Submitted:
16 April 2026
Posted:
17 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results and Discussion
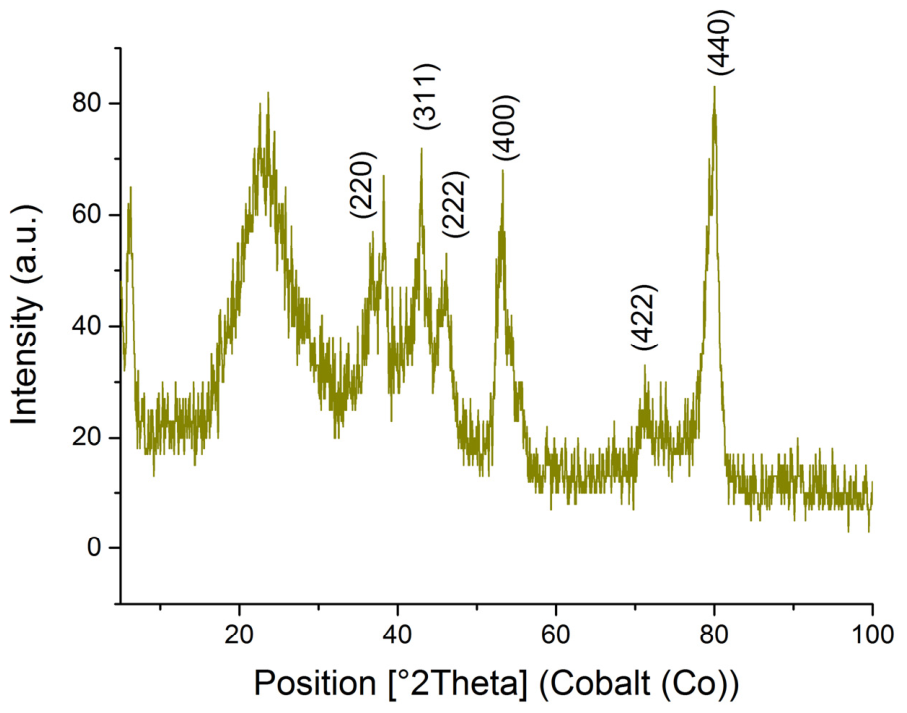
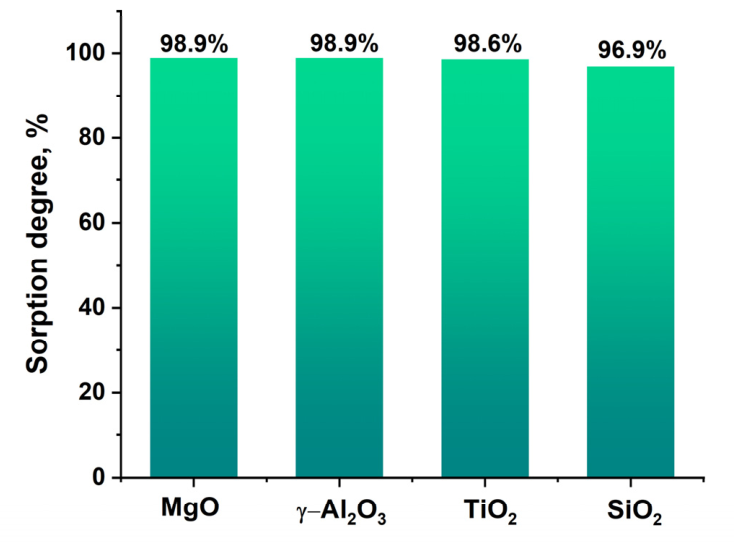
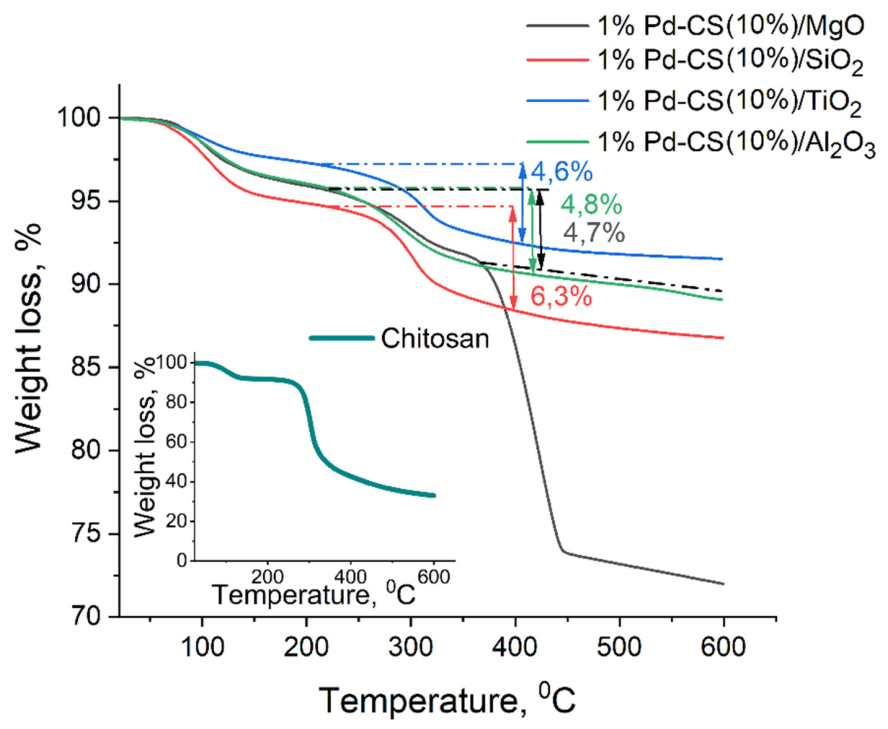
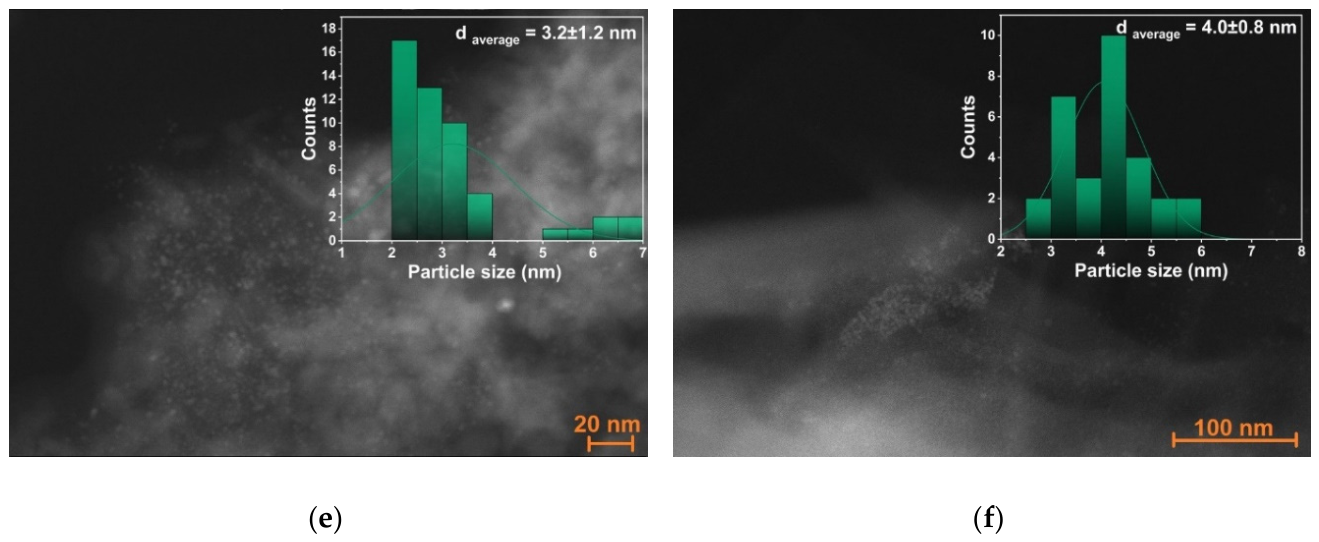
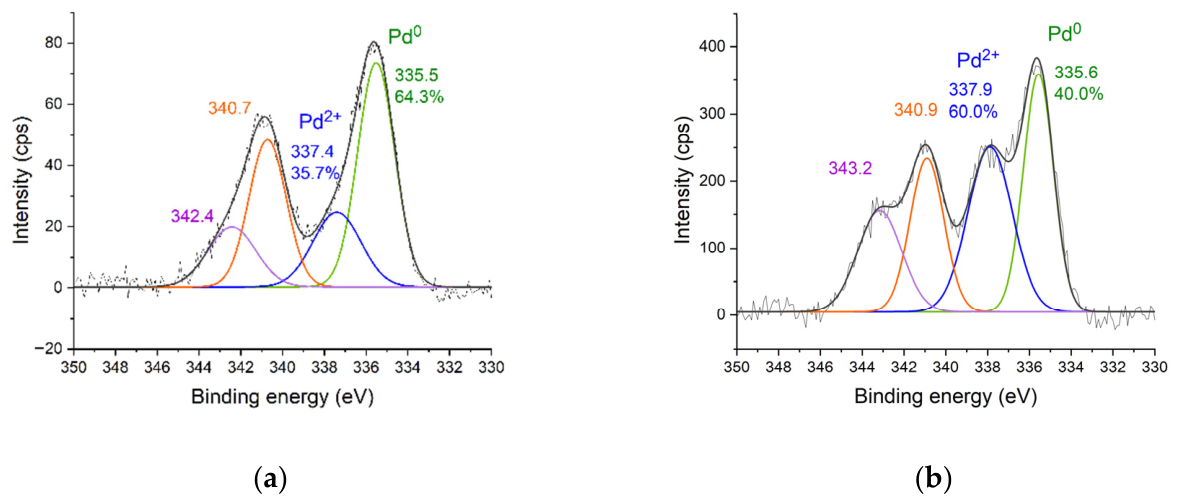
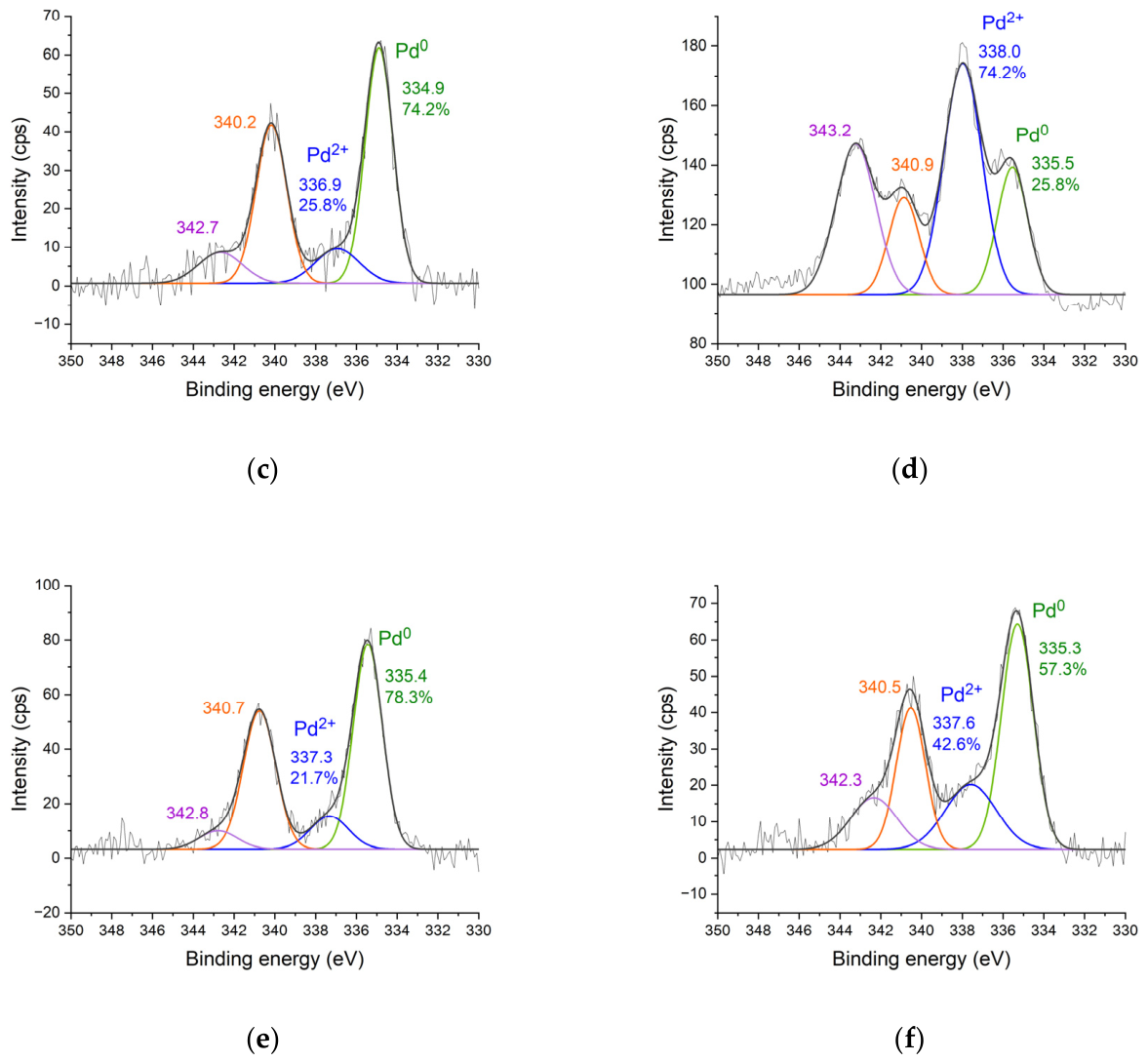
2.1. Synthesis of Palladium Catalysts Deposited on Support Materials and Their Characteristics
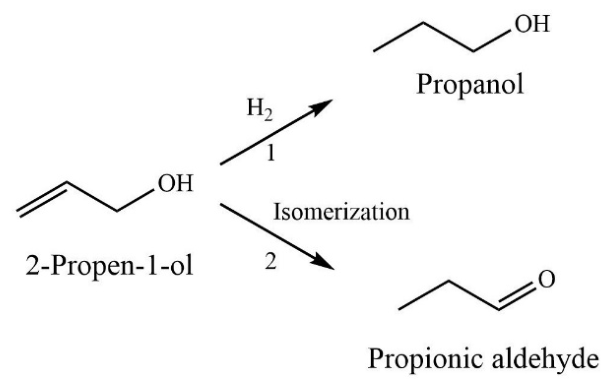
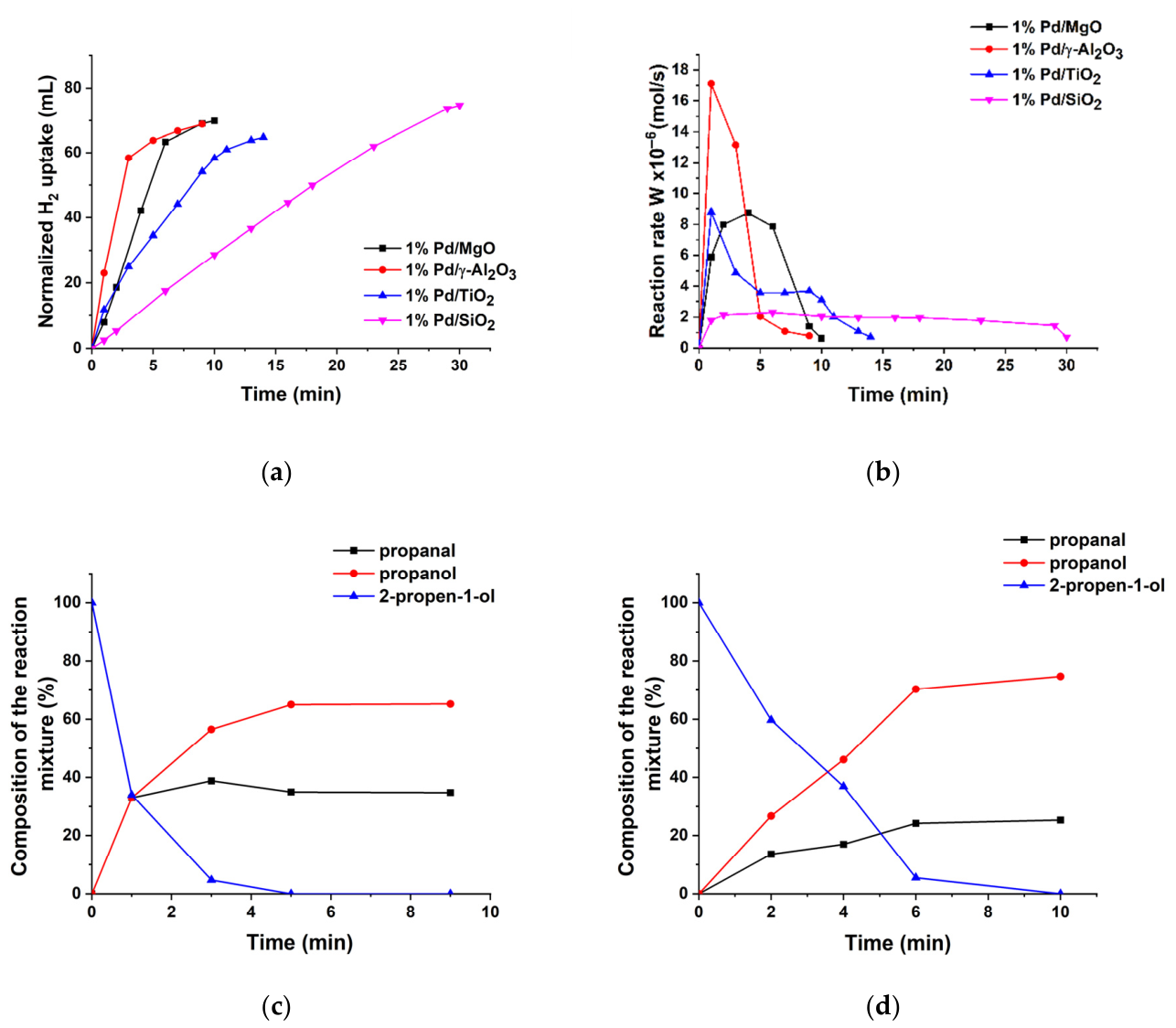
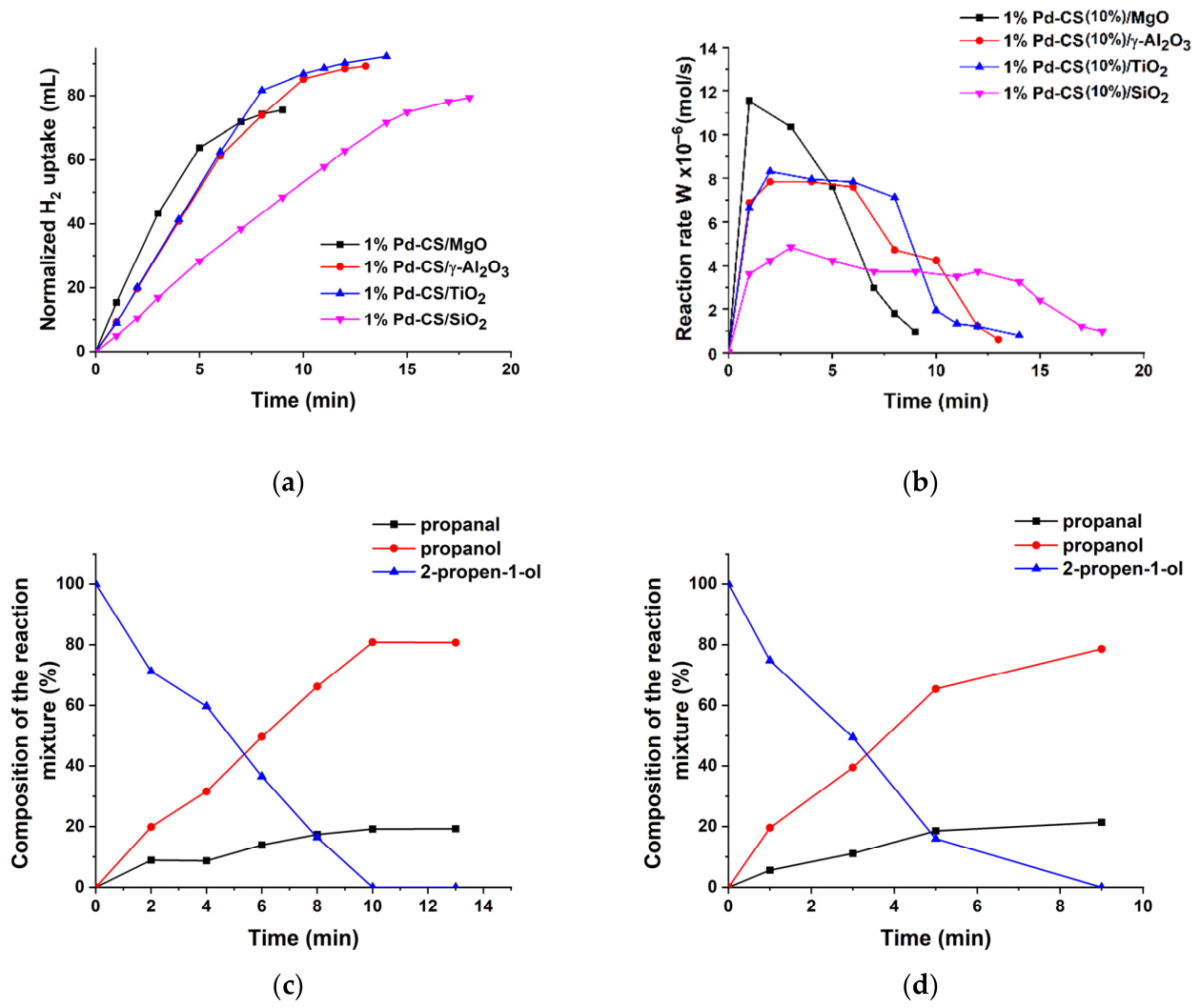
2.2. Catalytic Properties of the Supported Pd Catalysts in Hydrogenation of 2-Propen-1-ol
3. Materials and Methods
3.1. Materials
3.2. Synthesis of Alumina
3.3. Synthesis of Pd Catalysts
3.4. Characterisation of the Catalysts by Physicochemical Methods
3.5. Methodology of Hydrogenation
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Stoffels, M.A.; Klauck, F.J.R.; Hamadi, T.; Glorius, F.; Leker, J. Technology Trends of Catalysts in Hydrogenation Reactions: A Patent Landscape Analysis. Adv. Synth. Catal. 2020, 362, 1258–1274. [CrossRef]
- Zhao, X.; Chang, Y.; Chen, W.-J.; Wu, Q.; Pan, X.; Chen, K.; Weng, B. Recent Progress in Pd-Based Nanocatalysts for Selective Hydrogenation. ACS Omega 2022, 7, 17–31. [CrossRef]
- Bonrath, W.; Medlock, J.; Schutz, J.; Wustenberg, B.; Netscher, T. Hydrogenation in the Vitamins and Fine Chemicals Industry—An Overview. In Hydrogenation; Karamé, I., Ed.; IntechOpen: London, UK, 2012; рр. 17–47. [CrossRef]
- Monguchi, Y.; Ichikawa, T.; Sajiki, H. Recent Development of Palladium-Supported Catalysts for Chemoselective Hydrogenation. Chem. Pharm. Bull. 2017, 65, 2–9. [CrossRef]
- Luo, Q.; Wang, T.; Beller, M.; Jiao, H. Acrolein Hydrogenation on Ni(111). J. Phys. Chem. C 2013, 117, 12715–12724. [CrossRef]
- Pietrantonio, K.; Coccia, F.; Tonucci, L.; d’Alessandro, N.; Bressan, M. Hydrogenation of allyl alcohols catalyzed by aqueous palladium and platinum nanoparticles. RSC Adv. 2015, 5, 68493–68499. [CrossRef]
- Nakagawa, Y.; Yabushita, M.; Tomishige, K. A perspective on catalytic production of olefinic compounds from biomass. RSC Sustainability 2023, 1, 814–837. [CrossRef]
- Mon, M.; Leyva-Pérez, A. Industrial Synthesis of Allyl Alcohols: Outlook and Challenges for a Sustainable Production. ACS Sustainable Chem. Eng. 2025, 13, 19916–19936. [CrossRef]
- Condotta, R.; Gomes, E.L.; de Freitas, D.A.; Poco, J.G.R. Acrolein Production from Glycerol: A Systematic Investigation of Metal-Oxides and Zeolite Catalysts. Ind. Eng. Chem. Res. 2025, 64, 4300–4308. [CrossRef]
- Tabassum, N.; Pothu, R.; Pattnaik, A.; Boddula, R.; Balla, P.; Gundeboyina, R.; Challa, P.; Rajesh, R.; Perugopu, V.; Mameda, N.; et al. Heterogeneous Catalysts for Conversion of Biodiesel-Waste Glycerol into High-Added-Value Chemicals. Catalysts 2022, 12, 767. [CrossRef]
- Talebian-Kiakalaieh, A.; Amin, N.A.S.; Hezaveh, H. Glycerol for renewable acrolein production by catalytic dehydration. Renewable and Sustainable Energy Reviews 2014, 40, 28–59. [CrossRef]
- Wu, H.; Wu, Ch.; He, Q.; Liao, X.; Shi, B. Collagen fiber with surface-grafted polyphenol as a novel support for Pd(0) nanoparticles: Synthesis, characterization and catalytic application. Materials Science and Engineering C 2010, 30, 770–776. [CrossRef]
- Mammadov, B.; Mammadova, U.; Aslanova, H.; Hasanova, K.; Rahimli, N. Hydrogenation of allyl alcohol using a polymer supported nickel-based catalyst. Slovak International Scientific Journal 2025, 94, 13–15. [CrossRef]
- Bukharbayeva, F.; Zharmagambetova, A.; Talgatov, E.; Auyezkhanova, A.; Akhmetova, S.; Jumekeyeva, A.; Naizabayev, A.; Kenzheyeva, A.; Danilov, D. The Synthesis of Green Palladium Catalysts Stabilized by Chitosan for Hydrogenation. Molecules 2024, 29, 4584. [CrossRef]
- Sadeghmoghaddam, E.; Gu, H.; Shon, Y.-S. Pd Nanoparticle-Catalyzed Isomerization vs Hydrogenation of Allyl Alcohol: Solvent-Dependent Regioselectivity. ACS Catalysis 2012, 2, 1838–1845. [CrossRef]
- Zharmagambetova, A.K.; Ergozhin, E.E.; Sheludyakov, Y.L.; Mukhamedzhanova, S.G.; Kurmanbayeva, I.A.; Selenova, B.A.; Utkelov, B.A. 2-Propen-1-ol hydrogenation and isomerization on polymer-palladium complexes — effect of polymer matrix. J. Mol. Catal. A: Chem. 2001, 177, 165−170. [CrossRef]
- Zamanbekova, A.; Zharmagambetova, A.; Jumekeyeva, A.; Akhmetova, S.; Auyezkhanova, A., Kenzheyeva, A. Perspectives on the Catalytic Processes for the Deep Valorization of Carbohydrates into Fuels and Chemicals. Molecules 2025, 30, 3498. [CrossRef]
- Luneau, M.; Lim, J.S.; Patel, D.A.; Sykes, E.Ch.H.; Friend, C.M.; Sautet, P. Guidelines to Achieving High Selectivity for the Hydrogenation of α,β-Unsaturated Aldehydes with Bimetallic and Dilute Alloy Catalysts: A Review. Chem. Rev. 2020, 120, 12834–12872. [CrossRef]
- San, K.A.; Shon, Y.-S. Synthesis of Alkanethiolate-Capped Metal Nanoparticles Using Alkyl Thiosulfate Ligand Precursors: A Method to Generate Promising Reagents for Selective Catalysis. Nanomaterials 2018, 8, 346. [CrossRef]
- Moreno, M.; Kissell, L.N.; Jasinski, J.B.; Zamborini, F.P. Selectivity and Reactivity of Alkylamine- and Alkanethiolate-Stabilized Pd and PdAg Nanoparticles for Hydrogenation and Isomerization of Allyl Alcohol. ACS Catalysis 2012, 2, 2602–2613. [CrossRef]
- Gavia, D.J.; Maung, M.S.; Shon, Y.-S. Water-Soluble Pd Nanoparticles Synthesized from ω-Carboxyl-S-Alkanethiosulfate Ligand Precursors as Unimolecular Micelle Catalysts. ACS Appl. Mater. Interfaces 2013, 5, 12432–12440. [CrossRef]
- Baimuratova, R.K.; Andreeva, A.V.; Uflyand, I.E.; Shilov, G.V.; Bukharbayeva, F.U.; Zharmagambetova, A.K.; Dzhardimalieva, G.I. Synthesis and Catalytic Activity in the Hydrogenation Reaction of Palladium-Doped Metal-Organic Frameworks Based on Oxo-Centered Zirconium Complexes. J. Compos. Sci. 2022, 6, 299. [CrossRef]
- Sibakoti, T.R.; Jasinski, J.B.; Nantz, M.H.; Zamborini, F.P. Iodine Activation: A General Method for Catalytic Enhancement of Thiolate Monolayer-Protected Metal Clusters. Nanoscale 2020, 12, 12027–12037. [CrossRef]
- Auyezkhanova, A.S.; Talgatov, E.T.; Akhmetova, S.N.; Jumekeyeva, A.I.; Naizabayev, A.A.; Zamanbekova, A.T.; Malgazhdarova, M.K. Effect of Support and Polymer Modifier on the Catalytic Performance of Supported Palladium Catalysts in Hydrogenation. Molecules 2025, 30, 3820. [CrossRef]
- Szubiakiewicz, E.; Modelska, M.; Brzezinska, M.; Binczarski, M.J.; Severino, C.J.; Stanishevsky, A.; Witonska, I. Influence of modification of supported palladium systems by polymers: PVP, AMPS and AcrAMPS on their catalytic properties in the reaction of transformation of biomass into fuel bio-components. Fuel 2020, 271, 117584. [CrossRef]
- Wissing, M.; Niehues, M.; Ravoo, B.J.; Studer, A. Synthesis and Immobilization of Metal Nanoparticles Using Photoactive Polymer-Decorated Zeolite L Crystals and Their Application in Catalysis. Adv. Synth. Catal. 2020, 362, 2245–2253. [CrossRef]
- Teixeira-Costa, B.E.; Andrade, C.T. Chitosan as a Valuable Biomolecule from Seafood Industry Waste in the Design of Green Food Packaging. Biomolecules 2021, 11, 1599. [CrossRef]
- Lewandowska, K.; Sionkowska, A.; Furtos, G.; Grabska, S.; Michalska, M. Structure and Interactions in Chitosan Composites. Key Eng. Mater. 2016, 672, 257–260. [CrossRef]
- Aranaz, I.; Acosta, N. Chitin- and Chitosan-Based Composite Materials. Biomimetics 2022, 7, 1. [CrossRef]
- Lee, M.; Chen, B.-Y.; Den, W. Chitosan as a Natural Polymer for Heterogeneous Catalysts Support: A Short Review on Its Applications. Appl. Sci. 2015, 5, 1272–1283. [CrossRef]
- Akhmetova, S.; Zharmagambetova, A.; Talgatov, E.; Auyezkhanova, A.; Malgazhdarova, M.; Zhurinov, M.; Abilmagzhanov, A.; Jumekeyeva, A.; Kenzheyeva, A. How the chemical properties of polysaccharides make it possible to design various types of organic–inorganic composites for catalytic applications. Molecules 2024, 29, 3214. [CrossRef]
- Wang, S.; Li, X.; Wang, S.; Li, Y.; Zhai, Y. Synthesis of γ-alumina via precipitation in ethanol. Materials Letters 2008, 62, 3552–3554. [CrossRef]
- Alkallas, F.H.; Ahmed, H.A.; Alrebdi, T.A.; Pashameah, R.A.; Alrefaee, S.H.; Alsubhe, E.; Trabelsi, A.B.G.; Mostafa, A.M.; Mwafy, E. A. Removal of Ni(II) ions by poly(vinyl alcohol)/Al2O3 nanocomposite film via laser ablation in liquid. Membranes 2022, 12, 660. [CrossRef]
- Yatsenko, D A.; Pakharukova, V.P.; Tsybulya, S. V. Low-temperature transitional aluminas: structure specifics and related X-ray diffraction features. Crystals 2021, 11, 690. [CrossRef]
- Conesa, J.M.; Morales, M.V.; Rodriguez-Ramos, I.; Rocha, M. CuPd Bimetallic Nanoparticles Supported on Magnesium Oxide as an Active and Stable Catalyst for the Reduction of 4-Nitrophenol to 4-Aminophenol. Int. J. Green Technol. 2018, 3, 51–62. [CrossRef]
- Morad, M.; Nowicka, E.; Douthwaite, M.; Iqbal, S.; Miedziak, P.; Edwards, J.K.; Brett, G.L.; He, Q.; Knight, D.W.; Morgan, D.J.; et al. Multifunctional supported bimetallic catalysts for a cascade reaction with hydrogen auto transfer: Synthesis of 4-phenylbutan-2-ones from 4-methoxybenzyl alcohols. Catal. Sci. Technol. 2017, 7, 1928–1936. [CrossRef]
- Chen, Y., Soler, L.; Armengol-Profitós, M.; Xie, C.; Crespo, D.; Llorca, J. Enhanced photoproduction of hydrogen on Pd/TiO2 prepared by mechanochemistry. Applied Catalysis B: Environmental 2022, 309, 121275. [CrossRef]
- Zharmagambetova, A.; Auyezkhanova, A.; Talgatov, E.; Jumekeyeva, A.; Buharbayeva, F.; Akhmetova, S.; Myltykbayeva, Z.; Lopez Nieto, J.M. Synthesis of polymer protected Pd–Ag/ZnO catalysts for phenylacetylene hydrogenation. J. Nanopart. Res. 2022, 24, 236. [CrossRef]
- Parambhath, V.B.; Nagar, R.; Ramaprabhu, S. Effect of Nitrogen Doping on Hydrogen Storage Capacity of Palladium Decorated Graphene. Langmuir 2012, 28, 7826–7833. [CrossRef]
- Han, S.; Liu, Y.; Li, J.; Li, R.; Yuan, F.; Zhu, Y. Improvement Effect of Ni to Pd-Ni/SBA-15 Catalyst for Selective Hydrogenation of Cinnamaldehyde to Hydrocinnamaldehyde. Catalysts 2018, 8, 200. [CrossRef]
- Chen, H.; Zhang, P.; Tan, W.; Jiang, F.; Tang, R. Palladium supported on amino functionalized magnetic MCM-41 for catalytic hydrogenation of aqueous bromate. RSC Adv. 2014, 4, 38743–38749. [CrossRef]

| Sample | Mass, % | ||||||
| O | Mg | Al | Si | Ti | Na | Pd | |
| Pd/MgO | 39.1 | 60.0 | – | – | – | – | 0.9 |
| 1% Pd-CS(10%)/MgO | 49.2 | 49.4 | – | – | – | 0.3 | 1.1 |
| Pd/γ-Al2O3 | 47.9 | – | 49.9 | – | – | 0.9 | 1.3 |
| 1% Pd-CS(10%)/Al2O3 | 46.6 | – | 50.5 | 0.8 | 0.9 | – | 1.2 |
| Pd/SiO2 | 53.8 | 0.6 | 0.2 | 44.1 | – | 0.3 | 1.0 |
| 1% Pd-CS(10%)/SiO2 | 54.0 | 1.0 | 0.4 | 43.4 | – | – | 1.2 |
| Pd/TiO2 | 42.8 | – | 0.3 | – | 55.6 | – | 1.3 |
| 1% Pd-CS(10%)/TiO2 | 44.3 | – | – | 0.6 | 53.8 | – | 1.3 |
| Catalyst | W×10–6,mol/s | Selectivity, % | Conversion, % | |
| С=С | Propanal | Propanol | ||
| 1%Pd/Al2O3 | 17.1 | 35 | 65 | 100 |
| 1%Pd-CS(10%)/Al2O3 | 7.8 | 19 | 81 | 100 |
| 1%Pd/MgO | 8.7 | 25 | 75 | 100 |
| 1%Pd-CS(10%)/MgO | 11.5 | 21 | 79 | 100 |
| 1%Pd/TiO2 | 8.8 | 33 | 67 | 100 |
| 1%Pd-CS(10%)/TiO2 | 8.3 | 22 | 78 | 100 |
| 1%Pd/SiO2 | 2.3 | 28 | 72 | 100 |
| 1%Pd-CS(10%)/SiO2 | 4.8 | 24 | 76 | 100 |
| Catalyst | T, °C | Pressure, MPa | TOF, h−1 |
Propanol selectivity, % |
Conversion, % | Ref. |
| Pd CaLig NPs | 20 | 0.1 | - | 74.0 | 100 | [6] |
| Commercial Pd/C | 20 | 0.1 | - | 71.0 | 100 | [6] |
| C16NH2 Pd | r.t. | 0.1 | 114 | 73.0 | 100 | [20] |
| Pd/Zr6O4(OH)6(C8H4O4)6(H2O)4 | 40 | 0.1 | - | 78.0 | 100 | [22] |
| Pd(0)–EGCG0.2–CF | 30 | 0.5 | 9703 | 89.0 | 99.8 | [12] |
| 1%Pd-CS(10%)/MgO | 40 | 0.1 | 8640 | 78.6 | 100 | This study |
| 1%Pd-CS(10%)/Al2O3 | 40 | 0.1 | 6120 | 80.7 | 100 | This study |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




