Submitted:
16 April 2026
Posted:
17 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Statistical Analysis and Post Hoc Effect Size Estimation
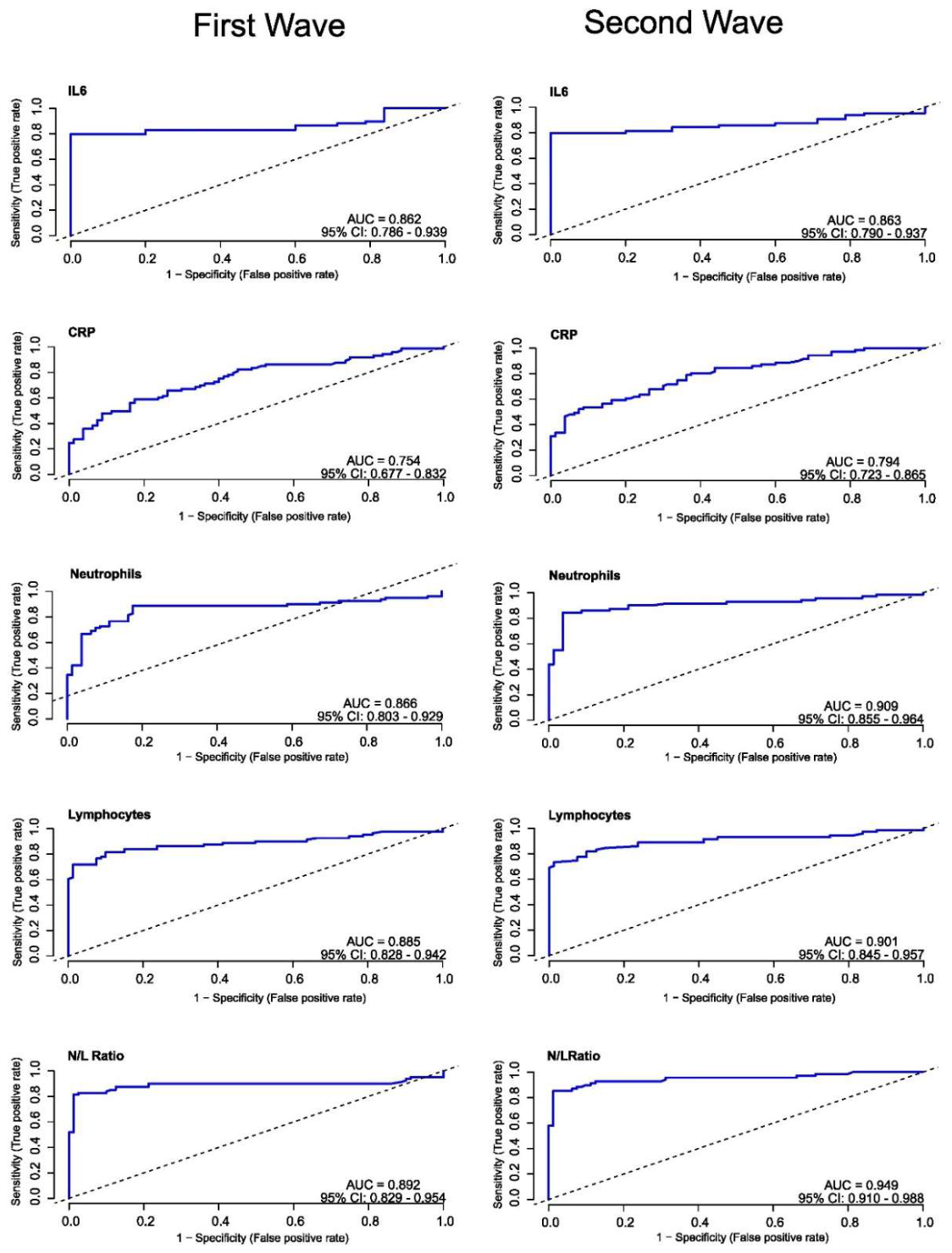
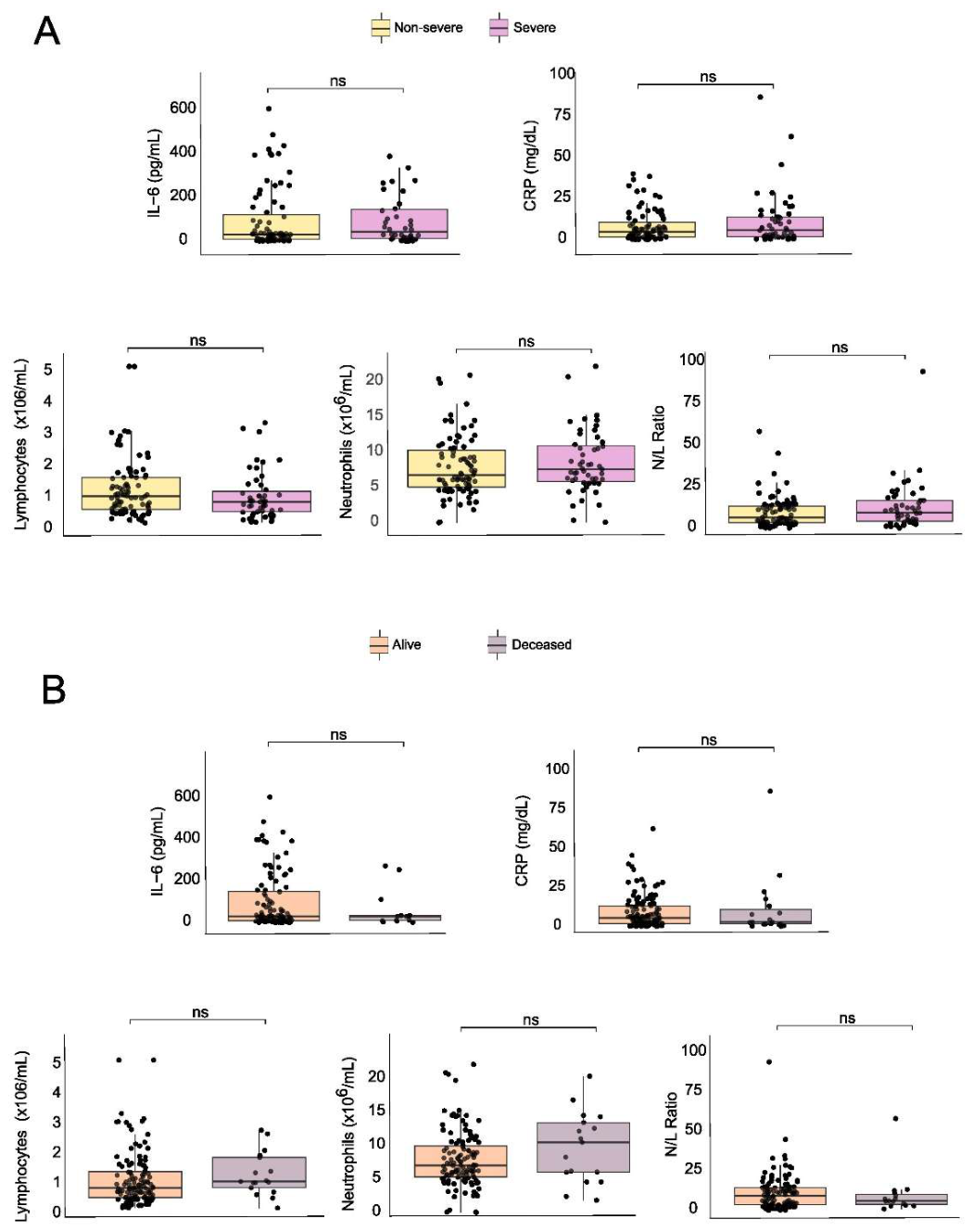
3. Results
4. Discussion
5. Conclusion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AUC | Area under the curve |
| CRP | C-reactive protein |
| ICU | Intensive care unit |
| IL-6 | Interleukin-6 |
| N/L ratio | Neutrophil-to-lymphocyte ratio |
| NPV | Negative predictive value |
| PPV | Positive predictive value |
| ROC | Receiver operating characteristics |
| RSV | Respiratory syncytial virus |
References
- Symes, R.; Keddie, S.H.; Walker, J.; McKeever, T.; Ahmad, S.; Arnold, D.; Evans, C.M.; Pelosi, E.; Rahman, N.M.; Sapey, E., et al. Burden of respiratory syncytial virus infection in older adults hospitalised in England during 2023/24. J. Infect. 2025, 91, 106570. [CrossRef]
- Shi, T.; Denouel, A.; Tietjen, A.K.; Campbell, I.; Moran, E.; Li, X.; Campbell, H.; Demont, C.; Nyawanda, B.O.; Chu, H.Y., et al. Global disease burden estimates of respiratory syncytial virus-associated acute respiratory infection in older adults in 2015: A systematic review and meta-analysis. J. Infect. Dis. 2020, 222 (Suppl. 7), S577-S583. [CrossRef]
- Shi, T.; Arnott, A.; Semogas, I.; Falsey, A.R.; Openshaw, P.; Wedzicha, J.A.; Campbell, H.; Nair, H; RESCEU Investigators. The etiological role of common respiratory viruses in acute respiratory infections in older adults: A systematic review and meta-analysis. J. Infect. Dis. 2020, 222 (Suppl. 7), S563-S569. [CrossRef]
- Center for Diseases and Control. Respiratory syncytial virus infection (RSV). RSV in adults. https://www.cdc.gov/rsv/adults/index.html#:~:text=other%20respiratory%20viruses.-,Severe%20RSV%20illness,older%20against%20severe%20RSV%20illness. (accessed on 15 April 2026).
- Yayan, J. Impact of RSV infection on mortality, rehospitalization, and long-term pulmonary, cardiovascular, and functional outcomes in hospitalized adults: a systematic review and meta-analysis. Virol. J. 2025, 22, 160. [CrossRef]
- Wildenbeest, J.G.; Lowe, D.M.; Standing, J.F.; Butler, C.C. Respiratory syncytial virus infections in adults: a narrative review. Lancet Respir. Med. 2024, 12, 822−836. [CrossRef]
- Perčinić, A.; Vuletić, T.; Lizzul, N.; Vukić Dugac, A.; Gverić Grginić, A.; Tabain, I.; Jurić, D.; Budimir, A. Epidemiological and clinical characteristics of adult RSV infections: A retrospective analysis at University Hospital Center Zagreb (2022-2024). Pathogens 2025, 14, 284. [CrossRef]
- Bermúdez Barrezueta, L.; Matías Del Pozo, V.; López-Casillas, P.; Brezmes Raposo, M.; Gutiérrez Zamorano, M.; Pino Vázquez, M.A. Variation in the seasonality of the respiratory syncytial virus during the COVID-19 pandemic. Infection 2022, 50, 1001-1005. [CrossRef]
- Rios-Guzman, E.; Simons, L.M.; Dean, T.J.; Agnes, F.; Pawlowski, A.; Alisoltanidehkordi, A.; Nam, H.H.; Ison, M.G.; Ozer, E.A.; Lorenzo-Redondo, R., et al. Deviations in RSV epidemiological patterns and population structures in the United States following the COVID-19 pandemic. Nat. Commun. 2024, 15, 3374. [CrossRef]
- Raffaldi, I.; Castagno, E. The epidemiology of respiratory syncytial virus: New trends and future perspectives. Viruses 2024, 16, 1100. [CrossRef]
- Iftimie, S.; Gabaldó-Barrios, X.; Penadés-Nadal, J.; Canela-Capdevila, M.; Piñana, R.; Jiménez-Franco, A.; López-Azcona, A.F.; Castañé, H.; Cárcel, M-; Camps, J., et al. Serum Levels of arachidonic acid, interleukin-6, and C-reactive protein as potential indicators of pulmonary viral infections: Comparative analysis of influenza A, respiratory syncytial virus infection, and COVID-19. Viruses 2024, 16, 1065. [CrossRef]
- Chandra, K.; Das, A.K.; Upadhyay, B.; Uddin, A.; Hussain, S.M.; Islam, F.; Alvi, Y.; Gautam, R.; Kaur, J.; Khan, S., et al. Circulating viral antigenic proteins and inflammatory biomarkers in influenza and respiratory syncytial virus infections: Associations with disease severity and transmission. Cureus 2025, 17, e100037. [CrossRef]
- Kreger, B.E.; Craven, D.E.; Carling, P.C.; McCabe, W.R. Gram-negative bacteremia. III. Reassessment of etiology, epidemiology and ecology in 612 patients. Am. J. Med. 1980, 68, 332–343. [CrossRef]
- Fort-Gallifa, I.; García-Heredia, A.; Hernández-Aguilera, A.; Simó, J.M.; Sepúlveda, J.; Martín-Paredero, V.; Camps, J.; Joven, J. Biochemical indices of oxidative stress and inflammation in the evaluation of peripheral artery disease. Free Radic. Biol. Med. 2016, 97, 568−576. [CrossRef]
- Zweig M.H., Campbell G. Receiver-operating characteristic (ROC) plots: A fundamental evaluation tool in clinical medicine. Clin. Chem. 1993, 39, 561–577. [CrossRef]
- Solidoro, P.; Curtoni, A.; Costa, C.; De Rosa, F.G.; Bondi, A.; Sidoti, F.; Shbaklo, N.; Patrucco, F.; Favre, D.; Zanotto, E., et al. The epidemiology of respiratory syncytial virus and the impact of the COVID-19 pandemic in a retrospective evaluation. Pathogens 2025, 14, 375. [CrossRef]
- Kenmoe, S.; Nair, H. The disease burden of respiratory syncytial virus in older adults. Curr. Opin. Infect. Dis. 2024, 37, 129−136. [CrossRef]
- Abrams, E.M.; Doyon-Plourde, P.; Davis, P.; Lee, L.; Rahal, A.; Brousseau, N.; Siu, W.; Killikelly, A. Burden of disease of respiratory syncytial virus in older adults and adults considered at high risk of severe infection. Can. Commun. Dis. Rep. 2025, 51, 26−34. [CrossRef]
- Asseri, A.A. Respiratory syncytial virus: A narrative review of updates and recent advances in epidemiology, pathogenesis, diagnosis, management and prevention. J. Clin. Med. 2025, 14, 3880. [CrossRef]
- Kim, T.; Choi, S.H. Epidemiology and disease burden of respiratory syncytial virus infection in adults. Infect. Chemother. 2024, 56, 1−12. [CrossRef]
- Han, J.S.; Jang, S.H.; Jeon, J.S.; Lee, K.B.; Kim, J.K. Epidemiological shifts in respiratory virus infections among older adults (≥65 Years) before and after the COVID-19 pandemic: An 18-year retrospective study in the Republic of Korea. Microorganisms 2025, 13, 2301. [CrossRef]
- Carvajal, J.J.; Avellaneda, A.M.; Salazar-Ardiles, C.; Maya, J.E.; Kalergis, A.M.; Lay, M.K. Host components contributing to respiratory syncytial virus pathogenesis. Front. Immunol. 2019, 10, 2152. [CrossRef]
- Zhou, Y.; Xu, L.; Zhong, X.; Guo, X.; Ma, Q. Differentiating bacterial from viral respiratory tract infections using CRP, SAA, and blood routine parameters: A retrospective cohort study. Clinics (Sao Paulo) 2025, 80, 100845. [CrossRef]
- Brown, P.M.; Schneeberger, D.L.; Piedimonte, G. Biomarkers of respiratory syncytial virus (RSV) infection: specific neutrophil and cytokine levels provide increased accuracy in predicting disease severity. Paediatr. Respir. Rev. 2015, 16, 232−240. [CrossRef]
- Tan, L.; He, Z.; Liang, Y.; Wang, K.; Chen, X. Correlation analysis between the severity of respiratory syncytial virus pneumonia and the expression levels of inflammatory cytokines in bronchoalveolar lavage fluid among infants and young children. Front. Pediatr. 2025, 13, 1482029. [CrossRef]
- Lui, G.; Wong, C.K.; Chan, M.; Chong, K.C.; Wong, R.; Chu, I.; Zhang, M.; Li, T.; Hui, D.; Lee, N., et al. Host inflammatory response is the major marker of severe respiratory syncytial virus infection in older adults. J. Infect. 2021, 83, 686−692. [CrossRef]
- GSK’s RSV vaccine, Arexvy, receives European approval for expanded use in all adults 18 years and older. https://www.gsk.com/en-gb/media/press-releases/gsk-s-rsv-vaccine-arexvy-receives-european-approval-for-expanded-use-in-all-adults-18-years-and-older/. (accessed on 15 April 2026).
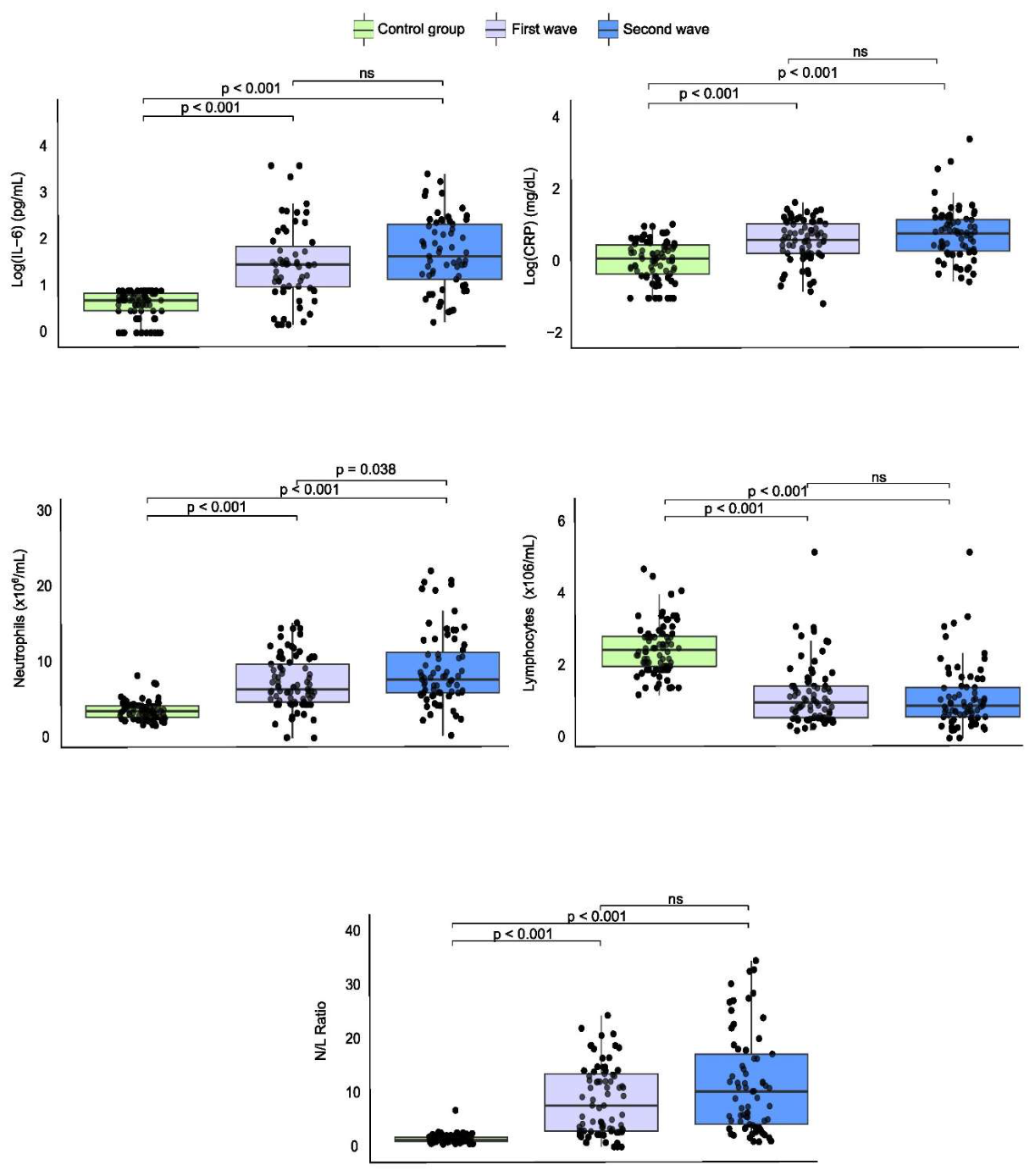
| Variable | 1st wave (n = 81) | 2nd wave (n = 71) | p-value |
| Demographic characteristics and coinfections | |||
| Age, years | 74 (22 – 96) | 78 (20 – 96) | 0.009 |
| Sex, female | 43 (53.1) | 39 (54.9) | 0.820 |
| Tobacco habit | 9 (11.1) | 10 (14.1) | < 0.001 |
| Alcohol drinking | 12 (14.8) | 5 (7.0) | < 0.001 |
| Influenza A | 2 (2.5) | 3 (4.2) | 0.545 |
| Influenza B | 0 (0.0) | 0 (0.0) | 1.000 |
| COVID-19 | 0 (0.0) | 0 (0.0) | 1.000 |
| Admission department | |||
| Geriatrics | 8 (9.9) | 20 (28.2) | < 0.001 |
| Internal Medicine | 29 (35.8) | 38 (53.5) | |
| Emergency | 37 (45.7) | 9 (12.7) | |
| Oncology | 4 (4.9) | 3 (4.2) | |
| Surgery | 0 (0.0) | 1 (1.4) | |
| Direct ICU admissions | 2 (2.5) | 0 (0.0) | |
| Ginecology | 1 (1.2) | 0 (0.0) | |
| Ward-to-ICU transfers | 5 (6.2) | 5 (7.0) | 0.829 |
| ICU length of stay, days | 3 (2 – 10) | 6 (5 – 11) | 0.151 |
| Hospital length of stay, days | 7 (1 – 35) | 7 (1 – 29) | 0.658 |
| Symptoms | |||
| Pneumonia | 18 (22.2) | 26 (36.6) | 0.050 |
| Bronchitis | 16 (19.8) | 46 (64.8) | < 0.001 |
| Cough | 61 (75.3) | 54 (76.1) | 0.915 |
| Fever | 33 (40.7) | 29 (40.8) | 0.990 |
| Odynophagia | 3 (3.7) | 3 (4.2) | 0.869 |
| Headache | 2 (2.5) | 4 (5.6) | 0.317 |
| Anorexia or hyporexia | 2 (2.5) | 4 (5.6) | 0.317 |
| Myalgia | 16 (19.8) | 2 (2.8) | 0.001 |
| Arthralgia | 13 (16.0) | 2 (2.8) | 0.006 |
| Pulmonary embolism | 0 (0.0) | 1 (1.4) | 1.000 |
| Other symptoms | 45 (55.6) | 61 (85.9) | < 0.001 |
| Comorbidities | |||
| Diabetes mellitus | 27 (33.3) | 27 (38.0) | 0.546 |
| Cardiovascular disease | 48 (59.3) | 56 (78.9) | 0.009 |
| Chronic liver disease | 3 (3.7) | 3 (4.2) | 0.495 |
| Chronic lung disease | 36 (44.4) | 29 (40.8) | 0.655 |
| Chronic kidney disease | 18 (22.2) | 17 (23.9) | 0.802 |
| CNMD | 14 (17.3) | 18 (25.4) | 0.046 |
| Cancer | 7 (8.6) | 6 (8.5) | 0.988 |
| Charlson index | |||
| 0 | 25 (30.9) | 17 (23.9) | 0.191 |
| 1 | 22 (27.2) | 21 (29.6) | |
| 2 | 15 (18.5) | 23 (32.4) | |
| 3 | 10 (12.3) | 9 (12.7) | |
| 4 | 5 (6.2) | 1 (1.4) | |
| 5 | 2 (2.5) | 0 (0.0) | |
| 6 | 1 (1.2) | 0 (0.0) | |
| 7 | 1 (1.2) | 0 (0.0) | |
| McCabe score | |||
| 1 | 71 (87.7) | 26 (36.6) | < 0.001 |
| 2 | 6 (7.4) | 24 (33.8) | |
| 3 | 4 (4.9) | 21 (29.6) | |
| Treatments | |||
| IMV | 1 (1.2) | 3 (4.2) | 0.342 |
| NIMV | 3 (3.7) | 11 (15.5) | 0.022 |
| Conventional oxygen therapy* | 58 (71.6) | 61 (85.9) | 0.049 |
| Anticoagulants | 16 (19.8) | 3 (4.2) | 0.004 |
| Corticosteroids | 50 (61.7) | 59 (83.1) | 0.004 |
| Radiotherapy | 0 (0.0) | 2 (2.8) | < 0.001 |
| Deceased | 11 (13.6) | 8 (11.3) | 0.668 |
| Parameter | AUC | 95% CI | Cut-off | Sensitivity | Specificity | PPV | NPV |
| IL-6 | 0.863 | 0.809 – 0.916 | 8.00 pg/mL | 0.835 | 1.000 | 1.000 | 0.761 |
| CRP | 0.774 | 0.713 – 0.834 | 1.85 mg/dL | 0.769 | 0.625 | 0.795 | 0.588 |
| Neutrophils | 0.886 | 0.842 – 0.931 | 5.0 x 106/mL | 0.794 | 0.912 | 0.946 | 0.695 |
| Lymphocytes | 0.894 | 0.853 – 0.935 | 1.7 x 106/mL | 0.816 | 0.875 | 0.925 | 0.714 |
| N/L ratio | 0.992 | 0.885 – 0.960 | 2.3 | 0.882 | 0.875 | 0.931 | 0.805 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




