Submitted:
15 April 2026
Posted:
16 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
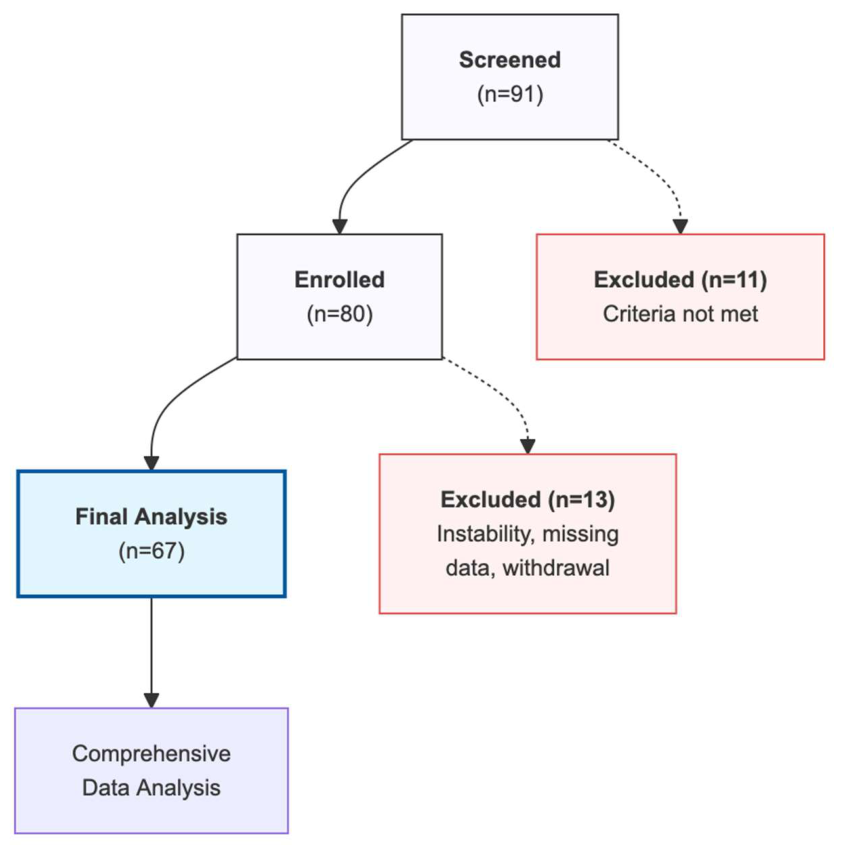
2. Materials and Methods
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| CSA-AKI | Cardiac Surgery-Associated Acute Kidney Injury |
| AKI | Acute Kidney Injury |
| NGAL | Neutrophil Gelatinase-Associated Lipocalin |
| KIM-1 | Kidney Injury Molecule-1 |
| L-FABP | Liver-type Fatty Acid-Binding Protein |
| IL-18 | Interleukin-18 |
| pKDIGO | Paediatric Kidney Disease Improving Global Outcomes |
| AUC | Area Under the Curve |
| ROC | Receiver Operating Characteristic |
| CHD | Congenital Heart Disease |
| CPB | Cardiopulmonary Bypass |
| RRT | Renal Replacement Therapy |
| SIRS | Systemic Inflammatory Response Syndrome |
| SD | Standard Deviation |
| ELISA | Enzyme-Linked Immunosorbent Assay |
| OR | Odds Ratio |
| CI | Confidence Interval |
| SpO2 | Peripheral Oxygen Saturation |
| ECG | Electrocardiogram |
| BP | Blood Pressure |
| MAC | Minimum Alveolar Concentration |
| CVP | Central Venous Pressure |
| EtCO2 | End-tidal Carbon Dioxide |
| NIRS | Near-Infrared Spectroscopy |
| IU | International Units |
| ACT | Activated Clotting Time |
| Z-BUF | Zero-balance Ultrafiltration |
| MUF | Modified Ultrafiltration |
| PCICU | Paediatric Cardiac Intensive Care Unit |
| eGFR | Estimated Glomerular Filtration Rate |
| BSA | Body Surface Area |
| NLR | Neutrophil-to-Lymphocyte Ratio |
| ASD | Atrial Septal Defect |
| VSD | Ventricular Septal Defect |
| RBC | Red Blood Cells |
| PaO2 | Partial Pressure of Arterial Oxygen |
| SvO2 | Mixed Venous Oxygen Saturation |
| VIS | Vasoactive-Inotropic Score |
| VI | Ventilation Index |
| VVR | Vasoactive-Ventilation-Renal score |
| MV | Mechanical Ventilation |
| IVF | Intravenous Fluids |
| LoS | Length of Stay |
References
- LoBasso, M.; Schneider, J.; Sanchez-Pinto L.N.; et al. Acute kidney injury and kidney recovery after cardiopulmonary bypass in children. Pediatr Nephrol. 2022, 37(3), 659-665. [CrossRef]
- Van den Eynde, J.; Rotbi, H.; Gewillig, M.; et al. In-hospital outcomes of acute kidney injury after pediatric cardiac surgery: a meta-analysis. Front Pediatr. 2021, 9:733744. [CrossRef]
- Zinchuk, P.V.; Ivkin, A.A.; Grigoriev, E.V.; et al. The role of nitric oxide in nephroprotection during surgical correction of heart defects in children (review). Messenger of Anesthesiology and Resuscitation. 2024, 21(6), 122-129. [Russian]. [CrossRef]
- Van den Eynde, J.; Salaets, T.; Louw, J.J.; et al. Persistent markers of kidney injury in children who developed acute kidney injury after pediatric cardiac surgery: a prospective cohort study. J Am Heart Assoc. 2022, 11(6):e024266. [CrossRef]
- Polushin, Y.S.; Dreval, R.O.; Zabotina, A.N.; et al. Clinical and economic assessment of therapy for sepsis-associated acute kidney injury with continuous combined renal replacement therapy. Messenger of Anesthesiology and Resuscitation. 2021, 18(5), 7-20. [Russian]. [CrossRef]
- Ronco, C.; Haapio, M.; House, A.A.; et al. Cardiorenal syndrome. J Am Coll Cardiol. 2008, 52(19), 1527-1539. [CrossRef]
- Yu, Y.; Li, C.; Zhu, S.; et al. Diagnosis, pathophysiology and preventive strategies for cardiac surgery-associated acute kidney injury: a narrative review. Eur J Med Res. 2023, 28(1):45. [CrossRef]
- Milne, B.; Gilbey, T.; De Somer, F.; et al. Adverse renal effects associated with cardiopulmonary bypass. Perfusion. 2024, 39(3), 452-468. [CrossRef]
- Sidebotham, D. Novel biomarkers for cardiac surgery-associated acute kidney injury: a skeptical assessment of their role. J Extra Corpor Technol. 2012, 44(4) 235-240.
- Ostermann, M.; Kunst, G.; Baker, E.; et al. Cardiac surgery associated AKI prevention strategies and medical treatment for CSA-AKI. J Clin Med. 2021, 10(22):5285. [CrossRef]
- Balakhnin, D.G.; Chermnykh, I.I.; Ivkin, A.A.; et al. The problem of diagnosing acute kidney injury in children operated on under cardiopulmonary bypass. Messenger of Anesthesiology and Resuscitation. 2023, 20(6), 106-115. [Russian]. [CrossRef]
- Sebastian, R.; Ahmed, M.I. Blood conservation and hemostasis management in pediatric cardiac surgery. Front Cardiovasc Med. 2021, 8:689623. [CrossRef]
- Chen, Q.H.; Wang, H.L.; Liu, L.; et al. Effects of restrictive red blood cell transfusion on the prognoses of adult patients undergoing cardiac surgery: a meta-analysis of randomized controlled trials. Crit Care. 2018, 22(1):142. [CrossRef]
- Borisenko, D.V.; Ivkin, A.A.; Shukevich, D.L.; et al. Significance of red blood cell components of donor blood in the priming volume of the cardiopulmonary bypass circuit in the development of systemic inflammation in the correction of congenital heart defects in children. General Reanimatology. 2022, 18(3), 30-37. [Russian]. [CrossRef]
- Ivkin, A.A.; Borisenko, D.V.; Grigoriev, E.V. Cardiopulmonary bypass without donor blood components as a way to prevent brain damage in children. Complex Issues of Cardiovascular Diseases. 2021, 10(2S), 21-26. [Russian]. [CrossRef]
- Ivkin, A.A.; Grigoriev, E.V.; Balakhnin, D.G.; et al. Intraoperative transfusion as a risk factor for cerebral injury after cardiac surgery in children: a prospective observational study. Annals of Critical Care. 2023, 1, 101-114. [Russian]. [CrossRef]
- Suieubekov, B.; Sepbayeva, A.; Yeshmanova, A.; et al. Cardiac surgery-associated acute kidney injury in newborns: a meta-analysis. Electron J Gen Med. 2023, 20(2):em448. [CrossRef]
- Gulia, M.; Sethi, S.K.; Sharma, R.; et al. WCN25-1159 Acute kidney injury in infants post cardiac surgery - incidence, risk factors and outcome. Kidney Int Rep. 2025, 10(2 Suppl) S26-S27. [CrossRef]
- Sharma, A.; Chakraborty, R.; Sharma, K.; et al. Development of acute kidney injury following pediatric cardiac surgery. Kidney Res Clin Pract. 2020, 39(3), 259-268. [CrossRef]
- Zubarioglu, A.U.; Yıldırım, Ö.; Zeybek, C.; et al. Validation of the vasoactive-ventilation-renal score for neonatal heart surgery. Cureus. 2021, 13(5):e15110. [CrossRef]
- Yoneyama, F.; Okamura, T.; Takigiku, K.; et al. Novel urinary biomarkers for acute kidney injury and prediction of clinical outcomes after pediatric cardiac surgery. Pediatr Cardiol. 2020, 41(4), 695-702. [CrossRef]
- Khan, U.A.; Coca, S.G.; Hong, K.; et al. Blood transfusions are associated with urinary biomarkers of kidney injury in cardiac surgery. J Thorac Cardiovasc Surg. 2014, 148(2), 726-732. [CrossRef]
| Characteristics | Group | N | Median (Q1-Q3) | p-value | |
| Height, cm | No AKI | 47 | 78 (70-85) | <0.001 | |
| AKI | 20 | 67 (60-70.5) | |||
| Weight, kg | No AKI | 47 | 9.4 (7.95-12.2) | 0.001 | |
| AKI | 20 | 7.05 (5.65-8.2) | |||
| BSA, m2 | No AKI | 47 | 0.438 (0.384-0.54) | 0.001 | |
| AKI | 20 | 0.345 (0.29-0.393) | |||
| Age at the procedure, mo |
No AKI | 47 | 15 (9-26) | <0.001 | |
| AKI | 20 | 5.5 (3.75-10) | |||
| Creatinine baseline, micromole/l |
No AKI | 47 | 29 (25-35.5) | <0.001 | |
| AKI | 20 | 21.5 (18-25) | |||
| eGFR baseline, ml/min/m2 |
No AKI | 47 | 94.9 (82.8-109.5) | 0.001 | |
| AKI | 20 | 119.5 (103.4-139.3) | |||
| Erythrocytes baseline, 1/1012 |
No AKI | 47 | 4.56 (4.35-4.755) | 0.004 | |
| AKI | 20 | 4.2 (4.008-4.558) | |||
| Neutrophils baseline, 1/109 |
No AKI | 47 | 26.9 (18.7-36.45) | 0.01 | |
| AKI | 20 | 18.55 (16.1-25.425) | |||
| Lymphocytes baseline, 1/109 |
No AKI | 47 | 62.2 (49.8-69.45) | 0.168 | |
| AKI | 20 | 66.2 (59.7-72.975) | |||
| Neutrophil-to-lymphocyte ratio | No AKI | 47 | 0.43 (0.266-0.696) | 0.02 | |
| AKI | 20 | 0.265 (0.216-0.419) | |||
| Haemoglobin, g/l | No AKI | 47 | 121 (116.5-126.5) | 0.022 | |
| AKI | 20 | 113 (106-120.75) | |||
| Haematocrit, % | No AKI | 47 | 36 (34.6-37.4) | 0.002 | |
| AKI | 20 | 33.5 (31.825-34.975) | |||
| Diagnosis, n (%) | ASD | No AKI | 31 (83.8%) | 0.008 | |
| AKI | 6 (16.2%) | ||||
| VSD | No AKI | 16 (53.3%) | |||
| AKI | 14 (46.7%) | ||||
| Sex, n (%) | Male | No AKI | 15 (62.5%) | 0.307 | |
| AKI | 9 (37.5%) | ||||
| Female | No AKI | 32 (74.4%) | |||
| AKI | 11 (25.6%) | ||||
| Characteristics | Group | N | Median (Q1-Q3) | p-value |
| CPB time, min | No AKI | 47 | 54 (42-71.5) | 0.023 |
| AKI | 20 | 73.5 (52-99.5) | ||
| Aortic cross-clamp time, min | No AKI | 47 | 29 (21-41.5) | 0.014 |
| AKI | 20 | 38.5 (30.5-60.25) | ||
| Lactate baseline, mmol/l | No AKI | 47 | 1.05 (0.825-1.37) | 0.099 |
| AKI | 20 | 0.9 (0.7-1.1) | ||
| Lactate peak concentration, mmol/l | No AKI | 47 | 1.9 (1.6-2.4) | 0.994 |
| AKI | 20 | 1.9 (1.6-2.4) | ||
| PaO2 baseline, mm Hg |
No AKI | 47 | 139 (117-162) | 0.568 |
| AKI | 20 | 133 (114.25-141.75) | ||
| PaO2 the lowest value, mm Hg | No AKI | 47 | 140 (120-174) | 0.528 |
| AKI | 20 | 123 (100.025-181.75) | ||
| Hb preoperatively, g/l | No AKI | 47 | 104 (99-112.5) | 0.046 |
| AKI | 20 | 100 (88-107) | ||
| HCT baseline, % | No AKI | 47 | 32 (30.725-34.8) | 0.059 |
| AKI | 20 | 31 (27.1-33.2) | ||
| Hb after CPB initiation, g/l |
No AKI | 47 | 78 (72-83) | 0.058 |
| AKI | 20 | 72 (69-75.5) | ||
| HCT after CPB initiation, % |
No AKI | 47 | 24.2 (22.1-26) | 0.041 |
| AKI | 20 | 23 (21.85-23.65) | ||
| Weight-adjusted transfusion volume, ml/kg | No AKI | 47 | 12 (0-16.08) | 0.002 |
| AKI | 20 | 20.714 (13.622-43.58) | ||
| SvO2 the lowest value, % | No AKI | 47 | 63.5 (61-66) | 0.023 |
| AKI | 20 | 61 (57-63) | ||
| NIRS the lowest value, % | No AKI | 47 | 71 (68.25-74) | 0.12 |
| AKI | 20 | 69 (63-73) |
| Characteristics | Group | N | Median (Q1-Q3) | p-value |
| Lactate 16h postoperatively, mmol/l | No AKI | 47 | 1.2 (1-1.4) | 0.002 |
| AKI | 20 | 1.5 (1.35-1.6) | ||
| Creatinine, peak concentration, micromole/l | No AKI | 47 | 31 (27.5-39) | 0.135 |
| AKI | 20 | 35 (30-43.25) | ||
| eGFR, the lowest value, ml/min/m2 | No AKI | 47 | 87.85 (77.5254-104.75) | <0.001 |
| AKI | 20 | 67.49 (56.2331-77.84) | ||
| Neutrophils 16h postoperatively, 1/109 |
No AKI | 47 | 73.1 (63.75-77.65) | 0.995 |
| AKI | 20 | 73.9 (63.15-77.75) | ||
| Lymphocytes 16h postoperatively, 1/109 |
No AKI | 47 | 17.9 (13.3-23.5) | 0.934 |
| AKI | 20 | 16.55 (14.15-23.13) | ||
| Neutrophil-to-lymphocyte ratio 16h postoperatively | No AKI | 47 | 3.99 (2.6945-5.82) | 0.853 |
| AKI | 20 | 4.53 (2.7115-5.53) | ||
| Hb 16h postoperatively, g/l | No AKI | 47 | 111 (104-120) | 0.451 |
| AKI | 20 | 113 (106.75-118.25) | ||
| HCT 16h postoperatively, % | No AKI | 47 | 33 (30.85-35.2) | 0.924 |
| AKI | 20 | 33.15 (30.8-34.55) | ||
| VI-Score value | No AKI | 47 | 0 (0-5) | 0.021 |
| AKI | 20 | 5 (3.75-10) | ||
| VVR-Score value | No AKI | 47 | 1.58 (0.0565-5.9) | <0.001 |
| AKI | 20 | 7.6 (5.2755-11.47) | ||
| MV time, h | No AKI | 47 | 6 (3-9) | 0.17 |
| AKI | 20 | 8 (5-12.5) | ||
| IVF volume over 16 h, ml/kg | No AKI | 47 | 69.3 (53.9091-80.96) | 0.075 |
| AKI | 20 | 81.37 (64.8776-96.3) | ||
| PCICU LoS, h | No AKI | 47 | 24 (23.5-48) | 0.005 |
| AKI | 20 | 48 (43.75-84.5) | ||
| Hospital LoS, day | No AKI | 47 | 8 (5-9) | 0.088 |
| AKI | 20 | 9 (5-11.25) |
| Predictor variable | AUC | Se | Sp | 95% CI | Youden index | Cut-off value | p-value |
| Weight, kg | 0.694 | 0.615 | 0.810 | 0.527-0.862 | 0.425 | 7.68 | 0.023 |
| Age at the procedure, mo | 0.745 | 0.846 | 0.738 | 0.582-0.909 | 0.584 | 10.5 | 0.003 |
| BSA, m2 | 0.701 | 0.769 | 0.667 | 0.539-0.864 | 0.436 | 0.397 | 0.015 |
| CPB time, min | 0.66 | 0.563 | 0.744 | 0.512-0.809 | 0.307 | 69 | 0.035 |
| Aortic cross-clamp time, min | 0.673 | 0.75 | 0.605 | 0.528-0.818 | 0.355 | 30.5 | 0.019 |
| Weight-adjusted transfusion volume, ml/kg | 0.737 | 0.575 | 0.698 | 0.587-0.888 | 0.448 | 13.763 | 0.002 |
| Predictor variable | B | OR | 95% CI | R2 | AUC | Se | Sp | p-value |
| Weight, kg | -0.25 | 0.779 | 0.631-0.961 | 0.11 | 0.751 | 0.7 | 0.766 | 0.02 |
| Age at the procedure, mo | -0.06036 | 0.941 | 0.886-1 | 0.0942 | 0.772 | 0.85 | 0.723 | 0.051 |
| CPB time, min | 0.0257 | 1.0261 | 1.0053-1.047 | 0.081 | 0.677 | 0.5 | 0.766 | 0.014 |
| Aortic cross-clamp time, min | 0.0334 | 1.034 | 1.0043-1.064 | 0.0655 | 0.691 | 0.4 | 0.787 | 0.024 |
| Weight-adjusted transfusion volume, ml/kg | 0.0423 | 1.043 | 1.0071-1.081 | 0.0853 | 0.74 | 0.45 | 0.891 | 0.019 |
| Biomarker | Group | N | Median (Q1-Q3) | p-value |
| L-FABPu – 2, ng/ml | No AKI | 47 | 0.386 (0.22-0.753) | 0.009 |
| AKI | 20 | 0.952 (0.437-2.84) | ||
| L-FABPu – 2/L-FABPu – 1 | No AKI | 47 | 1.171 (0.811-2.267) | 0.039 |
| AKI | 20 | 1.546 (1.055-7.552) | ||
| L-FABPu – 3/L-FABPu – 2 | No AKI | 47 | 0.933 (0.7-1.236) | 0.014 |
| AKI | 20 | 0.606 (0.28-0.813) |
| Biomarker | Statistical parameters | Correlation with creatinine increase |
| NGALb – 3/NGALb – 2 | Rho | -0.236 |
| p-value | 0.065 | |
| NGALu – 3, ng/ml | Rho | -0.232 |
| p-value | 0.069 | |
| NGALu – 3/NGALu – 2 | Rho | -0.288 |
| p-value | 0.024 | |
| L-FABPb – 2, ng/ml | Rho | 0.231 |
| p-value | 0.067 | |
| KIM-1b – 2, ng/ml | Rho | -0.213 |
| p-value | 0.090 | |
| KIM-1b – 3, ng/ml | Rho | -0.222 |
| p-value | 0.081 | |
| KIM-1b – 2/KIM-1b – 1 | Rho | 0.256 |
| p-value | 0.043 | |
| IL-18b – 3/IL-18b – 2 | Rho | -0.264 |
| p-value | 0.040 | |
| IL-18b – 3/IL-18b – 1 | Rho | -0.248 |
| p-value | 0.052 | |
| IL-18u – 3, ng/ml | Rho | -0.284 |
| p-value | 0.025 |
| Biomarker | Statistical parameters | Correlation with transfusion |
| L-FABPb – 2, ng/ml | Rho | 0.214 |
| p-value | 0.092 | |
| L-FABPb – 3, ng/ml | Rho | 0.329 |
| p-value | 0.009 | |
| L-FABPu – 2, ng/ml | Rho | 0.352 |
| p-value | 0.005 | |
| L-FABPu – 2/L-FABPu – 1 | Rho | 0.258 |
| p-value | 0.043 | |
| L-FABPu – 3/L-FABPu – 2 | Rho | -0.54 |
| p-value | <0.001 | |
| KIM-1b – 3/KIM-1b – 2 | Rho | 0.251 |
| p-value | 0.053 | |
| KIM-1b – 3/KIM-1b – 1 | Rho | 0.244 |
| p-value | 0.059 | |
| IL-18u – 2, ng/ml | Rho | 0.290 |
| p-value | 0.022 | |
| IL-18u – 3/IL-18u – 2 | Rho | -0.253 |
| p-value | 0.052 |
| Predictor variable | AUC | Se | Sp | 95% CI | Youden index | Cut-off value | p-value |
| L-FABPu – 2, ng/ml | 0.702 | 0.706 | 0.636 | 0.560-0.844 | 0.342 | 0.497 | 0.005 |
| L-FABPu – 2/ L-FABPu– 1 |
0.667 | 0.941 | 0.341 | 0.518-0.816 | 0.282 | 0.884 | 0.028 |
| L-FABPu – 3/ L-FABPu– 2 |
0.747 | 0.846 | 0.714 | 0.595-0.899 | 0.56 | 0.754 | 0.001 |
| IL-18u – 2, ng/ml | 0.725 | 0.615 | 0.905 | 0.531-0.92 | 0.52 | 18.24 | 0.023 |
| IL-18u – 3/ IL-18u – 2 |
0.703 | 0.538 | 0.905 | 0.501-0.906 | 0.443 | 0.497 | 0.049 |
| Predictor variable | B | OR | 95% CI | R2 | AUC | Se | Sp | p-value |
| L-FABPu – 2, нг/мл | 0.122 | 1.13 | 0.997-1.28 | 0.102 | 0.711 | 0.611 | 0.689 | 0.056 |
| L-FABPu – 2/L-FABPu– 1 | 0.0917 | 1.096 | 0.99-1.213 | 0.101 | 0.668 | 0.556 | 0.644 | 0.077 |
| L-FABPu – 3/L-FABPu – 2 | -1.3 | 0.272 | 0.0729-1.02 | 0.0844 | 0.705 | 0.882 | 0.409 | 0.053 |
| IL-18u – 2, ng/ml | 0.0549 | 1.056 | 1.0095-1.106 | 0.128 | 0.626 | 0.444 | 0.533 | 0.018 |
| Predictor variable | B | OR | 95% CI | AUC | Se | Sp | p-value |
| Constant | -2.2966 | - | - | 0.755 | 0.667 | 0.705 | 0.002 |
| Weight-adjusted transfusion volume, ml/kg | 0.0285 | 1.029 | 0.9953-1.064 | ||||
| IL-18u – 2, ng/ml | 0.051 | 1.052 | 1.0049-1.102 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




