Submitted:
15 April 2026
Posted:
16 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Search Strategy
2.3. Eligibility Criteria
2.4. Study Selection
2.5. Data Extraction
2.6. Synthesis Strategy
3. Results
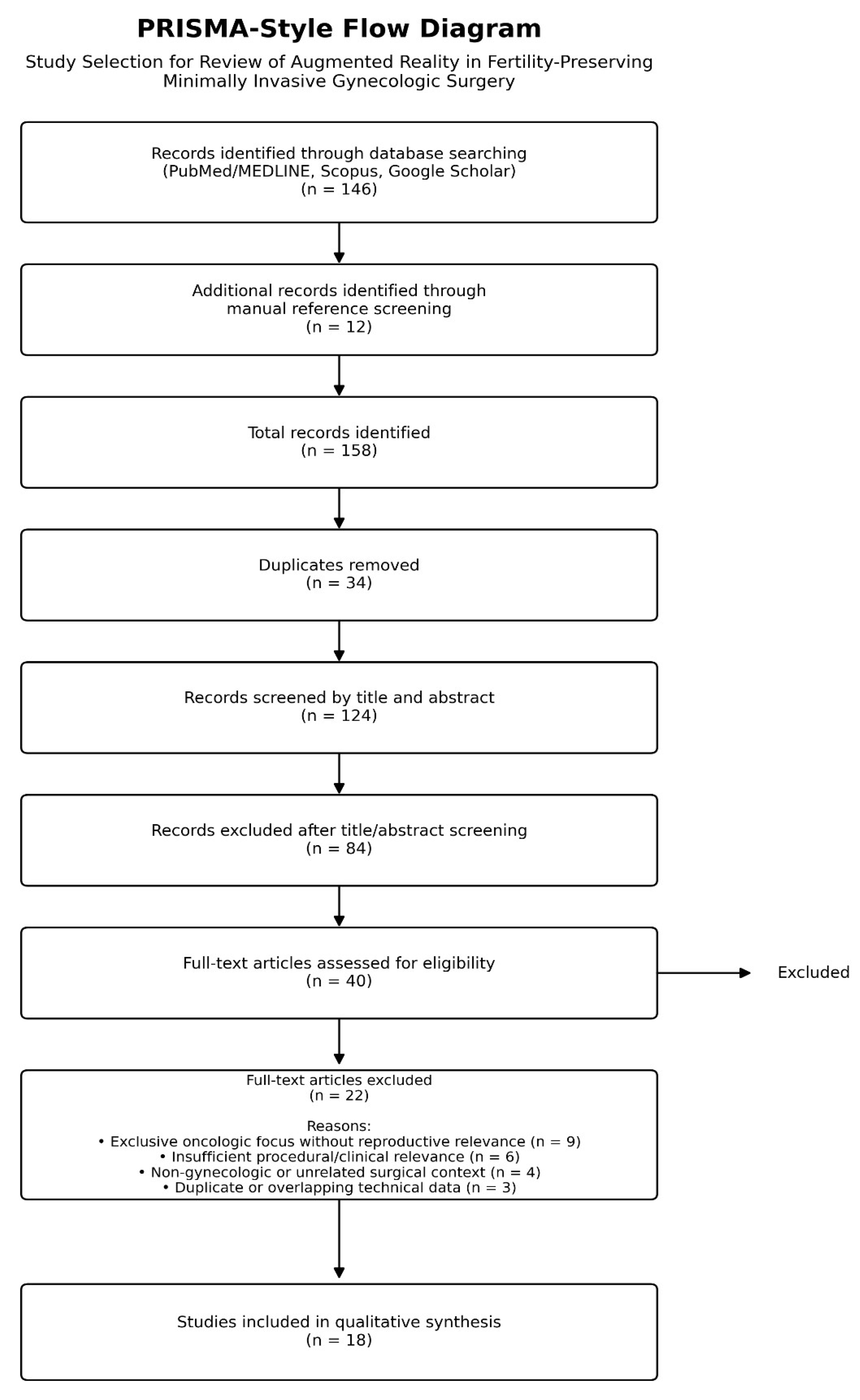
3.1. Study Selection
3.2. General Overview of the Evidence
3.3. Augmented Reality in Myomectomy
3.4. Augmented Reality in Adenomyomectomy and Adenomyosis Surgery
3.5. Augmented Reality in Endometriosis Surgery
3.6. Can Augmented Reality Prevent Complications in Reproductive Surgery?
3.7. Reproductive Outcomes: The Most Important Missing Evidence
4. Discussion
5. Future Directions
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Moawad, G.; Tyan, P.; Louie, M. Artificial intelligence and augmented reality in gynecology. Curr. Opin. Obstet. Gynecol. 2019, 31, 345–348. [CrossRef]
- Iftikhar, P.; Kuijpers, M.V.; Khayyat, A.; et al. Artificial intelligence: A new paradigm in obstetrics and gynecology research and clinical practice. Cureus 2020, 12, e7124. [CrossRef]
- Khamisy-Farah, R.; Furstenau, L.B.; Kong, J.D.; et al. Gynecology meets big data. Int. J. Environ. Res. Public Health 2021, 18, 5058. [CrossRef]
- Akladios, C.; Gabriele, V.; Agnus, V.; et al. Augmented reality in gynecologic laparoscopic surgery. Surg. Endosc. 2020, 34, 1077–1087. [CrossRef]
- Leaf, M.C.; Musselman, K.; Wang, K.C. Cutting-edge care in gynecologic surgery. Curr. Opin. Obstet. Gynecol. 2024, 36, 255–259.
- Sato, M.; Koizumi, M.; Nakabayashi, M.; et al. Computer vision for total laparoscopic hysterectomy. Asian J. Endosc. Surg. 2019, 12, 294–300. [CrossRef]
- Bourdel, N.; Collins, T.; Pizarro, D.; et al. Use of augmented reality in laparoscopic gynecology to visualize myomas. Fertil. Steril. 2017, 107, 737–739. [CrossRef]
- Chauvet, P.; Bourdel, N.; Calvet, L.; et al. Augmented reality with diffusion tensor imaging and tractography during laparoscopic myomectomies. J. Minim. Invasive Gynecol. 2020, 27, 973–976. [CrossRef]
- Bourdel, N.; Chauvet, P.; Calvet, L.; et al. Use of augmented reality in gynecologic surgery to visualize adenomyomas. J. Minim. Invasive Gynecol. 2019, 26, 1177–1180. [CrossRef]
- Ochi, Y.; Yanai, S.; Yoshino, Y.; et al. Mixed reality in laparoscopic myomectomy. Int. J. Gynaecol. Obstet. 2023, 162, 364–365.
- Comptour, A.; Chauvet, P.; Grémeau, A.S.; et al. Retrospective case control study on the evaluation of the impact of augmented reality in gynecological laparoscopy on patients operated for myomectomy or adenomyomectomy. Gynecol. Surg. 2025, 30, 2509686. [CrossRef]
- Netter, A.; Noorzadeh, S.; Duchateau, F.; et al. Initial results in the automatic visual recognition of endometriosis lesions by artificial intelligence during laparoscopy: A proof-of-concept study. J. Minim. Invasive Gynecol. 2025, 32, 1118–1125. [CrossRef]
- Zhang, C.; Martel, C.; Host, A.; et al. Three-dimensional modeling of deep endometriosis from pelvic MRI: Feasibility, clinical perception, and preliminary prospective evaluation. J. Minim. Invasive Gynecol. 2026, ahead of print. [CrossRef]
- Chauvet, P.; Collins, T.; Debize, C.; et al. Augmented reality in a tumor resection model. Surg. Endosc. 2018, 32, 1192–1201. [CrossRef]
- Lecointre, L.; Verde, J.; Goffin, L.; et al. Augmented reality for sentinel lymph node identification. Surg. Endosc. 2022, 36, 9224–9233.
- Lecointre, L.; Verde, J.; Hubele, F.; et al. Augmented reality-guided sentinel node targeting in endometrial cancer. EJNMMI Phys. 2022, 9, 81.
- Pisano, G.; Wendler, T.; Valdés Olmos, R.A.; et al. Molecular image-guided surgery in gynecologic cancer. Eur. J. Nucl. Med. Mol. Imaging 2024, 51, 3026–3039.
- Madad Zadeh, S.; François, T.; Comptour, A.; et al. SurgAI3.8K: A labeled dataset of gynecologic organs in laparoscopy with application to automatic augmented reality surgical guidance. J. Minim. Invasive Gynecol. 2023, 30, 397–405.
- Panichyawat, N.; Duchon, M.; Chauvet, P.; Bourdel, N. Intraureteric indocyanine green in laparoscopic endometriosis surgery: 10 steps. J. Minim. Invasive Gynecol. 2026, ahead of print. [CrossRef]
- Warshafsky, C.; Flaxman, T.; Duigenan, S.; Singh, S.S. Future of endometriosis visualization. J. Minim. Invasive Gynecol. 2024, ahead of print.
| Component | Description | Reproductive Surgical Relevance |
|---|---|---|
| Imaging acquisition | MRI, CT, SPECT/CT, or intraoperative ultrasound | Defines lesion anatomy and relationships to reproductive structures |
| Segmentation | Manual, semi-automated, or AI-assisted delineation | Identifies fibroids, adenomyotic lesions, endometrial cavity, ureters, and endometriotic nodules |
| Registration | Alignment of virtual and real anatomy | Determines AR accuracy during surgery |
| Tracking | Camera-, instrument-, or landmark-based tracking | Maintains overlay during operative movement |
| Visualization | Laparoscopic monitor, console, or head-mounted display | Delivers augmented information to the surgeon |
| Study | Country / Setting | Design | Procedure / Pathology | No. of Patients | Mean / Median Age | Imaging / Platform | Proposed AR Benefit | Reported Perioperative Benefit | Complications | Fertility Outcome Data | Evidence Level | Main Limitation |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Bourdel et al. (2017) [7] | France, tertiary center | Feasibility clinical report | Laparoscopic myomectomy | 3 | NR | MRI-based 3D uterine and myoma modeling with intraoperative overlay | Localization of occult/intramural fibroids; improved spatial orientation | Feasibility of lesion localization during laparoscopy | No major complications specifically highlighted | None reported | IV–V | Very small sample; no comparative outcomes; no fertility follow-up |
| Chauvet et al. (2020) [8] | France | Case report / pilot application | Laparoscopic myomectomy | 2 | NR | MRI + DTI + tractography AR | Visualization of uterine fiber architecture to guide incision planning | Proposed improvement in myometrial incision strategy and tissue-sparing dissection | NR | None reported | V | Two cases only; no clinical validation |
| Bourdel et al. (2019) [9] | France | Case report / technical application | Fertility-preserving adenomyomectomy / adenomyosis surgery | 1 | NR | MRI-based 3D lesion and cavity modeling with AR | Delineation of adenomyoma and endometrial cavity preservation | Proposed support for targeted excision and safer cavity-preserving dissection | NR | None reported | V | Single case; no comparative or reproductive outcome data |
| Ochi et al. (2023) [10] | Japan | Case report / mixed reality application | Laparoscopic myomectomy | 1 | NR | Mixed reality headset with holographic uterine model | Intraoperative lesion guidance and residual myoma awareness | Feasibility of mixed reality integration into workflow | NR | None reported | V | No comparative metrics; no fertility data |
| Comptour et al. (2025) [11] | France, single-center | Retrospective matched case-control study | Laparoscopic myomectomy / adenomyomectomy | 34 total (17 AR vs 17 controls) | 32.7 ± 4.3 years (AR) vs 33.4 ± 3.8 years (controls) | Preoperative uterine modeling with AR-guided laparoscopic navigation | Improve lesion targeting without increasing operative burden | Operative time: 135 ± 39 min (AR) vs 149 ± 62 min (controls); blood loss ≤200 mL in 82.3% vs 75% | No intraoperative or postoperative complications reported in either group | None reported | III | Non-randomized; small sample; no fertility endpoints |
| Akladios et al. (2020) [4] | France / technical framework | Technical / translational report | General gynecologic laparoscopy with relevance to myomectomy | NA | NA | AR workflow and navigation framework | Improve anatomical guidance and target localization | No direct procedure-specific perioperative outcomes | NA | NA | V | Not a direct clinical outcome study |
| Study | Country / Setting | Design | Surgical / Disease Context | No. of Patients / Cases | Mean / Median Age | Technology | Intended Use | Proposed or Reported Perioperative Benefit | Complications | Fertility Outcome Data | Evidence Level | Main Limitation |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Zhang et al. (2026) [13] | MRI modeling study | Preliminary prospective feasibility study | Deep infiltrating endometriosis | NR | NR | Pelvic MRI-based 3D modeling | Preoperative lesion mapping and anatomical relationship visualization | Improved understanding of lesion extent and adjacent organ involvement; planning aid for complex DIE surgery | NR | None reported | IV | Real-time intraoperative AR validation not established |
| Netter et al. (2025) [12] | International proof-of-concept collaboration | Proof-of-concept AI study | Endometriosis lesion recognition during laparoscopy | NR | NR | AI-assisted visual recognition relevant to future AR workflows | Automatic lesion detection as precursor to AR-enabled guidance | Potential future support for lesion awareness and dissection planning | Not a direct complication study | None reported | IV–V | Not a direct intraoperative AR outcome study |
| Panichyawat et al. (2026) [19] | Technical surgical application | Technical report | Laparoscopic endometriosis surgery | NR | NR | Intraureteric indocyanine green visualization | Ureteral identification in complex endometriosis dissection | Potential reduction in ureteral injury risk; improved ureteral awareness | Technical application only; formal complication reduction not proven | None reported | V | Not AR per se; image-guidance adjunct rather than overlay navigation |
| Warshafsky et al. (2024) [20] | Review article | Narrative / conceptual review | Endometriosis visualization and future imaging | NA | NA | Visualization technologies including AR-adjacent approaches | Improve future lesion visualization and surgical understanding | Conceptual support for future image-guided endometriosis surgery | NA | NA | V | No primary procedural data |
| Madad Zadeh et al. (2023) [18] | Translational development | Technical dataset study | Gynecologic laparoscopy with future AR guidance applications | NA | NA | SurgAI3.8K labeled laparoscopic dataset | Development of automatic gynecologic organ recognition for future AR guidance | Foundational step toward real-time anatomy-aware overlays | NA | NA | V | No direct clinical surgical outcomes |
| Surgical Challenge | Procedure(s) Most Relevant | Potential AR Contribution | Proposed Perioperative Benefit | Reproductive Relevance | Current Evidence Strength |
|---|---|---|---|---|---|
| Occult intramural fibroid localization | Myomectomy | Localization of hidden lesions before uterine incision | May reduce blind dissection, repeated uterine entry attempts, and unnecessary tissue trauma | Important for uterine preservation and reconstruction | Moderate conceptual support; limited clinical comparative evidence |
| Endometrial cavity preservation | Myomectomy, adenomyomectomy | Visualization of lesion–cavity relationship | May reduce inadvertent cavity breach and improve precision of resection | Relevant to uterine healing and fertility planning | Strong theoretical relevance; very limited direct clinical data |
| Poor adenomyoma margin definition | Adenomyomectomy | Improved lesion delineation using MRI-based modeling | May reduce over-resection or incomplete excision | Critical for balancing disease treatment and uterine preservation | Early case-based evidence only |
| Excessive myometrial trauma | Myomectomy, adenomyomectomy | More selective and anatomy-guided dissection | Potentially improved tissue preservation and reconstruction conditions | Important for future pregnancy safety | Conceptually strong; not outcome-proven |
| Ureteral injury risk | DIE surgery | Improved awareness of ureteral course or relation to disease | May support safer parametrial and pelvic sidewall dissection | Important in fertility-preserving pelvic surgery | Indirect or adjacent evidence only |
| Bowel or bladder interface uncertainty | DIE surgery | Better lesion-interface understanding | May support safer dissection planning and multidisciplinary surgery | Relevant to pelvic organ preservation and postoperative morbidity | Emerging translational support |
| Distorted pelvic anatomy / fibrosis | Endometriosis surgery | Enhanced spatial orientation and lesion mapping | Potentially reduces technical uncertainty and avoidable dissection error | Highly relevant in reproductive-age surgery | Translational / conceptual evidence |
| Workflow-related technical ambiguity | All complex reproductive surgeries | Improved surgeon orientation and procedural confidence | More deliberate dissection and potentially fewer technical errors | Relevant across fertility-preserving procedures | Plausible but unquantified |
| Domain | Current State of Evidence | Relevance | Implication for Future Research |
|---|---|---|---|
| Sample size and external validity | Most studies are case reports, small series, or single-center cohorts | Limits generalizability | Multicenter prospective cohorts are needed |
| Comparative perioperative outcomes | Rarely reported; only one small matched case-control study identified | Unclear whether AR improves surgery beyond feasibility | Controlled comparative studies are needed |
| Complication reporting | Often absent or underreported | Safety claims remain preliminary | Standardized complication reporting is required |
| Fertility outcomes | Essentially absent | Core rationale for use in reproductive surgery remains unproven | Pregnancy, live birth, uterine integrity, and obstetric outcomes should be reported |
| Registration accuracy | Poorly standardized | Misregistration may create false confidence | Validation metrics for pelvic AR are needed |
| Workflow burden / usability | Limited formal evaluation | Clinical adoption depends on practicality | Human-factors and workflow studies are needed |
| Cost-effectiveness | Not meaningfully studied | Resource burden may limit implementation | Economic analyses are required |
| Standardization across platforms | Highly heterogeneous | Difficult to compare studies or reproduce results | Consensus methodological frameworks are needed |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




