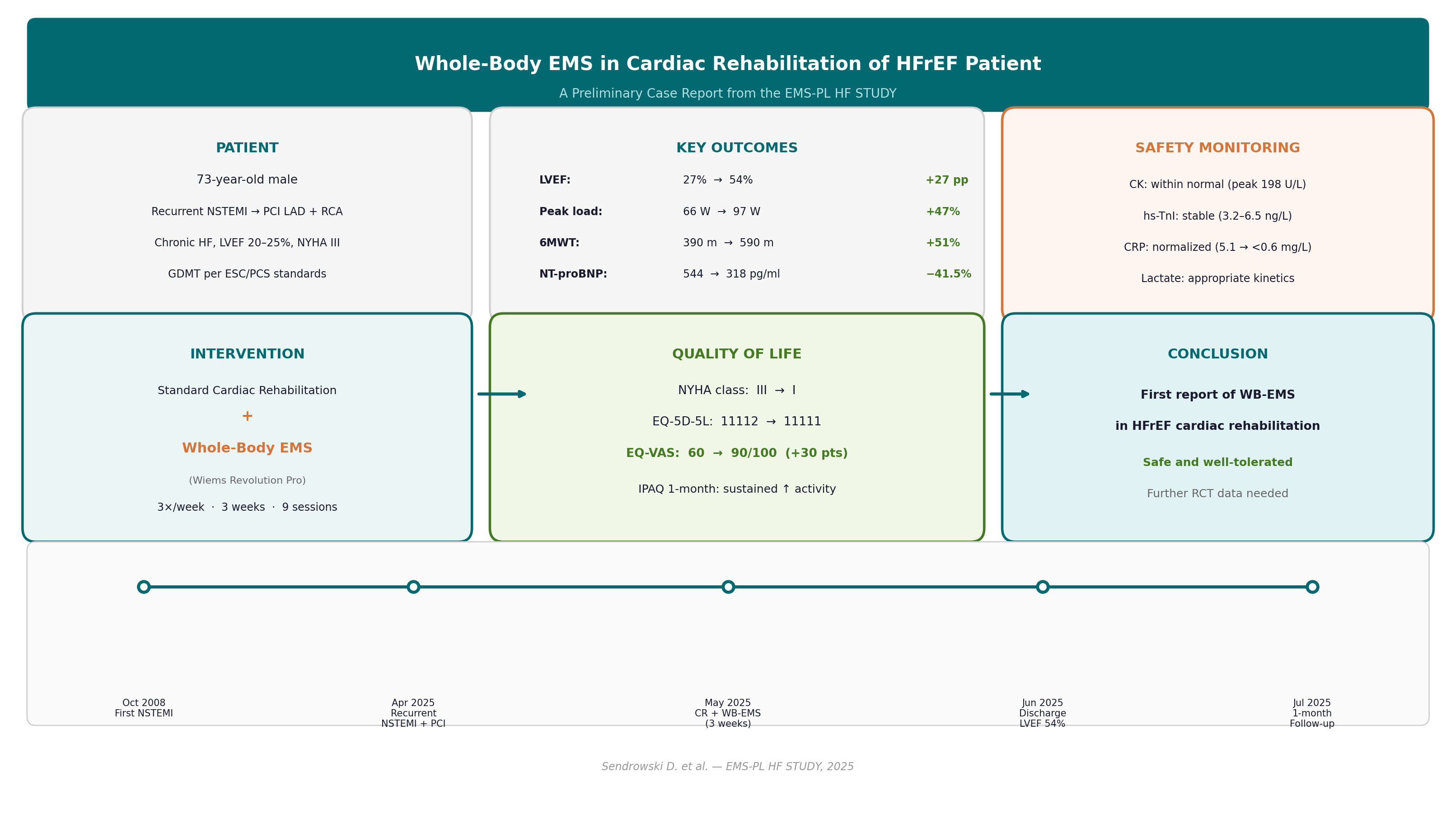
Background and Purpose: Whole-body electrical muscle stimulation (WB-EMS) is an emerging modality that activates multiple large muscle groups simultaneously via a wearable electrode suit. Although neuromuscular electrical stimulation (NMES) has demonstrated efficacy as an adjunctive strategy in cardiac rehabilitation (CR), WB-EMS has not previously been investigated in patients with heart failure and reduced ejection fraction (HFrEF) undergoing CR. This report presents preliminary findings from the active arm of an ongoing randomized trial. Methods: A 73-year-old male with recurrent non-ST-elevation myocardial infarction (NSTEMI), chronic HF (left ventricular ejection fraction [LVEF] 20–25%), and NYHA functional class III was admitted for inpatient CR. Standard CR (cycle ergometry, treadmill, resistance exercises, respiratory physiotherapy, and patient education, five days/week for 24 days) was augmented with WB-EMS (Wiems Revolution Pro device; 85 Hz bipolar, 350 μs, 4s on/4s off, 20-minute sessions) three times per week (~9 sessions). Safety was monitored with serial creatine kinase (CK), high-sensitivity troponin I (hs-TnI), NT-proBNP, C-reactive protein (CRP), and venous blood gas with lactate, measured before and 2–3 hours after each session. Results: LVEF improved from 27% to 54% (Simpson biplane); peak workload from 66 to 97 W (+47%); six-minute walk test (6MWT) distance from 390 to 590 m (+51%); and NT-proBNP declined from 544 to 318 pg/ml (−41.5%). EQ-5D-5L EQ-VAS improved from 60 to 90/100. One-month IPAQ data demonstrated sustained increases in daily physical activity. All safety biomarkers remained within acceptable limits throughout the programme. No adverse events were recorded. Discussion: To our knowledge, this is the first report of WB-EMS combined with standard CR in an HFrEF patient. The comprehensive serial biomarker monitoring confirmed a favorable safety profile. The striking improvements observed across functional, echocardiographic, neurohormonal, and quality-of-life domains are promising but require confirmation in the ongoing randomized trial, as multiple concurrent therapeutic interventions preclude causal attribution to WB-EMS alone.