Submitted:
14 April 2026
Posted:
15 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
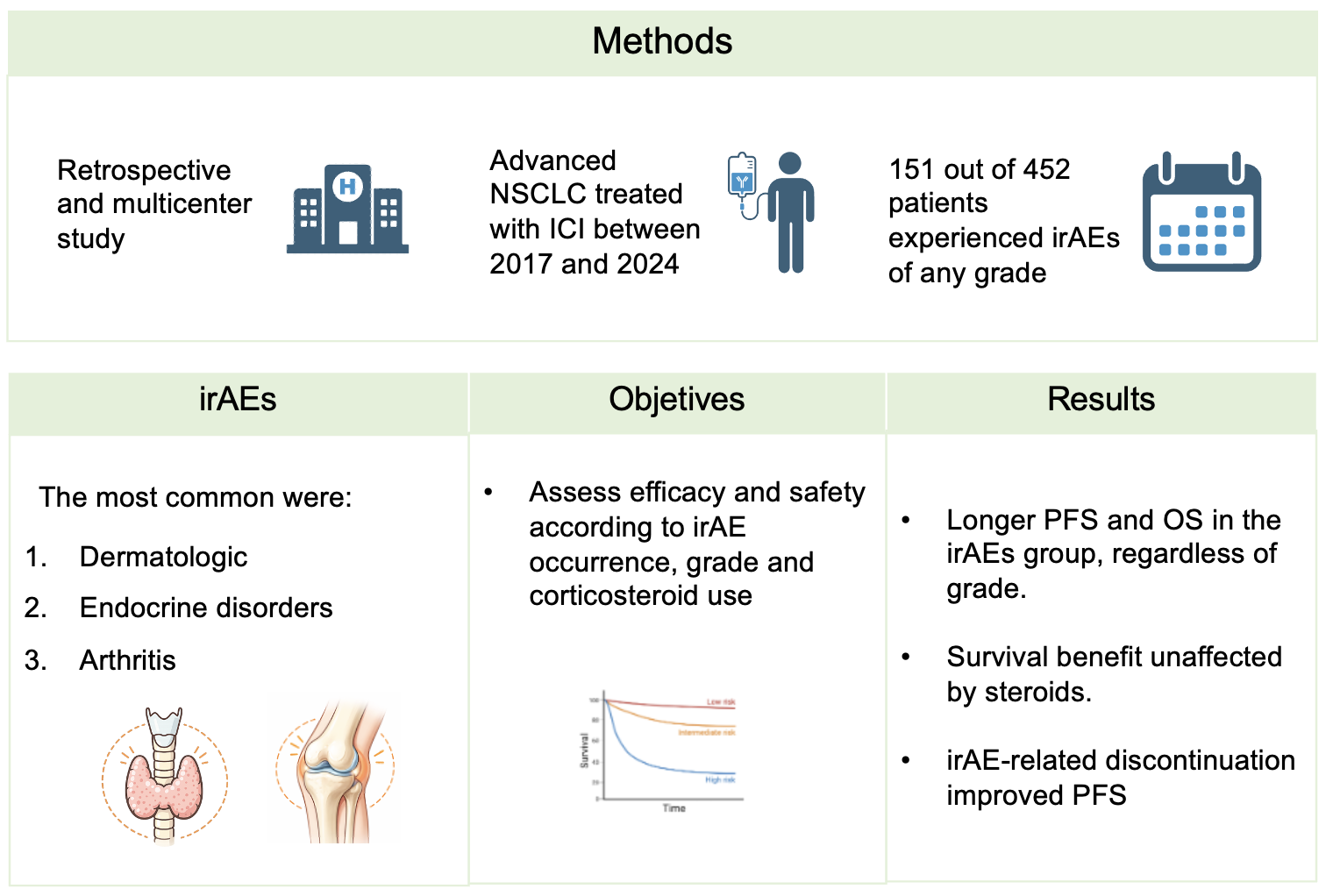
2.1. Study Design and Participants
2.2. Objectives
2.3. Statiscal Analysis
2.4. Review
3. Results
3.1. Patient Characteristics
3.2. Characterization of irAEs
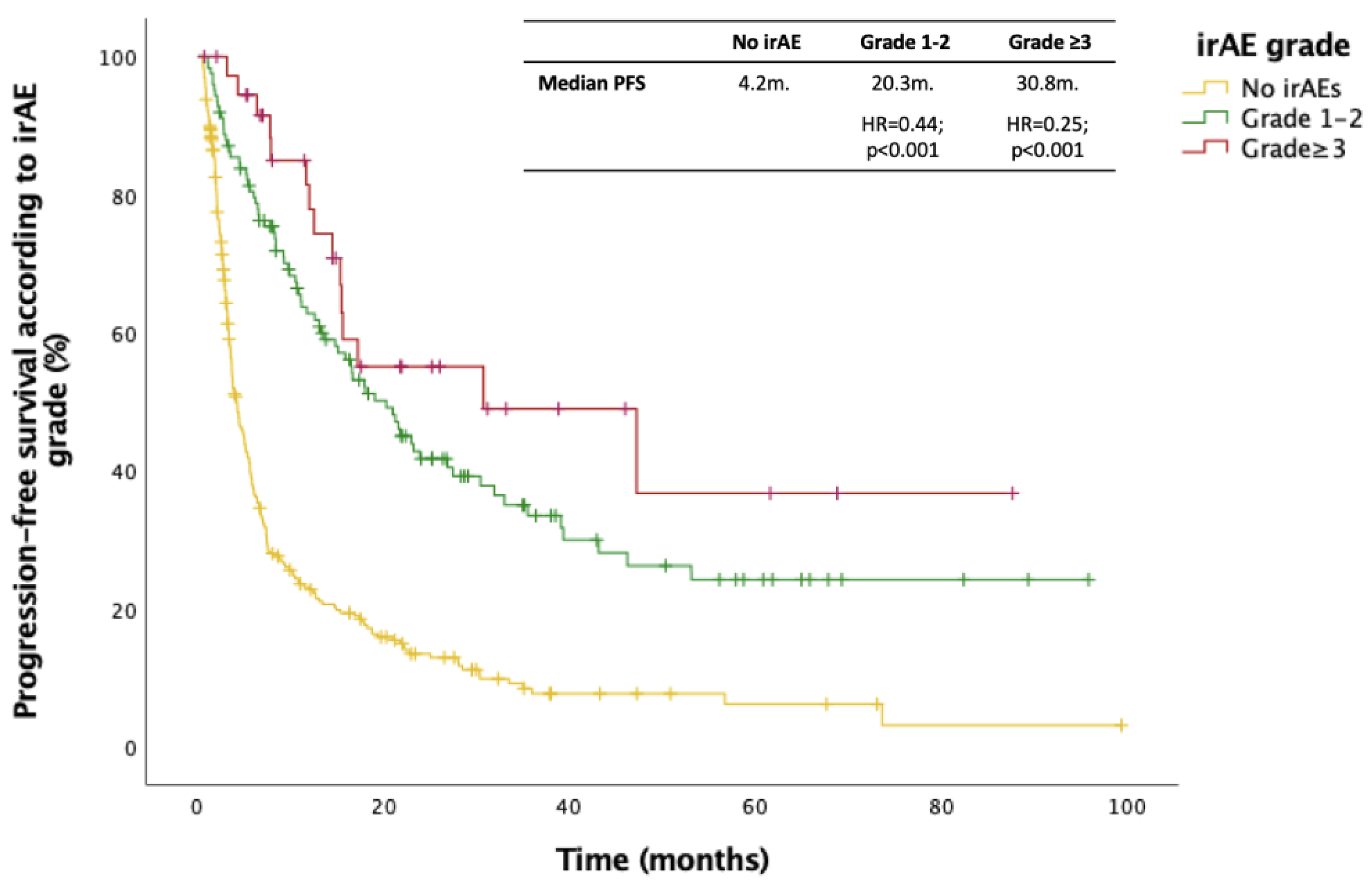
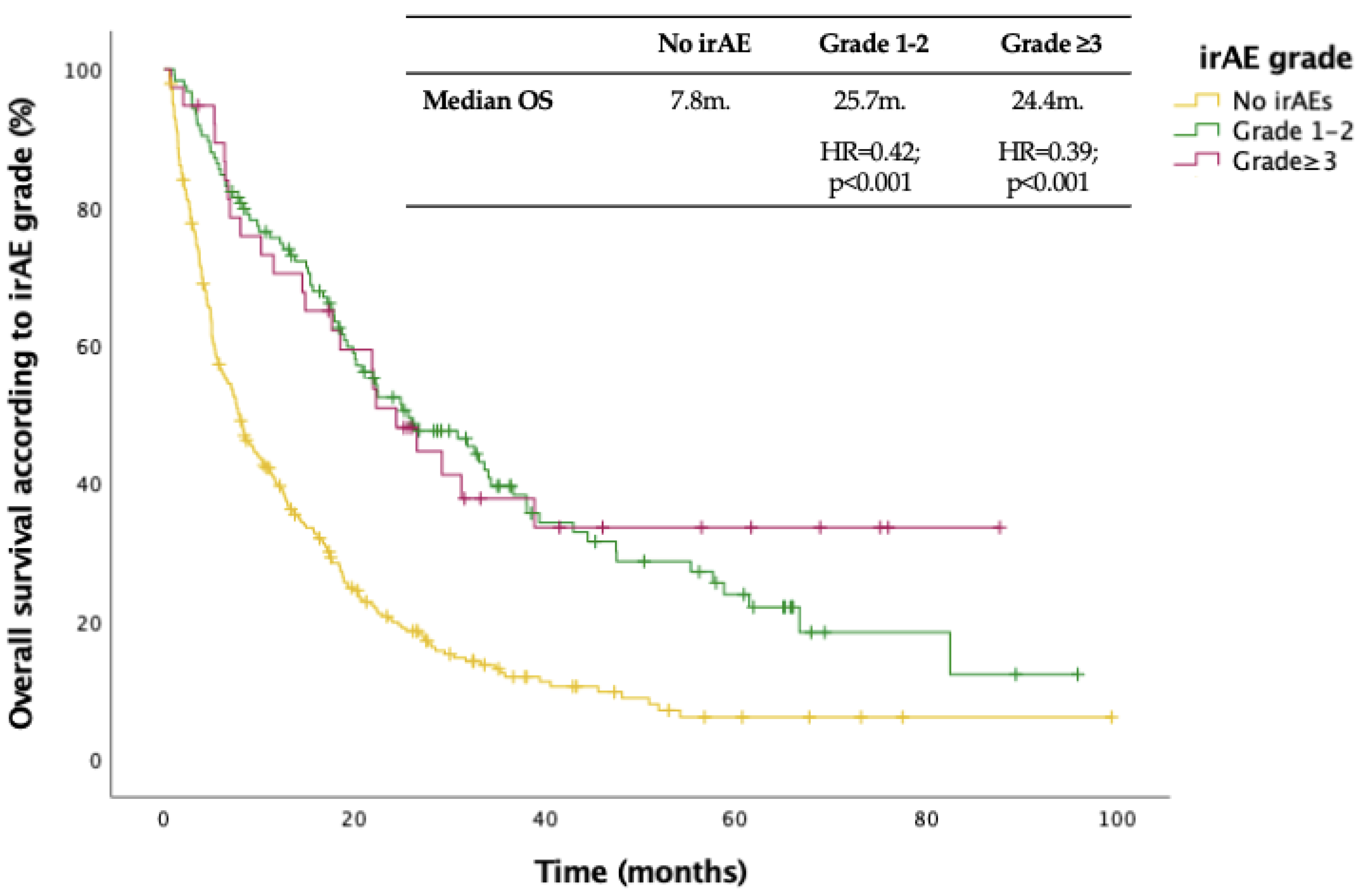
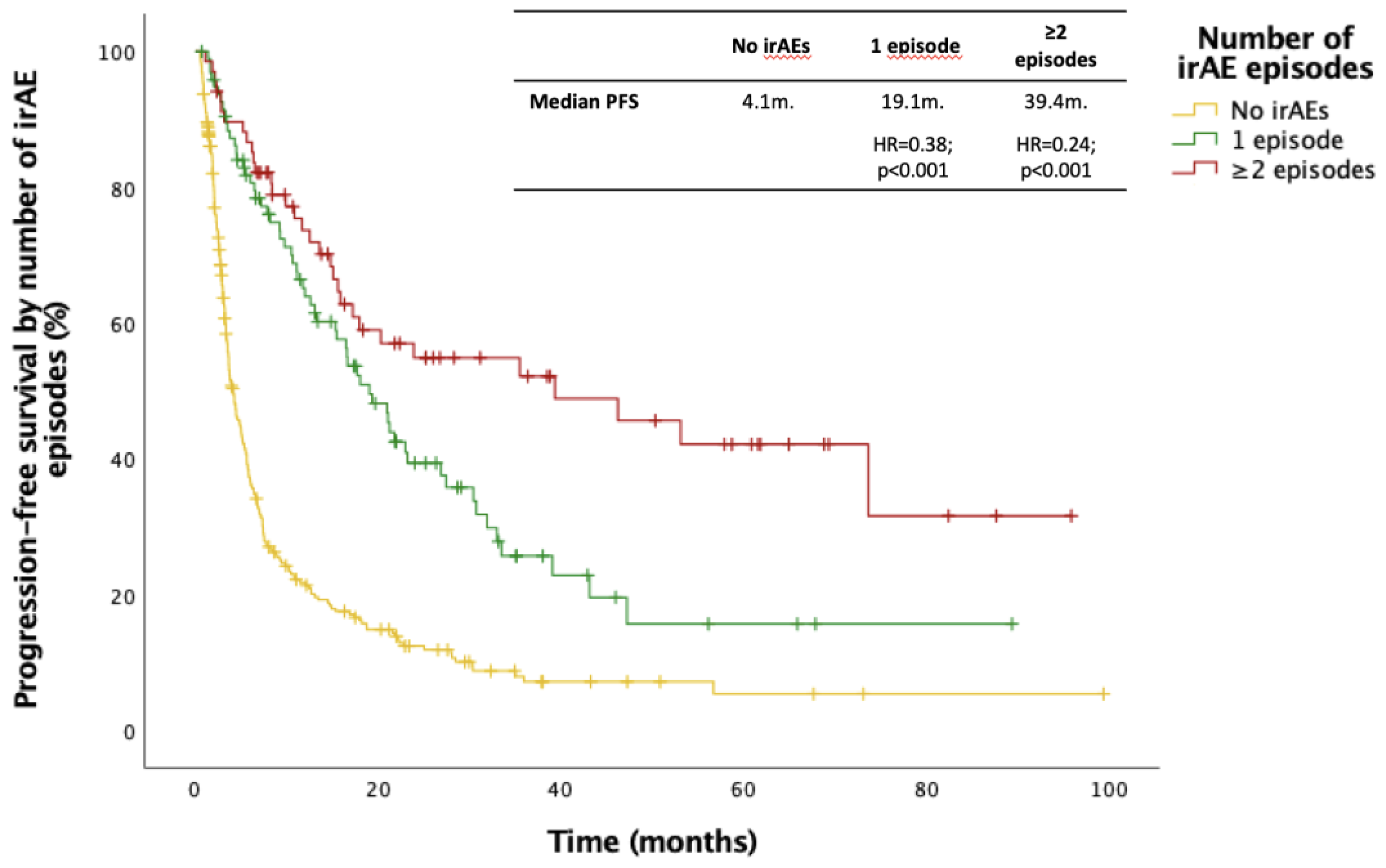
3.3. Impact of irAEs on Efficacy of ICIs
3.4. Impact of Corticosteroids on Effectiveness of ICIs
3.5. Multivariant Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ICIs | Immune checkpoint inhibitors |
| NSCLC | Non-small cell lung cancer |
| IRAEs | Immune-related adverse events |
| NCI CTCAE | National cancer institute common toxicity criteria for adverse events |
| CS | Corticosteroids |
| ORR | Objective response rate |
| PFS | Progression-free survival |
| OS | Overall survival |
| CR | Complete responses |
| PR | Partial responses |
| DCR | Disease control rate |
| SD | Stable disease |
| HR | Hazard ratios |
| CIs | Confidence intervals |
| ICI | Immune-checkpoints inhibitors |
| ECOG PS | Eastern cooperative oncology group performance status |
| GI | Gastrointestinal |
| NR | Not reached |
| NLR | Neutrophil-lymphocyte ratio |
| TTNT | Time to next therapy |
References
- Hendriks, L.; Kerr, K.; Menis, J.; Mok, T.; Nestle, U.; Passaro, A.; Peters, S.; Planchard, D.; Smit, E.; Solomon, B.; et al. Non-oncogene-addicted metastatic non-small-cell lung cancer: ESMO Clinical Practice Guideline for diagnosis, treatment and follow-up. Ann. Oncol. 2023, 34, 358–376. [Google Scholar] [CrossRef]
- Di Federico, A.; De Giglio, A.; Parisi, C.; Gelsomino, F.; Ardizzoni, A. PD-1/PD-L1 inhibitor monotherapy or in combination with chemotherapy as upfront treatment for advanced NSCLC with PD-L1 expression ≥ 50%: Selecting the best strategy. Crit. Rev. Oncol. 2021, 160, 103302. [Google Scholar] [CrossRef]
- Grangeon, M.; Tomasini, P.; Chaleat, S.; Jeanson, A.; Souquet-Bressand, M.; Khobta, N.; Bermudez, J.; Trigui, Y.; Greillier, L.; Blanchon, M.; et al. Association Between Immune-related Adverse Events and Efficacy of Immune Checkpoint Inhibitors in Non–small-cell Lung Cancer. Clin. Lung Cancer 2019, 20, 201–207. [Google Scholar] [CrossRef]
- National Cancer Institute. Common Terminology Criteria for Adverse Events (CTCAE) v5.0 [Internet]. 2017. Available online: https://www.meddra.org/.
- Haanen, J.; Obeid, M.; Spain, L.; Carbonnel, F.; Wang, Y.; Robert, C.; Lyon, A.; Wick, W.; Kostine, M.; Peters, S.; et al. Management of toxicities from immunotherapy: ESMO Clinical Practice Guideline for diagnosis, treatment and follow-up. Ann. Oncol. 2022, 33, 1217–1238. [Google Scholar] [CrossRef] [PubMed]
- Şen, G.A.; Öztaş, N.Ş; Değerli, E.; Safarov, S.; Guliyev, M.; Bedir, Ş; Can, G.; Turna, H.; Özgüroğlu, M. Effects of immune related adverse events and corticosteroids on the outcome of patients treated with immune checkpoint inhibitors. Sci. Rep. 2025, 15, 1–9. [Google Scholar] [CrossRef]
- Arbour, K.C.; Mezquita, L.; Long, N.; Rizvi, H.; Auclin, E.; Ni, A.; Martínez-Bernal, G.; Ferrara, R.; Lai, W.V.; Hendriks, L.E.L.; et al. Impact of Baseline Steroids on Efficacy of Programmed Cell Death-1 and Programmed Death-Ligand 1 Blockade in Patients With Non–Small-Cell Lung Cancer. J. Clin. Oncol. 2018, 36, 2872–2878. [Google Scholar] [CrossRef]
- Petrelli, F.; Signorelli, D.; Ghidini, M.; Ghidini, A.; Pizzutilo, E.G.; Ruggieri, L.; Cabiddu, M.; Borgonovo, K.; Dognini, G.; Brighenti, M.; et al. Association of Steroids Use with Survival in Patients Treated with Immune Checkpoint Inhibitors: A Systematic Review and Meta-Analysis. Cancers 2020, 12, 546. [Google Scholar] [CrossRef]
- Verheijden, R.J.; de Groot, J.S.; Fabriek, B.O.; Hew, M.N.; May, A.M.; Suijkerbuijk, K.P. Corticosteroids for Immune-Related Adverse Events and Checkpoint Inhibitor Efficacy: Analysis of Six Clinical Trials. J. Clin. Oncol. 2024, 42, 3713–3724. [Google Scholar] [CrossRef]
- Skribek, M.; Rounis, K.; Afshar, S.; Grundberg, O.; Friesland, S.; Tsakonas, G.; Ekman, S.; De Petris, L. Effect of corticosteroids on the outcome of patients with advanced non–small cell lung cancer treated with immune-checkpoint inhibitors. Eur. J. Cancer 2021, 145, 245–254. [Google Scholar] [CrossRef] [PubMed]
- Berti, A.; Bortolotti, R.; Dipasquale, M.; Kinspergher, S.; Prokop, L.; Grandi, G.; Inchiostro, S.; Paolazzi, G.; Caffo, O.; Veccia, A. Meta-analysis of immune-related adverse events in phase 3 clinical trials assessing immune checkpoint inhibitors for lung cancer. Crit. Rev. Oncol. 2021, 162, 103351. [Google Scholar] [CrossRef] [PubMed]
- Das, S.; Johnson, D.B. Immune-related adverse events and anti-tumor efficacy of immune checkpoint inhibitors. J. Immunother. Cancer 2019, 7, 306. [Google Scholar] [CrossRef]
- Sung, M.; Zer, A.; Walia, P.; Khoja, L.; Maganti, M.; Labbe, C.; Shepherd, F.A.; Bradbury, P.A.; Liu, G.; Leighl, N.B. Correlation of immune-related adverse events and response from immune checkpoint inhibitors in patients with advanced non-small cell lung cancer. J. Thorac. Dis. 2020, 12, 2706–2712. [Google Scholar] [CrossRef]
- Daniello, L.; Elshiaty, M.; Bozorgmehr, F.; Kuon, J.; Kazdal, D.; Schindler, H.; Shah, R.; Volckmar, A.-L.; Lusky, F.; Diekmann, L.; et al. Therapeutic and Prognostic Implications of Immune-Related Adverse Events in Advanced Non-Small-Cell Lung Cancer. Front. Oncol. 2021, 11, 703893. [Google Scholar] [CrossRef]
- Cortellini, A.; Chiari, R.; Ricciuti, B.; Metro, G.; Perrone, F.; Tiseo, M.; Bersanelli, M.; Bordi, P.; Santini, D.; Giusti, R.; et al. Correlations Between the Immune-related Adverse Events Spectrum and Efficacy of Anti-PD1 Immunotherapy in NSCLC Patients. Clin. Lung Cancer 2019, 20, 237–247.e1. [Google Scholar] [CrossRef]
- Grangeon, M.; Tomasini, P.; Chaleat, S.; Jeanson, A.; Souquet-Bressand, M.; Khobta, N.; Bermudez, J.; Trigui, Y.; Greillier, L.; Blanchon, M.; et al. Association Between Immune-related Adverse Events and Efficacy of Immune Checkpoint Inhibitors in Non–small-cell Lung Cancer. Clin. Lung Cancer 2019, 20, 201–207. [Google Scholar] [CrossRef]
- Guezour, N.; Soussi, G.; Brosseau, S.; Abbar, B.; Naltet, C.; Vauchier, C.; Poté, N.; Hachon, L.; Namour, C.; Khalil, A.; et al. Grade 3–4 Immune-Related Adverse Events Induced by Immune Checkpoint Inhibitors in Non-Small-Cell Lung Cancer (NSCLC) Patients Are Correlated with Better Outcome: A Real-Life Observational Study. Cancers 2022, 14, 3878. [Google Scholar] [CrossRef]
- Eggermont, A.M.M.; Kicinski, M.; Blank, C.U.; Mandala, M.; Long, G.V.; Atkinson, V.; Dalle, S.; Haydon, A.; Khattak, A.; Carlino, M.S.; et al. Association Between Immune-Related Adverse Events and Recurrence-Free Survival Among Patients With Stage III Melanoma Randomized to Receive Pembrolizumab or Placebo. JAMA Oncol. 2020, 6, 519–527. [Google Scholar] [CrossRef] [PubMed]
- Schweizer, C.; Schubert, P.; Rutzner, S.; Eckstein, M.; Haderlein, M.; Lettmaier, S.; Semrau, S.; Gostian, A.-O.; Frey, B.; Gaipl, U.S.; et al. Prospective evaluation of the prognostic value of immune-related adverse events in patients with non-melanoma solid tumour treated with PD-1/PD-L1 inhibitors alone and in combination with radiotherapy. Eur. J. Cancer 2020, 140, 55–62. [Google Scholar] [CrossRef] [PubMed]
- Osorio, J.C.; Ni, A.; Chaft, J.E.; Pollina, R.; Kasler, M.K.; Stephens, D.; Rodriguez, C.; Cambridge, L.; Rizvi, H.; Wolchok, J.D.; et al. Antibody-mediated thyroid dysfunction during T-cell checkpoint blockade in patients with non-small-cell lung cancer. Ann. Oncol. 2017, 28, 583–589. [Google Scholar] [CrossRef] [PubMed]
- Lin, L.; Liu, Y.; Chen, C.; Wei, A.; Li, W. Association between immune-related adverse events and immunotherapy efficacy in non-small-cell lung cancer: a meta-analysis. Front. Pharmacol. 2023, 14. [Google Scholar] [CrossRef]
- Ma, S.; Nie, H.; Wei, C.; Jin, C.; Wang, L. Association between immune-related adverse events and prognosis in patients with advanced non-small cell lung cancer: a systematic review and meta-analysis. Front. Oncol. 2024, 14, 1402017. [Google Scholar] [CrossRef] [PubMed]
- Liang, Y.; Xu, H.; Liu, F.; Li, L.; Lin, C.; Zhang, Y.; Wang, N.; Wang, L. Immune-related adverse events and their effects on survival outcomes in patients with non-small cell lung cancer treated with immune checkpoint inhibitors: a systematic review and meta-analysis. Front. Oncol. 2024, 14, 1281645. [Google Scholar] [CrossRef] [PubMed]
- Naqash, A.R.; Ricciuti, B.; Owen, D.H.; Florou, V.; Toi, Y.; Cherry, C.; Hafiz, M.; De Giglio, A.; Muzaffar, M.; Patel, S.H.; et al. Correction to: Outcomes associated with immune-related adverse events in metastatic non-small cell lung cancer treated with nivolumab: a pooled exploratory analysis from a global cohort. Cancer Immunol. Immunother. 2020, 69, 1189–1189. [Google Scholar] [CrossRef] [PubMed]
- Weber, J.S.; Hodi, F.S.; Wolchok, J.D.; Topalian, S.L.; Schadendorf, D.; Larkin, J.; Sznol, M.; Long, G.V.; Li, H.; Waxman, I.M.; et al. Safety Profile of Nivolumab Monotherapy: A Pooled Analysis of Patients With Advanced Melanoma. J. Clin. Oncol. 2017, 35, 785–792. [Google Scholar] [CrossRef]
- Lisberg, A.; Tucker, D.A.; Goldman, J.W.; Wolf, B.; Carroll, J.; Hardy, A.; Morris, K.; Linares, P.; Adame, C.; Spiegel, M.L.; et al. Treatment-Related Adverse Events Predict Improved Clinical Outcome in NSCLC Patients on KEYNOTE-001 at a Single Center. Cancer Immunol. Res. 2018, 6, 288–294. [Google Scholar] [CrossRef]
- van Dijk, E.; Smeenk, M.; Bensch, F.; Sadowska, A.; Verschueren, M.; Verhaert, M.; Llobell, A.; Suzuki, Y.; Liew, D.; Takeji, A.; et al. Checkpoint inhibitor effectiveness after corticosteroids and second-line immunosuppressants for immune-related adverse events in non-small-cell lung cancer. ESMO Open 2026, 11, 106052. [Google Scholar] [CrossRef]
| Overall, n (%) (n=452) | irAEs (%) (n=167) | No irAEs (%) (n=285) | p- value | |
|---|---|---|---|---|
| Age, y | 0.384 | |||
| ≥65 | 269 (59.5%) | 95 (56.9% | 174 (61.1%) | |
| <65 | 183 (40.5%) | 72 (43.1%) | 111 (38.9%) | |
| Sex | 0.814 | |||
| Male | 371 (82.1%) | 138 (82.6%) | 233 (81.8%) | |
| Female | 81 (17.9%) | 29 (17.4%) | 52 (18.2%) | |
| Smoking status | 0.008 | |||
| Current smoker | 202 (44.7%) | 91 (54.5%) | 113 (39.6%) | |
| Never-smoker | 40 (8.8%) | 11 (6.6%) | 29 (10.2%) | |
| Former smoker | 210 (46.5%) | 65 (38.9%) | 143 (50.2%) | |
| Histology | 0.18 | |||
| Adenocarcinoma | 264 (58.4%) | 101 (60.5%) | 163 (57.2%) | |
| Squamous cell carc. | 152 (33.6%) | 50 (29.9%) | 102 (35.8%) | |
| NOS | 33 (7.3%) | 14 (8.4%) | 19 (6.7%) | |
| Other | 3 (0.9%) | 2 (1.2%) | 1 (0.4%) | |
| Stage | 0.197 | |||
| Recurrent IIIA | 17 (3.8%) | 6 (3.6%) | 11 (3.9%) | |
| Recurrent IIIB | 17 (3.8%) | 8 (4.8%) | 9 (3.2%) | |
| Recurrent IIIC | 2 (0.4%) | 0 (0.0%) | 2 (0.7%) | |
| M1a | 158 (34.9%) | 64 (38.3%) | 94 (33.0%) | |
| M1b | 85 (18.8%) | 37 (22.2%) | 48 (16.8%) | |
| M1c | 173 (38.3%) | 52 (31.1%) | 121 (42.5%) | |
| M1 location | ||||
| Brain | 73 (16.2%) | 25 (15.0%) | 48 (16.8%) | 0.602 |
| Liver | 59 (10.1%) | 20 (12.0%) | 39 (13.7%) | 0.603 |
| Bone | 115 (25.4%) | 33 (19.8%) | 82 (28.8%) | 0.034 |
| Adrenal | 73 (16.2%) | 32 (19.2%) | 41 (14.4%) | 0.183 |
| PDL1 | <0.001 | |||
| ≥ 50% | 183 (40.4%) | 93 (55.7%) | 90 (31.6%) | |
| 1-49% | 101 (22.4%) | 33 (19.8%) | 68 (23.9%) | |
| <1% | 101 (22.4%) | 23 (13.8%) | 78 (27.4%) | |
| Unknown | 67 (14.8%) | 18 (10.8%) | 49 (17.2%) | |
| Treatment line | 0.128 | |||
| First line | 263 (58.4%) | 109 (65.3%) | 154 (54.0%) | |
| Second line | 153 (33.8%) | 50 (29.9%) | 103 (36.1%) | |
| Third and beyond | 36 (7.7%) | 8 (4.8%) | 28 (9.8%) | |
| Treatment regimen | 0.008 | |||
| Chemotherapy-ICI | 149 (32.9%) | 51 (30.5%) | 98 (34.4%) | |
| Pembrolizumab | 134 (29.9%) | 65 (38.9%) | 69 (24.2%) | |
| Atezolizumab | 116 (25.7%) | 33 (19.8%) | 83 (29.1%) | |
| Nivolumab | 53 (11.5%) | 18 (10.8%) | 35 (12.3%) | |
| ECOG PS | 0.003 | |||
| 0-1 | 398 (88.1%) | 157 (94%) | 241 (84.6%) | |
| ≥ 2 | 54 (11.9%) | 10 (6.0%) | 44 (15.4%) | |
| Subsequent therapy | 0.266 | |||
| 0 subsequent line | 276 (61.7%) | 106 (63.5%) | 170 (59.6%) | |
| 1 subsequent line | 136 (29.6%) | 43 (25.7%) | 93 (32.6%) | |
| ≥2 subsequent lines | 39 (8.6%) | 17 (10.2%) | 22 (7.7%) | |
| Driver mutations | 0.044 | |||
| ALK translocation | 2 (0.4%) | 2 (1.2%) | 0 (0.0%) | |
| EGFR | 18 (4%) | 2 (1.2%) | 16 (5.6%) | |
| KRAS | 52 (10%) | 15 (9.0%) | 37 (13.0%) | |
| BRAF | 8 (2%) | 2 (1.2%) | 6 (2.1%) | |
| MET | 3 (0.4%) | 1 (0.6%) | 2 (0.7%) |
| Total, n=452 (%) | Grade 1-2 | Grade ≥3 | |
|---|---|---|---|
| Affected organ | |||
| Skin | 50 (11.1%) | 38 (8.4%) | 12 (2.7%) |
| Endocrine | 41 (9.1%) | 39 (8.6%) | 2 (0.4%) |
| Arthritis | 25 (5.5%) | 21 (4.6%) | 4 (0.9%) |
| Pneumonitis | 25 (5.5%) | 17 (3.8%) | 8 (1.8%) |
| GI | 23 (5.1%) | 21 (4.6%) | 2 (0.4%) |
| Hepatitis | 18 (4.0%) | 10 (2.2%) | 8 (1.8%) |
| Nephritis | 8 (1.8%) | 7 (1.5%) | 1 (0.2%) |
| Mucositis | 6 (1.3%) | 6 (1.3%) | 0 (0.0%) |
| Neurologic | 5 (1.1%) | 4 (0.9%) | 1 (0.2%) |
| irAEs | irAEs and others | Others | p value | |
|---|---|---|---|---|
| DCR | 90% | 93.3% | 49.4% | <0.001 |
| ORR | 65% | 60% | 31.3% | <0.001 |
| PFS, months | 46.3 | 21.3 | 4.5 | – |
| HR for PFS vs. others (95% CI) | 0.26 (0.17–0.41) |
0.39 (0.18–0.83) |
Reference | <0.001 and 0.014 |
| PFS | OS | |||
|---|---|---|---|---|
| HR | p-value | HR | p-value | |
| Sex | ||||
| Male | ||||
| Female | 1.12 (0.71-1.75) | 0.639 | 1.04 (0.67-1.63) | 0.857 |
| Age | ||||
| <65 | ||||
| ≥65 | 0.94 (0.64-1.39) | 0.753 | 1.26 (0.86-1.85) | 0.242 |
| Histology | ||||
| Squamous cell carc. | ||||
| Adenocarcinoma | 1.11 (0.70-1.76) | 0.658 | 1.15 (0.75-1.78) | 0.522 |
| ECOG | ||||
| 0-1 | ||||
| ≥2 | 2.29 (1.38-3.80) | 0.001 | 3.05 (1.86-5.00) | <0.001 |
| NLR | ||||
| <5 | ||||
| ≥5 | 0.88 (0.60-1.31) | 0.533 | 1.04 (0.71-1.52) | 0.830 |
| Stage | ||||
| III/IVA | ||||
| IVB | 1.46 (0.95-2.25) | 0.083 | 1.62 (1.08-2.42) | 0.019 |
| Brain metastases | ||||
| No | ||||
| Yes | 0.88 (0.55-1.40) | 0.594 | 0.81 (0.52-1.26) | 0.343 |
| PD-L1 (%) | ||||
| <1% | ||||
| 1-49% | 0.67 (0.40-1.11) | 0.123 | 0.55 (0.34-0.90) | 0.017 |
| ≥50% | 0.47 (0.29-0.76) | 0.003 | 0.48 (0.30-0.76) | 0.002 |
| Treatment line | ||||
| First line | ||||
| Second line | 0.75 (0.47-1.20) | 0.229 | 0.83 (0.53-1.31) | 0.421 |
| Third and beyond | 1.13 (0.55-2.35) | 0.736 | 0.93 (0.45-1.93) | 0.840 |
| irAEs | ||||
| No irAEs | ||||
| irAEs | 0.40 (0.23-0.71) | 0.002 | 0.44 (0.26-0.77) | 0.004 |
| CS indication | ||||
| Non-irAE | ||||
| irAE and non-irAE | 0.55 (0.18-1.69) | 0.299 | 0.77 (0.30-2.01) | 0.602 |
| irAE | 0.49 (0.25-0.97) | 0.041 | 0.52 (0.27-0.99) | 0.045 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




