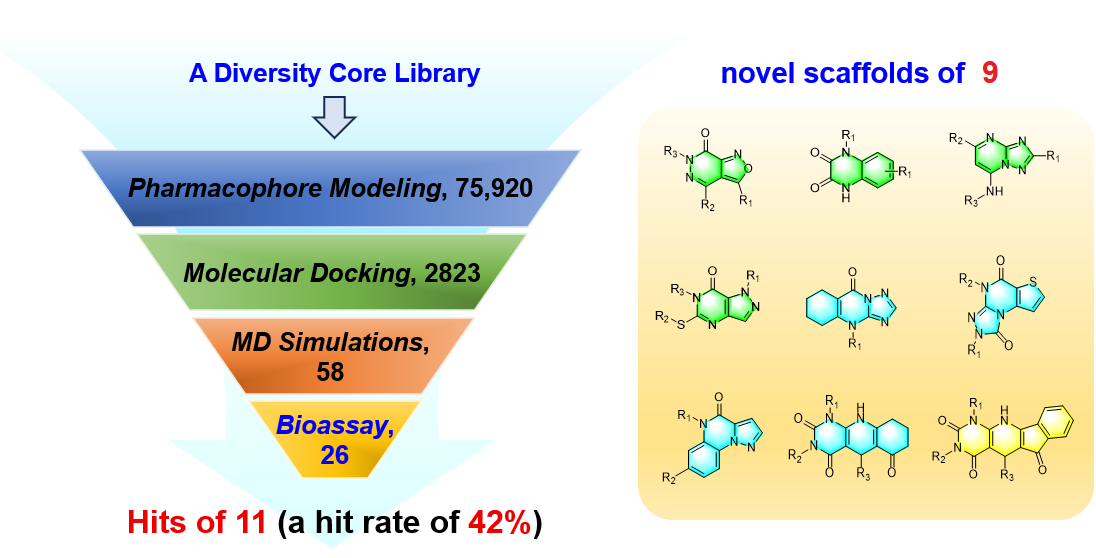
Phosphodiesterase-1 (PDE1) represents an attractive target for the treatment of idiopathic pulmonary fibrosis (IPF). However, the limited chemical diversity of current PDE1 inhibitors has hindered the development of potential anti-IPF drugs, primarily due to an ambiguous understanding of interactions between inhibitors and PDE1. Herein, we report an integrated virtual screening strategy containing pharmacophore modeling, molecular docking, and molecular dynamics simulations, which markedly accelerated the discovery of novel PDE1 inhibitors. Enzymatic assays identified eleven active compounds with moderate inhibition from twenty-six purchased candidates, encompassing nine distinct scaffold types. Notably, 6484-0008 and 6484-0032 exhibited more than 50% inhibition at a concentration of 1 μM. Hydrogen bonding analysis and residue-based energy decompositions revealed key recognition mechanisms involving crucial residues Gln421, His373, and Phe424, as well as the unique Thr271 in the flexible H-loop region, providing insights for the rational design of inhibitors with enhanced potency.