Submitted:
14 April 2026
Posted:
15 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
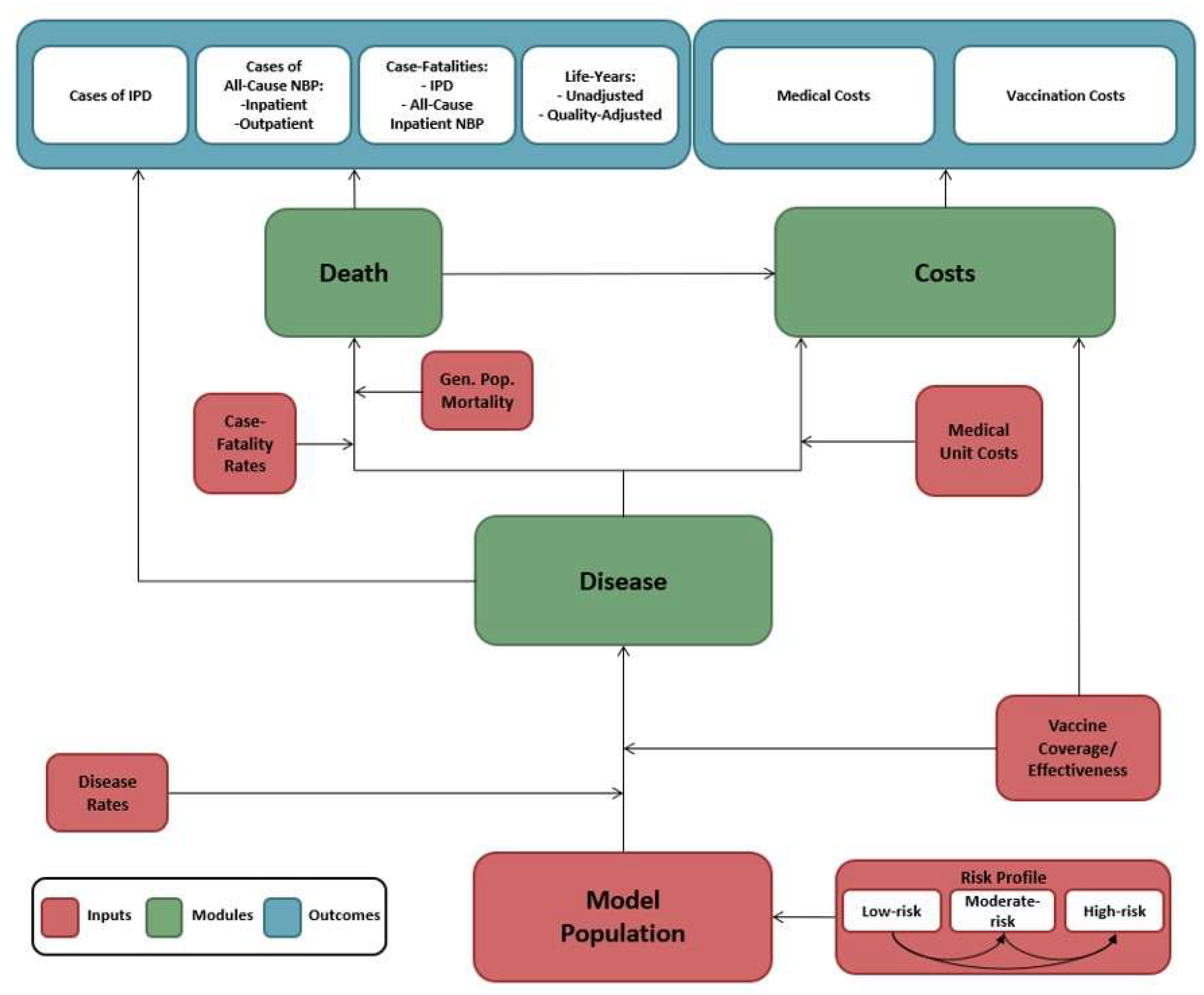
2.1. Model Description
2.2. Inputs Estimation
2.3. Cost-Effectiveness Analyses
2.4. Budget Impact Analyses
3. Results
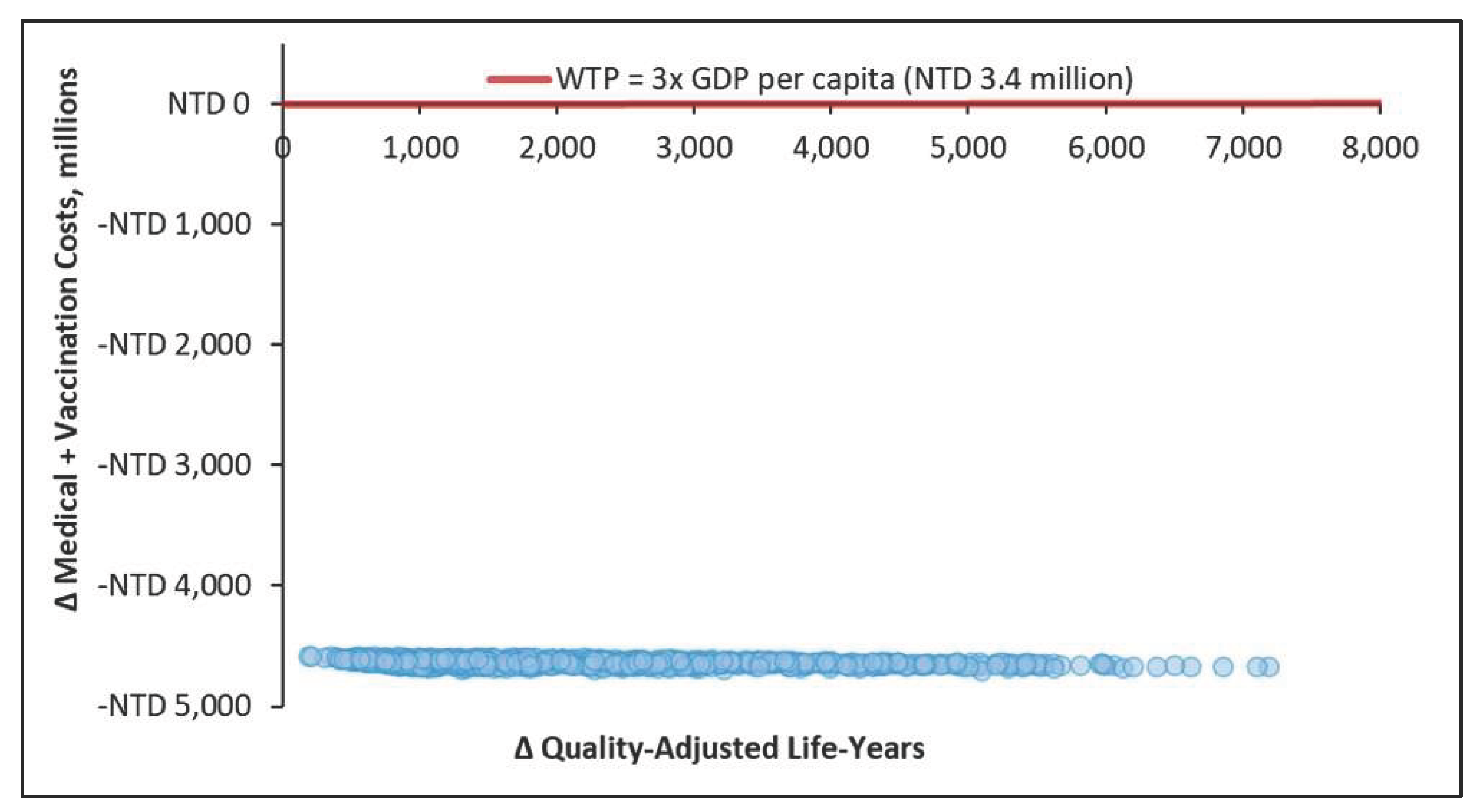
3.1. Cost-Effectiveness Analysis
3.2. Budget Impact Analysis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Ethics approval and consent to participate
Data Availability Statement
Acknowledgements
Conflicts of Interest
Abbreviations
| BIA | Budget impact analysis |
| BIM | Budget impact model |
| CAP | Community-acquired pneumonia |
| CAPiTA | Community-Acquired Pneumonia Immunization Trial in Adults |
| CDC | Centers for Disease Control |
| CEA | Cost-effectiveness analysis |
| CEM | Cost-effectiveness model |
| CFR | Case fatality rate |
| GDP | Gross domestic product |
| ICER | Incremental cost-effectiveness ratio |
| IPD | Invasive pneumococcal disease |
| LY | Life year |
| NBP | Non-bacteremic pneumonia |
| NT$/NTD | New Taiwan dollar |
| PCV13 | 13-valent pneumococcal conjugate vaccine |
| PCV15 | 15-valent pneumococcal conjugate vaccine |
| PCV20 | 20-valent pneumococcal conjugate vaccine |
| PPV23 | 23-valent pneumococcal polysaccharide vaccine |
| PSA | Probabilistic sensitivity analysis |
| QALY | Quality-adjusted life year |
| RR | Relative rate |
| VE | Vaccine effectiveness |
| VT | Vaccine-type |
| WTP | Willingness-to-pay |
References
- Maeda, H.; Morimoto, K. Global distribution and characteristics of pneumococcal serotypes in adults. Hum Vaccin Immunother From NLM. 2025, 21(1), 2469424. [Google Scholar] [CrossRef]
- Varon, E.; Mainardi, J. L.; Gutmann, L. Streptococcus pneumoniae: still a major pathogen. Clin Microbiol Infect 2010, 16(5), 401. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Fang, Y.; Dong, W.; Lau, M.; Mo, P. K. H. Illness representations on pneumonia and pneumococcal vaccination uptake among community-living Chinese people with high-risk conditions aged >/=65 years --- a population-based study. Hum Vaccin Immunother 2021, 17(5), 1455–1462. [Google Scholar] [CrossRef] [PubMed]
- Said, M. A.; Johnson, H. L.; Nonyane, B. A.; Deloria-Knoll, M.; O'Brien, K. L.; Team, A. A. P. B. S.; Andreo, F.; Beovic, B.; Blanco, S.; Boersma, W. G.; et al. Estimating the burden of pneumococcal pneumonia among adults: a systematic review and meta-analysis of diagnostic techniques. PLoS One 2013, 8(4), e60273. [Google Scholar] [CrossRef] [PubMed]
- Lansbury, L.; Lim, B.; McKeever, T. M.; Lawrence, H.; Lim, W. S. Non-invasive pneumococcal pneumonia due to vaccine serotypes: A systematic review and meta-analysis. EClinicalMedicine 2022, 44, 101271. [Google Scholar] [CrossRef]
- Lauderdale, T. L.; Chang, F. Y.; Ben, R. J.; Yin, H. C.; Ni, Y. H.; Tsai, J. W.; Cheng, S. H.; Wang, J. T.; Liu, Y. C.; Cheng, Y. W.; et al. Etiology of community acquired pneumonia among adult patients requiring hospitalization in Taiwan. Respir Med From NLM. 2005, 99(9), 1079–1086. [Google Scholar] [CrossRef] [PubMed]
- Taiwan Centers for Disease Control (CDC). Invasive Pneumococcal Disease. 2017. Available online: https://www.cdc.gov.tw/En/Category/ListContent/bg0g_VU_Ysrgkes_KRUDgQ?uaid=lS42udX_s0u2fN0qLcdrnw (accessed on 2 May 2025).
- The Advisory Committee on Immunization Practice (ACIP). Recommendations on pneumococcal vaccination for adults. Taiwan Centers for Disease Control. Ministry of Health and Welfare. 2015. Available online: https://www.cdc.gov.tw/File/Get/RZNFvB_786v_kT6tQ2HTBQ (accessed on 2 May 2025).
- Taiwan Centers for Disease Control (CDC). Adult Publicly Funded Pneumococcal Vaccination Program. Jan 2026 version. 2026. Available online: https://www.cdc.gov.tw/Category/Page/U_hY1WCfa5BU6fuqW9FIXg (accessed on 15 February 2026).
- Taiwan Centers for Disease Control (CDC). Minutes of the 3rd Advisory Committee on Immunization Practices (ACIP) Meeting. September 2, 2025. 2026. Available online: https://www.cdc.gov.tw/Category/MPage/FWEo643r7uqDO3-xM-zQ_g (accessed on 27 January 2026).
- Shapiro, E. D.; Berg, A. T.; Austrian, R.; Schroeder, D.; Parcells, V.; Margolis, A.; Adair, R. K.; Clemens, J. D. The protective efficacy of polyvalent pneumococcal polysaccharide vaccine. N Engl J Med 1991, 325(21), 1453–1460. [Google Scholar] [CrossRef]
- Taiwan Centers for Disease Control (CDC). Research on serotype and antigenicity surveillance of vaccine-preventable bacterial pathogens; 2018. [Google Scholar]
- Taiwan Centers for Disease Control (CDC). Invasive Streptococcus pneumoniae infection: Disease introduction. 2018. Available online: https://www.cdc.gov.tw/Category/Page/MEYvHLbHiWOcLfQKKF6dpw (accessed on 26 August 2024).
- Essink, B.; Sabharwal, C.; Cannon, K.; Frenck, R.; Lal, H.; Xu, X.; Sundaraiyer, V.; Peng, Y.; Moyer, L.; Pride, M. W.; et al. Pivotal Phase 3 Randomized Clinical Trial of the Safety, Tolerability, and Immunogenicity of 20-Valent Pneumococcal Conjugate Vaccine in Adults Aged ≥18 Years. Clin Infect Dis 2022, 75(3), 390–398. [Google Scholar] [CrossRef] [PubMed]
- Taiwan Food and Drug Administration. Taiwan Food and Drug Administration Assessment Report: Prevenar 20, Pneumococcal 20-valent Conjugate Vaccine (20vPnC). 2024. Available online: https://www.fda.gov.tw/tc/includes/GetFile.ashx?id=f638747767069838018&type=2&cid=48283 (accessed on 9 June 2025).
- Klein, N. P.; Peyrani, P.; Yacisin, K.; Caldwell, N.; Xu, X.; Scully, I. L.; Scott, D. A.; Jansen, K. U.; Gruber, W. C.; Watson, W. A phase 3, randomized, double-blind study to evaluate the immunogenicity and safety of 3 lots of 20-valent pneumococcal conjugate vaccine in pneumococcal vaccine-naive adults 18 through 49 years of age. Vaccine From NLM. 2021, 39(38), 5428–5435. [Google Scholar] [CrossRef] [PubMed]
- Cannon, K.; Elder, C.; Young, M.; Scott, D. A.; Scully, I. L.; Baugher, G.; Peng, Y.; Jansen, K. U.; Gruber, W. C.; Watson, W. A trial to evaluate the safety and immunogenicity of a 20-valent pneumococcal conjugate vaccine in populations of adults ≥65 years of age with different prior pneumococcal vaccination. Vaccine;PubMed 2021, 39(51), 7494–7502. [Google Scholar] [CrossRef]
- U.S. Food and Drug Administration. PCV13 Package Insert. 2019. 2019. Available online: https://www.fda.gov/vaccines-blood-biologics/vaccines/prevnar-13 (accessed on 2 May 2025).
- U.S. Food and Drug Administration. PCV15 Package Insert. 2021. Available online: https://www.fda.gov/vaccines-blood-biologics/vaccines/vaxneuvance (accessed on 2 May 2025).
- U.S. Food and Drug Administration. PCV20 Package Insert. 2021. Available online: https://www.fda.gov/vaccines-blood-biologics/vaccines/pneumovax-23-pneumococcal-vaccine-polyvalent (accessed on 2 May 2025).
- Pfizer Inc. Pfizer Announces Serotypes Included in 20-Valent Pneumococcal Conjugate Vaccine Candidate Being Investigated for the Prevention of Invasive Disease and Pneumonia in Adults Aged 18 Years and Older. 2019.
- Shah, A. A. Simplifying Pneumococcal Immunizations for Adults. Am Fam Physician 2022, 105(6), 580–581. [Google Scholar] [PubMed]
- Clutterbuck, E. A.; Lazarus, R.; Yu, L.-M.; Bowman, J.; Bateman, E. A. L.; Diggle, L.; Angus, B.; Peto, T. E.; Beverley, P. C.; Mant, D.; et al. Pneumococcal Conjugate and Plain Polysaccharide Vaccines Have Divergent Effects on Antigen-Specific B Cells. The Journal of Infectious Diseases 2012, 205(9), 1408–1416. [Google Scholar] [CrossRef]
- Taiwan Centers for Disease Control (CDC). Research on serotype and antigenicity surveillance of vaccine-preventable bacterial pathogens; 2019. [Google Scholar]
- National Statistics Republic of China (Taiwan). Total Population (end of year, persons). 2025. 2025. Available online: https://eng.stat.gov.tw/Point.aspx?sid=t.9&n=4208&sms=11713 (accessed on 12 May 2025).
- Population Pyramids. Population Pyramid of Taiwan at 2025. 2025. Available online: https://population-pyramid.net/en/pp/taiwan (accessed on 12 May 2025).
- Hu, R. H.; Hsiao, F. Y.; Chen, L. J.; Huang, P. T.; Hsu, W. W. Increasing age- and gender-specific burden and complexity of multimorbidity in Taiwan, 2003-2013: a cross-sectional study based on nationwide claims data From NLM. BMJ Open 2019, 9(6), e028333. [Google Scholar] [CrossRef]
- Huang, S. T.; Huang, Y. C.; Kuo, E.; Yang, Y. M.; Hsiao, F. Y. Impacts of Catch-Up Immunization program with the 13-Valent pneumococcal Conjugate vaccine in Taiwan: Focus on age-stratified differences and high-risk population (2001-2015). Vaccine 2022, 40(43), 6225–6234. [Google Scholar] [CrossRef]
- Lu, C. Y.; Chung, C. H.; Huang, L. M.; Kruger, E.; Tan, S. C.; Zhang, X. H.; Chiu, N. C. Cost-effectiveness evaluation of the 10-valent pneumococcal non-typeable Haemophilus influenzae protein D conjugate vaccine for children in Taiwan. Cost Eff Resour Alloc 2020, 18, 30. [Google Scholar] [CrossRef]
- Pelton, S. I.; Bornheimer, R.; Doroff, R.; Shea, K. M.; Sato, R.; Weycker, D. Decline in Pneumococcal Disease Attenuated in Older Adults and Those With Comorbidities Following Universal Childhood PCV13 Immunization. Clin Infect Dis 2019, 68(11), 1831–1838. [Google Scholar] [CrossRef]
- Weycker, D.; Farkouh, R. A.; Strutton, D. R.; Edelsberg, J.; Shea, K. M.; Pelton, S. I. Rates and costs of invasive pneumococcal disease and pneumonia in persons with underlying medical conditions. BMC Health Serv Res From NLM. 2016, 16, 182. [Google Scholar] [CrossRef]
- Averin, A.; Shaff, M.; Weycker, D.; Lonshteyn, A.; Sato, R.; Pelton, S. I. Mortality and readmission in the year following hospitalization for pneumonia among US adults. Respir Med From NLM. 2021, 185, 106476. [Google Scholar] [CrossRef]
- Ministry of the Interior Republic of China (Taiwan). Abridged life table in Republic of China Area, 2023 . Available online: https://www.moi.gov.tw/english/cl.aspx?n=7780 (accessed on 12 May 2025).
- Bonten, M. J. M.; Huijts, S. M.; Bolkenbaas, M.; Webber, C.; Patterson, S.; Gault, S.; van Werkhoven, C. H.; van Deursen, A. M. M.; Sanders, E. A. M.; Verheij, T. J. M.; et al. Polysaccharide Conjugate Vaccine against Pneumococcal Pneumonia in Adults. New England Journal of Medicine 2015, 372(12), 1114–1125. [Google Scholar] [CrossRef] [PubMed]
- Mangen, M. J.; Rozenbaum, M. H.; Huijts, S. M.; van Werkhoven, C. H.; Postma, D. F.; Atwood, M.; van Deursen, A. M.; van der Ende, A.; Grobbee, D. E.; Sanders, E. A.; et al. Cost-effectiveness of adult pneumococcal conjugate vaccination in the Netherlands. Eur Respir J 2015, 46(5), 1407–1416. [Google Scholar] [CrossRef] [PubMed]
- Djennad, A.; Ramsay, M. E.; Pebody, R.; Fry, N. K.; Sheppard, C.; Ladhani, S. N.; Andrews, N. J. Effectiveness of 23-Valent Polysaccharide Pneumococcal Vaccine and Changes in Invasive Pneumococcal Disease Incidence from 2000 to 2017 in Those Aged 65 and Over in England and Wales. EClinicalMedicine 2018, 6, 42–50. [Google Scholar] [CrossRef] [PubMed]
- van Hoek, A. J.; Andrews, N.; Waight, P. A.; Stowe, J.; Gates, P.; George, R.; Miller, E. The effect of underlying clinical conditions on the risk of developing invasive pneumococcal disease in England. Journal of Infection 2012, 65(1), 17–24. [Google Scholar] [CrossRef]
- Atwood, M.; Beausoleil, L.; Breton, M. C.; Laferriere, C.; Sato, R.; Weycker, D. Cost-effectiveness of alternative strategies for use of 13-valent pneumococcal conjugate vaccine (PCV13) in Canadian adults. Can J Public Health 2018, 109(5-6), 756–768. [Google Scholar] [CrossRef]
- Marbaix, S.; Peetermans, W. E.; Verhaegen, J.; Annemans, L.; Sato, R.; Mignon, A.; Atwood, M.; Weycker, D. Cost-effectiveness of PCV13 vaccination in Belgian adults aged 65-84 years at elevated risk of pneumococcal infection. PLoS One 2018, 13(7), e0199427. [Google Scholar] [CrossRef]
- Weycker, D.; Sato, R.; Strutton, D.; Edelsberg, J.; Atwood, M.; Jackson, L. A. Public health and economic impact of 13-valent pneumococcal conjugate vaccine in US adults. Vaccine 2012, 30(36), 5437–5444. [Google Scholar] [CrossRef] [PubMed]
- Kulkarni, N.; Averin, A.; Taur, S.; Huang, L.; Hariharan, D.; Atwood, M.; Gupta, N. Cost-effectiveness of 13-valent pneumococcal polysaccharide conjugate vaccine in Indian adults aged ≥50 years. Expert Rev Pharmacoecon Outcomes Res From NLM. 2025, 1–13. [CrossRef]
- Mendes, D.; Averin, A.; Atwood, M.; Sato, R.; Vyse, A.; Campling, J.; Weycker, D.; Slack, M.; Ellsbury, G.; Mugwagwa, T. Cost-effectiveness of using a 20-valent pneumococcal conjugate vaccine to directly protect adults in England at elevated risk of pneumococcal disease. Expert Rev Pharmacoecon Outcomes Res 2022, 22(8), 1285–1295. [Google Scholar] [CrossRef]
- Rey-Ares, L.; Averin, A.; Mac Mullen, M.; Hariharan, D.; Atwood, M.; Carballo, C.; Huang, L. Cost-Effectiveness of 20-Valent Pneumococcal Conjugate Vaccine in Argentinean Adults. Infect Dis Ther 2024, 13(6), 1235–1251. [Google Scholar] [CrossRef]
- Essink, B.; Sabharwal, C.; Cannon, K.; Frenck, R.; Lal, H.; Xu, X.; Sundaraiyer, V.; Peng, Y.; Moyer, L.; Pride, M. W.; et al. Pivotal Phase 3 Randomized Clinical Trial of the Safety, Tolerability, and Immunogenicity of 20-Valent Pneumococcal Conjugate Vaccine in Adults. Clin Infect Dis 2022, 75(3), 390–398. [Google Scholar] [CrossRef] [PubMed]
- Simberkoff, M. S.; Cross, A. P.; Al-Ibrahim, M.; Baltch, A. L.; Geiseler, P. J.; Nadler, J.; Richmond, A. S.; Smith, R. P.; Schiffman, G.; Shepard, D. S.; et al. Efficacy of Pneumococcal Vaccine in High-Risk Patients. New England Journal of Medicine 1986, 315(21), 1318–1327. [Google Scholar] [CrossRef]
- Örtqvist, Å; Hedlund, J.; Burman, L.-Å; Elbel, E.; Höfer, M.; Leinonen, M.; Lindblad, I.; Sundelöf, B.; Kalin, M. Randomised trial of 23-valent pneumococcal capsular polysaccharide vaccine in prevention of pneumonia in middle-aged and elderly people. The Lancet acccessed. 1998, 351(9100), 399–403. [Google Scholar] [CrossRef] [PubMed]
- Fry, A. M.; Zell, E. R.; Schuchat, A.; Butler, J. C.; Whitney, C. G. Comparing potential benefits of new pneumococcal vaccines with the current polysaccharide vaccine in the elderly. Vaccine 2002, 21(3), 303–311. [Google Scholar] [CrossRef]
- Sisk, J. E.; Whang, W.; Butler, J. C.; Sneller, V.-P.; Whitney, C. G. Cost-Effectiveness of Vaccination against Invasive Pneumococcal Disease among People 50 through 64 Years of Age: Role of Comorbid Conditions and Race. Annals of Internal Medicine 2003, 138(12), 960–968. [Google Scholar] [CrossRef]
- Evers, S. M. A. A.; Ament, A. J. H. A.; Colombo, G. L.; Konradsen, H. B.; Reinert, R. R.; Sauerland, D.; Wittrup-Jensen, K.; Loiseau, C.; Fedson, D. S. Cost-effectiveness of pneumococcal vaccination for prevention of invasive pneumococcal disease in the elderly: an update for 10 Western European countries. European Journal of Clinical Microbiology & Infectious Diseases 2007, 26(8), 531–540. [Google Scholar] [CrossRef]
- Johnstone, J.; Marrie, T. J.; Eurich, D. T.; Majumdar, S. R. Effect of Pneumococcal Vaccination in Hospitalized Adults With Community-Acquired Pneumonia. Archives of Internal Medicine 2007, 167(18), 1938–1943. [Google Scholar] [CrossRef]
- Smith, K. J.; Zimmerman, R. K.; Lin, C. J.; Nowalk, M. P.; Ko, F.-S.; McEllistrem, M. C.; Roberts, M. S. Alternative strategies for adult pneumococcal polysaccharide vaccination: A cost-effectiveness analysis. Vaccine 2008, 26(11), 1420–1431. [Google Scholar] [CrossRef] [PubMed]
- Kuhlmann, A.; Theidel, U.; Pletz, M. W.; von der Schulenburg, J. M. G. Potential cost-effectiveness and benefit-cost ratios of adult pneumococcal vaccination in Germany. Health Economics Review 2012, 2(1), 4. [Google Scholar] [CrossRef] [PubMed]
- Boccalini, S.; Bechini, A.; Levi, M.; Tiscione, E.; Gasparini, R.; Bonanni, P. Cost-effectiveness of new adult pneumococcal vaccination strategies in Italy. Human Vaccines & Immunotherapeutics 2013, 9(3), 699–706. [Google Scholar] [CrossRef]
- Cho, B.-H.; Stoecker, C.; Link-Gelles, R.; Moore, M. R. Cost-effectiveness of administering 13-valent pneumococcal conjugate vaccine in addition to 23-valent pneumococcal polysaccharide vaccine to adults with immunocompromising conditions. Vaccine 2013, 31(50), 6011–6021. [Google Scholar] [CrossRef]
- Moberley, S.; Holden, J.; Tatham, D. P.; Andrews, R. M. Vaccines for preventing pneumococcal infection in adults. Cochrane Database of Systematic Reviews 2013, (1). [Google Scholar] [CrossRef] [PubMed]
- Smith, K. J.; Wateska, A. R.; Nowalk, M. P.; Raymund, M.; Lee, B. Y.; Zimmerman, R. K. Modeling of Cost Effectiveness of Pneumococcal Conjugate Vaccination Strategies in U.S. Older Adults. American Journal of Preventive Medicine acccessed. 2013, 44(4), 373–381. [Google Scholar] [CrossRef]
- Hoshi, S.-l.; Kondo, M.; Okubo, I. Economic Evaluation of Immunisation Programme of 23-Valent Pneumococcal Polysaccharide Vaccine and the Inclusion of 13-Valent Pneumococcal Conjugate Vaccine in the List for Single-Dose Subsidy to the Elderly in Japan. PLOS ONE 2015, 10(10), e0139140. [Google Scholar] [CrossRef]
- Stoecker, C.; Kim, L.; Gierke, R.; Pilishvili, T. Incremental Cost-Effectiveness of 13-valent Pneumococcal Conjugate Vaccine for Adults Age 50 Years and Older in the United States. Journal of General Internal Medicine 2016, 31(8), 901–908. [Google Scholar] [CrossRef]
- Heo, J. Y.; Seo, Y. B.; Choi, W. S.; Lee, J.; Noh, J. Y.; Jeong, H. W.; Kim, W. J.; Kim, M. J.; Lee, H. Y.; Song, J. Y. Cost-effectiveness of pneumococcal vaccination strategies for the elderly in Korea. PLOS ONE 2017, 12(5), e0177342. [Google Scholar] [CrossRef]
- Stoecker, C.; Kobayashi, M.; Matanock, A.; Cho, B.-H.; Pilishvili, T. Cost-effectiveness of continuing pneumococcal conjugate vaccination at age 65 in the context of indirect effects from the childhood immunization program. Vaccine 2020, 38(7), 1770–1777. [Google Scholar] [CrossRef] [PubMed]
- Taiwan Centers for Disease Control (CDC). Change trends of Streptococcus pneumoniae serotypes before and after the promotion of pneumococcal vaccine policy, 2017.
- Taiwan Centers for Disease Control (CDC). Change trends of Streptococcus pneumoniae serotypes before and after the promotion of pneumococcal vaccine policy; 2016. [Google Scholar]
- Taiwan Centers for Disease Control (CDC). Change trends of Streptococcus pneumoniae serotypes before and after the promotion of pneumococcal vaccine policy. 2015. [Google Scholar]
- Lu, C. Y.; Tang, C. H.; Fu, T.; Pwu, R. F.; Ho, Y. F. Pneumococcal conjugate vaccines in Taiwan: optimizing health gains in children and older adults through constrained optimization modeling: Pneumococcal conjugate vaccines optimization in Taiwan. Int J Infect Dis 2022, 114, 155–164. [Google Scholar] [CrossRef]
- National Statistics Republic of China (Taiwan). CPI Change Rate. 2024. Available online: https://eng.stat.gov.tw/News_Content.aspx?n=2329&s=234351 (accessed on 2 May 2025).
- Wong, E. L.; Cheung, A. W.; Wong, A. Y.; Xu, R. H.; Ramos-Goni, J. M.; Rivero-Arias, O. Normative Profile of Health-Related Quality of Life for Hong Kong General Population Using Preference-Based Instrument EQ-5D-5L. Value Health 2019, 22(8), 916–924. [Google Scholar] [CrossRef]
- Mangen, M.-J. J.; Huijts, S. M.; Bonten, M. J. M.; de Wit, G. A. The impact of community-acquired pneumonia on the health-related quality-of-life in elderly. BMC Infectious Diseases 2017, 17(1), 208. [Google Scholar] [CrossRef]
- Melegaro, A.; Edmunds, W. J. Cost-effectiveness analysis of pneumococcal conjugate vaccination in England and Wales. Vaccine From NLM. 2004, 22(31-32), 4203–4214. [Google Scholar] [CrossRef] [PubMed]
- Taiwan News. Taiwan's per capita GDP highest in East Asia, says IMF. 2022. Available online: https://www.taiwannews.com.tw/news/4685794 (accessed on 21 August 2024).
- Kuhne, F.; Achtert, K.; Puschner, F.; Urbanski-Rini, D.; Schiller, J.; Mahar, E.; Friedrich, J.; Atwood, M.; Sprenger, R.; Vietri, J.; et al. Cost-effectiveness of use of 20-valent pneumococcal conjugate vaccine among adults in Germany. Expert Rev Vaccines 2023, 22(1), 921–932. [Google Scholar] [CrossRef]
- Leidner, A. J. Summary of three economic models assessing pneumococcal vaccines in US adults; Centers for Disease Control and Prevention (CDC), 2021; file:///C:/Users/DHWANI~1/AppData/Local/Temp/MicrosoftEdgeDownloads/c8b78e48-1029-4b02-9eb0-708c95b442b7/cdc_110717_DS1.pdf. [Google Scholar]
- Zhang, J.; Vietri, J.; Averin, A.; Hariharan, D.; Atwood, M.; Huang, L. Cost-Effectiveness of 20-Valent Pneumococcal Conjugate Vaccine in Adults Aged ≥18 Years in Singapore. Value in Health Regional Issues acccessed. 2025, 49. [Google Scholar] [CrossRef]
- Malene, B. M.; Oyvind, H.; Tor, M.; David, N. M.; Jens, O.; Nanna, V. K.; Jeffrey, V. Cost-effectiveness of 20-valent pneumococcal conjugate vaccine compared with 23-valent pneumococcal polysaccharide vaccine among adults in a Norwegian setting. Cost Eff Resour Alloc 2023, 21(1), 52. [Google Scholar] [CrossRef]
- Olsen, J.; Schnack, H.; Skovdal, M.; Vietri, J.; Mikkelsen, M. B.; Poulsen, P. B. Cost-effectiveness of 20-valent pneumococcal conjugate vaccine in Denmark compared with PPV23. J Med Econ 2022, 25(1), 1240–1254. [Google Scholar] [CrossRef]
- Nakamura, S.; Mikami, M.; Hayamizu, T.; Yonemoto, N.; Moyon, C.; Gouldson, M.; Crossan, C.; Vietri, J.; Kamei, K. Cost-effectiveness analysis of adult pneumococcal conjugate vaccines for pneumococcal disease in Japan. Expert Rev Vaccines 2024, 23(1), 546–560. [Google Scholar] [CrossRef]
- Zayed, M.; Joury, J.; Farghaly, M.; Al Dallal, S.; Mahboub, B.; Kutrieb, E.; Averin, A. Budgetary Impact of 20-Valent Pneumococcal Conjugate Vaccine Use for Adult Expatriates Living in Dubai. Current Therapeutic Research 2023, 98, 100698. [Google Scholar] [CrossRef]
- Mugwagwa, T.; Averin, A.; Atwood, M.; Sato, R.; Vyse, A.; Campling, J.; Weycker, D.; Slack, M.; Ellsbury, G.; Mendes, D. Public health and budgetary impact of 20-valent pneumococcal conjugate vaccine for adults in England. Expert Rev Vaccines From NLM. 2022, 21(9), 1331–1341. [Google Scholar] [CrossRef]
- Essink, B.; Sabharwal, C.; Xu, X.; Sundaraiyer, V.; Peng, Y.; Moyer, L.; Pride, M. W.; Scully, I. L.; Jansen, K. U.; Gruber, W. C.; et al. 3. Phase 3 Pivotal Evaluation of 20-valent Pneumococcal Conjugate Vaccine (PCV20) Safety, Tolerability, and Immunologic Noninferiority in Participants 18 Years and Older. Open Forum Infectious Diseases 2020, 7 (Supplement_1), S2–S2. [Google Scholar] [CrossRef]
- Lawrence, H.; Pick, H.; Baskaran, V.; Daniel, P.; Rodrigo, C.; Ashton, D.; Edwards-Pritchard, R. C.; Sheppard, C.; Eletu, S. D.; Litt, D.; et al. Effectiveness of the 23-valent pneumococcal polysaccharide vaccine against vaccine serotype pneumococcal pneumonia in adults: A case-control test-negative design study. PLOS Medicine 2020, 17(10), e1003326. [Google Scholar] [CrossRef]
- Suzuki, M.; Dhoubhadel, B. G.; Ishifuji, T.; Yasunami, M.; Yaegashi, M.; Asoh, N.; Ishida, M.; Hamaguchi, S.; Aoshima, M.; Ariyoshi, K.; et al. Serotype-specific effectiveness of 23-valent pneumococcal polysaccharide vaccine against pneumococcal pneumonia in adults aged 65 years or older: a multicentre, prospective, test-negative design study. The Lancet Infectious Diseases acccessed. 2017, 17(3), 313–321. [Google Scholar] [CrossRef]
- Klugman, K. P.; Madhi, S. A.; Huebner, R. E.; Kohberger, R.; Mbelle, N.; Pierce, N. A Trial of a 9-Valent Pneumococcal Conjugate Vaccine in Children with and Those without HIV Infection. New England Journal of Medicine 2003, 349(14), 1341–1348. [Google Scholar] [CrossRef] [PubMed]
- French, N.; Gordon, S. B.; Mwalukomo, T.; White, S. A.; Mwafulirwa, G.; Longwe, H.; Mwaiponya, M.; Zijlstra, E. E.; Molyneux, M. E.; Gilks, C. F. A Trial of a 7-Valent Pneumococcal Conjugate Vaccine in HIV-Infected Adults. New England Journal of Medicine 2010, 362(9), 812–822. [Google Scholar] [CrossRef] [PubMed]
|
|
Age/Risk Profile | ||||||||||||||||
| 18-49 years | 50-64 years | 65-74 years | 75-84 years | ≥85 years | |||||||||||||
| High | High | Low | Moderate | High | Low | Moderate | High | Low | Moderate | High | |||||||
| No. of adults | 361,455 | 452,216 | 1,373,521 | 1,200,807 | 381,838 | 479,038 | 562,725 | 212,962 | 169,480 | 211,068 | 88,419 | ||||||
| Annual disease incidence (per 100,000) | |||||||||||||||||
| IPD | 4.6 | 6.2 | 1.9 | 6.0 | 8.1 | 2.9 | 6.9 | 7.6 | 4.6 | 8.2 | 6.5 | ||||||
| Inpatient NBP | 271.9 | 859.3 | 212.5 | 1,041.3 | 2,358.9 | 683.0 | 1,980.8 | 2,868.8 | 1,056.1 | 3,062.6 | 4,435.4 | ||||||
| Outpatient NBP | 888.6 | 2,260.3 | 1,028.2 | 3,106.4 | 4,166.4 | 1,422.1 | 4,296.5 | 5,762.6 | 1,853.4 | 5,599.9 | 7,510.7 | ||||||
| Annual mortality/case-fatality (per 100) | |||||||||||||||||
| General population | 0.2 | 1.0 | 1.2 | 1.8 | 2.2 | 2.7 | 4.0 | 5.2 | 5.1 | 7.4 | 9.7 | ||||||
| IPD | 8.6 | 9.8 | 6.8 | 12.9 | 12.0 | 13.2 | 17.0 | 13.2 | 17.4 | 21.3 | 17.9 | ||||||
| Inpatient NBP | 10.5 | 11.4 | 4.5 | 6.7 | 10.0 | 8.6 | 10.7 | 11.4 | 11.6 | 11.7 | 15.6 | ||||||
| Outpatient NBP | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | ||||||
| Medical care costs (per case, NTD) | |||||||||||||||||
| IPD | 119,179 | 119,179 | 119,179 | 119,179 | 119,179 | 119,179 | 119,179 | 119,179 | 119,179 | 119,179 | 119,179 | ||||||
| Inpatient NBP | 48,195 | 48,195 | 48,195 | 48,195 | 48,195 | 48,195 | 48,195 | 48,195 | 48,195 | 48,195 | 48,195 | ||||||
| Outpatient NBP | 785 | 785 | 785 | 785 | 785 | 785 | 785 | 785 | 785 | 785 | 785 | ||||||
| CEA Utilities | |||||||||||||||||
| General population health-state utilities | 0.7190 | 0.6632 | 0.8872 | 0.6984 | 0.6768 | 0.8805 | 0.6711 | 0.6521 | 0.8733 | 0.6438 | 0.6017 | ||||||
| IPD | 0.1300 | 0.1300 | 0.1300 | 0.1300 | 0.1300 | 0.1300 | 0.1300 | 0.1300 | 0.1300 | 0.1300 | 0.1300 | ||||||
| Inpatient NBP | 0.1300 | 0.1300 | 0.1300 | 0.1300 | 0.1300 | 0.1300 | 0.1300 | 0.1300 | 0.1300 | 0.1300 | 0.1300 | ||||||
| Outpatient NBP | 0.0040 | 0.0040 | 0.0040 | 0.0040 | 0.0040 | 0.0040 | 0.0040 | 0.0040 | 0.0040 | 0.0040 | 0.0040 | ||||||
| PCV13→PPV23 | PCV20 | Difference | ||
| Clinical Outcomes | ||||
| No. of Cases | ||||
| IPD | 5,511 | 5,482 | -29 | |
| All-Cause NBP | ||||
| Inpatient | 1,934,376 | 1,932,893 | -1,483 | |
| Requiring Outpatient Care Only | 3,716,882 | 3,713,710 | -3,171 | |
| No. of Deaths | 236,583 | 236,423 | -160 | |
| Life-Years (discounted)* | 69,827,105 | 69,828,283 | 1,178 | |
| Quality-Adjusted Life-Years (discounted)* | 50,672,027 | 50,672,971 | 944 | |
| Economic Outcomes | ||||
| Total Costs (millions) | ||||
| Medical Care | NTD 63,671.5 | NTD 63,607.1 | -NTD 64.4 | |
| Vaccination | NTD 11,482.8 | NTD 9,199.2 | -NTD 2,283.6 | |
| Medical + Vaccination | NTD 75,154.3 | NTD 72,806.3 | -NTD 2,348.0 | |
| Cost-Effectiveness | ||||
| Cost per LY | --- | --- | Dominant | |
| Cost per QALY | --- | --- | Dominant | |
| PCV13→PPV23 | PCV20 | Difference | ||
| Clinical Outcomes | ||||
| No. of Cases | ||||
| IPD | 5,453 | 5,429 | -23 | |
| All-Cause NBP | ||||
| Inpatient | 1,930,861 | 1,929,593 | -1,268 | |
| Requiring Outpatient Care Only | 3,710,239 | 3,707,514 | -2,725 | |
| No. of Deaths | 236,140 | 236,004 | -135 | |
| Life-Years (discounted)* | 69,829,068 | 69,830,085 | 1,017 | |
| Quality-Adjusted Life-Years (discounted)* | 50,673,581 | 50,674,395 | 815 | |
| Economic Outcomes | ||||
| Total Costs (millions) | ||||
| Medical Care | NTD 63,557.1 | NTD 63,501.7 | -NTD 55.3 | |
| Vaccination | NTD 11,482.8 | NTD 9,199.2 | -NTD 2,283.6 | |
| Medical + Vaccination | NTD 75,039.8 | NTD 72,700.9 | -NTD 2,338.9 | |
| Cost-Effectiveness | ||||
| Cost per LY | --- | --- | Dominant | |
| Cost per QALY | --- | --- | Dominant | |
| PCV13→PPV23 | PCV20 | Difference | ||
| Clinical Outcomes | ||||
| No. of Cases | ||||
| IPD | 5,521 | 5,482 | -39 | |
| All-Cause NBP | ||||
| Inpatient | 1,934,376 | 1,932,893 | -1,483 | |
| Requiring Outpatient Care Only | 3,716,881 | 3,713,710 | -3,171 | |
| No. of Deaths | 236,584 | 236,423 | -161 | |
| Life-Years (discounted)* | 69,827,093 | 69,828,283 | 1,190 | |
| Quality-Adjusted Life-Years (discounted)* | 50,672,018 | 50,672,971 | 953 | |
| Economic Outcomes | ||||
| Total Costs (millions) | ||||
| Medical Care | NTD 63,672.5 | NTD 63,607.1 | -NTD 65.5 | |
| Vaccination | NTD 9,922.8 | NTD 9,199.2 | -NTD 723.6 | |
| Medical + Vaccination | NTD 73,595.4 | NTD 72,806.3 | -NTD 789.1 | |
| Cost-Effectiveness | ||||
| Cost per LY | --- | --- | Dominant | |
| Cost per QALY | --- | --- | Dominant | |
| PCV20 vs. PCV13-> PPV23 | |||||||
| Year 1 | Year 2 | Year 3 | Year 4 | Year 5 | Total | ||
| Cumulative vaccine uptake | 40% | 65% | 80% | 88% | 93% | --- | |
| Clinical Outcomes | |||||||
| No. of Cases | |||||||
| IPD | -2 | -3 | -4 | -5 | -6 | -20 | |
| All-Cause NBP | |||||||
| Inpatient | -111 | -188 | -236 | -265 | -281 | -1,080 | |
| Requiring Outpatient Care Only | -253 | -420 | -518 | -571 | -596 | -2,357 | |
| No. of Deaths | -11 | -19 | -25 | -28 | -30 | -113 | |
| Economic Outcomes | |||||||
| Total Costs (millions) | |||||||
| Medical Care | -NTD 5.8 | -NTD 9.8 | -NTD 12.3 | -NTD 13.8 | -NTD 14.7 | -NTD 56.3 | |
| Vaccination | -NTD 2,361.7 | -NTD 1,453.8 | -NTD 833.1 | -NTD 457.0 | -NTD 251.3 | -NTD 5,356.8 | |
| Medical + Vaccination | -NTD 2,367.5 | -NTD 1,463.5 | -NTD 845.4 | -NTD 470.8 | -NTD 265.9 | -NTD 5,413.1 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any in-jury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




