Submitted:
14 April 2026
Posted:
15 April 2026
You are already at the latest version
Abstract
Keywords:
Introduction
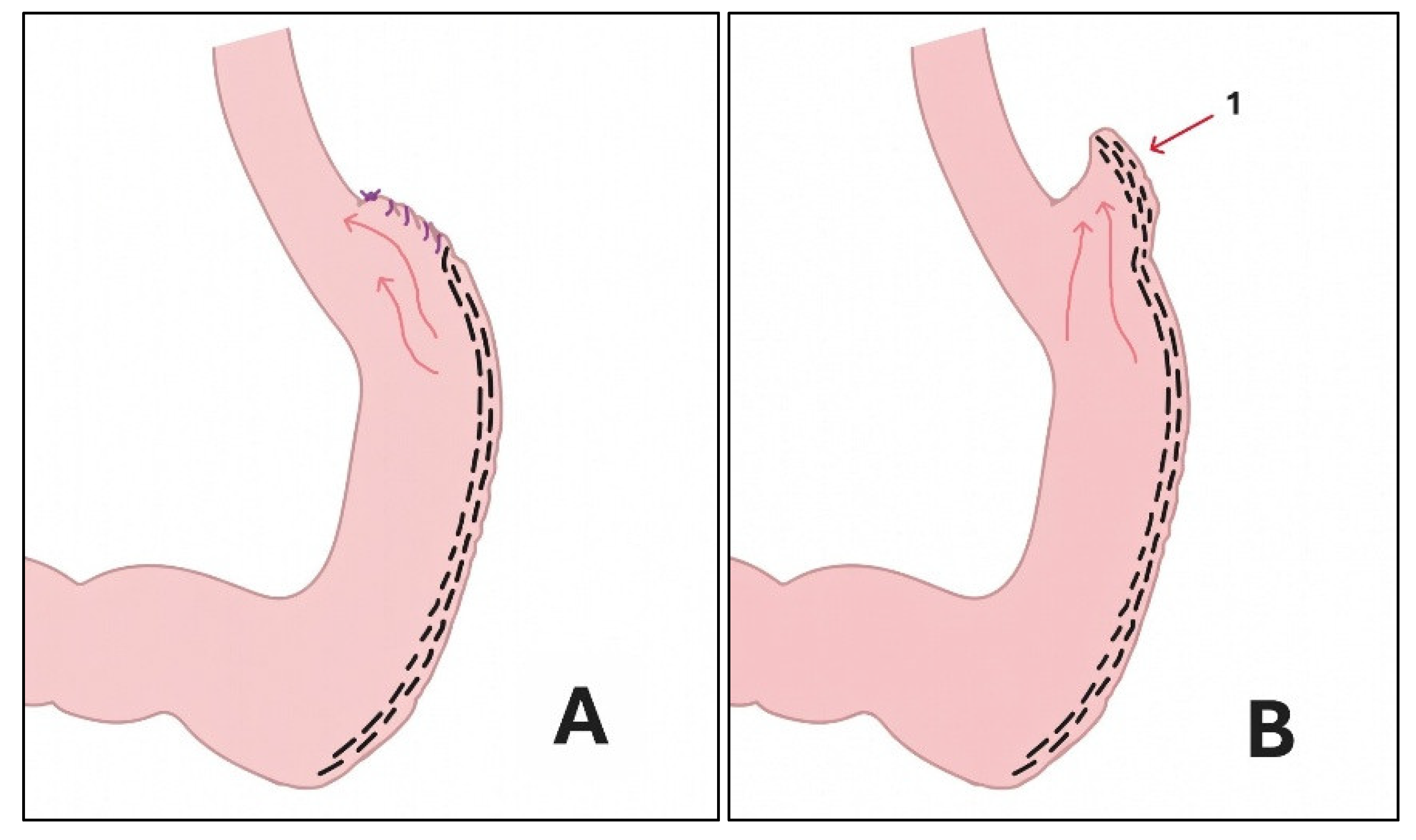
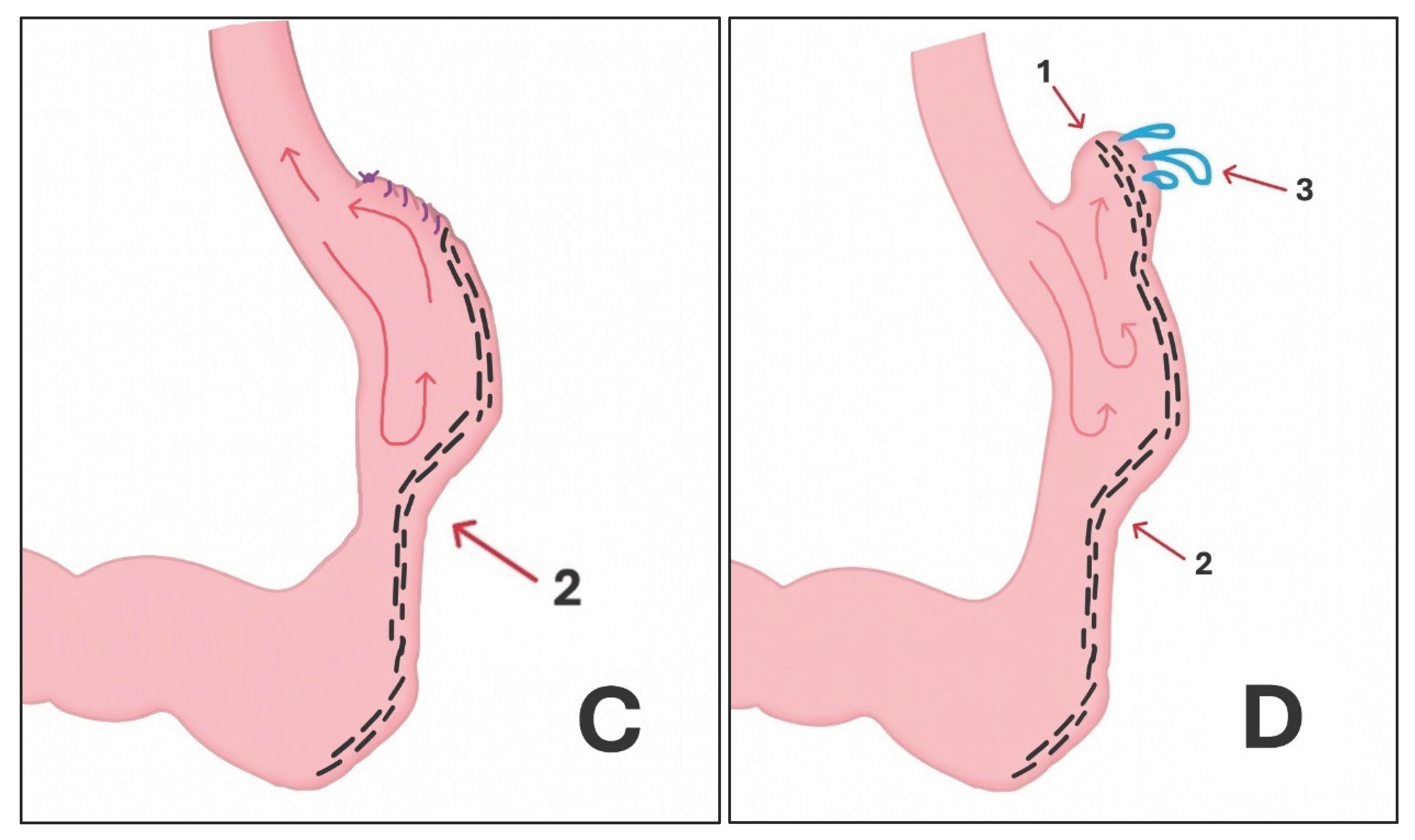
The Proposed Leak Formula: Leakage = Obstruction & "Dog Ear"
Discussion
Staple Line Ischemia
The Stronger the Anti-Reflux Mechanism, the Higher the Leak Risk
Timing of Leak Onset
Practical Conclusions and Technical Recommendations
Conclusion
References
- Galloro, G.; Giglio, M.C.; Chini, A.; Maione, R.; Pollastro, M.; Vitale, R.; et al. Endoscopic treatment of staple-line leaks after sleeve gastrectomy in patients with obesity: Which one is the best option, if any? A systematic review with meta-analysis and meta-regression. Obes Surg. 2025, 35(12), 5496–5514. [Google Scholar] [CrossRef] [PubMed]
- Hughes, D.; Hughes, I.; Khanna, A. Management of staple line leaks following sleeve gastrectomy — a systematic review. Obes Surg. 2019, 29(9), 2759–2772. [Google Scholar] [CrossRef] [PubMed]
- Iossa, A.; Martini, L.; De Angelis, F.; et al. Leaks after laparoscopic sleeve gastrectomy: 2024 update on risk factors. Langenbecks Arch Surg. 2024, 409, 249. [Google Scholar] [CrossRef] [PubMed]
- Verras, G.I.; Mulita, F.; Lampropoulos, C.; Kehagias, D.; Curwen, O.; Antzoulas, A.; et al. Risk factors and management approaches for staple line leaks following sleeve gastrectomy: A single-center retrospective study of 402 patients. J Pers Med. 2023, 13(9), 1422. [Google Scholar] [CrossRef]
- Frattini, F.; Delpini, R.; Inversini, D.; Pappalardo, V.; Rausei, S.; Carcano, G. Gastric leaks after sleeve gastrectomy: Focus on pathogenetic factors. Surg Technol Int. 2017, 31, 123–126. [Google Scholar] [PubMed]
- Niaz, O.; Askari, A.; Currie, A.; McGlone, E.R.; Zakeri, R.; Khan, O.; et al. Analysis of the effect of staple line reinforcement on leaking and bleeding after sleeve gastrectomy from the UK National Bariatric Surgery Registry. World J Surg. 2024, 48, 1950–1957. [Google Scholar] [CrossRef]
- Russo, M.F.; Castagneto-Gissey, L.; Illuminati, G.; D’Andrea, V.; Genco, A.; Casella, G. Leaks after sleeve gastrectomy: Is it still an issue? — single-center experience and systematic literature review. Ann Laparosc Endosc Surg. 2025, 10, 1. [Google Scholar] [CrossRef]
- Li, M.; Zeng, N.; Liu, Y.; Sun, X.; Yang, W.; Liu, Y.; et al. GC-MBD Study Group. Management and outcomes of gastric leak after sleeve gastrectomy: Results from the 2010–2020 national registry. Chin Med J. 2023, 136(16), 1967–1976. [Google Scholar] [CrossRef]
- Kim, J.; Azagury, D.; Eisenberg, D.; DeMaria, E.; Campos, G.M.; ASMBS Clinical Issues Committee. ASMBS position statement on prevention, detection, and treatment of gastrointestinal leak after gastric bypass and sleeve gastrectomy, including the roles of imaging, surgical exploration, and nonoperative management. Surg Obes Relat Dis. 2015, 11(4), 739–748. [Google Scholar] [CrossRef]
- Gagner, M.; Buchwald, J.N. Comparison of laparoscopic sleeve gastrectomy leak rates in four staple-line reinforcement options: A systematic review. Surg Obes Relat Dis. 2014, 10(4), 713–723. [Google Scholar] [CrossRef]
- Caiazzo, R.; Marciniak, C.; Wallach, N.; Devienne, M.; Baud, G.; Cazauran, J.B.; et al. Malignant leakage after sleeve gastrectomy: Endoscopic and surgical approach. Obes Surg. 2020, 30(11), 4459–4466. [Google Scholar] [CrossRef] [PubMed]
- Sillén, L.; Andersson, E.; Olbers, T.; Edholm, D. Obstruction after sleeve gastrectomy, prevalence, and interventions: A cohort study of 9,726 patients from the Scandinavian Obesity Surgery Registry (SOReg). Obes Surg. 2021, 31(11), 4701–4707. [Google Scholar] [CrossRef] [PubMed]
- Gipe, J.; Agathis, A.Z.; Nguyen, S.Q. Managing leaks and fistulas after laparoscopic sleeve gastrectomy: Challenges and solutions. Clin Exp Gastroenterol. 2025, 18, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Dang, J.; Chaivanijchaya, K.; Farah, A.; Kroh, M. Endoscopic management of complications after sleeve gastrectomy: A narrative review. Mini-invasive Surg. 2024, 8, 18. [Google Scholar] [CrossRef]
- Catchlove, W.; Liao, S.; Lim, G.; Brown, W.; Burton, P. Mechanism of staple line leak after sleeve gastrectomy via isobaric pressurisation concentrating stress forces at the proximal staple line. Obes Surg. 2022, 32(8), 2525–2536. [Google Scholar] [CrossRef]
- Negm, S.; Mousa, B.; Shafiq, A.; Abozaid, M.; Allah, E.A.; Attia, A.; et al. Endoscopic management of refractory leak and gastro-cutaneous fistula after laparoscopic sleeve gastrectomy: A randomized controlled trial. Surg Endosc. 2023, 37(3), 2173–2181. [Google Scholar] [CrossRef]
- Aurora, A.R.; Khaitan, L.; Saber, A.A. Sleeve gastrectomy and the risk of leak: A systematic analysis of 4,888 patients. Surg Endosc. 2012, 26(6), 1509–1515. [Google Scholar] [CrossRef]
- Di Furia, M.; Romano, L.; Salvatorelli, A.; Brandolin, D.; Lomanto, D.; Cianca, G.; et al. Indocyanine green fluorescent angiography during laparoscopic sleeve gastrectomy: Preliminary results. Obes Surg. 2019, 29(12), 3786–3790. [Google Scholar] [CrossRef]
- Pavone, G.; Fersini, A.; Pacilli, M.; De Fazio, M.; Panzera, P.; Ambrosi, A.; et al. Can indocyanine green during laparoscopic sleeve gastrectomy be considered a new intraoperative modality for leak testing? BMC Surg. 2022, 22(1), 341. [Google Scholar] [CrossRef]
- Natoudi, M.; Theodorou, D.; Papaloos, A.; Drymousis, P.; Alevizos, L.; Katsaragakis, S.; et al. Does tissue ischemia actually contribute to leak after sleeve gastrectomy? An experimental study. Obes Surg. 2014, 24(5), 675–683. [Google Scholar] [CrossRef]
- Smith, M.D.; Adeniji, A.; Wahed, A.S.; Patterson, E.; Chapman, W.; Courcoulas, A.P.; et al. Technical factors associated with anastomotic leak after Roux-en-Y gastric bypass. Surg Obes Relat Dis. 2015, 11(2), 313–320. [Google Scholar] [CrossRef] [PubMed]
- Mocanu, V.; Dang, J.; Ladak, F.; Switzer, N.; Birch, D.W.; Karmali, S. Predictors and outcomes of leak after Roux-en-Y gastric bypass: An analysis of the MBSAQIP data registry. Surg Obes Relat Dis. 2019, 15(3), 396–403. [Google Scholar] [CrossRef] [PubMed]
- Jacobsen, H.J.; Nergard, B.J.; Leifsson, B.G.; Frederiksen, S.G.; Agajahni, E.; Ekelund, M.; et al. Management of suspected anastomotic leak after bariatric laparoscopic Roux-en-Y gastric bypass. Br J Surg. 2014, 101(4), 417–423. [Google Scholar] [CrossRef] [PubMed]
- Sakran, N.; Goitein, D.; Raziel, A.; Keidar, A.; Beglaibter, N.; Grinbaum, R.; et al. Gastric leaks after sleeve gastrectomy: A multicenter experience with 2,834 patients. Surg Endosc. 2013, 27(1), 240–245. [Google Scholar] [CrossRef]
- Parmer, M.; Wang, Y.H.W.; Hersh, E.H.; Zhang, L.; Chin, E.; Nguyen, S.Q. Management of staple line leaks after laparoscopic sleeve gastrectomy. JSLS 2022, 26(3), e2022.00029. [Google Scholar] [CrossRef]
- Rebibo, L.; Tricot, M.; Dembinski, J.; Dhahri, A.; Brazier, F.; Regimbeau, J.M. Gastric leak after sleeve gastrectomy: Risk factors for poor evolution under conservative management. Surg Obes Relat Dis. 2021, 17(5), 947–955. [Google Scholar] [CrossRef]
- Montuori, M.; Benavoli, D.; D’Ugo, S.; Di Benedetto, L.; Bianciardi, E.; Gaspari, A.L.; et al. Integrated approaches for the management of staple line leaks following sleeve gastrectomy. J Obes. 2017, 2017, 4703236. [Google Scholar] [CrossRef]
- Park, S.; Kim, S.; Kim, S.; Shin, A.R.; Park, Y. Nutritional intervention for a patient with sleeve gastrectomy. Clin Nutr Res. 2023, 12(3), 177–183. [Google Scholar] [CrossRef]
- Frias-Toral, E.; Chapela, S.; Gonzalez, V.; Martinuzzi, A.; Locatelli, J.; Llobera, N.; et al. Optimizing nutritional management before and after bariatric surgery: A comprehensive guide for sustained weight loss and metabolic health. Nutrients 2025, 17(4), 688. [Google Scholar] [CrossRef]
- Bettini, S.; Belligoli, A.; Fabris, R.; Busetto, L. Diet approach before and after bariatric surgery. Rev Endocr Metab Disord. 2020, 21(3), 297–306. [Google Scholar] [CrossRef]
- Sista, F.; Abruzzese, V.; Clementi, M.; Carandina, S.; Cecilia, M.; Amicucci, G. The effect of sleeve gastrectomy on GLP-1 secretion and gastric emptying: A prospective study. Surg Obes Relat Dis. 2017, 13(1), 7–14. [Google Scholar] [CrossRef]
- Wickremasinghe, A.C.; Johari, Y.; Laurie, C.; Shaw, K.; Playfair, J.; Beech, P.; et al. Delayed gastric emptying after sleeve gastrectomy is associated with poor weight loss. Obes Surg. 2022, 32(12), 3922–3931. [Google Scholar] [CrossRef]
- Sethi, M.; Zagzag, J.; Patel, K.; Magrath, M.; Somoza, E.; Parikh, M.S.; et al. Intraoperative leak testing has no correlation with leak after laparoscopic sleeve gastrectomy. Surg Endosc. 2016, 30(3), 883–891. [Google Scholar] [CrossRef]
- Nimeri, A.; Maasher, A.; Salim, E.; et al. The use of intraoperative endoscopy may decrease postoperative stenosis in laparoscopic sleeve gastrectomy. Obes Surg. 2016, 26(6), 1398–1401. [Google Scholar] [CrossRef]
- Kockerling, F.; Schug-Pass, C. Gastroscopically controlled laparoscopic sleeve gastrectomy. Obes Facts 2009, 2, 15–18. [Google Scholar] [CrossRef]
- Chen, I.S.; Tsai, M.S.; Chen, J.H.; Chen, C.Y.; Chen, I.L.; Tai, C.M. The utility of intraoperative endoscopy to assist novice surgeons in the detection of gastric stenosis during laparoscopic sleeve gastrectomy. BMC Surg. 2022, 22(1), 323. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.



