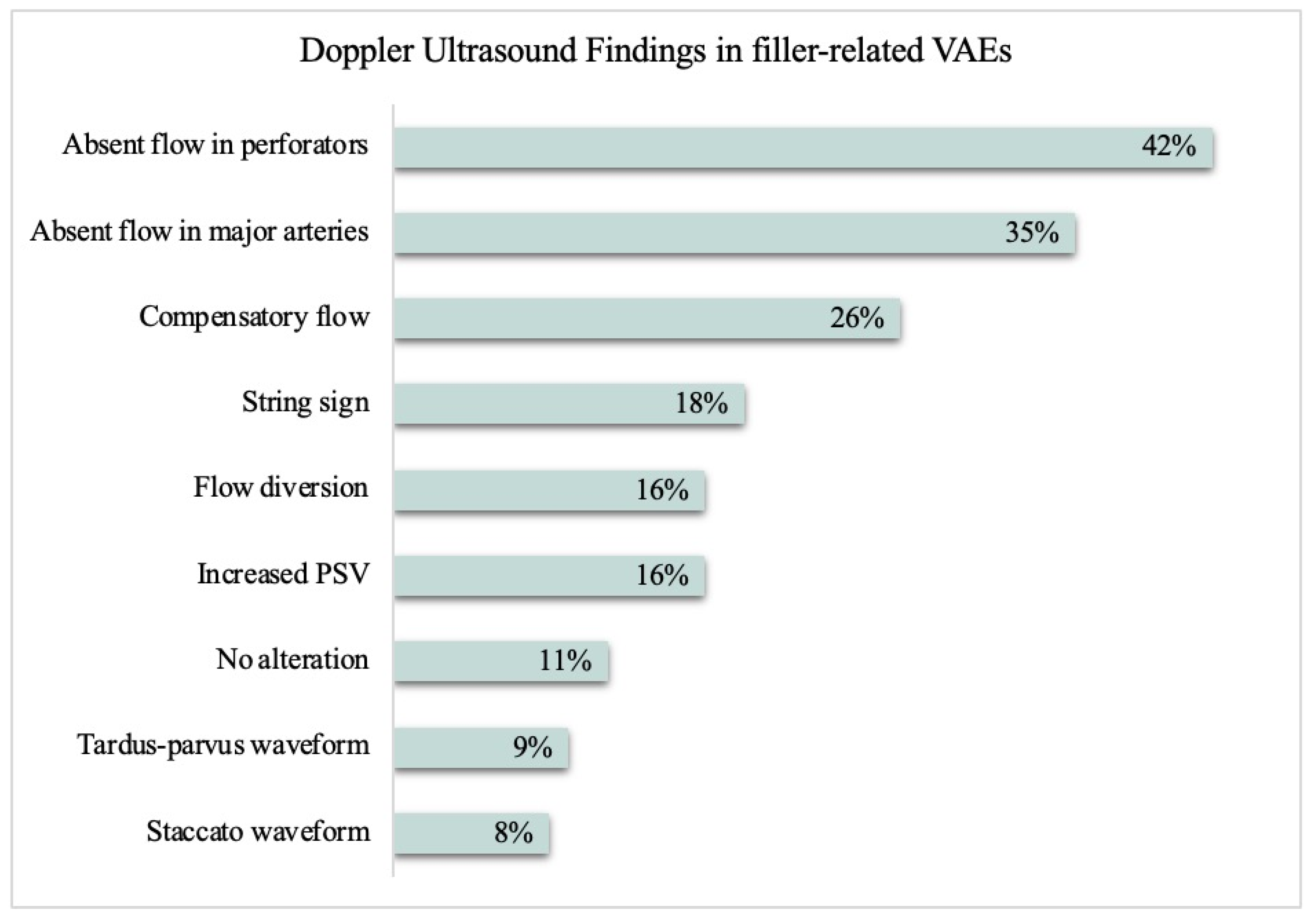
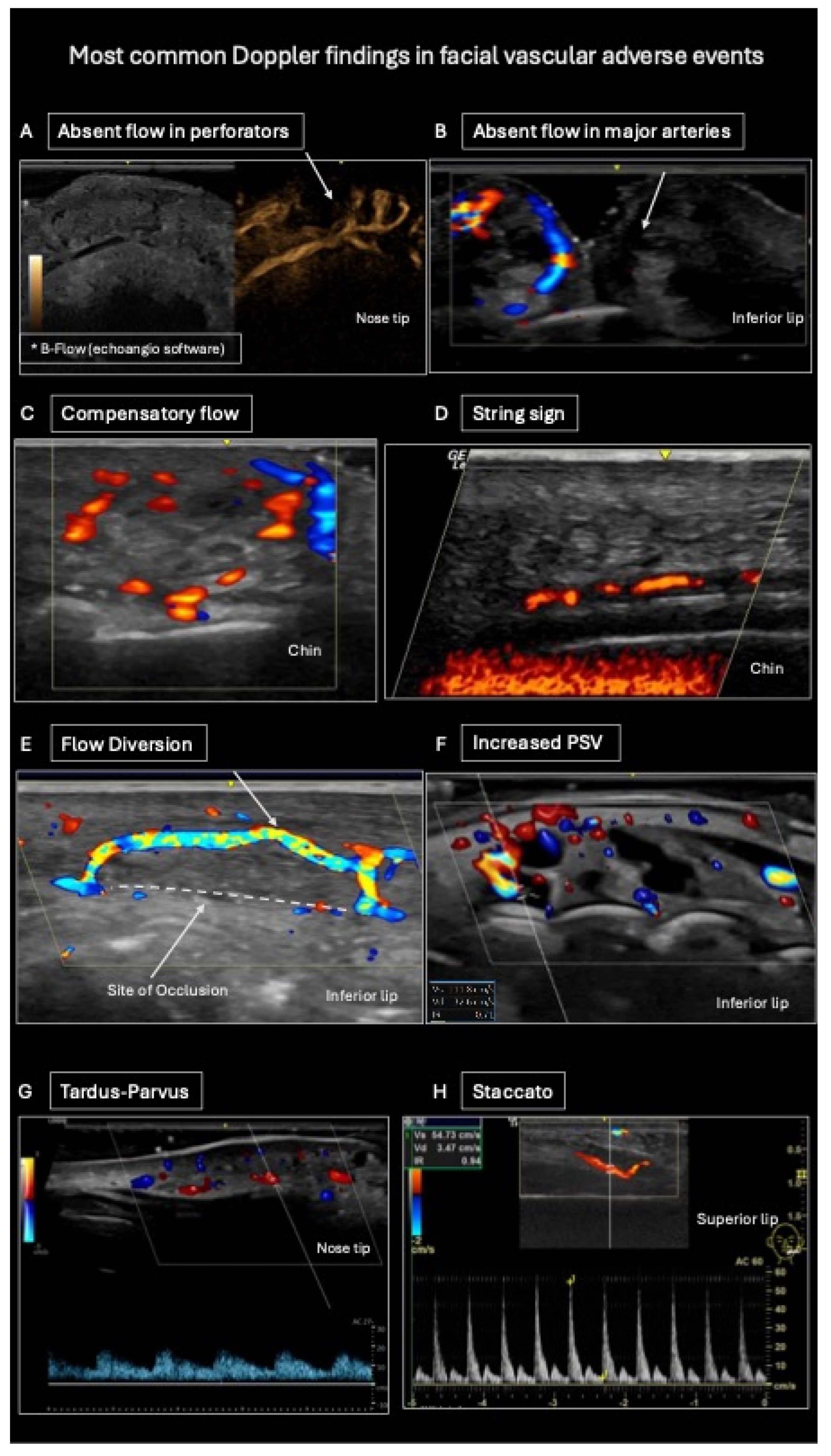
3.2. Doppler Ultrasound Findings
3.3. Doppler Findings According to Prior Hyaluronidase Use
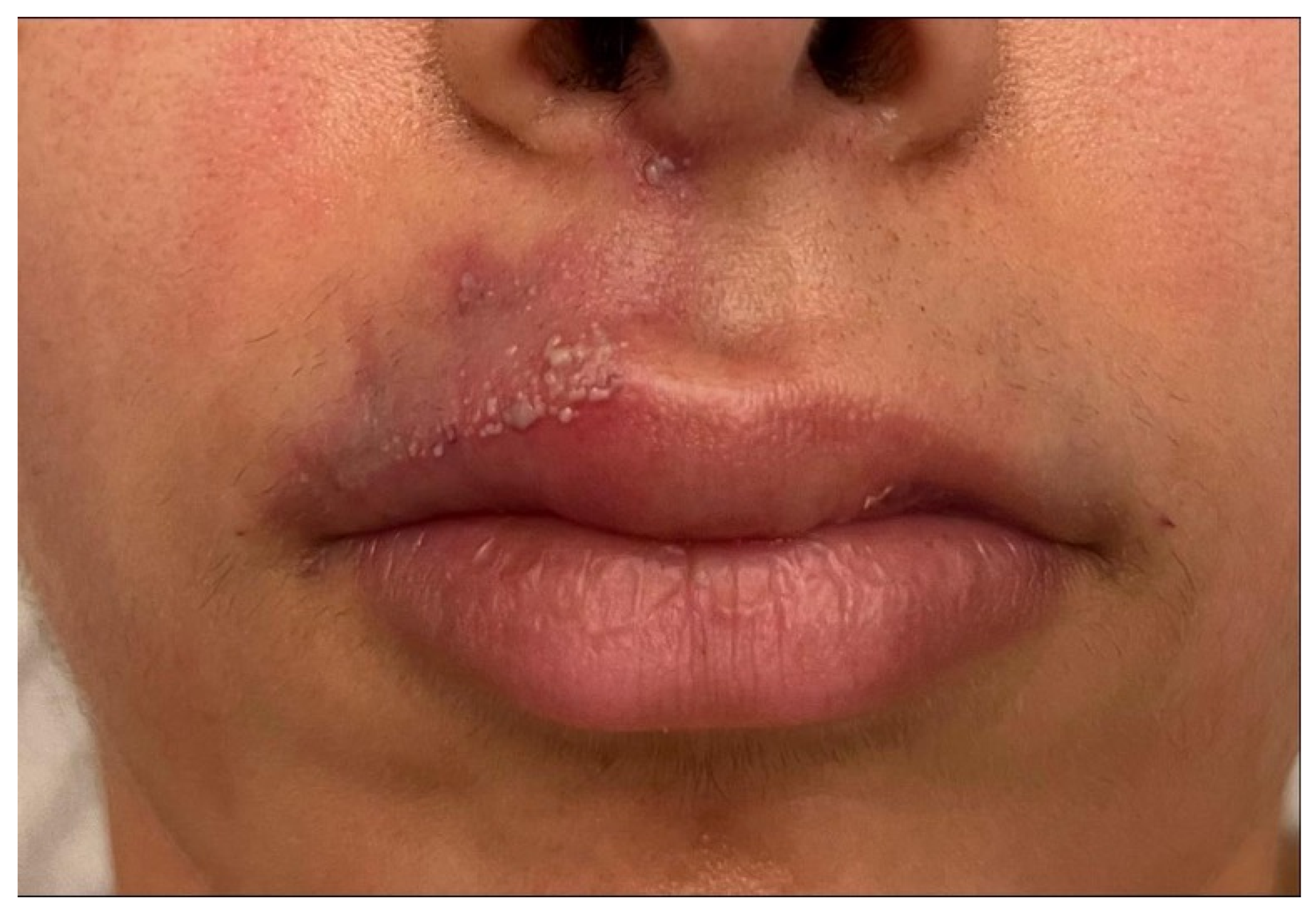
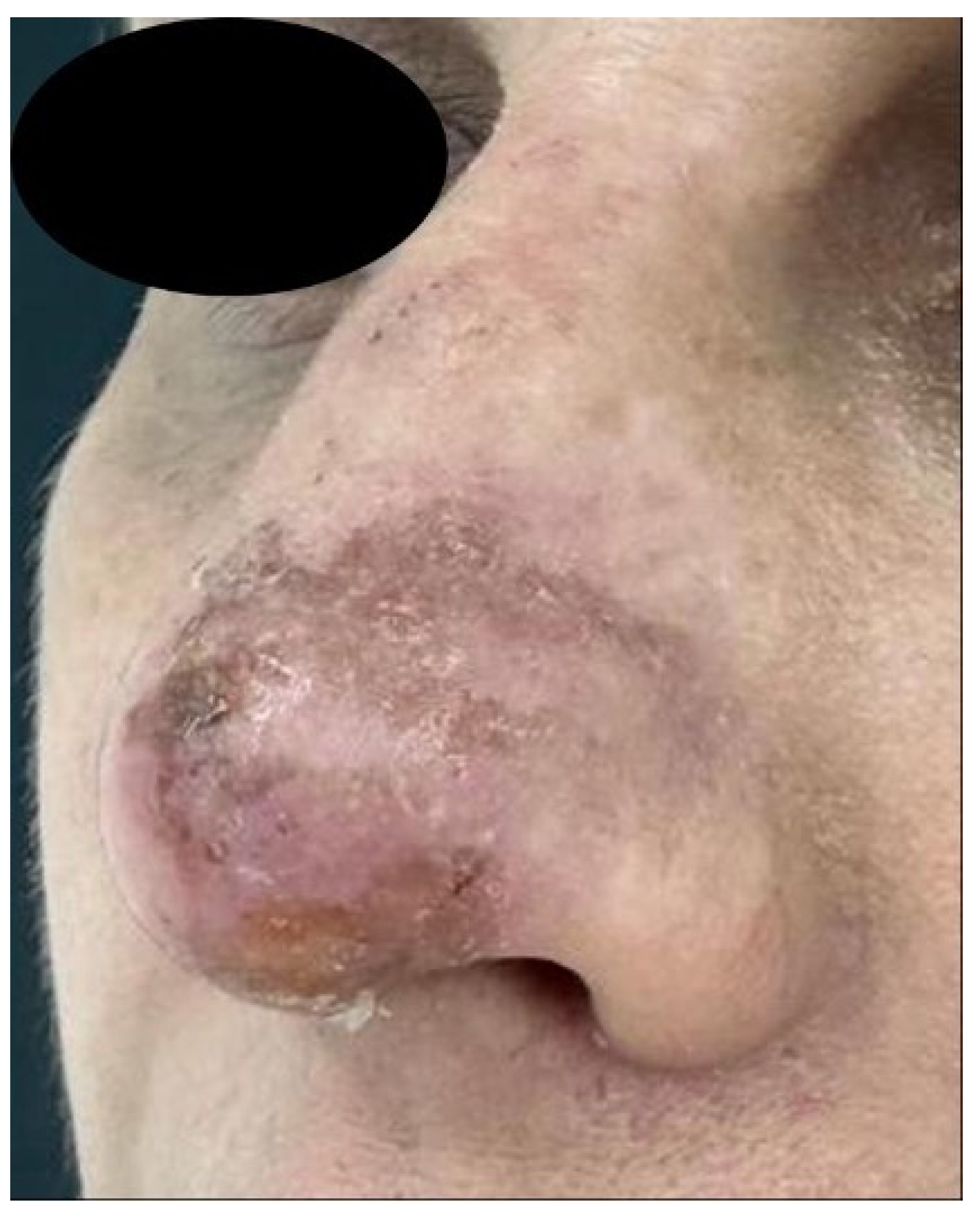
3.4. Clinical Signs and Associations
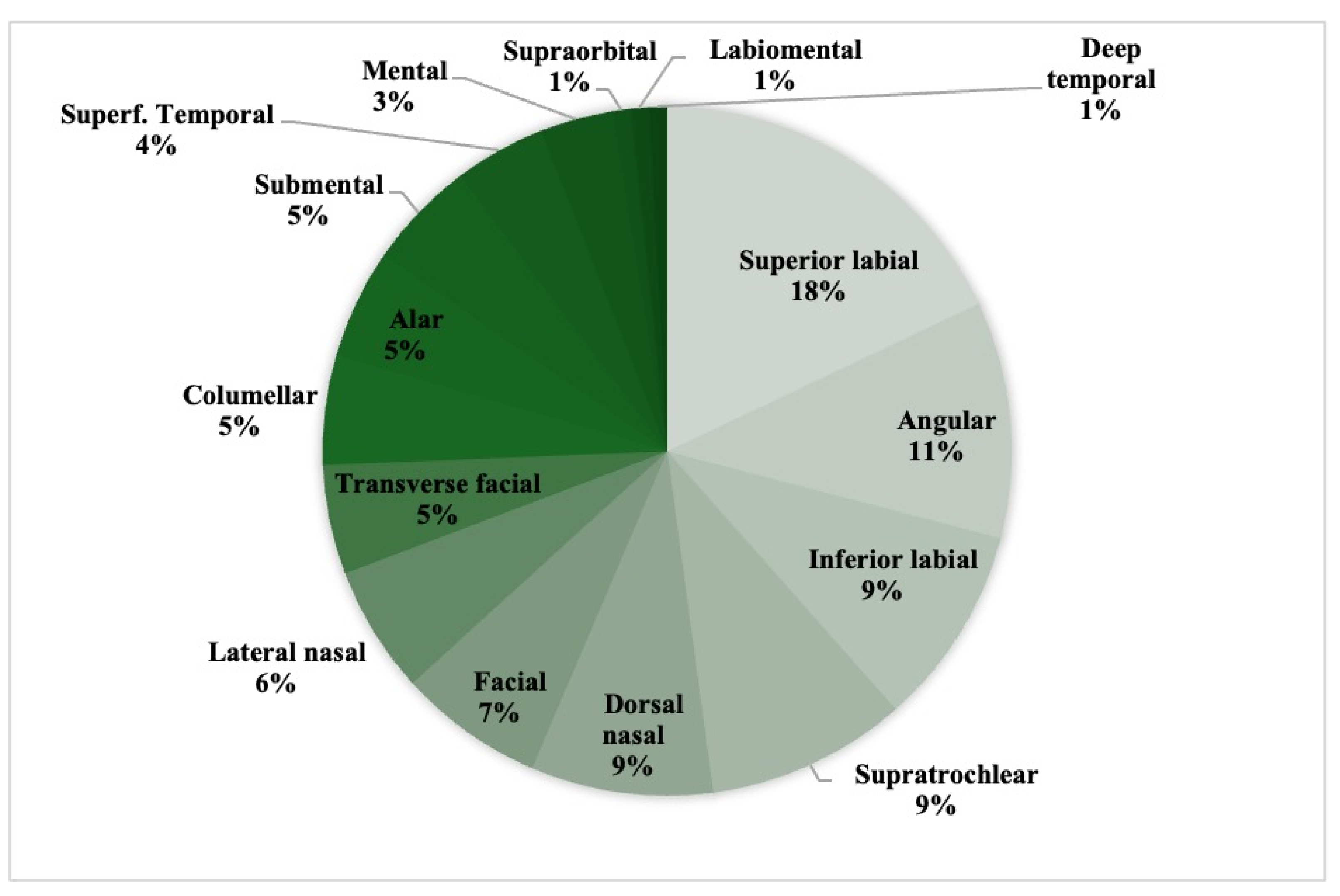
3.5. Arterial Territories Involved
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| VAEs | Vascular adverse events |
| PSV | Peak systolic velocity |
References
- Triana, L.; Palacios Huatuco, R.M.; Campilgio, G.; Liscano, E. Trends in Surgical and Nonsurgical Aesthetic Procedures: A 14-Year Analysis of the International Society of Aesthetic Plastic Surgery-ISAPS. Aesthetic Plast Surg 2024, 48, 4217-4227. [CrossRef]
- Schelke, L.; Decates, T.; Kadouch, J.; Velthuis, P. Incidence of Vascular Obstruction After Filler Injections. Aesthet Surg J 2020, 40, NP457-NP460. [CrossRef]
- Soares, D.M. Bridging a Century-Old Problem: The Pathophysiology and Molecular Mechanisms of HA Filler-Induced Vascular Occlusion (FIVO)-Implications for Therapeutic Interventions. Molecules 2022, 27. [CrossRef]
- Kim, Y.K.; Jung, C.; Woo, S.J.; Park, K.H. Cerebral Angiographic Findings of Cosmetic Facial Filler-related Ophthalmic and Retinal Artery Occlusion. J Korean Med Sci 2015, 30, 1847-1855. [CrossRef]
- Yang, Q.; Lu, B.; Guo, N.; Li, L.; Wang, Y.; Ma, X.; Su, Y. Fatal Cerebral Infarction and Ophthalmic Artery Occlusion After Nasal Augmentation with Hyaluronic Acid-A Case Report and Review of Literature. Aesthetic Plast Surg 2020, 44, 543-548. [CrossRef]
- Schelke, L.W.; Velthuis, P.; Kadouch, J.; Swift, A. Early ultrasound for diagnosis and treatment of vascular adverse events with hyaluronic acid fillers. J Am Acad Dermatol 2023, 88, 79-85. [CrossRef]
- Schelke, L.W.; Velthuis, P.J.; Mojallal, A.; Henry, G.; Hofer, S.O.P.; Cotofana, S.C. Reticulated livedoid skin patterns after soft-tissue filler-related vascular adverse events. J Am Acad Dermatol 2024, 91, 37-42. [CrossRef]
- Mojallal, A.; Schelke, L.; Velthuis, P.J.; Boucher, F.; Henry, G. Ultrasound assisted hyaluronic acid vascular adverse event management based on the vascular anatomy. Ann Chir Plast Esthet 2024, 69, 674-680. [CrossRef]
- Albricker, A.C.L.; Freire, C.M.V.; Santos, S.N.D.; Alcantara, M.L.; Cantisano, A.L.; Porto, C.L.L.; Amaral, S.I.D.; Veloso, O.C.G.; Morais Filho, D.; Teodoro, J.A.R.; et al. Recommendation Update for Vascular Ultrasound Evaluation of Carotid and Vertebral Artery Disease: DIC, CBR and SABCV - 2023. Arq Bras Cardiol 2023, 120, e20230695. [CrossRef]
- Dhanoa, D.; Baerlocher, M.O.; Benko, A.J.; Benenati, J.F.; Kuo, M.D.; Dariushnia, S.R.; Faintuch, S.; Midia, M.; Nikolic, B. Position Statement on Noninvasive Imaging of Peripheral Arterial Disease by the Society of Interventional Radiology and the Canadian Interventional Radiology Association. J Vasc Interv Radiol 2016, 27, 947-951. [CrossRef]
- Taylor, G.I.; Corlett, R.J.; Ashton, M.W. The Functional Angiosome: Clinical Implications of the Anatomical Concept. Plast Reconstr Surg 2017, 140, 721-733. [CrossRef]
- Taylor, G.I.; Palmer, J.H. The vascular territories (angiosomes) of the body: experimental study and clinical applications. Br J Plast Surg 1987, 40, 113-141. [CrossRef]
- DeLorenzi, C. Complications of injectable fillers, part 2: vascular complications. Aesthet Surg J 2014, 34, 584-600. [CrossRef]
- Murray, G.; Convery, C.; Walker, L.; Davies, E. Guideline for the Management of Hyaluronic Acid Filler-induced Vascular Occlusion. J Clin Aesthet Dermatol 2021, 14, E61-E69.
- Wortsman, X. Guidelines and protocols. In Atlas of Dermatologic Ultrasound, Wortsman, X., Ed.; Springer International Publishing: New York, NY, 2018; pp. 23-34.
- Demchuk, A.M.; Christou, I.; Wein, T.H.; Felberg, R.A.; Malkoff, M.; Grotta, J.C.; Alexandrov, A.V. Specific transcranial Doppler flow findings related to the presence and site of arterial occlusion. Stroke 2000, 31, 140-146. [CrossRef]
- Kim, E.S.; Sharma, A.M.; Scissons, R.; Dawson, D.; Eberhardt, R.T.; Gerhard-Herman, M.; Hughes, J.P.; Knight, S.; Marie Kupinski, A.; Mahe, G.; et al. Interpretation of peripheral arterial and venous Doppler waveforms: A consensus statement from the Society for Vascular Medicine and Society for Vascular Ultrasound. Vasc Med 2020, 25, 484-506. [CrossRef]
- Hwang, J.Y. Doppler ultrasonography of the lower extremity arteries: anatomy and scanning guidelines. Ultrasonography 2017, 36, 111-119. [CrossRef]
- Giller, C.A.; Mathews, D.; Purdy, P.; Kopitnik, T.A.; Batjer, H.H.; Samson, D.S. The transcranial Doppler appearance of acute carotid artery occlusion. Ann Neurol 1992, 31, 101-103. [CrossRef]
- Lee, S.J.; Lee, T.K.; Moon, J.E. Vertebral artery foraminal segment doppler sonography to detect vertebral and basilar artery stenosis or occlusion. J Neuroimaging 2023, 33, 852-859. [CrossRef]
- AbuRahma, A.F.; Avgerinos, E.D.; Chang, R.W.; Darling, R.C., 3rd; Duncan, A.A.; Forbes, T.L.; Malas, M.B.; Perler, B.A.; Powell, R.J.; Rockman, C.B.; et al. The Society for Vascular Surgery implementation document for management of extracranial cerebrovascular disease. J Vasc Surg 2022, 75, 26S-98S. [CrossRef]
- Saito, K.; Kimura, K.; Nagatsuka, K.; Nagano, K.; Minematsu, K.; Ueno, S.; Naritomi, H. Vertebral artery occlusion in duplex color-coded ultrasonography. Stroke 2004, 35, 1068-1072. [CrossRef]
- Keller, H.; Meier, W.; Yonekawa, Y.; Kumpe, D. Noninavasive angiography for the diagnosis of carotid artery disease using Doppler ultrasound (carotid artery Doppler). Stroke 1976, 7, 354-363. [CrossRef]
- Munia, M.A.; Munia, C.G.; Parada, M.B.; Ben-Hurferraz Parente, J.; Wolosker, N. Doppler Ultrasound in the Management of Vascular Complications Associated with Hyaluronic Acid Dermal Fillers. J Clin Aesthet Dermatol 2022, 15, 40-43.
- Jagus, D.; Skrzypek, E.; Migda, B.; Wozniak, W.; Mlosek, R.K. Usefulness of Doppler sonography in aesthetic medicine. J Ultrason 2021, 20, e268-e272. [CrossRef]
- Huang, P.; Liu, A.; Ren, H.; Xue, K. Color Doppler Flow Imaging of Retrobulbar Ocular Blood Flow Changes in Retinal Artery Occlusions Caused by Cosmetic Facial Filler Injections. Ophthalmic Plast Reconstr Surg 2019, 35, 227-231. [CrossRef]
- Myers, B.; Cohen, J.L. Reviewing the Role of Angiosomes in Filler-Induced Tissue Necrosis: Observed Patterns and Clinical Signs. Dermatol Surg 2026, 52, 368-372. [CrossRef]
- Houseman, N.D.; Taylor, G.I.; Pan, W.R. The angiosomes of the head and neck: anatomic study and clinical applications. Plast Reconstr Surg 2000, 105, 2287-2313. [CrossRef]
- Jajoria, H.; Venkataram, A.; Mysore, V. Importance of Choke Vessels in Injectable Fillers. J Cutan Aesthet Surg 2020, 13, 185-190. [CrossRef]
- Ashton, M.W.; Taylor, G.I.; Corlett, R.J. The Role of Anastomotic Vessels in Controlling Tissue Viability and Defining Tissue Necrosis with Special Reference to Complications following Injection of Hyaluronic Acid Fillers. Plast Reconstr Surg 2018, 141, 818e-830e. [CrossRef]
- Zierler, R.E.; Jordan, W.D.; Lal, B.K.; Mussa, F.; Leers, S.; Fulton, J.; Pevec, W.; Hill, A.; Murad, M.H. The Society for Vascular Surgery practice guidelines on follow-up after vascular surgery arterial procedures. J Vasc Surg 2018, 68, 256-284. [CrossRef]
- Schelke, L.W.; Velthuis, P.J.; Decates, T.; Kadouch, J.; Alfertshofer, M.; Frank, K.; Cotofana, S. Ultrasound-Guided Targeted vs Regional Flooding: A Comparative Study for Improving the Clinical Outcome in Soft Tissue Filler Vascular Adverse Event Management. Aesthet Surg J 2023, 43, 86-96. [CrossRef]
- Boey, J.J.E.; Boey, J.J.J.; Chen, Z.; Cao, T.; Tan, A.S.M.; Ng, Z.Y. Superior Outcomes with Ultrasound-Guided Hyaluronidase for Impending Filler-Induced Facial Skin Necrosis: A Systematic Review and Pilot Meta-Analysis. Aesthetic Plast Surg 2025, 49, 3519-3525. [CrossRef]
- Yi, K.H. Understanding Vascular Complications Arising From Dermal Filler Injection. J Craniofac Surg 2025, 36, 1168-1174. [CrossRef]
- Wollina, U.; Goldman, A. Facial vascular danger zones for filler injections. Dermatol Ther 2020, 33, e14285. [CrossRef]
- Soares, D.J.; Bowhay, A.; Blevins, L.W.; Patel, S.M.; Zuliani, G.F. Patterns of Filler-Induced Facial Skin Ischemia: A Systematic Review of 243 Cases and Introduction of the FOEM Scoring System and Grading Scale. Plast Reconstr Surg 2023, 151, 592e-608e. [CrossRef]
- Foster, J.; Aakalu, V.K.; Freitag, S.K.; McCulley, T.J.; Tao, J.P.; Vagefi, M.R.; Yen, M.T.; Kim, S.J.; Wladis, E.J. Vision-Threatening Complications of Soft Tissue Fillers: A Report by the American Academy of Ophthalmology. Ophthalmology 2025, 132, 935-944. [CrossRef]
- Li, Z.H.; Alfertshofer, M.; Hong, W.J.; Li, X.R.; Zhang, Y.L.; Moellhoff, N.; Frank, K.; Luo, S.K.; Cotofana, S. Upper Facial Anastomoses Between the External and Internal Carotid Vascular Territories - A 3D Computed Tomographic Investigation. Aesthet Surg J 2022, 42, 1145-1151. [CrossRef]
- Ozturk, C.N.; Li, Y.; Tung, R.; Parker, L.; Piliang, M.P.; Zins, J.E. Complications following injection of soft-tissue fillers. Aesthet Surg J 2013, 33, 862-877. [CrossRef]
| Characteristic | N = 100¹ |
| Age, median (Q1, Q3) | 38 (30, 50) |
| Missing data | 1 |
| Sex | |
| Female | 88 (88%) |
| Male | 12 (12%) |
| Filler | |
| Hyaluronic acid | 98 (98%) |
| Calcium hydroxyapatite | 2 (2%) |
| Hyaluronidase before US | |
| Yes | 79 (79%) |
| No | 21 (21%) |
| Category | n | % | |
| No hyaluronidase | 21 | 21% | |
| 1–1500 UI | 26 | 26% | |
| Hyaluronidase dose | 1501–3000 UI | 11 | 11% |
| 3001–4500 UI | 9 | 9% | |
| >4500 UI | 24 | 24% | |
| Dose not recorded | 9 | 9% |
| Overall | No | Yes | ||
| Doppler finding | N = 100¹ | N = 21 | N = 79 | p-value² |
| Absent flow in perforator | 42 (42%) | 10 (48%) | 32 (41%) | 0.6 |
| Absent flow in major artery | 35 (35%) | 5 (24%) | 30 (38%) | 0.3 |
| Compensatory flow | 26 (26%) | 9 (43%) | 17 (22%) | 0.056 |
| String sign | 18 (18%) | 4 (19%) | 14 (18%) | >0.9 |
| Flow diversion | 16 (16%) | 6 (29%) | 10 (13%) | 0.10 |
| Increased PSV | 16 (16%) | 2 (9.5%) | 14 (18%) | 0.5 |
| No alteration | 12 (12%) | 2 (9.5%) | 10 (13%) | >0.9 |
| Tardus–parvus waveform | 9 (9.0%) | 3 (14%) | 6 (7.6%) | 0.4 |
| Staccato waveform | 8 (8.0%) | 1 (4.8%) | 7 (8.9%) | >0.9 |
| ¹n(%) | ||||
| ²Fisher’s exact test |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




