Submitted:
14 April 2026
Posted:
15 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
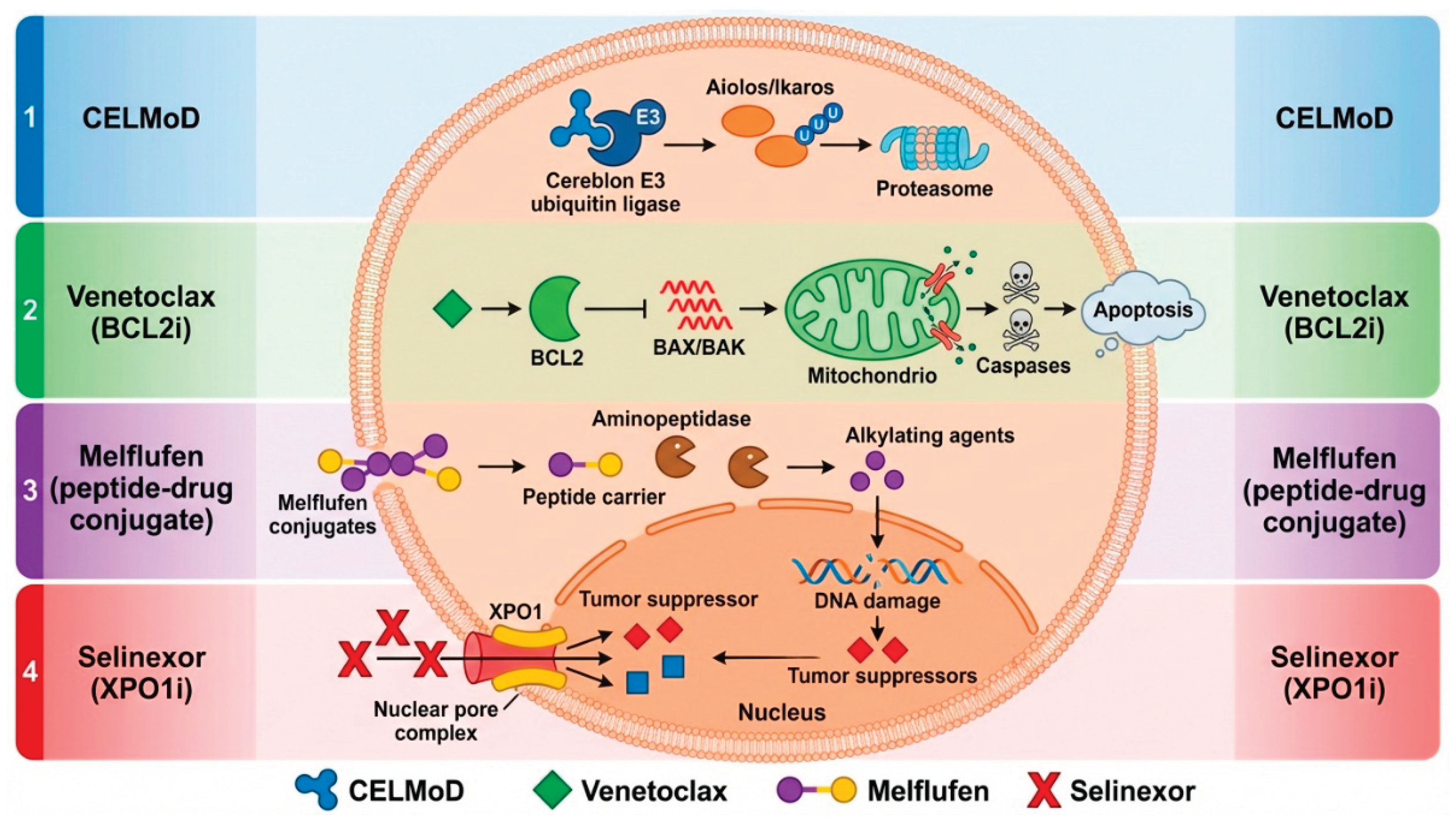
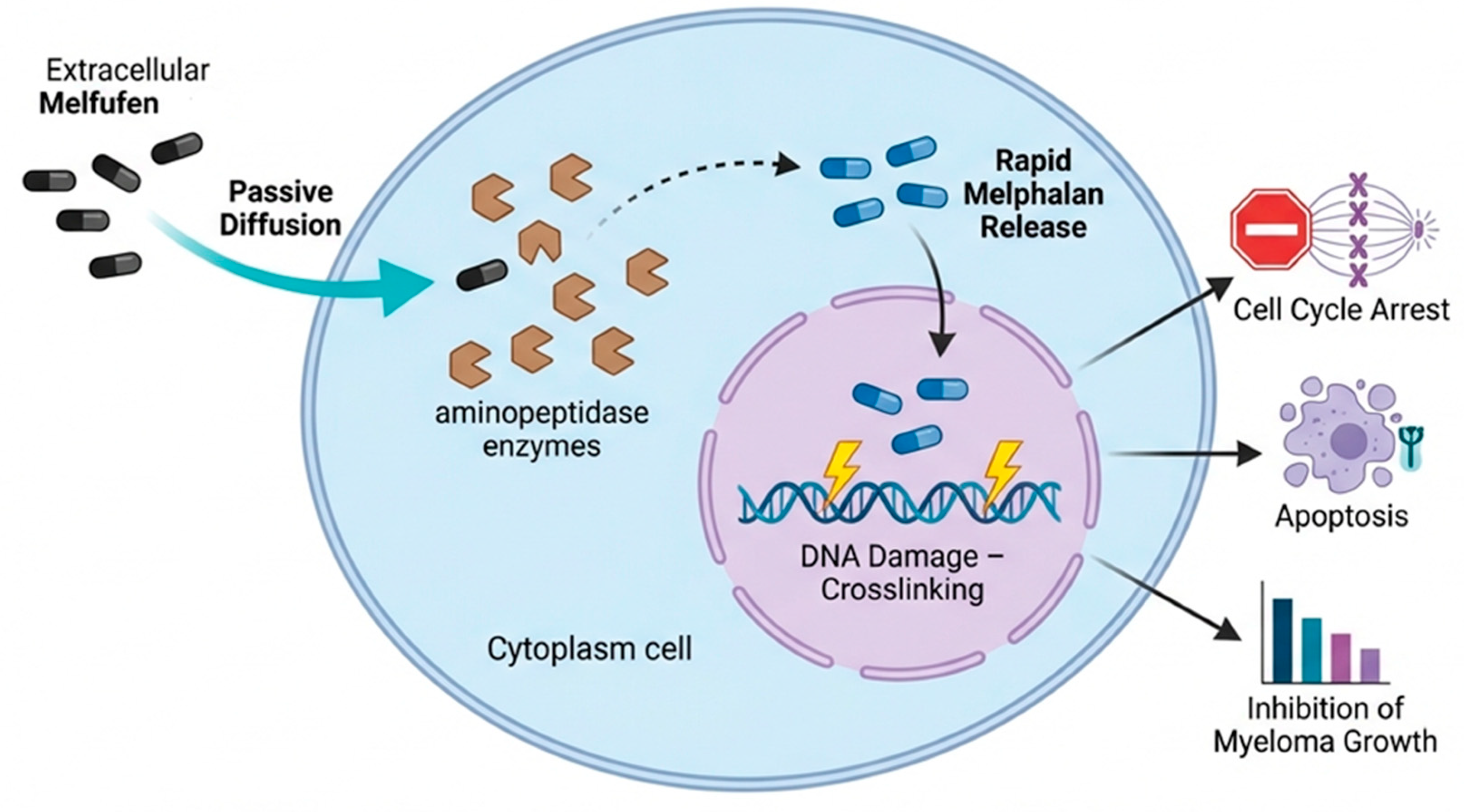
2. Mechanism of Action of Melflufen
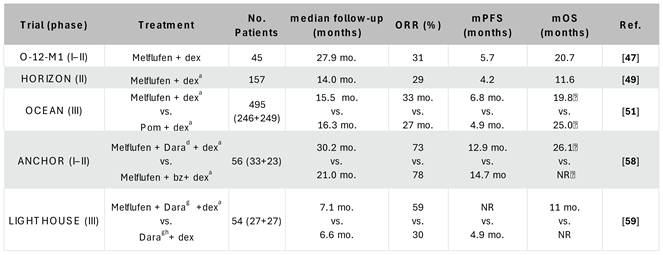
3. Clinical Development
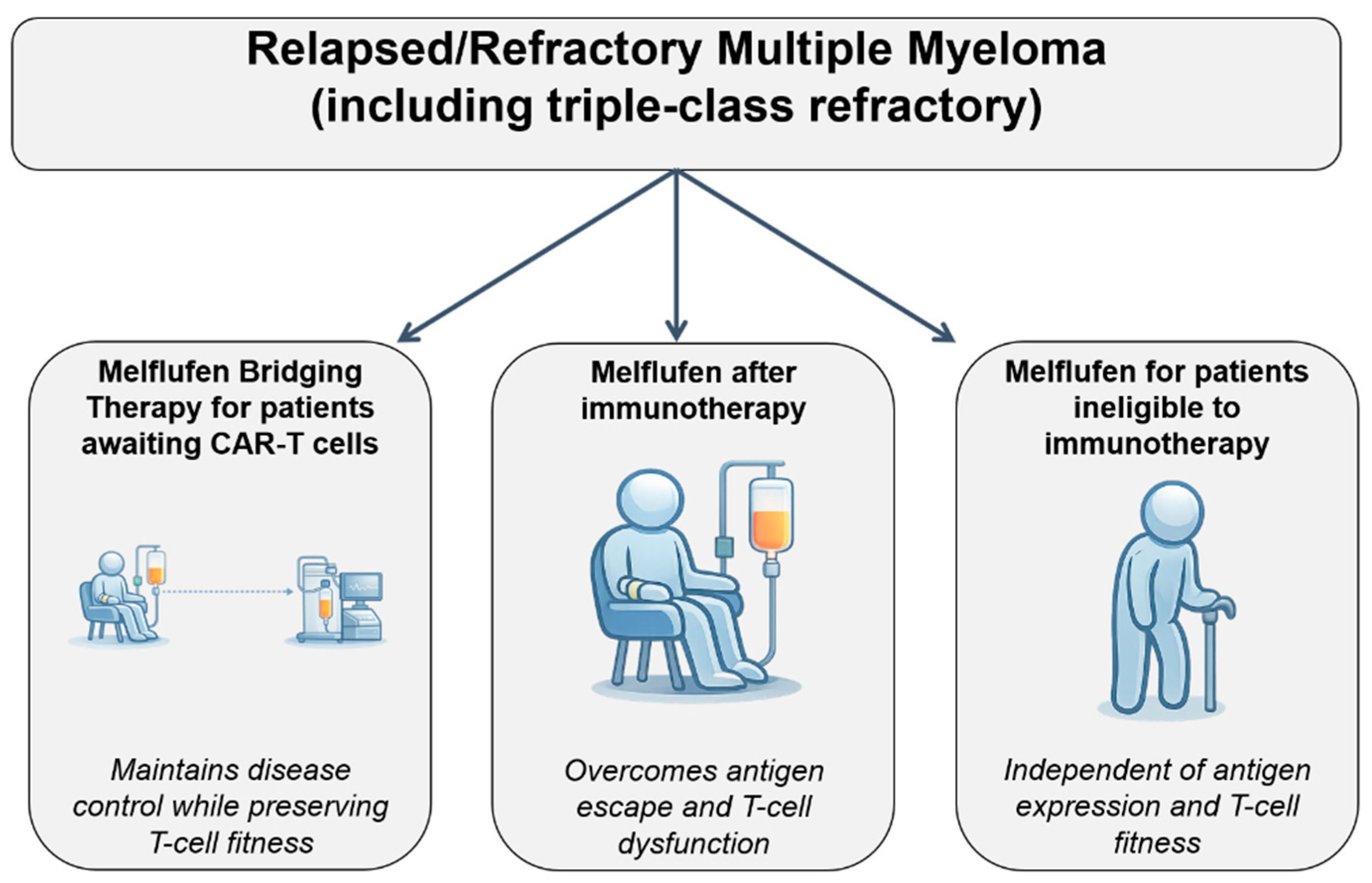
4. Real-World Evidence and Special Populations
5. Positioning of Melflufen in the Evolving Treatment Landscape of MM
6. Safety Profile
7. Future Perspectives
8. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| ADC | Antibody-drug Conjugate |
| ASCT | autologous stem cell transplantation |
| BM | bone marrow |
| Bz | bortezomib |
| CAR-T | chimeric antigen receptor T |
| CELMoDs | cereblon E3 ligase modulators |
| Dara | Daratumumab |
| DEX | dexamethasone |
| DOAJ | Directory of open access journals |
| DOR | duration of response |
| LD | Linear dichroism |
| MDR1/ABCB1 | multidrug resistance protein 1 |
| MM | Multiple Myeloma |
| NR | Not reached |
| ORR | overall response rate |
| OS | Overall Survival |
| PFS | progression-free survival |
| PI | Proteasome inhibitor |
| Pom | Pomalidomide |
| RRMM | relapsed or refractory multiple myeloma |
| SINE | selective inhibitors of nuclear export |
| TCE | T-cell engagers |
References
- Palumbo, A.; Anderson, K. Multiple myeloma. N Engl J Med 2011, vol. 364(no. 11), 1046–1060. [Google Scholar] [CrossRef]
- Rajkumar, S. V. International Myeloma Working Group updated criteria for the diagnosis of multiple myeloma. Lancet Oncol 2014, vol. 15(no. 12), e538–548. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S. K. Multiple myeloma. Nat Rev Dis Primers 2017, vol. 3, 17046. [Google Scholar] [CrossRef] [PubMed]
- Malard, F. Multiple myeloma. Nat Rev Dis Primers 2024, vol. 10(no. 1), 45. [Google Scholar] [CrossRef] [PubMed]
- Bray, F.; et al. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 2024, vol. 74(no. 3), 229–263. [Google Scholar] [CrossRef]
- Zanwar, S.; Rajkumar, S. V. Current risk stratification and staging of multiple myeloma and related clonal plasma cell disorders. Leukemia 2025, vol. 39(no. 11), 2610–2617. [Google Scholar] [CrossRef]
- Rajkumar, S. V. Multiple myeloma: 2024 update on diagnosis, risk-stratification, and management. Am J Hematol 2024, vol. 99(no. 9), 1802–1824. [Google Scholar] [CrossRef]
- Kumar, S.; et al. Gaps and opportunities in the treatment of relapsed-refractory multiple myeloma: Consensus recommendations of the NCI Multiple Myeloma Steering Committee. Blood Cancer Journal 2022, vol. 12(no. 6), 98. [Google Scholar] [CrossRef]
- Chen, Q.; Zhang, M.; Zheng, S.; Tong, Y.; Tan, Y. Therapeutic progress in relapsed/refractory multiple myeloma. Annals of Hematology 2024, vol. 103(no. 6), 1833–1841. [Google Scholar] [CrossRef]
- van de Donk, N. W. C. J.; Pawlyn, C.; Yong, K. L. Multiple myeloma. The Lancet 2021, vol. 397(no. 10272), 410–427. [Google Scholar] [CrossRef]
- Wang, P. F. Treatment patterns and overall survival of patients with double-class and triple-class refractory multiple myeloma: a US electronic health record database study. Leukemia & Lymphoma 2023, vol. 64(no. 2), 398–406. [Google Scholar] [CrossRef]
- Dimopoulos, M. A. EHA–EMN Evidence-Based Guidelines for diagnosis, treatment and follow-up of patients with multiple myeloma. Nature Reviews Clinical Oncology 2025, vol. 22(no. 9), 680–700. [Google Scholar] [CrossRef]
- Mateos, M.-V. LocoMMotion: a study of real-life current standards of care in triple-class exposed patients with relapsed/refractory multiple myeloma - 2-year follow-up (final analysis). Leukemia 2024, vol. 38(no. 12), 2554–2560. [Google Scholar] [CrossRef]
- Riedhammer, C.; et al. The evolution to hepta-refractory myeloma involves sequential loss of CD38, BCMA and GPRC5D. Leukemia. 2026. [CrossRef]
- Talarico, M.; et al. Exceptional long-term responses from OCEAN and HORIZON trials: melflufen-dexamethasone as an expansion of treatment options for relapsed/refractory multiple myeloma in the era of new immunotherapies? J Cancer Res Clin Oncol 2025, vol. 151(no. 11), 288. [Google Scholar] [CrossRef] [PubMed]
- Neupane, K.; et al. Profile and management of toxicity of selinexor and belantamab mafodotin for the treatment of triple class refractory multiple myeloma. Journal of Blood Medicine 2021, 529–550. [Google Scholar] [CrossRef] [PubMed]
- Bobin, A. Novel Non-Immunologic Agents for Relapsed and Refractory Multiple Myeloma: A Review Article. Cancers 2021, vol. 13(no. 20), 5210. [Google Scholar] [CrossRef] [PubMed]
- Dhillon, S. Melphalan Flufenamide (Melflufen): First Approval. Drugs 2021, vol. 81(no. 8), 963–969. [Google Scholar] [CrossRef]
- Sgherza, N.; Curci, P.; Rizzi, R.; Musto, P. Novel Approaches Outside the Setting of Immunotherapy for the Treatment of Multiple Myeloma: The Case of Melflufen, Venetoclax, and Selinexor. Front Oncol 2021, vol. 11, 716751. [Google Scholar] [CrossRef]
- Schjesvold, F.; Oriol, A. Current and Novel Alkylators in Multiple Myeloma. Cancers 2021, vol. 13(no. 10), 2465. [Google Scholar] [CrossRef]
- Kazandjian, D.; Dew, A.; Hill, E. The changing role of high dose melphalan with stem cell rescue in the treatment of newly diagnosed multiple myeloma in the era of modern therapies—back to the future! Best Practice & Research Clinical Haematology 2020, vol. 33(no. 1), 101150. [Google Scholar] [CrossRef]
- Wickström, M. Melflufen - a peptidase-potentiated alkylating agent in clinical trials. Oncotarget 2017, vol. 8(no. 39), 66641–66655. [Google Scholar] [CrossRef]
- National Center for Biotechnology Information. “PubChem Compound Summary for CID 9935639, Melphalan Flufenamide.”. 2026. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/Melphalan_flufenamide (accessed on 16 March 2026).
- Cliff, E. R. S.; Janakiram, M.; Kesselheim, A. S. Melflufen: post-hoc subgroup analyses and the US FDA Oncologic Drugs Advisory Committee. The Lancet Haematology 2023, vol. 10(no. 5), e314–e317. [Google Scholar] [CrossRef] [PubMed]
- Westermark, U.; et al. A rapid intracellular enrichment of alkylating payload is essential for melphalan flufenamide potency and mechanism of action. Biochemical and Biophysical Research Communications 2023, vol. 656, 122–130. [Google Scholar] [CrossRef] [PubMed]
- Chauhan, D.; et al. In vitro and in vivo antitumor activity of a novel alkylating agent, melphalan-flufenamide, against multiple myeloma cells. Clin Cancer Res 2013, vol. 19(no. 11), 3019–3031. [Google Scholar] [CrossRef] [PubMed]
- Miettinen, J. J. Aminopeptidase Expression in Multiple Myeloma Associates with Disease Progression and Sensitivity to Melflufen. Cancers (Basel) 2021, vol. 13(no. 7). [Google Scholar] [CrossRef]
- Bianchi, G.; Anderson, K. C. Contribution of Inhibition of Protein Catabolism in Myeloma. Cancer J 2019, vol. 25(no. 1), 11–18. [Google Scholar] [CrossRef]
- Wallace, D. C. Mitochondria and cancer. Nat Rev Cancer 2012, vol. 12(no. 10), 685–698. [Google Scholar] [CrossRef]
- Porporato, P. E.; Filigheddu, N.; Pedro, J. M. B.-S.; Kroemer, G.; Galluzzi, L. Mitochondrial metabolism and cancer. Cell Res 2018, vol. 28(no. 3), 265–280. [Google Scholar] [CrossRef]
- Acs, K. Efficacy of melflufen in multiple myeloma with mutated or deleted TP53. Experimental Hematology & Oncology 2025, vol. 14(no. 1), 138. [Google Scholar] [CrossRef]
- Kühne, A.; Tzvetkov, M. V.; Hagos, Y.; Lage, H.; Burckhardt, G.; Brockmöller, J. Influx and efflux transport as determinants of melphalan cytotoxicity: Resistance to melphalan in MDR1 overexpressing tumor cell lines. Biochem Pharmacol 2009, vol. 78(no. 1), 45–53. [Google Scholar] [CrossRef]
- Byrgazov, K.; et al. Novel Peptide-drug Conjugate Melflufen Efficiently Eradicates Bortezomib-resistant Multiple Myeloma Cells Including Tumor-initiating Myeloma Progenitor Cells. HemaSphere 2021, vol. 5(no. 7). [Google Scholar] [CrossRef] [PubMed]
- Byrgazov, K.; et al. Up-regulation of multidrug resistance protein MDR1/ABCB1 in carfilzomib-resistant multiple myeloma differentially affects efficacy of anti-myeloma drugs. Leukemia Research 2021, vol. 101, 106499. [Google Scholar] [CrossRef] [PubMed]
- Ocio, E. M. Melflufen for the treatment of multiple myeloma. Expert Rev Clin Pharmacol 2022, vol. 15(no. 4), 371–382. [Google Scholar] [CrossRef] [PubMed]
- Flanagan, K.; et al. The Peptide-Drug Conjugate Melflufen Modulates the Unfolded Protein Response of Multiple Myeloma and Amyloidogenic Plasma Cells and Induces Cell Death. Hemasphere 2022, vol. 6(no. 3), e687. [Google Scholar] [CrossRef]
- Giliberto, M. Ex vivo drug sensitivity screening in multiple myeloma identifies drug combinations that act synergistically. Mol Oncol 2022, vol. 16(no. 6), 1241–1258. [Google Scholar] [CrossRef]
- Strese, S.; et al. In vitro and in vivo anti-leukemic activity of the peptidase-potentiated alkylator melflufen in acute myeloid leukemia. Oncotarget 2017, vol. 8(no. 4), 6341–6352. [Google Scholar] [CrossRef]
- Gebraad, A. Growth Response and Differentiation of Bone Marrow-Derived Mesenchymal Stem/Stromal Cells in the Presence of Novel Multiple Myeloma Drug Melflufen. Cells 2022, vol. 11(no. 9). [Google Scholar] [CrossRef]
- Strese, S.; et al. The novel alkylating prodrug melflufen (J1) inhibits angiogenesis in vitro and in vivo. Biochemical Pharmacology 2013, vol. 86(no. 7), 888–895. [Google Scholar] [CrossRef]
- Byrgazov, K. Melphalan flufenamide inhibits osteoclastogenesis by suppressing proliferation of monocytes. Bone Rep 2021, vol. 15, 101098. [Google Scholar] [CrossRef]
- Wickström, M.; Larsson, R.; Nygren, P.; Gullbo, J. Aminopeptidase N (CD13) as a target for cancer chemotherapy. Cancer Sci 2011, vol. 102(no. 3), 501–508. [Google Scholar] [CrossRef]
- Sevcikova, S.; Minarik, J.; Stork, M.; Jelinek, T.; Pour, L.; Hajek, R. Extramedullary disease in multiple myeloma – controversies and future directions. Blood Reviews 2019, vol. 36, 32–39. [Google Scholar] [CrossRef] [PubMed]
- Bladé, J.; Fernández de Larrea, C.; Rosiñol, L.; Cibeira, M. T.; Jiménez, R.; Powles, R. Soft-tissue plasmacytomas in multiple myeloma: incidence, mechanisms of extramedullary spread, and treatment approach. J Clin Oncol 2011, vol. 29(no. 28), 3805–3812. [Google Scholar] [CrossRef] [PubMed]
- Schepsky, A. Melflufen, a peptide-conjugated alkylator, is an efficient anti-neo-plastic drug in breast cancer cell lines. Cancer Med 2020, vol. 9(no. 18), 6726–6738. [Google Scholar] [CrossRef] [PubMed]
- Bringhen, S.; et al. Melflufen plus dexamethasone in relapsed/refractory multiple myeloma: long-term survival follow-up from the Phase II study O-12-M1. Br J Haematol 2021, vol. 193(no. 6), 1105–1109. [Google Scholar] [CrossRef]
- Richardson, P. G.; et al. Melflufen plus dexamethasone in relapsed and refractory multiple myeloma (O-12-M1): a multicentre, international, open-label, phase 1-2 study. Lancet Haematol 2020, vol. 7(no. 5), e395–e407. [Google Scholar] [CrossRef]
- Richardson, P. G. Melflufen and Dexamethasone in Heavily Pretreated Relapsed and Refractory Multiple Myeloma. J Clin Oncol 2021, vol. 39(no. 7), 757–767. [Google Scholar] [CrossRef]
- Larocca, A.; et al. Patient-reported outcomes in relapsed/refractory multiple myeloma treated with melflufen plus dexamethasone: analyses from the Phase II HORIZON study. Br J Haematol 2022, vol. 196(no. 3), 639–648. [Google Scholar] [CrossRef]
- Schjesvold, F.; Robak, P.; Pour, L.; Aschan, J.; Sonneveld, P. OCEAN: a randomized Phase III study of melflufen + dexamethasone to treat relapsed refractory multiple myeloma. Future Oncol 2020, vol. 16(no. 11), 631–641. [Google Scholar] [CrossRef]
- Schjesvold, F. H.; et al. Melflufen or pomalidomide plus dexamethasone for patients with multiple myeloma refractory to lenalidomide (OCEAN): a randomised, head-to-head, open-label, phase 3 study. Lancet Haematol 2022, vol. 9(no. 2), e98–e110. [Google Scholar] [CrossRef]
- Schjesvold, F. H.; et al. Health-related quality of life in relapsed/refractory multiple myeloma treated with melflufen and dexamethasone: analyses from the phase III OCEAN study. Haematologica 2024, vol. 109(no. 7), 2331–2336. [Google Scholar] [CrossRef]
- Sonneveld, P.; et al. Benefit Versus Risk Assessment of Melflufen and Dexamethasone in Relapsed/Refractory Multiple Myeloma: Analyses From Longer Follow-up of the OCEAN and HORIZON Studies. Clin Lymphoma Myeloma Leuk 2023, vol. 23(no. 9), 687–696. [Google Scholar] [CrossRef]
- Schjesvold, F. H.; et al. Melflufen in relapsed/refractory multiple myeloma refractory to prior alkylators: A subgroup analysis from the OCEAN study. Eur J Haematol 2024, vol. 112(no. 3), 402–411. [Google Scholar] [CrossRef] [PubMed]
- “FDA (2024) Final Decision on the Proposal to Withdraw Approval of Pepaxto (melphalan flufenamide) for Injection.”. Available online: https://www.fda.gov/media/176510/download?attachment (accessed on 10 February 2026).
- EMA (2022) Pepaxti: melphalan flufenamide. European Medicines Agency]. Available online: https://www.ema.europa.eu/en/documents/smop-initial/chmp-summary-positive-opinion-pepaxti_en.pdf (accessed on 10 February 2026).
- EMA (2025) Pepaxti—Annex 1—Summary of Product Characteristics. EMA. Available online: https://www.ema.europa.eu/en/documents/product-information/pepaxti-epar-product-information_en.pdf (accessed on 10 February 2026).
- Ocio, E. M.; et al. ANCHOR: melflufen plus dexamethasone and daratumumab or bortezomib in relapsed/refractory multiple myeloma: final results of a phase I/IIa study. Haematologica 2024, vol. 109(no. 3), 867–876. [Google Scholar] [CrossRef] [PubMed]
- Pour, L.; et al. Efficacy and safety of melflufen plus daratumumab and dexamethasone in relapsed/refractory multiple myeloma: results from the randomized, open-label, phase III LIGHTHOUSE study. Haematologica 2024, vol. 109(no. 3), 895–905. [Google Scholar] [CrossRef] [PubMed]
- Hossain, S. Outcomes of Melflufen Treatment in Patients With Relapsed/Refractory Multiple Myeloma. Eur J Haematol 2025, vol. 114(no. 6), 982–989. [Google Scholar] [CrossRef]
- Gálvez Ruiz, A.; et al. Experiencia en vida real con melflufen en pacientes con mieloma múltiple en recaída/refractario en un centro español. in proceedings of the XIX Reunión Anual de la Asociación Valenciana de Hematología y Hemoterapia (AVHH), Valencia, Spain, 26-28 February 2025. [Google Scholar]
- Martínez-Campuzano, D.; Lamas, C.; Sopeña, B.; et al. Real-World Use of Melflufen Plus Dexamethasone in Relapsed/Refractory Multiple Myeloma: A Retrospective Study from the Valencian Community and the Region of Murcia (Spain). in proceedings of the International Myeloma Society Annual Meeting 2025, Toronto, Canada, 17-20 September 2025. [Google Scholar]
- Mancuso, K. Positioning of Melflufen in Heavily Pretreated RRMM Patients: Real-World Evidence in a Rapidly Evolving Therapeutic Landscape. Eur J Haematol 2026. [Google Scholar] [CrossRef]
- Giunta, G.; Longo, E.; Lo Faro, C.; et al. Efficacy and tolerability of melflufen in relapsed multiple myeloma: a single-center real-life experience. in proceedings of the European Myeloma Network (EMN) Meeting, Athens, Greece, 10-12 April, 2025. [Google Scholar]
- Pour, L.; et al. BRIDGE (OP-107): A Phase 2 pharmacokinetic study of melflufen plus dexamethasone in patients with relapsed/refractory multiple myeloma and impaired renal function. In EHA Library; 2021. [Google Scholar]
- Costa, L. J.; et al. International myeloma working group immunotherapy committee recommendation on sequencing immunotherapy for treatment of multiple myeloma. Leukemia 2025, vol. 39(no. 3), 543–554. [Google Scholar] [CrossRef]
- Manier, S.; Jurczyszyn, A.; Vesole, D. H. “Bridging Chemotherapy: Multiple Myeloma.,” in The EBMT/EHA CAR-T Cell Handbook; Kröger, N., Gribben, J., Chabannon, C., Yakoub-Agha, I., Einsele, H., Eds.; Springer: Cham (CH), 2022; pp. 127–129. [Google Scholar]
- Da Vià, M. C. Homozygous BCMA gene deletion in response to anti-BCMA CAR T cells in a patient with multiple myeloma. Nat Med 2021, vol. 27(no. 4), 616–619. [Google Scholar] [CrossRef]
- Lee, H. Impact of soluble BCMA and non-T-cell factors on refractoriness to BCMA-targeting T-cell engagers in multiple myeloma. Blood 2024, vol. 144(no. 25), 2637–2651. [Google Scholar] [CrossRef]
- Lee, H.; et al. Mechanisms of antigen escape from BCMA- or GPRC5D-targeted immunotherapies in multiple myeloma. Nat Med 2023, vol. 29(no. 9), 2295–2306. [Google Scholar] [CrossRef]
- Friedrich, M. J. The pre-existing T cell landscape determines the response to bispecific T cell engagers in multiple myeloma patients. Cancer Cell 2023, vol. 41(no. 4), 711–725.e6. [Google Scholar] [CrossRef]
- Ludwig, H.; Mai, E. K.; Högner, M.; Welslau, M.; Waldschmidt, J. M. Optimizing the use of melflufen (melphalan flufenamide) in relapsed or refractory multiple myeloma: recommendations for clinical practice. Annals of Hematology 2025, vol. 104(no. 11), 5593–5603. [Google Scholar] [CrossRef]
- Moreau, P. Teclistamab in Relapsed or Refractory Multiple Myeloma. N Engl J Med 2022, vol. 387(no. 6), 495–505. [Google Scholar] [CrossRef]
- Lesokhin, A. M.; et al. Elranatamab in relapsed or refractory multiple myeloma: phase 2 MagnetisMM-3 trial results. Nat Med 2023, vol. 29(no. 9), 2259–2267. [Google Scholar] [CrossRef]
- Berdeja, J. G.; et al. Ciltacabtagene autoleucel, a B-cell maturation antigen-directed chimeric antigen receptor T-cell therapy in patients with relapsed or refractory multiple myeloma (CARTITUDE-1): a phase 1b/2 open-label study. Lancet 2021, vol. 398(no. 10297), 314–324. [Google Scholar] [CrossRef]
- Rodriguez-Otero, P. Ide-cel or Standard Regimens in Relapsed and Refractory Multiple Myeloma. N Engl J Med 2023, vol. 388(no. 11), 1002–1014. [Google Scholar] [CrossRef]
- Wallington-Beddoe, C. T.; Mynott, R. L. “Prognostic and predictive biomarker developments in multiple myeloma,”. J Hematol Oncol 2021, vol. 14(no. 1), 151. [Google Scholar] [CrossRef] [PubMed]
 |
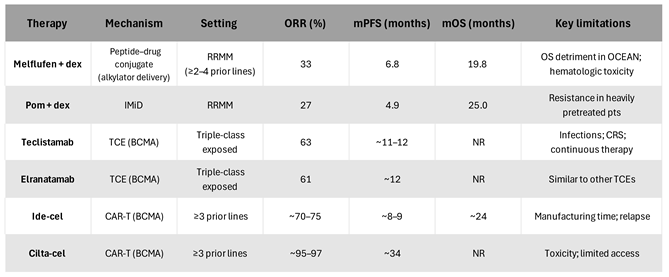 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




