Submitted:
15 April 2026
Posted:
15 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Nanoparticle Characterization
2.1.1. Silver Nanoparticle (AgNP) Characterization
2.1.2. Titanium Dioxide Nanoparticle (TiO₂NP) Characterization
2.1.3. Carbon Dot Nanoparticle (CD NP) Characterization
2.2. Nanoparticle Preparations
2.3. RAW 264.7 Macrophage Cultures
2.4. WST-1 Cell Proliferation Assay
2.5. Ellman Acetylcholinesterase Analysis
3. Results and Discussion
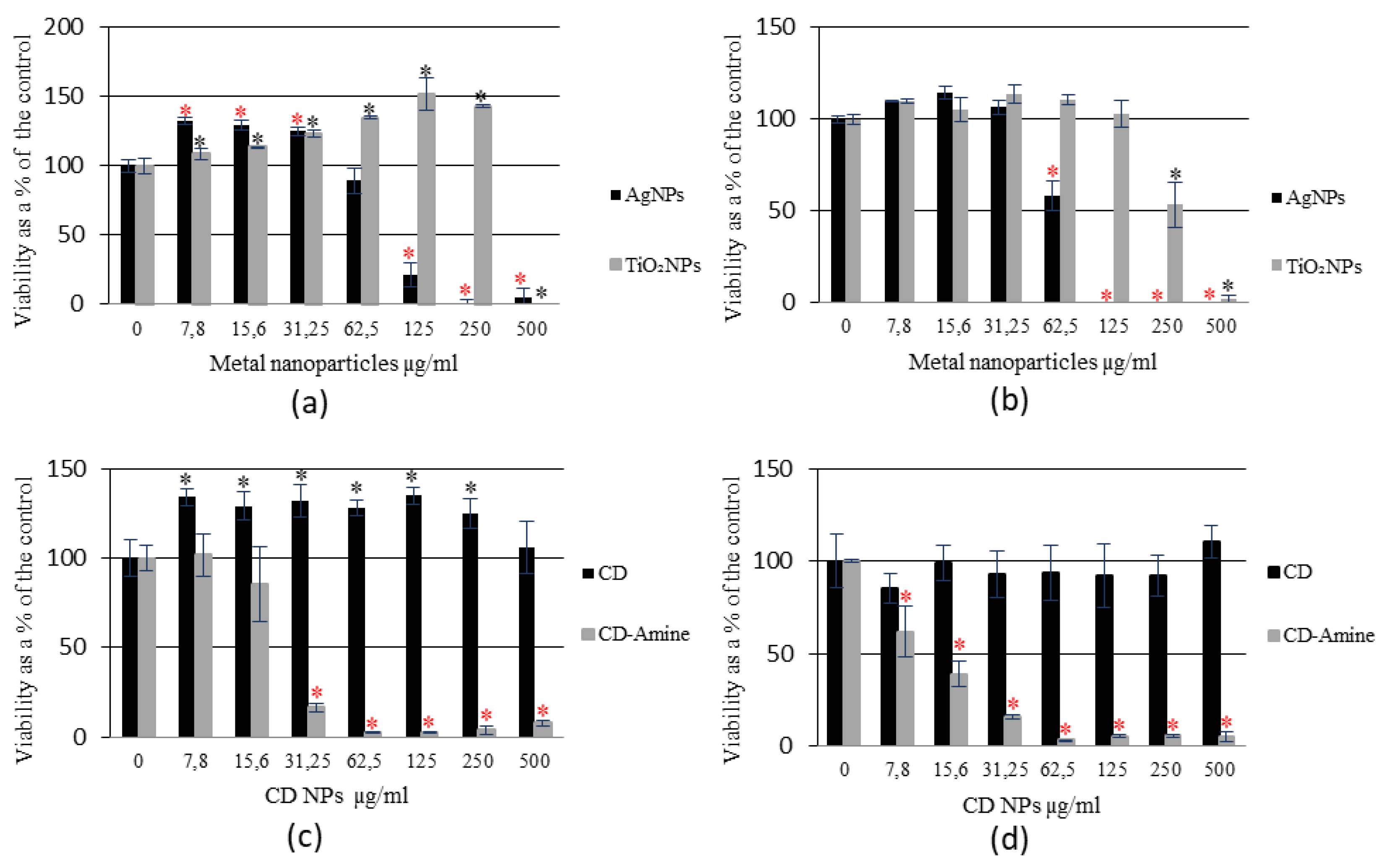
3.1. Assessment of NPs Cytotoxicity on RAW 264.7 Cells
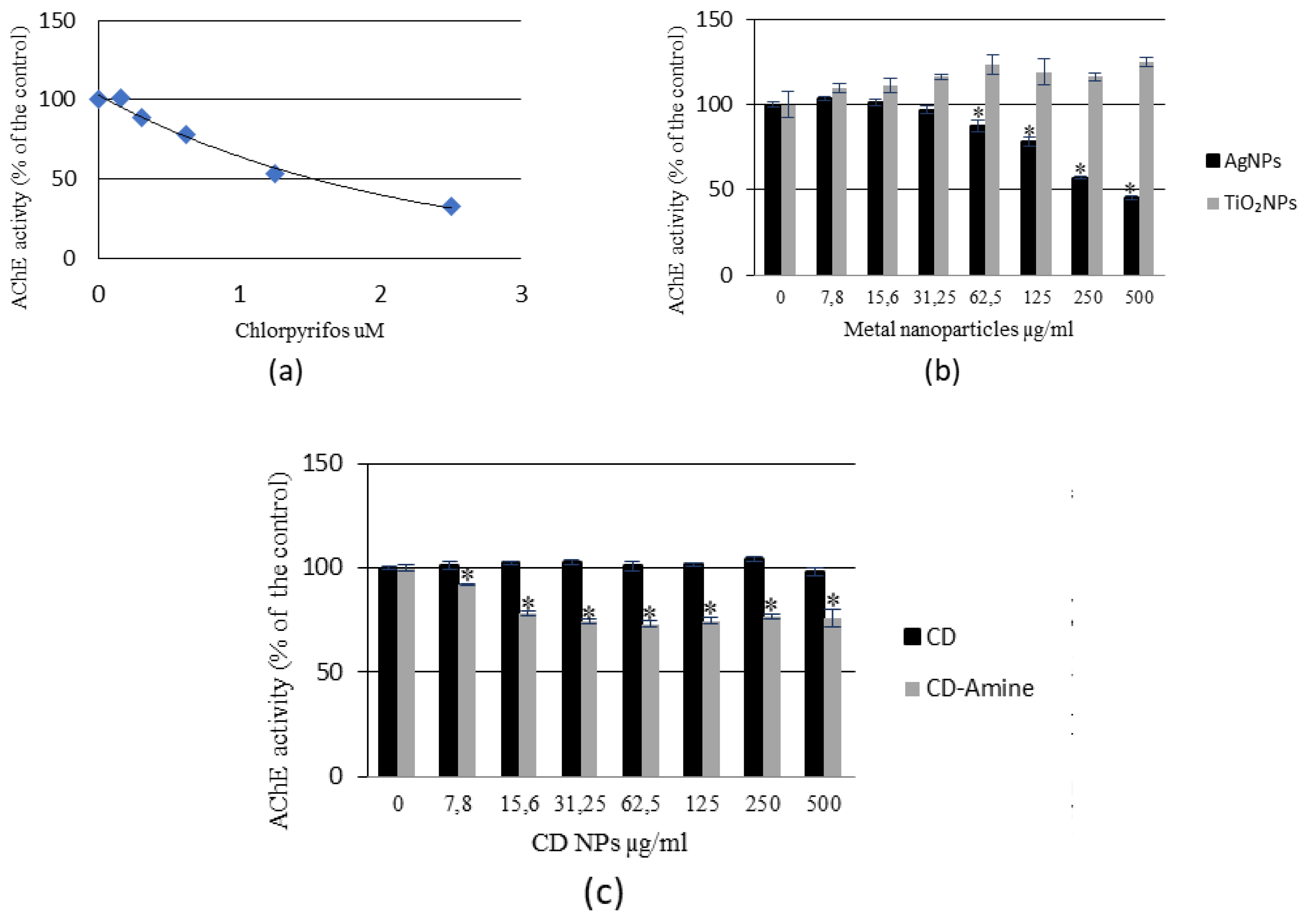
3.2. Acetylcholinesterase Activity Analysis
4. Conclusion
Declarations
Data availability
References
- Aggarwal P, Hall JB, McLeland CB, Dobrovolskaia MA, McNeil SE (2009) Nanoparticle interaction with plasma proteins as it relates to particle biodistribution, biocompatibility and therapeutic efficacy. In Advanced Drug Delivery Reviews 61(6): 428–437. [CrossRef]
- Auría-Soro C, Nesma T, Juanes-Velasco P, Landeira-Viñuela A, Fidalgo-Gomez H, Acebes-Fernandez V, Gongora R, Jesus M, Parra A, Manzano-Roman R, Fuentes M (2019) Interactions of Nanoparticles and biosystems: microenvironment of nanoparticles and biomolecules in nanomedicine. [CrossRef]
- Avinash Chunduri LA, Madhusudhana Rao K, Vijaya Sai K, Kurdekar AD, Prathibha C, Kamisetti V (2020) Influence of Surface Charge on the Functional Properties of Silica Nanoparticles and Cellular Toxicity. Letters in Applied NanoBioScience 9(3): 1225–1238. [CrossRef]
- Barabadi H, Najafi M, Samadian H, Azarnezhad A, Vahidi H, Mahjoub MA, Koohiyan M, Ahmadi A (2019). A systematic review of the genotoxicity and antigenotoxicity of biologically synthesized metallic nanomaterials: Are green nanoparticles safe enough for clinical marketing? In Medicina (Lithuania) (Vol. 55, Issue 8). MDPI AG. [CrossRef]
- Bayati M, Dai J, Zambrana A, Rees C, Fidalgo de Cortalezzi M (2018) Effect of water chemistry on the aggregation and photoluminescence behavior of carbon dots. Journal of Environmental Sciences (China) 65:223–235. [CrossRef]
- Bin-Jumah M, Al-Abdan M, Albasher G, Alarifi S (2020) Effects of green silver nanoparticles on apoptosis and oxidative stress in normal and cancerous human hepatic cells in vitro. International Journal of Nanomedicine 15:1537–1548. [CrossRef]
- Chatonnet A, Lockridgetl O (1989) Comparison of butyrylcholinesterase and acetylcholinesterase. In Biochem. J (Vol. 260).
- Chaturvedi S, Singh AK, Keshari AK, Maity S, Sarkar S, Saha S (2016) Human Metabolic Enzymes Deficiency: A Genetic Mutation Based Approach. In Scientifica (Vol. 2016). Hindawi Limited. [CrossRef]
- Coban A, Carr RL, Chambers HW, Willeford KO, Chambers JE (2016) Comparison of inhibition kinetics of several organophosphates, including some nerve agent surrogates, using human erythrocyte and rat and mouse brain acetylcholinesterase. Toxicology Letters 248:39–45. [CrossRef]
- Čolović MB, Krsti DZ, Lazarevi-Pati TD, Bondi AM, Vasi VM (2013) Acetylcholinesterase Inhibitors: Pharmacology and Toxicology.
- Dai H (2002) Carbon nanotubes: opportunities and challenges. www.elsevier.com/locate/susc.
- David OM, Lategan KL, de Cortalezzi MF, Pool EJ (2022) The Stability and Anti-Angiogenic Properties of Titanium Dioxide Nanoparticles (TiO₂NPs) Using Caco-2 Cells. Biomolecules 12(10). [CrossRef]
- Davis L, Britten JJ, Morgan M (1997) Cholinesterase. Its significance in anaesthetic practice. In Anaesthesia 52(3):244–260, Blackwell Publishing Ltd. [CrossRef]
- Eyer P, Worek F, Kiderlen D, Sinko G, Stuglin A, Simeon-Rudolf V, Reiner E (2003) Molar absorption coefficients for the reduced Ellman reagent. www.elsevier.com/locate/yabio.
- Faul AK, Julies E, Pool EJ (2013) Oestrogen, testosterone, cytotoxin and cholinesterase inhibitor removal during reclamation of sewage to drinking water. Water SA 39(4):499–506. [CrossRef]
- Fischer NO, Mcintosh CM, Simard JM, Rotello VM (2002) Inhibition of chymotrypsin through surface binding using nanoparticle-based receptors. PNAS 99(8):5018–5023. www.pnas.orgcgidoi10.1073pnas.082644099.
- Fröhlich E (2012) The role of surface charge in cellular uptake and cytotoxicity of medical nanoparticles. In International Journal of Nanomedicine 7:5577–5591. [CrossRef]
- Fu PP, Xia Q, Hwang HM, Ray PC, Yu H (2014) Mechanisms of nanotoxicity: Generation of reactive oxygen species. In Journal of Food and Drug Analysis 22(1):64–75. Elsevier Taiwan LLC. [CrossRef]
- Giri K, Shameer K, Zimmermann MT, Saha S, Chakraborty PK, Sharma A, Arvizo RR, Madden BJ, McCormick DJ, Kocher JPA, Bhattacharya R, Mukherjee P (2014) Understanding protein-nanoparticle interaction: A new gateway to disease therapeutics. Bioconjugate Chemistry 25(6):1078–1090. [CrossRef]
- Guillama Barroso G, Narayan M, Alvarado M, Armendariz I, Bernal J, Carabaz X, Chavez S, Cruz P, Escalante V, Estorga S, Fernandez D, Lozano C, Marrufo M, Ahmad N, Negrete S, Olvera K, Parada X, Portillo B, Ramirez A, Viel S (2020) Nanocarriers as Potential Drug Delivery Candidates for Overcoming the Blood-Brain Barrier: Challenges and Possibilities. In ACS Omega 5(22):12583–12595, American Chemical Society. [CrossRef]
- Hebeish A, El-Naggar ME, Fouda MMG, Ramadan MA, Al-Deyab SS, El-Rafie MH (2011) Highly effective antibacterial textiles containing green synthesized silver nanoparticles. Carbohydrate Polymers 86(2):936–940. [CrossRef]
- Hendricks R, Pool EJ (2012) Rapid in vitro tests to determine the toxicity of raw wastewater and treated sewage effuents. Water SA 38(5):807–812. [CrossRef]
- Hersh AM, Alomari S, Tyler BM (2022) Crossing the Blood-Brain Barrier: Advances in Nanoparticle Technology for Drug Delivery in Neuro-Oncology. International Journal of Molecular Sciences 23(8). [CrossRef]
- Huang YW, Cambre M, Lee HJ (2017) The Toxicity of Nanoparticles Depends on Multiple Molecular and Physicochemical Mechanisms. In International journal of molecular sciences 18(12). [CrossRef]
- Inshakova E, Inshakova A, Goncharov A (2020) Engineered nanomaterials for energy sector: Market trends, modern applications and future prospects. IOP Conference Series: Materials Science and Engineering 971(3). [CrossRef]
- Joudeh N, Linke D (2022) Nanoparticle classification, physicochemical properties, characterization, and applications: a comprehensive review for biologists. In Journal of Nanobiotechnology 20(1), BioMed Central Ltd. [CrossRef]
- Karthick S, Karthick S, Namasivayam R, Lazar F, Karthick S, Aroma R, Manikanta M, Gopinath P, Francis AL (2016) Evaluation of enzyme activity inhibition of biogenic silver nanoparticles against microbial extracellular enzymes. https://www.researchgate.net/publication/316286330.
- Khan I, Saeed K, Khan I (2019) Nanoparticles: Properties, applications and toxicities. In Arabian Journal of Chemistry 12(7):908–931. Elsevier B.V. [CrossRef]
- Khan Y, Sadia H, Ali Shah SZ, Khan MN, Shah AA, Ullah N, Ullah MF, Bibi H, Bafakeeh OT, Khedher N, Eldin SM, Fadhl BM, Khan MI (2022) Classification, Synthetic, and Characterization Approaches to Nanoparticles, and Their Applications in Various Fields of Nanotechnology: A Review. In Catalysts 12(11). MDPI. [CrossRef]
- Lategan KL, Alghadi H, Bayati M, de Cortalezzi MF, Pool EJ (2018) Effects of graphene oxide nanoparticles on the immune system biomarkers produced by RAW 264.7 and human whole blood cell cultures. Nanomaterials 8(2). [CrossRef]
- Lategan KL (2017) The in vitro effects of heavy metals and nanoparticles on the immune system. Food and Chemical Toxicology 24(6–7):545–549. [CrossRef]
- Lategan KL, Walters CR, Pool EJ (2019) The in vitro effects of silver nanoparticles on the immune system. In Frontiers In Bioscience (Vol. 24).
- Li Y, Sun Y, Li J, Tang R, Miu Y, Ma X (2021) Research on the influence of microplastics on marine life. IOP Conference Series: Earth and Environmental Science 631(1). [CrossRef]
- Liu W, Worms I, Slaveykova VI (2020) Interaction of silver nanoparticles with antioxidant enzymes. Environmental Science: Nano 7(5):1507–1517. [CrossRef]
- Liu, Y., Pan, B., Li, H., Lang, D., Zhao, Q., Zhang, D., Wu, M., Steinberg, C. E. W., & Xing, B. (2020). Can the properties of engineered nanoparticles be indicative of their functions and effects in plants? Ecotoxicology and Environmental Safety, 205. [CrossRef]
- Murthy SK (2007) Nanoparticles in modern medicine: State of the art and future challenges. In International Journal of Nanomedicine (Vol. 2, Issue 2).
- Myrzakhanova M, Gambardella C, Falugi C, Gatti AM, Tagliafierro G, Ramoino P, Bianchini P, Diaspro A (2013) Effects of nanosilver exposure on cholinesterase activities, CD41, and CDF/LIF-like expression in zebraFish (Danio rerio) larvae. BioMed Research International, 2013. [CrossRef]
- Nagano T, Nagano K, Nabeshi H, Yoshida T, Kamada, H, Tsunoda, S-I, Gao J-Q, Higashisaka K, Yoshioka Y, Tsutsumi Y (2017) Modifying the Surface of Silica Nanoparticles with Amino or Carboxyl Groups Decreases Their Cytotoxicity to Parenchymal Hepatocytes. In Biol. Pharm. Bull 40(5).
- Nandhakumar S, Dhanaraju MD, Sundar VD, Heera B (2017) Influence of surface charge on the in vitro protein adsorption and cell cytotoxicity of paclitaxel loaded poly(ε-caprolactone) nanoparticles. Bulletin of Faculty of Pharmacy, Cairo University 55(2):249–258. [CrossRef]
- Ng KK, Zheng G (2015) Molecular Interactions in Organic Nanoparticles for Phototheranostic Applications. In Chemical Reviews 115(19):11012–11042, American Chemical Society. [CrossRef]
- Ngamwongsatit P, Banada PP, Panbangred W, Bhunia AK (2008) WST-1-based cell cytotoxicity assay as a substitute for MTT-based assay for rapid detection of toxigenic Bacillus species using CHO cell line. Journal of Microbiological Methods 73(3):211–215. [CrossRef]
- Oberle C, Huai J, Reinheckel T, Tacke M, Rassner M, Ekert PG, Buellesbach J, Borner C (2010) Lysosomal membrane permeabilization and cathepsin release is a Bax/Bak-dependent, amplifying event of apoptosis in fibroblasts and monocytes. Cell Death and Differentiation 17(7):1167–1178. [CrossRef]
- Odaudu OR, Akinsiku AA (2022) Toxicity and Cytotoxicity Effects of Selected Nanoparticles: A Review. IOP Conference Series: Earth and Environmental Science 1054(1). [CrossRef]
- Poh TY, Ali NATBM, Mac Aogáin M, Kathawala MH, Setyawati MI, Ng KW, Chotirmall SH (2018) Inhaled nanomaterials and the respiratory microbiome: Clinical, immunological and toxicological perspectives. Particle and Fibre Toxicology 15(1). [CrossRef]
- Rahman, A, Sarkar A, Yadav OP, Achari, G, Slobodnik J (2021) Potential human health risks due to environmental exposure to nano- and microplastics and knowledge gaps: A scoping review. In Science of the Total Environment (Vol. 757), Elsevier B.V. [CrossRef]
- Ray PC, Yu H, Fu PP (2009) Toxicity and environmental risks of nanomaterials: Challenges and future needs. In Journal of Environmental Science and Health - Part C Environmental Carcinogenesis and Ecotoxicology Reviews 27(1):1–35. [CrossRef]
- Ray S (2017) Different Synthesis Process of Carbon Nanomaterials for Biological Applications [Book]. In Carbon Nanomaterials for Biological and Medical Applications. [CrossRef]
- Saptarshi SR, Duschl A, Lopata AL (2013) Interaction of nanoparticles with proteins: Relation to bio-reactivity of the nanoparticle. Journal of Nanobiotechnology 11(1). [CrossRef]
- Sarı C (2019) A comparative study of MTT and WST-1 assays in cytotoxicity analysis. Haydarpasa Numune Training and Research Hospital Medical Journal. [CrossRef]
- Schaeublin NM, Braydich-Stolle LK, Schrand AM, Miller JM, Hutchison J, Schlager JJ, Hussain SM (2011) Surface charge of gold nanoparticles mediates mechanism of toxicity. Nanoscale 3(2):410–420. [CrossRef]
- Schultheiss E, Weiss S, Winterer E, Maas R, Heinzle E, Jose J (2008) Esterase autodisplay: Enzyme engineering and whole-cell activity determination in microplates with pH sensors. Applied and Environmental Microbiology 74(15):4782–4791. [CrossRef]
- Shukla RK, Badiye A, Vajpayee K, Kapoor N (2021) Genotoxic Potential of Nanoparticles: Structural and Functional Modifications in DNA. In Frontiers in Genetics (Vol. 12), Frontiers Media S.A. [CrossRef]
- Singh R (2021) Nanobiotechnology in animal production and health [Book]. In Advances in Animal Genomics. [CrossRef]
- Siqueira PR, Carmo TLL, Bonomo MM, Santos FA, Fernandes MN (2021) Proliferative response avoids mutagenic effects of titanium dioxide (TiO2) nanoparticles in a zebrafish hepatocyte cell line. Journal of Hazardous Materials Advances 4. [CrossRef]
- Smijs TG, Pavel S (2011) Titanium dioxide and zinc oxide nanoparticles in sunscreens: Focus on their safety and effectiveness. In Nanotechnology, Science and Applications 4(1):95–112. Dove Medical Press Ltd. [CrossRef]
- Smith PJ, Giroud M, Wiggins HL, Gower F, Thorley JA, Stolpe B, Mazzolini J, Dyson RJ, Rappoport JZ (2012) Cellular entry of nanoparticles via serum sensitive clathrin-mediated endocytosis, and plasma membrane permeabilization. International Journal of Nanomedicine 7:2045–2055. [CrossRef]
- Spring FA, Gardner B, Anstee DJ (1992) Evidence That the Antigens of the Yt Blood Group System Are Located on Human Erythrocyte Acetylcholinesterase. In Blood 80(8).
- Stensberg MC, Wei Q, Mclamore ES, Marshall D, Wei A, Porterfield DM, Sepulveda MS (2012) Toxicological studies on silver nanoparticles. 6(5):879–898. [CrossRef]
- Sutkovic JJ (2016) A review on Nanoparticle and Protein interaction in biomedical applications. Periodicals of Engineering and Natural Sciences (PEN) 4(2). [CrossRef]
- Teleanu, D. M., Chircov, C., Grumezescu, A. M., Volceanov, A., & Teleanu, R. I. (2018). Blood-brain delivery methods using nanotechnology. In Pharmaceutics (Vol. 10, Issue 4). MDPI AG. [CrossRef]
- Tianlu Zhang, Liming Wang, Qiang Chen (2012) Cytotoxic Potential of Silver Nanoparticles. 55(2):1–7.
- Van Wyk HM, Schoeman D, Kwembeya E, Hans RH, Pool EJ, Louw S (2022) In vitro acetylcholinesterase inhibitory activity and chemical composition of the dichloromethane extracts of the leaves and inflorescences of the tumbleweed, Trachyandra laxa (N.E.Br.) Oberm. var. rigida (Suesseng.) Roessl. South African Journal of Botany 147:231–237. [CrossRef]
- Walters C, Pool EJ, Somerset V (2013) Aggregation and dissolution of silver nanoparticles in a laboratory-based freshwater microcosm under simulated environmental conditions. Toxicological and Environmental Chemistry 95(10):1690–1701. [CrossRef]
- Wang F, Salvati A, Boya P (2018) Lysosome-dependent cell death and deregulated autophagy induced by amine-modified polystyrene nanoparticles. Open Biology 8(8). [CrossRef]
- Wang M, Li J, Zhang S, You Y, Zhu X, Xiang H, Yan L, Zhao F, Li Y (2022) Effects of Titanium Dioxide Nanoparticles on Cell Growth and Migration of A549 Cells under Simulated Microgravity. Nanomaterials 12(11). [CrossRef]
- Wang Y, Zhou B, Chen H, Yuan R, Wang F (2022) Distribution, biological effects and biofilms of microplastics in freshwater systems - A review. In Chemosphere (Vol. 299), Elsevier Ltd. [CrossRef]
- Widmaier EP, Raff H, Strang KT (2001) Vanders Human physiology: The mechanisms of body function. In Paper Knowledge. Toward a Media History of Documents 8th edn, the McGraw-Hill Companies.
- Wu Z, Zhang B, Yan B (2009) Regulation of enzyme activity through interactions with nanoparticles. In International Journal of Molecular Sciences 10(10):4198–4209. [CrossRef]
- Xiong P, Huang X, Ye N, Lu Q, Zhang G, Peng S, Wang H, Liu Y (2022) Cytotoxicity of Metal-Based Nanoparticles: From Mechanisms and Methods of Evaluation to Pathological Manifestations. In Advanced Science 9(16), John Wiley and Sons Inc. [CrossRef]
- Yang W, Wang L, Mettenbrink EM, Deangelis PL, Wilhelm S (2021) Nanoparticle Toxicology. Annu. Rev. Pharmacol. Toxicol. 61:269–289. [CrossRef]
| Nanoparticles | Sonication period (min) |
|---|---|
| Metal-based Nanoparticles | |
| Silver (AgNP) | 5 |
|
Titanium dioxide (TiO₂NP) |
15 |
| Carbon-based Nanoparticles | |
| Carbon dot (CD) | 5 |
| Amine-modified Carbon dot (CD-Amine) | 20 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




