Submitted:
15 April 2026
Posted:
16 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
- Elucidate the dynamic rheological behaviour of the composite hydrogels in relation to sol-gel thermal transitions,
- Investigate the structural and microstructural organisation of the gel−emulsion composites and
- Assess the 3D printing feasibility of the beeswax-structured gel-emulsions.
2. Materials and Methods
2.1. Materials
2.2. Preparation of the Beeswax Oil-In-Gel Emulsions
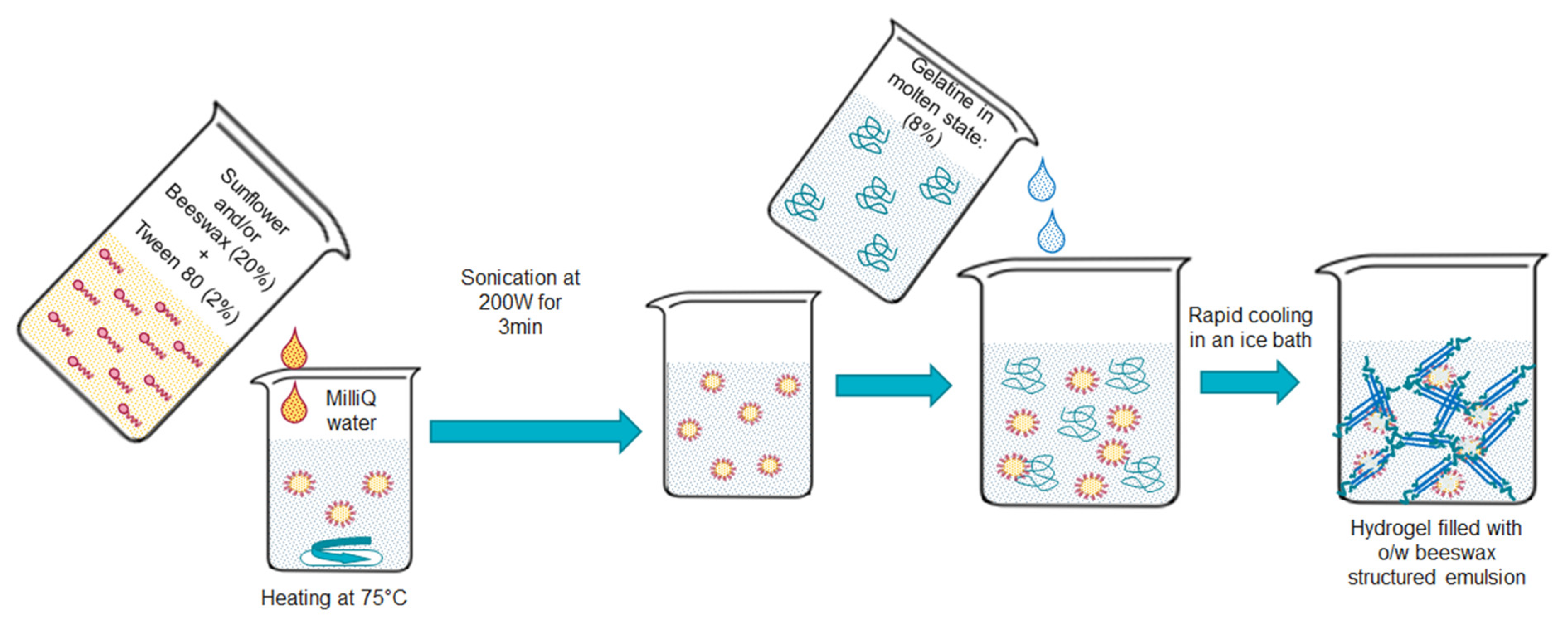
2.3. Lipid Droplet Size Distribution and Colloidal Stability Measurements
2.4. Confocal Laser Scanning Microscopy (CLSM) Measurements
2.5. Differential Scanning Calorimetry Measurements
2.6. Colour Measurements
2.7. Oscillatory Rheology Characterisation of the Beeswax Oil-In-Gel Emulsions
2.7.1. Oscillatory Thermo-Rheological (OTR) Measurements
2.7.2. Small Amplitude Oscillatory (SAOS) Rheological Behaviour
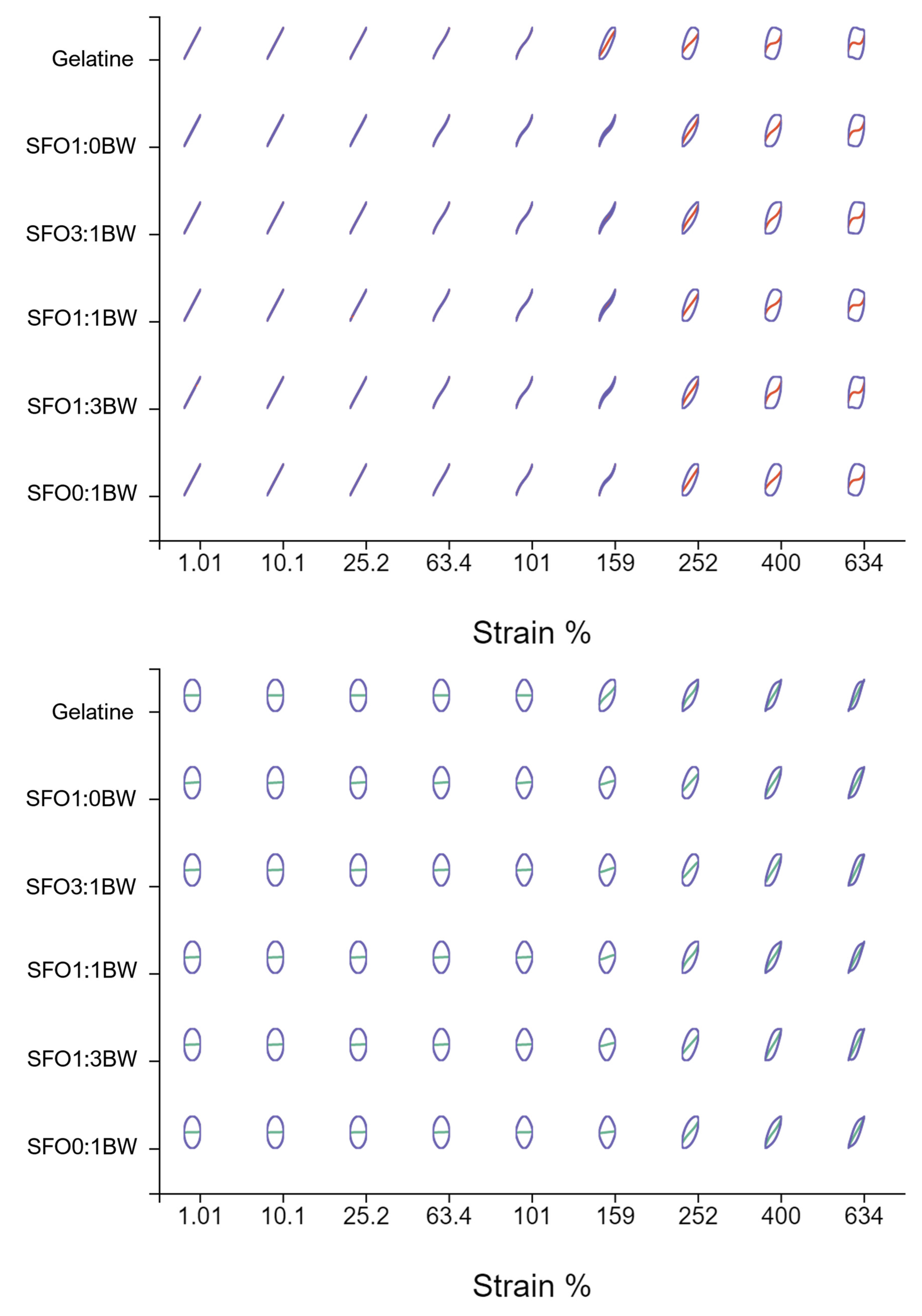
2.7.3. Large Amplitude Oscillatory (LAOS) Rheological Behaviour
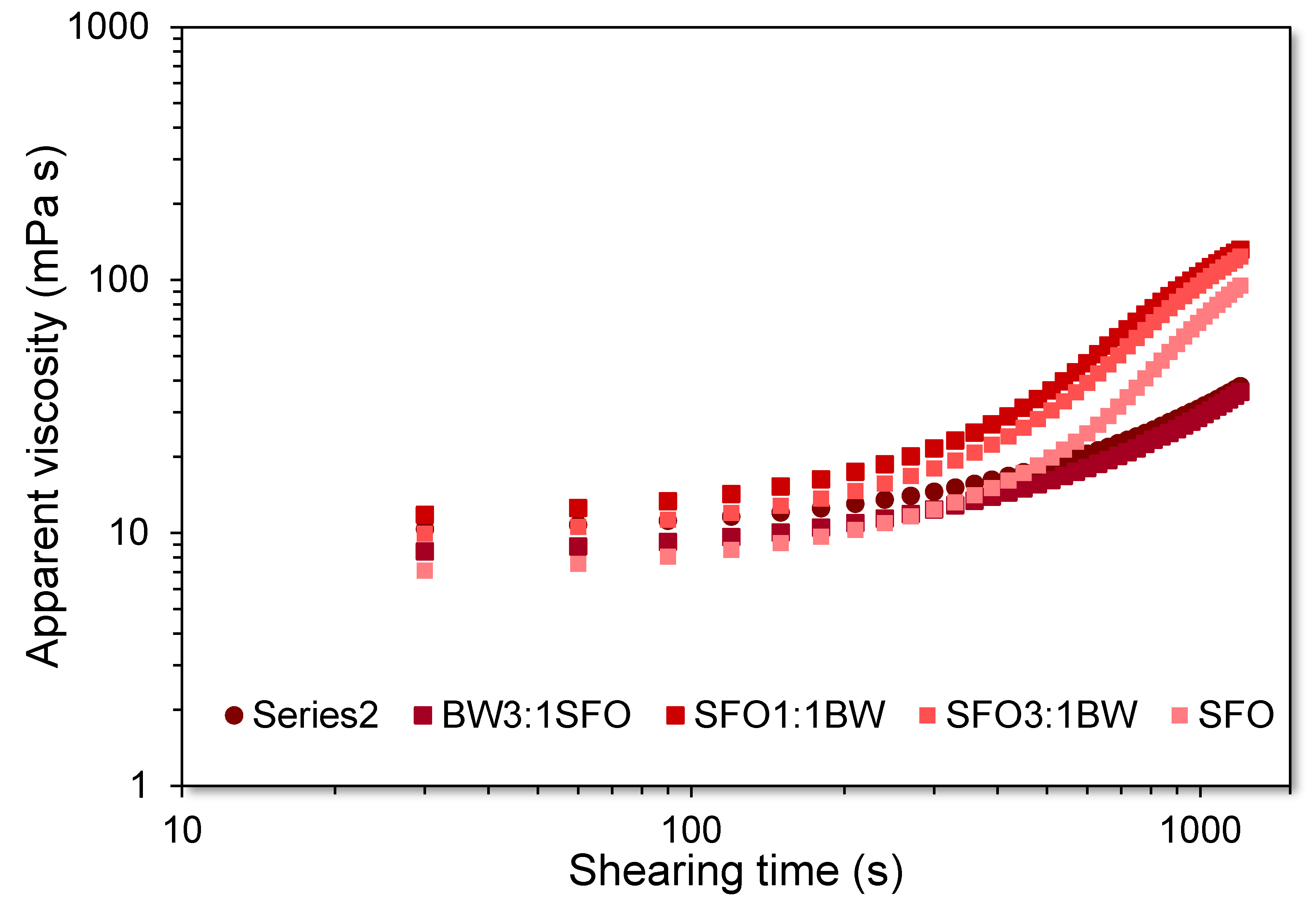
2.8. 3D Printability of the Beeswax Oil-In-Gel Emulsions
2.9. Statistical Analyses
3. Results and Discussion
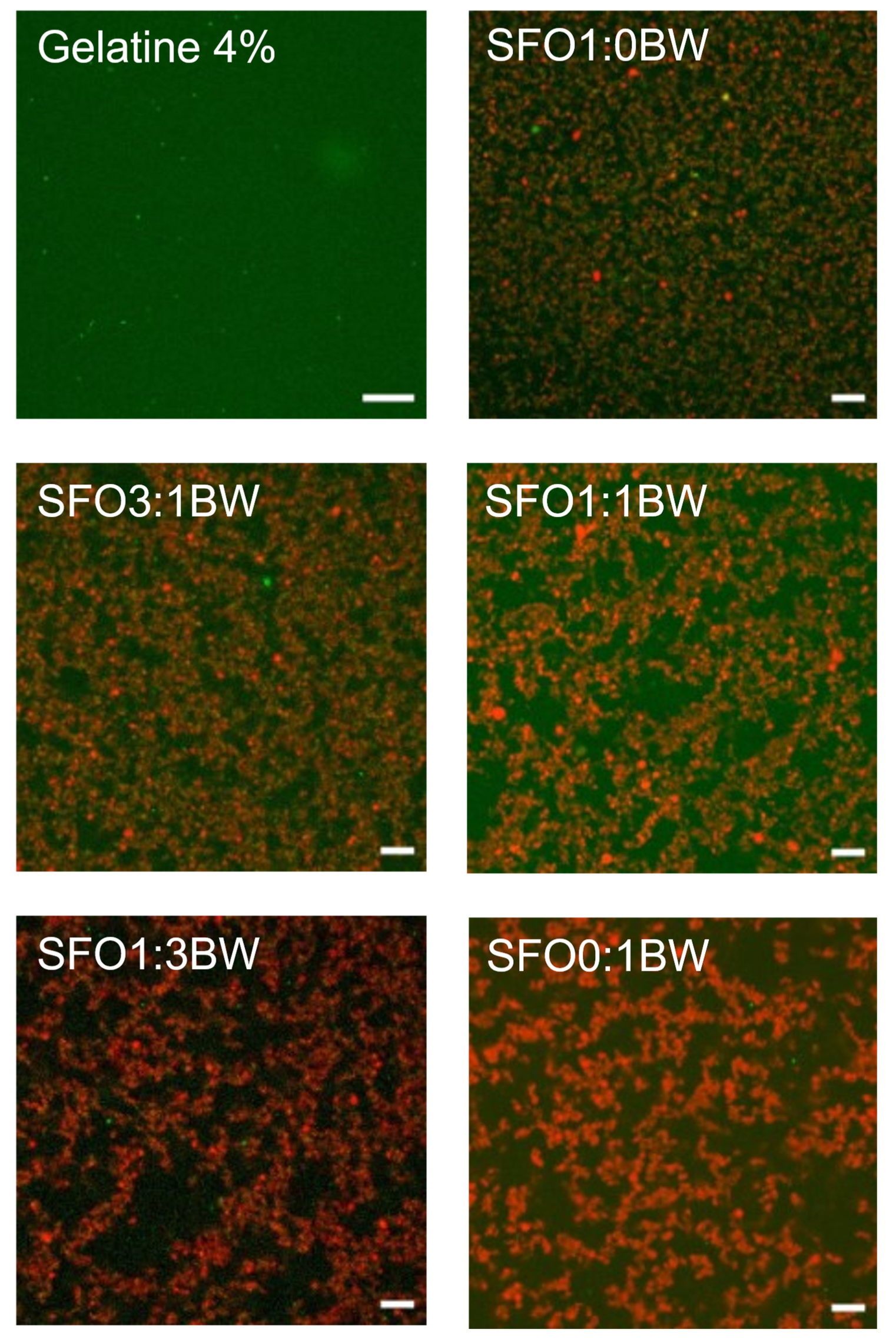
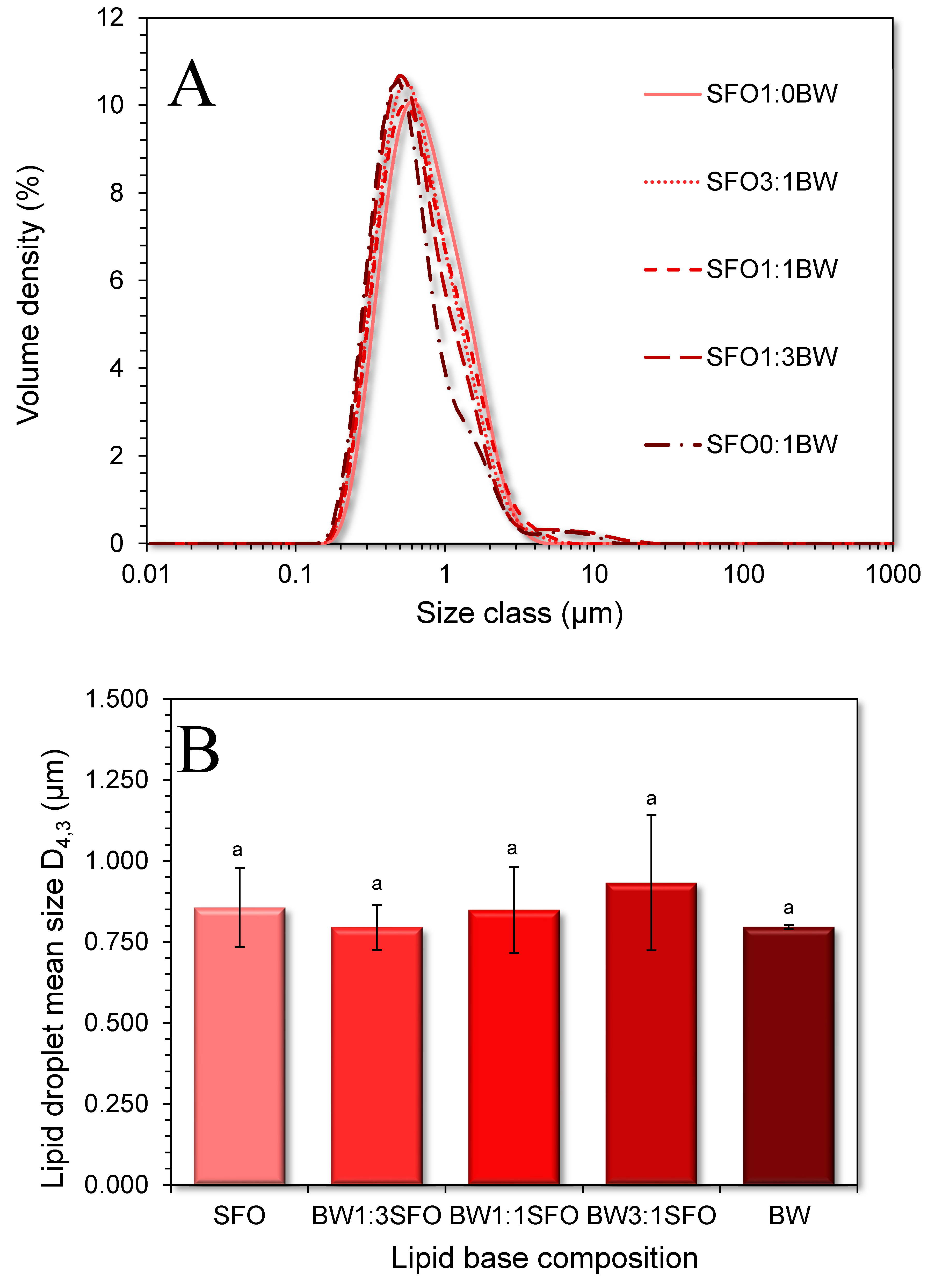
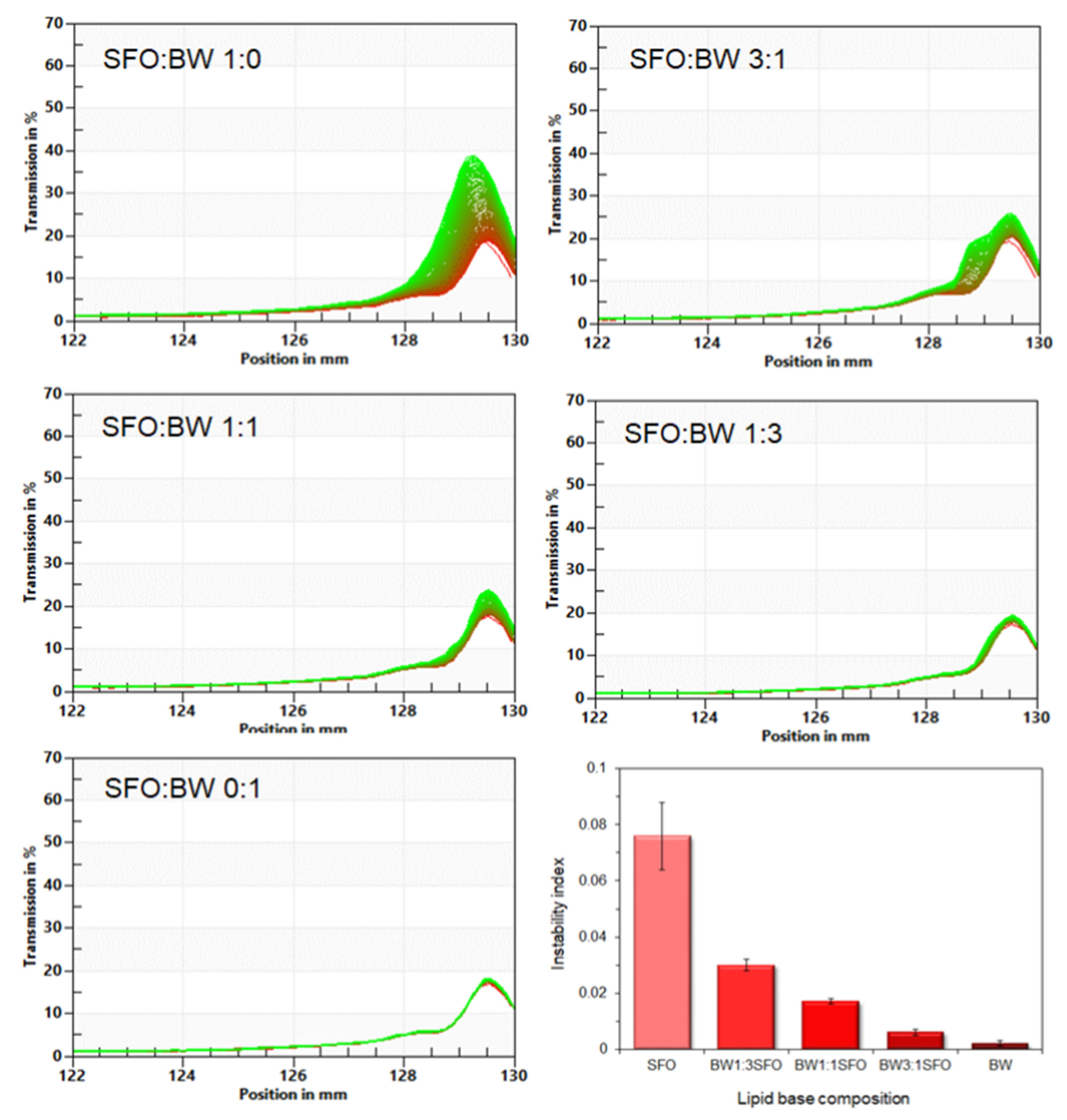
3.1. Microstructural and Physicochemical Characteristics of the BOGEs
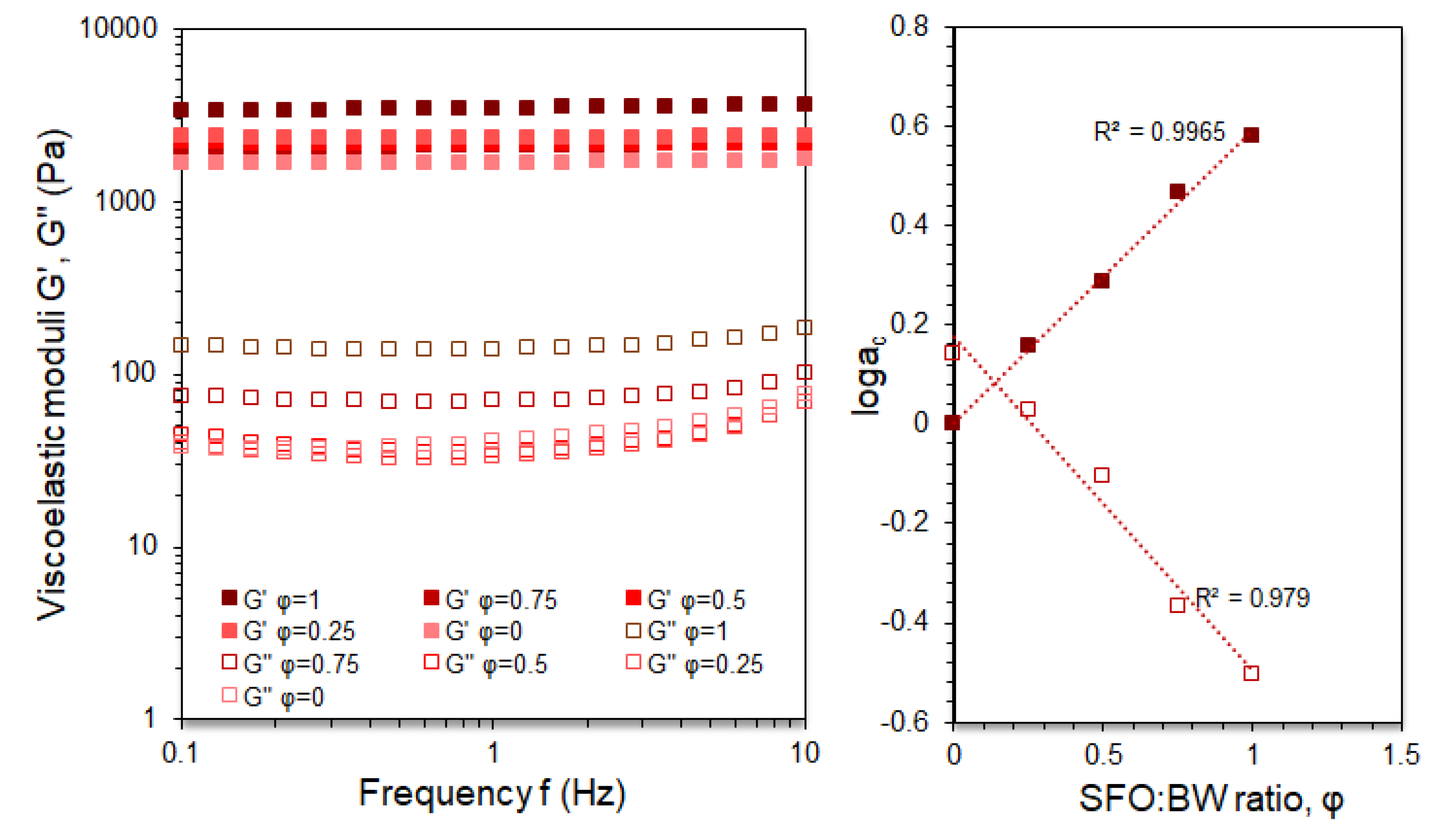
3.2. Thermophysical and Oscillatory Thermo-Rheological Behaviour of the BOGEs
| Gelatine | Beeswax solid fraction | |||||
|---|---|---|---|---|---|---|
| Sample | Tm, onset | Tm, midpoint | ΔH | Tm, onset | Tm, midpoint | ΔH |
| Gelatine | 31.3a | 35.4a | 1.39d | nd | nd | nd |
| SFO1:0BW | 32.0a | 35.9a | 0.90c | nd | nd | nd |
| SFO3:1BW | 31.8a | 35.9a | 0.58b | 46.6a | 56.8a | 1.59a |
| SFO1:1BW | 32.9a | 37.7b | 0.52b | 48.7ab | 59.8a | 3.25b |
| SFO1:3BW | 31.5a | 35.8a | 0.48b | 51.3b | 61.6ab | 6.24c |
| SFO0:1BW | 33.1a | 35.8a | 0.19a | 51.7b | 62.7b | 13.8d |
| Sample | Onset | Tm (mid) | Offset |
|---|---|---|---|
| SFO1:0BW | 16.21b | 23.05c | 29.89b |
| SFO3:1BW | 14.62a | 21.87a | 29.11b |
| SFO1:1BW | 14.89a | 21.63a | 28.37a |
| SFO1:3BW | 15.33a | 22.40b | 29.47b |
| SFO0:1BW | 15.12a | 22.10ab | 29.09b |
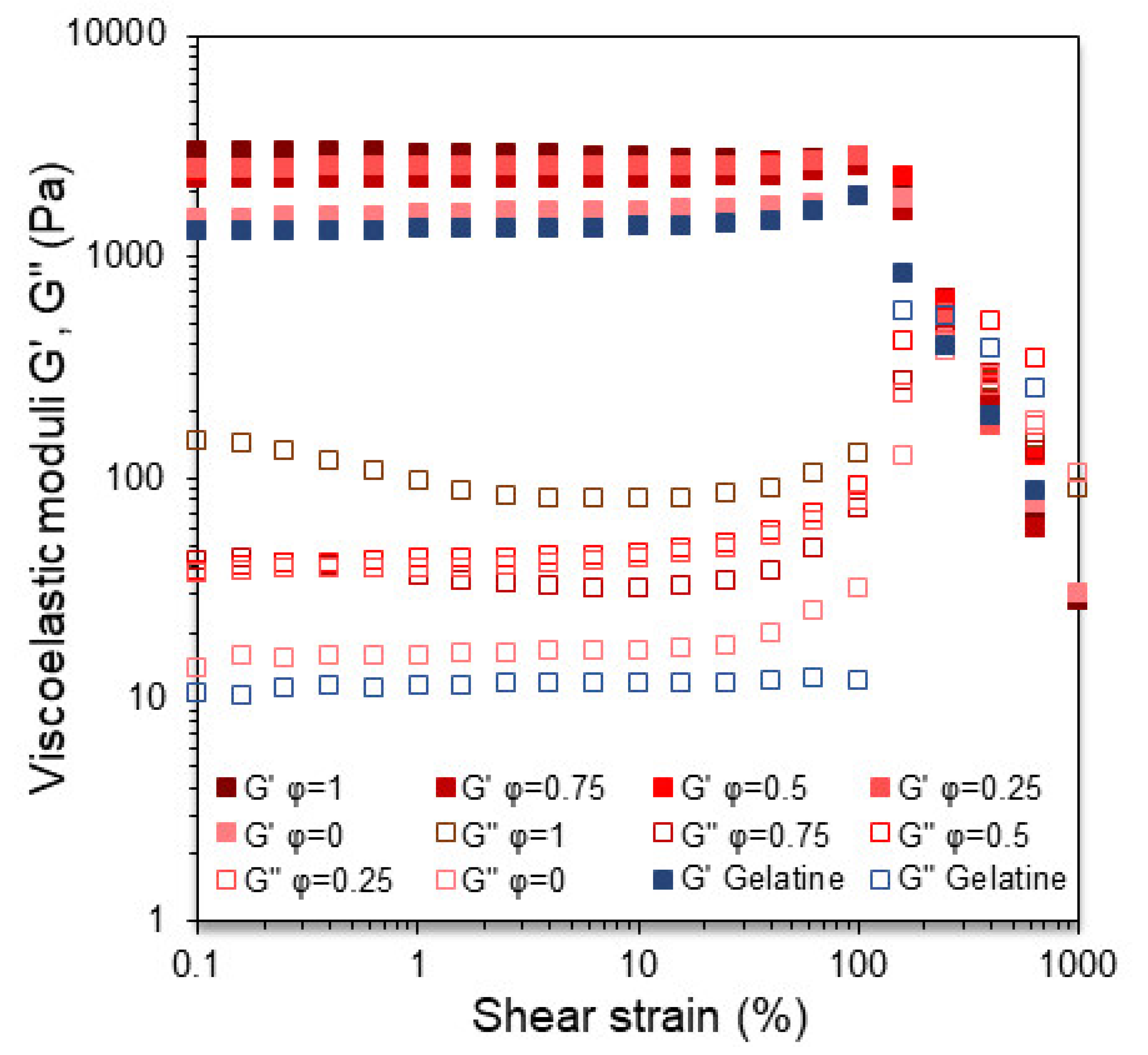
| Sample | γLVE (%) |
G’LVE (Pa) |
τf (Pa) |
γf (%) |
|---|---|---|---|---|
| GELATINE | 5.78d | 1330a | 1662d | 205a |
| SFO1:0BW | 0.191a | 1476b | 1448a | 286b |
| SFO3:1BW | 0.565b | 2524d | 1526b | 304c |
| SFO1:1BW | 1.74c | 2519d | 1587c | 321cd |
| SFO1:3BW | 15.5e | 2281c | 1679d | 332d |
| SFO0:1BW | 11.4e | 2867e | 1596c | 318cd |
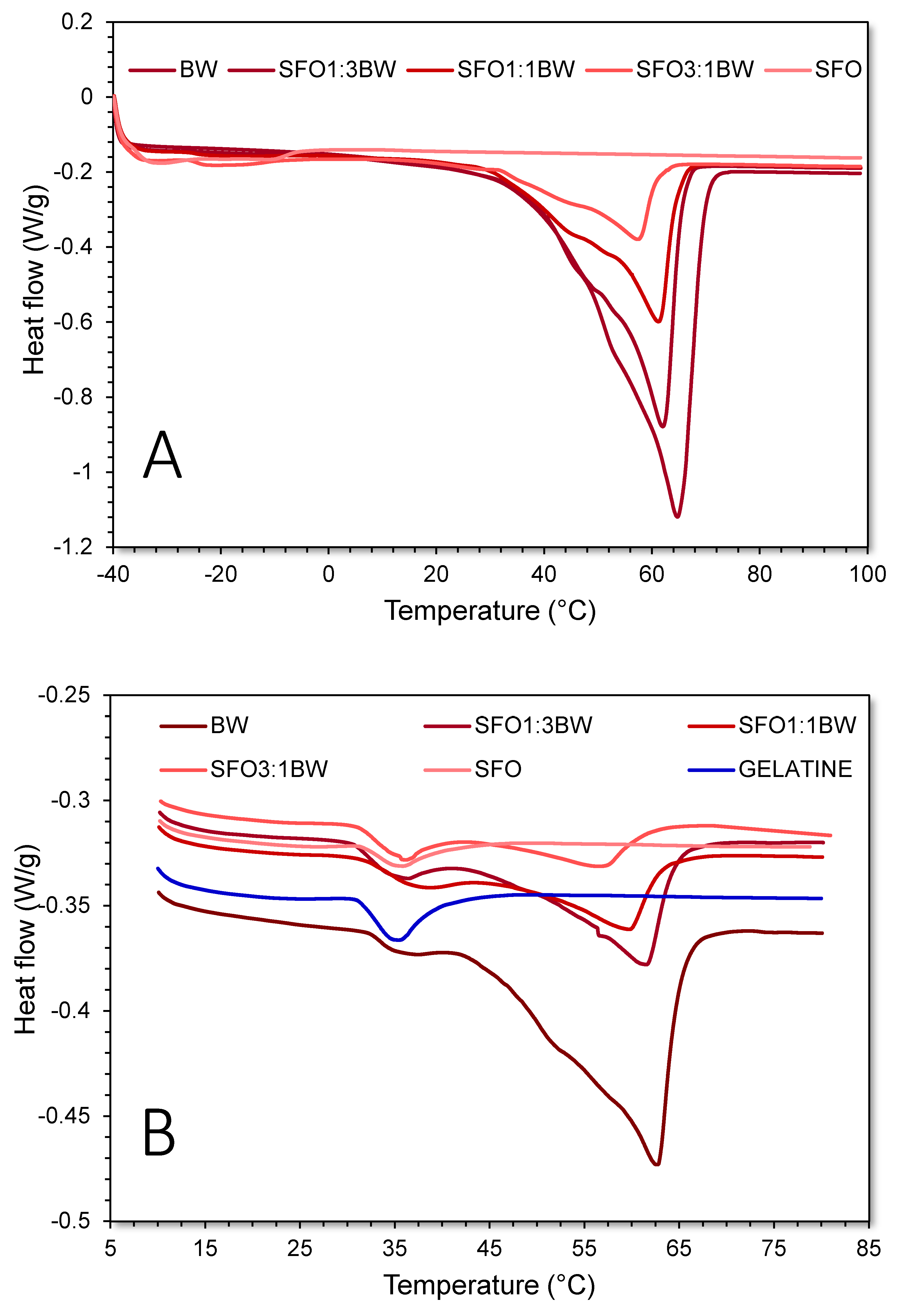
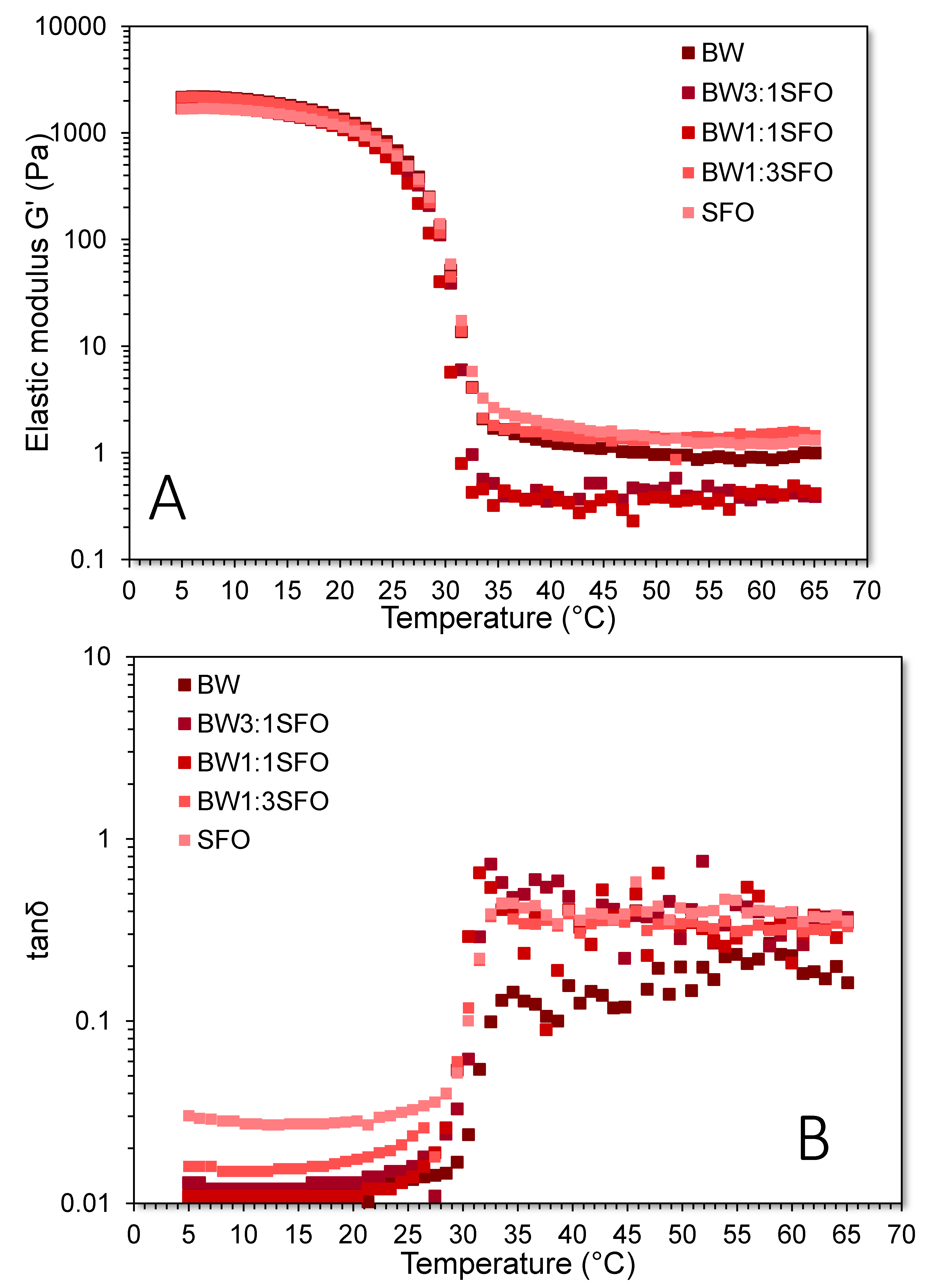
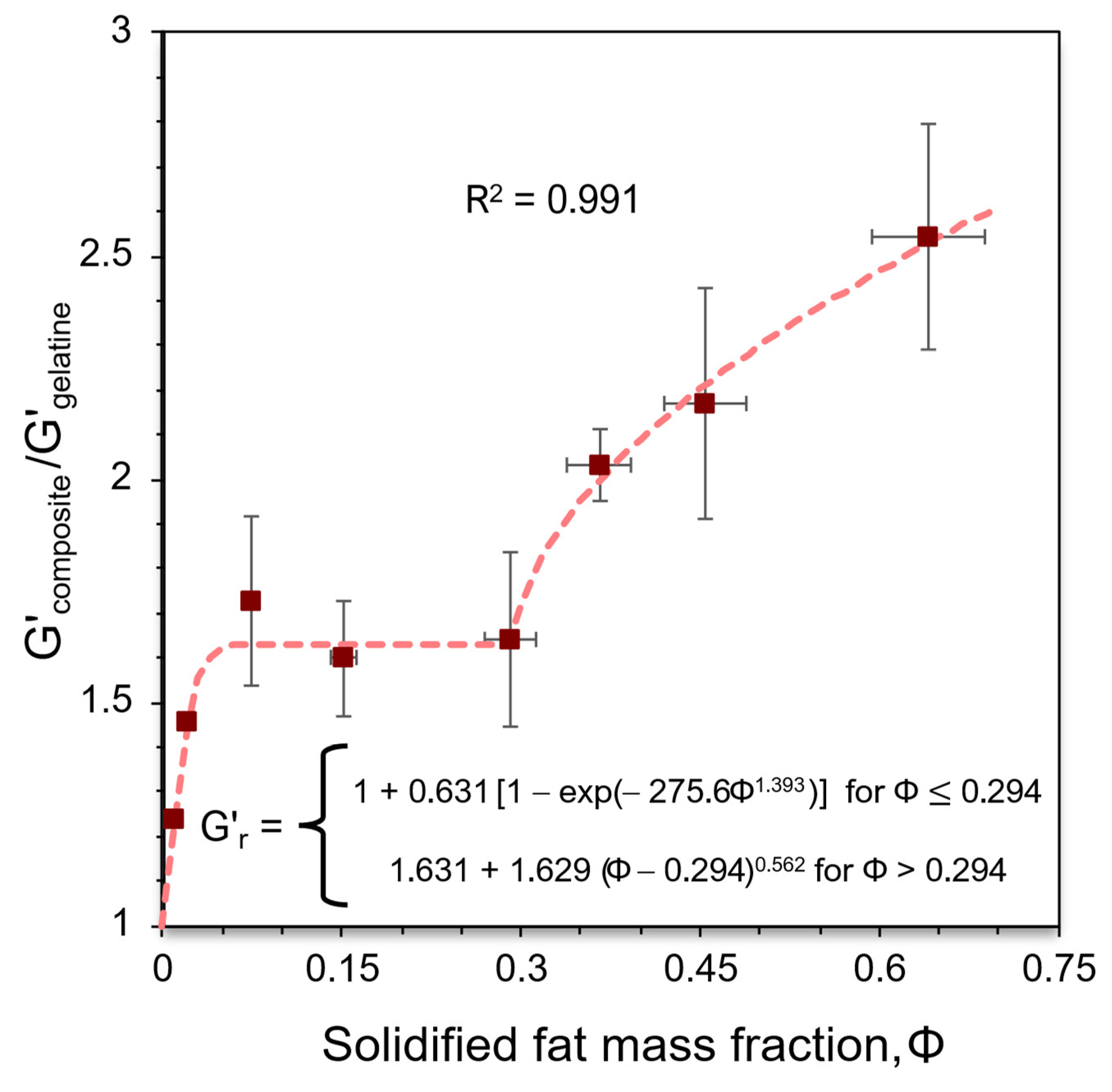
3.3. Viscoelastic Characterisation of the BOGEs
| Sample | L | a* | b* | C* | ΔΕ* |
|---|---|---|---|---|---|
| SFO1:0BW | 86.93a | −0.40a | 0.96a | 1.04a | - |
| SFO3:1BW | 88.99b | −0.73b | 0.84a | 1.11a | 2.09a |
| SFO1:1BW | 89.65bc | −1.05c | 2.04b | 2.30b | 3.00b |
| SFO1:3BW | 89.99c | −1.29c | 3.09c | 3.35c | 3.83c |
| SFO0:1BW | 90.21c | −1.47d | 4.09d | 4.35d | 4.66d |
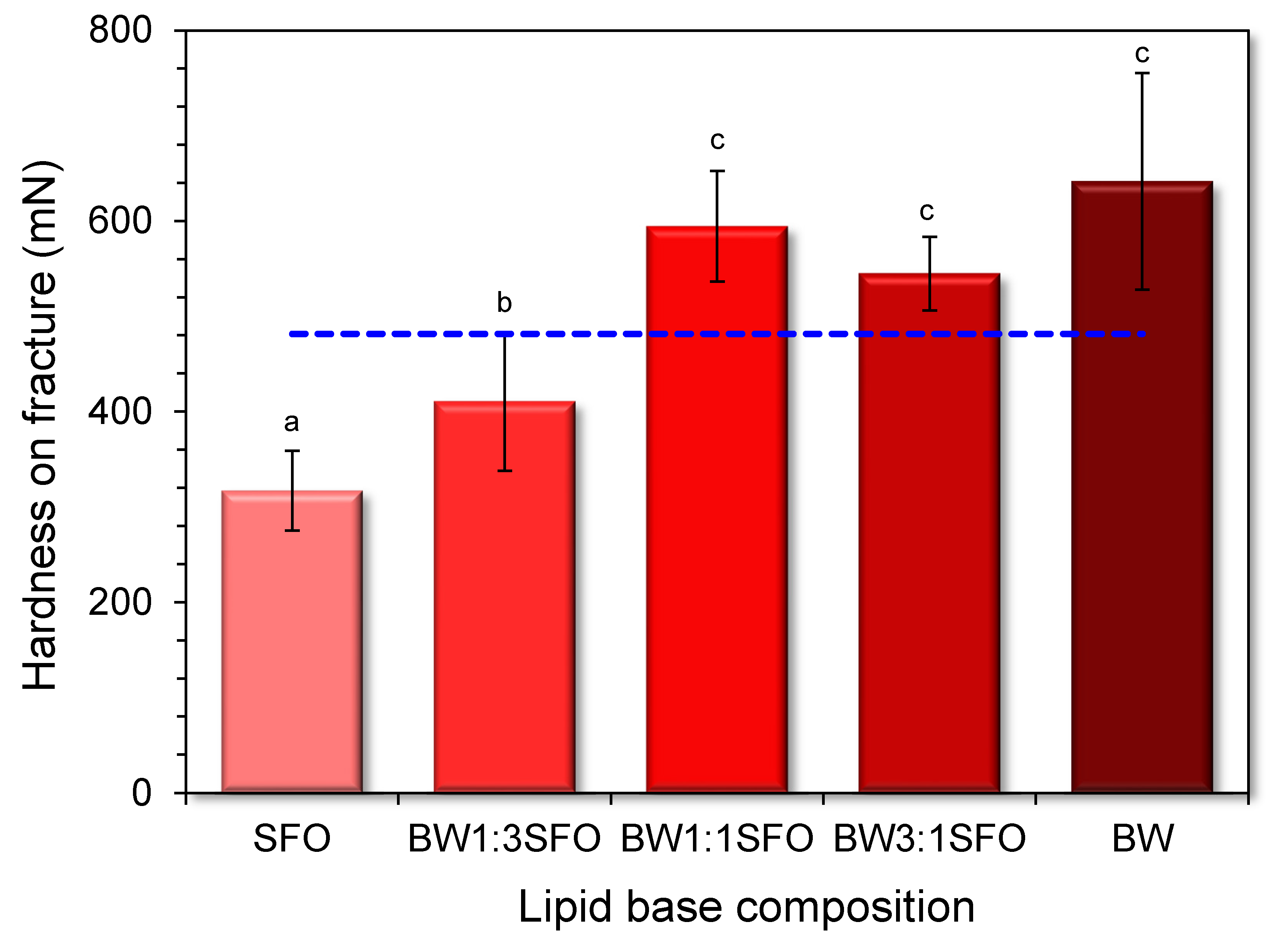
3.4. Instrumental Hardness
4. Conclusions
References
- Arredondo-Ochoa, T.; García-Almendárez, B. E.; Escamilla-García, M.; Martín-Belloso, O.; Rossi-Márquez, G.; Medina-Torres, L.; Regalado-González, C. Physicochemical and Antimicrobial Characterization of Beeswax–Starch Food-Grade Nanoemulsions Incorporating Natural Antimicrobials. International Journal of Molecular Sciences 2017, 18(12), 2712. [Google Scholar] [CrossRef]
- Blake, A. I.; Marangoni, A. G. The Effect of Shear on the Microstructure and Oil Binding Capacity of Wax Crystal Networks. Food Biophysics 2015, 10(4), 403–415. [Google Scholar] [CrossRef]
- Blake, A. I.; Toro-Vasquez, J.; Hwang, H. S. (2018). Wax Oleogels. In Edible Oleogels (pp. 133–171). AOCS Press. [CrossRef]
- Boudara, V. A. H.; Read, D. J.; Ramírez, J. reptate rheology software: Toolkit for the analysis of theories and experiments. Journal of Rheology 2020, 64(3), 709–722. [Google Scholar] [CrossRef]
- Chai, X.; Meng, Z.; Liu, C.; Liu, Y. Structural and mechanical behavior of colloidal fat crystal networks of fully hydrogenated lauric acid-rich fats and rapeseed oils mixtures. Food Chemistry 2019, 288, 108–116. [Google Scholar] [CrossRef]
- Ewoldt, R. H.; Winter, P.; Maxey, J.; McKinley, G. H. Large amplitude oscillatory shear of pseudoplastic and elastoviscoplastic materials. Rheologica Acta 2010, 49(2), 191–212. [Google Scholar] [CrossRef]
- Fan, Z.; Cheng, P.; Zhang, P.; Zhang, G.; Han, J. Rheological insight of polysaccharide/protein based hydrogels in recent food and biomedical fields: A review. International Journal of Biological Macromolecules 2022, 222, 1642–1664. [Google Scholar] [CrossRef] [PubMed]
- de Farias, B. S.; da Cunha, L. B.; Christ Ribeiro, A.; Jaeschke, D. P.; Gonçalves, J. O.; Fernandes, S. S.; Cadaval, T. R. S.; Pinto, L. A. de A. Designing Emulsion Gels for 3D Food Printing: Structure, Stability, and Functional Applications. Surfaces 2025, 8(3), 64. [Google Scholar] [CrossRef]
- Giannakaki, F.; Dimakopoulou-Papazoglou, D.; Zampouni, K.; Moschakis, T.; Katsanidis, E. Design and characterization of bigels composed of agar, κ-carrageenan, candelilla wax, and monoglycerides. International Journal of Biological Macromolecules 2025, 308, 142422. [Google Scholar] [CrossRef] [PubMed]
- Goff, H. D. Instability and Partial Coalescence in Whippable Dairy Emulsions. Journal of Dairy Science 1997, 80(10), 2620–2630. [Google Scholar] [CrossRef]
- Grossi, M.; Fang, B.; Rao, J.; Chen, B. Discovering novel cryo-oleofoams with excellent overrun: Synergistic effect between zein and monoglyceride at interface. Food Hydrocolloids 2024, 150, 109690. [Google Scholar] [CrossRef]
- Guo, Y.; Ma, C.; Xu, Y.; Du, L.; Yang, X. Food Gels Based on Polysaccharide and Protein: Preparation, Formation Mechanisms, and Delivery of Bioactive Substances. Gels 2024, 10(11), 735. [Google Scholar] [CrossRef]
- Hernández-Nolasco, Z.; Acateca-Hernández, M. I.; Rojas-López, M.; Ríos-Corripio, M. A.; Hidalgo-Contreras, J. V.; Hernández-Cázares, A. S. Structural and functional properties of beeswax–taro starch-based bigels: Effect of the oleogel:hydrogel ratio. Food Science and Biotechnology 2025, 34(13), 3099–3108. [Google Scholar] [CrossRef]
- Hyun, K.; Wilhelm, M.; Klein, C. O.; Cho, K. S.; Nam, J. G.; Ahn, K. H.; Lee, S. J.; Ewoldt, R. H.; McKinley, G. H. A review of nonlinear oscillatory shear tests: Analysis and application of large amplitude oscillatory shear (LAOS). Progress in Polymer Science 2011, 36(12), 1697–1753. [Google Scholar] [CrossRef]
- Jana, S.; Martini, S. Physical characterization of crystalline networks formed by binary blends of waxes in soybean oil. Food Research International 2016, 89, 245–253. [Google Scholar] [CrossRef] [PubMed]
- Kamlow, M.-A.; Spyropoulos, F.; Mills, T. 3D printing of kappa-carrageenan emulsion gels. Food Hydrocolloids for Health 2021, 1, 100044. [Google Scholar] [CrossRef]
- Li, B.; Zhou, M.; Qin, H.; Wu, A.; Li, J.; Wang, Y.; Hu, Z. Impact of gelatin/K-carrageenan ratio in the properties of bigels for food applications. Food and Bioproducts Processing 2025, 153, 315–321. [Google Scholar] [CrossRef]
- Lu, Y.; Zhang, Y.; Zhang, R.; Gao, Y.; Miao, S.; Mao, L. Different interfaces for stabilizing liquid–liquid, liquid–gel and gel–gel emulsions: Design, comparison, and challenges. Food Research International 2024, 187, 114435. [Google Scholar] [CrossRef]
- MacDougall, D. B. (2010). Colour measurement of food: Principles and practice. In Colour Measurement (pp. 312–342). Woodhead Publishing. [CrossRef]
- Madsen, J.; Sode, L.; Dahl, J. F.; Corredig, M.; Schulz, H.-J. (2022). Visual Exploration of Rheological Test Results from Soft Materials. Proceedings of the First International Workshop on Visualization in Testing of Hardware, Software, and Manufacturing (TestVis’22), 1–7. [CrossRef]
- Marangoni, A. G. On the use and misuse of the avrami equation in characterization of the kinetics of fat crystallization. Journal of the American Oil Chemists’ Society 1998, 75(10), 1465–1467. [Google Scholar] [CrossRef]
- Mezger, T. (2020). In For users of rotational and oscillatory rheometers. Vincentz Network. [CrossRef]
- Miłek, M.; Drogoń, A.; Pyda, M.; Czerniecka-Kubicka, A.; Tomczyk, M.; Dżugan, M. The use of infrared spectroscopy and thermal analysis for the quick detection of adulterated beeswax. Journal of Apicultural Research 2020, 59(4), 677–684. [Google Scholar] [CrossRef]
- Musollini, S.; Soukoulis, C.; Tolve, R.; You, L.; Zanoni, M.; Aprea, E.; Gasperi, F.; Favati, F. Viscoelastic characterisation of high protein ice cream: Predicting tactile sensory properties via time–concentration superposition and large amplitude oscillatory shear (LAOS) rheology. Food Hydrocolloids 2026, 172, 112028. [Google Scholar] [CrossRef]
- Narine, S. S.; Marangoni, A. G. Mechanical and structural model of fractal networks of fat crystals at low deformations. Physical Review E 1999, 60(6), 6991–7000. [Google Scholar] [CrossRef] [PubMed]
- Owayss, A. A.; Rady, M. M.; Gadallah, F. M. Pigmentation of Some Honeybee (Apis mellifera L.) Products. Fayoum Journal of Agricultural Research & Development 2004, 18(2), 121–132. [Google Scholar]
- Pang, M.; Shi, Z.; Lei, Z.; Ge, Y.; Jiang, S.; Cao, L. Structure and thermal properties of beeswax-based oleogels with different types of vegetable oil. Grasas y Aceites 2020, 71(4), e380–e380. [Google Scholar] [CrossRef]
- Pang, M.; Xu, L.; Ge, Y.; Cheng, J.; Zhang, Z.; Cao, L. Fabrication of beeswax/plant sterol ester-gelatin/whey protein isolate bigels with dual gelation effects as substitutes for traditional solid fats. Food Hydrocolloids 2024, 157, 110458. [Google Scholar] [CrossRef]
- Pang, Z.; Deeth, H.; Sopade, P.; Sharma, R.; Bansal, N. Rheology, texture and microstructure of gelatin gels with and without milk proteins. Food Hydrocolloids 2014, 35, 484–493. [Google Scholar] [CrossRef]
- Parker, N. G.; Povey, M. J. W. Ultrasonic study of the gelation of gelatin: Phase diagram, hysteresis and kinetics. Food Hydrocolloids 2012, 26(1), 99–107. [Google Scholar] [CrossRef]
- Penagos, I. A.; Murillo Moreno, J. S.; Dewettinck, K.; Van Bockstaele, F. Carnauba Wax and Beeswax as Structuring Agents for Water-in-Oleogel Emulsions without Added Emulsifiers. Foods 2023, 12(9), 1850. [Google Scholar] [CrossRef]
- Qiu, R.; Qiu, G.; Zhao, P.; Awais, M.; Fan, B.; Huang, Y.; Tong, L.; Wang, L.; Liu, L.; Wang, F. Regulation of rheological properties of soy protein isolate-beeswax based bigel inks for high-precision 3D printing. Food Hydrocolloids 2024, 153, 110052. [Google Scholar] [CrossRef]
- Ramel, P. R.; Marangoni, A. G. Engineering the microstructure of milk fat by blending binary and ternary mixtures of its fractions. RSC Advances 2016, 6(47), 41189–41194. [Google Scholar] [CrossRef]
- Sala, G.; van Vliet, T.; Cohen Stuart, M. A.; van Aken, G. A.; van de Velde, F. Deformation and fracture of emulsion-filled gels: Effect of oil content and deformation speed. Food Hydrocolloids 2009, 23(5), 1381–1393. [Google Scholar] [CrossRef]
- Shirzad, K.; Viney, C. A critical review on applications of the Avrami equation beyond materials science. Journal of The Royal Society Interface 2023, 20(203), 20230242. [Google Scholar] [CrossRef]
- Shu, J.; McClements, D. J.; Luo, S.; Liu, C.; Ye, J. Advances of biopolymer-based emulsion gels: Fabrication, design, and application. Trends in Food Science & Technology 2025, 165, 105335. [Google Scholar] [CrossRef]
- Soukoulis, C.; Bohn, T. A comprehensive overview on the micro- and nano-technological encapsulation advances for enhancing the chemical stability and bioavailability of carotenoids. Critical Reviews in Food Science and Nutrition 2018, 58(1), 1–36. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, S.; Singh, A. Bigels: A review on formulation, characterization, and application in the food industry. Trends in Food Science & Technology 2025, 165, 105285. [Google Scholar] [CrossRef]
- Svečnjak, L.; Chesson, L. A.; Gallina, A.; Maia, M.; Martinello, M.; Mutinelli, F.; Muz, M. N.; Nunes, F. M.; Saucy, F.; Tipple, B. J.; Wallner, K.; Waś, E.; Waters, T. A. Standard methods for Apis mellifera beeswax research. Journal of Apicultural Research 2019, 58(2), 1–108. [Google Scholar] [CrossRef]
- Tan, X.; Li, L.; Yang, W.; Wu, X.; Teng, F. Deciphering the competitive mechanisms of gelation and phase transition in bigels: From interfacial thermodynamics to precision structural control. Trends in Food Science & Technology 2026, 105693. [Google Scholar] [CrossRef]
- Tian, H.; Wang, K.; Lan, H.; Wang, Y.; Hu, Z.; Zhao, L. Effect of hybrid gelator systems of beeswax-carrageenan-xanthan on rheological properties and printability of litchi inks for 3D food printing. Food Hydrocolloids 2021, 113, 106482. [Google Scholar] [CrossRef]
- van Wetten, I. A.; van Herwaarden, A. W.; Splinter, R.; Boerrigter-Eenling, R.; van Ruth, S. M. Detection of sunflower oil in extra virgin olive oil by fast differential scanning calorimetry. Thermochimica Acta, Chip Calorimetry 2015, 603, 237–243. [Google Scholar] [CrossRef]
- Woszczak, L.; Khachatryan, K.; Krystyjan, M.; Lenart-Boroń, A.; Krzan, M.; Białecka, A.; Khachatryan, G. Chitosan composites with beeswax and olive oil nano-microcapsules physicochemical characterization and antibacterial activity against skin microbiota. Scientific Reports 2025, 15(1), 35927. [Google Scholar] [CrossRef]
- Xiao, J.; Wang, W.; Wang, K.; Liu, Y.; Liu, A.; Zhang, S.; Zhao, Y. Impact of melting point of palm oil on mechanical and water barrier properties of gelatin-palm oil emulsion film. Food Hydrocolloids 2016, 60, 243–251. [Google Scholar] [CrossRef]
- Yang, Z.; Hemar, Y.; Hilliou, L.; Gilbert, E. P.; McGillivray, D. J.; Williams, M. A. K.; Chaieb, S. Nonlinear Behavior of Gelatin Networks Reveals a Hierarchical Structure. Biomacromolecules 2016, 17(2), 590–600. [Google Scholar] [CrossRef]
- You, L.; Dieden, R.; Fleming, Y.; Fortuin, J.; Shaplov, A.; Odelli, D.; Pittois, D.; Ritzoulis, C.; Soukoulis, C. Structure conformational, physicochemical and rheological characterisation of red clover seed (Trifolium pratense L.) galactomannan. Food Hydrocolloids 2025, 168, 111517. [Google Scholar] [CrossRef]
- Zampouni, K.; Mouzakitis, C. K.; Lazaridou, A.; Moschakis, T.; Katsanidis, E. Physicochemical properties and microstructure of bigels formed with gelatin and κ-carrageenan hydrogels and monoglycerides in olive oil oleogels. Food Hydrocolloids 2023, 140, 108636. [Google Scholar] [CrossRef]
- Zhang, N.; Qavi, I.; Araneda, M.; Rumi, S. S.; Abidi, N.; Halder, S.; Tan, G. Z. 3D bioprinting of self-strengthening living materials using cellulose nanofiber-producing bacteria in sodium alginate hydrogel. Bioprinting 2025, 51, e00443. [Google Scholar] [CrossRef]
- Zhou, M.; Li, B.; Wu, A.; Hu, Z.; Liu, J.; Wang, Y.; Liu, H. Preparation of a bigel system based on k-carrageenan hydrogel and beeswax oleogel and the effect of starch on the bigel properties. LWT 2024, 205, 116516. [Google Scholar] [CrossRef]
- Zhou, M.; Li, B.; Wu, A.; Hu, Z.; Zhou, L.; Hu, H. Properties of beeswax–gelatin-based bigels and the influence of hydrogel particle size. LWT 2025, 228, 118087. [Google Scholar] [CrossRef]

| SFO | WAX | |||||
|---|---|---|---|---|---|---|
| Sample | Tm, onset (oC) |
Tm, midpoint (oC) |
ΔH (J g−1) |
Tm, onset (oC) |
Tm, midpoint (oC) |
ΔH (J g−1) |
| SFO1:0BW | −35.9a | −31.0a | 8.85a | nd | nd | nd |
| SFO3:1BW | −25.8b | −20.8b | 2.21b | 39.8a | 57.4a | 40.3a |
| SFO1:1BW | −25.7b | −20.7b | 0.74c | 46.0b | 61.1ab | 86.9b |
| SFO1:3BW | Nd | nd | nd | 49.9b | 62.0ab | 137.2c |
| SFO0:1BW | nd | nd | nd | 48.3b | 64.7b | 215.3d |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




