Submitted:
14 April 2026
Posted:
22 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Pathogenesis of DSRCT
3. Progress on Targeted Therapeutic
3.1. Receptor Tyrosine Kinase (RTK)
3.1.1. PDGF/PDGFR
3.1.2. VEGF/VEGFR
3.1.3. FGFR4
3.1.4. IGF /IGF1R
3.1.5. ERBB
3.1.6. c-KIT(CD117)
3.1.7. NTRK3
3.1.8. MERTK
3.2. mTOR
3.3. Androgen Receptors
3.4. B7H3(CD276)
3.5. CDK4/6
3.6. SIK1
3.7. Other Targets
4. Current Status and Advances in Immunotherapy
4.1. PD1-PDL1
4.2. B7H3
4.3. GD2
4.4. Other Immune Targets
5. DNA Damage Response (DDR)
6. Discussion
7. Conclusion and Outlook
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Reisner D, Brahee D, Patel S,et al. A Case of Desmoplastic Small Round Cell Tumor[J]. J Radiol Case Rep, 2015, 9(8): 1-7. [CrossRef]
- Gaffney E F, Breatnach F. Diverse immunoreactivity and metachronous ultrastructural variability in fatal primitive childhood tumor with rhabdoid features[J]. Arch Pathol Lab Med, 1989, 113(12): 1322.
- Gerald W L, Miller H K, Battifora H,et al. Intra-abdominal desmoplastic small round-cell tumor. Report of 19 cases of a distinctive type of high-grade polyphenotypic malignancy affecting young individuals[J]. Am J Surg Pathol, 1991, 15(6): 499-513.
- Mora J, Modak S, Cheung N K,et al. Desmoplastic small round cell tumor 20 years after its discovery[J]. Future Oncol, 2015, 11(7): 1071-81. [CrossRef]
- Zhang W D, Li C X, Liu Q Y,et al. CT, MRI, and FDG-PET/CT imaging findings of abdominopelvic desmoplastic small round cell tumors: correlation with histopathologic findings[J]. Eur J Radiol, 2011, 80(2): 269-73. [CrossRef]
- Arora V C, Price A P, Fleming S,et al. Characteristic imaging features of desmoplastic small round cell tumour[J]. Pediatr Radiol, 2013, 43(1): 93-102. [CrossRef]
- Hendricks A, Boerner K, Germer C T,et al. Desmoplastic Small Round Cell Tumors: A review with focus on clinical management and therapeutic options[J]. Cancer Treat Rev, 2021, 93: 102140. [CrossRef]
- Bulbul A, Shen J P, Xiu J,et al. Genomic and Proteomic Alterations in Desmoplastic Small Round Blue-Cell Tumors[J]. JCO Precis Oncol, 2018, 2. [CrossRef]
- Lae M E, Roche P C, Jin L,et al. Desmoplastic small round cell tumor: a clinicopathologic, immunohistochemical, and molecular study of 32 tumors[J]. Am J Surg Pathol, 2002, 26(7): 823-35. [CrossRef]
- Dufresne A, Cassier P, Couraud L,et al. Desmoplastic small round cell tumor: current management and recent findings[J]. Sarcoma, 2012, 2012: 714986. [CrossRef]
- Honoré C, Delhorme J B, Nassif E,et al. Can we cure patients with abdominal Desmoplastic Small Round Cell Tumor? Results of a retrospective multicentric study on 100 patients[J]. Surg Oncol, 2019, 29: 107-112. [CrossRef]
- Hatanaka K C, Takakuwa E, Hatanaka Y,et al. Desmoplastic small round cell tumor of the parotid gland-report of a rare case and a review of the literature[J]. Diagn Pathol, 2019, 14(1): 43. [CrossRef]
- Wang LL, Ji ZH, Gao Y, et al. Analysis of Clinical and Pathological Features of Desmoplastic Small Round Cell Tumor[J]. Chinese Journal of Clinical Oncology, 2020, 47(02): 72-76. [CrossRef]
- Bulbul A, Fahy B N, Xiu J,et al. Desmoplastic Small Round Blue Cell Tumor: A Review of Treatment and Potential Therapeutic Genomic Alterations[J]. Sarcoma, 2017, 2017: 1278268. [CrossRef]
- Scheer M, Vokuhl C, Blank B,et al. Desmoplastic small round cell tumors: Multimodality treatment and new risk factors[J]. Cancer Med, 2019, 8(2): 527-542. [CrossRef]
- Stiles Z E, Dickson P V, Glazer E S,et al. Desmoplastic small round cell tumor: A nationwide study of a rare sarcoma[J]. J Surg Oncol, 2018, 117(8): 1759-1767. [CrossRef]
- Sundaramoorthy S, Abraham G, Narayanan G,et al. Management and survival outcomes of desmoplastic small round cell tumor: a retrospective cohort study from a tertiary cancer center[J]. BMC Cancer, 2025, 26(1): 123. [CrossRef]
- Magrath J W, Goldberg I N, Truong D D,et al. Enzalutamide induces cytotoxicity in desmoplastic small round cell tumor independent of the androgen receptor[J]. Commun Biol, 2024, 7(1): 411. [CrossRef]
- Geyer F H, Ritter A, Kinn-Gurzo S,et al. Comprehensive DSRCT multi-omics analyses unveil CACNA2D2 as a diagnostic hallmark and super-enhancer-driven EWSR1::WT1 signature gene[J]. Cancer Commun (Lond), 2025, 45(6): 702-708. [CrossRef]
- Giani C, Radaelli S, Miceli R,et al. Long-term survivors with desmoplastic small round cell tumor (DSRCT): Results from a retrospective single-institution case series analysis[J]. Cancer Med, 2023, 12(9): 10694-10703. [CrossRef]
- Magrath J W, Sampath S S, Flinchum D A,et al. Comprehensive Transcriptomic Analysis of EWSR1::WT1 Targets Identifies CDK4/6 Inhibitors as an Effective Therapy for Desmoplastic Small Round Cell Tumors[J]. Cancer Res, 2024, 84(9): 1426-1442. [CrossRef]
- Magrath J W, Kang H J, Hartono A,et al. Desmoplastic small round cell tumor cancer stem cell-like cells resist chemotherapy but remain dependent on the EWSR1-WT1 oncoprotein[J]. Front Cell Dev Biol, 2022, 10: 1048709. [CrossRef]
- Gerald W L, Ladanyi M, De Alava E,et al. Clinical, pathologic, and molecular spectrum of tumors associated with t(11;22)(p13;q12): desmoplastic small round-cell tumor and its variants[J]. J Clin Oncol, 1998, 16(9): 3028-36. [CrossRef]
- Wang L L, Ji Z H, Gao Y,et al. Clinicopathological features of desmoplastic small round cell tumors: clinical series and literature review[J]. World J Surg Oncol, 2021, 19(1): 193. [CrossRef]
- Thway K, Noujaim J, Zaidi S,et al. Desmoplastic Small Round Cell Tumor: Pathology, Genetics, and Potential Therapeutic Strategies[J]. Int J Surg Pathol, 2016, 24(8): 672-684. [CrossRef]
- Schoolmeester J K, Folpe A L, Nair A A,et al. EWSR1-WT1 gene fusions in neoplasms other than desmoplastic small round cell tumor: a report of three unusual tumors involving the female genital tract and review of the literature[J]. Mod Pathol, 2021, 34(10): 1912-1920. [CrossRef]
- Sawyer J R, Tryka A F, Lewis J M. A novel reciprocal chromosome translocation t(11;22)(p13;q12) in an intraabdominal desmoplastic small round-cell tumor[J]. Am J Surg Pathol, 1992, 16(4): 411-6. [CrossRef]
- Ladanyi M, Gerald W. Fusion of the EWS and WT1 genes in the desmoplastic small round cell tumor[J]. Cancer Res, 1994, 54(11): 2837-40.
- Gerald W L, Rosai J, Ladanyi M. Characterization of the genomic breakpoint and chimeric transcripts in the EWS-WT1 gene fusion of desmoplastic small round cell tumor[J]. Proc Natl Acad Sci U S A, 1995, 92(4): 1028-32. [CrossRef]
- Dermawan J K, Slotkin E, Tap W D,et al. Chromoplexy Is a Frequent Early Clonal Event in EWSR1-Rearranged Round Cell Sarcomas That Can Be Detected Using Clinically Validated Targeted Sequencing Panels[J]. Cancer Res, 2024, 84(9): 1504-1516. [CrossRef]
- Gerald W L, Haber D A. The EWS-WT1 gene fusion in desmoplastic small round cell tumor[J]. Semin Cancer Biol, 2005, 15(3): 197-205. [CrossRef]
- Magro G, Broggi G, Zin A,et al. Desmoplastic Small Round Cell Tumor with "Pure" Spindle Cell Morphology and Novel EWS-WT1 Fusion Transcript: Expanding the Morphological and Molecular Spectrum of This Rare Entity[J]. Diagnostics (Basel), 2021, 11(3). [CrossRef]
- Magrath J W, Flinchum D A, Hartono A B,et al. Genomic Breakpoint Characterization and Transcriptome Analysis of Metastatic, Recurrent Desmoplastic Small Round Cell Tumor[J]. Sarcoma, 2023, 2023: 6686702. [CrossRef]
- Shi C, Feng Y, Zhang L C,et al. Effective treatment of apatinib in desmoplastic small round cell tumor: a case report and literature review[J]. BMC Cancer, 2018, 18(1): 338. [CrossRef]
- Yang L, Han Y, Suarez Saiz F,et al. A tumor suppressor and oncogene: the WT1 story[J]. Leukemia, 2007, 21(5): 868-76. [CrossRef]
- Anderson P M, Tu Z J, Kilpatrick S E,et al. Routine EWS Fusion Analysis in the Oncology Clinic to Identify Cancer-Specific Peptide Sequence Patterns That Span Breakpoints in Ewing Sarcoma and DSRCT[J]. Cancers (Basel), 2023, 15(5). [CrossRef]
- Lee J, Nguyen P T, Shim H S,et al. EWSR1, a multifunctional protein, regulates cellular function and aging via genetic and epigenetic pathways[J]. Biochim Biophys Acta Mol Basis Dis, 2019, 1865(7): 1938-1945. [CrossRef]
- Bleijs M, Pleijte C, Engels S,et al. EWSR1-WT1 Target Genes and Therapeutic Options Identified in a Novel DSRCT In Vitro Model[J]. Cancers (Basel), 2021, 13(23). [CrossRef]
- Boulay G, Broye L C, Dong R,et al. EWS-WT1 fusion isoforms establish oncogenic programs and therapeutic vulnerabilities in desmoplastic small round cell tumors[J]. Nat Commun, 2024, 15(1): 7460. [CrossRef]
- Bétrian S, Bergeron C, Blay J Y,et al. Antiangiogenic effects in patients with progressive desmoplastic small round cell tumor: data from the French national registry dedicated to the use of off-labeled targeted therapy in sarcoma (OUTC's)[J]. Clin Sarcoma Res, 2017, 7: 10. [CrossRef]
- Italiano A, Kind M, Cioffi A,et al. Clinical activity of sunitinib in patients with advanced desmoplastic round cell tumor: a case series[J]. Target Oncol, 2013, 8(3): 211-213. [CrossRef]
- Chen H M, Feng G. Use of anlotinib in intra-abdominal desmoplastic small round cell tumors: a case report and literature review[J]. Onco Targets Ther, 2019, 12: 57-61. [CrossRef]
- Jing X Y, Shen C Q, He G Q,et al. Effective Treatment of Anlotinib Combined With Chemotherapy in Children With Desmoplastic Small Round Cell Tumor: A Case Series in a Single-center and Literature Review[J]. J Pediatr Hematol Oncol, 2024, 46(3): 159-164. [CrossRef]
- Frezza A M, Benson C, Judson I R,et al. Pazopanib in advanced desmoplastic small round cell tumours: a multi-institutional experience[J]. Clin Sarcoma Res, 2014, 4: 7. [CrossRef]
- Menegaz B A, Cuglievan B, Benson J,et al. Clinical Activity of Pazopanib in Patients with Advanced Desmoplastic Small Round Cell Tumor[J]. Oncologist, 2018, 23(3): 360-366. [CrossRef]
- Van Der Graaf W T, Blay J Y, Chawla S P,et al. Pazopanib for metastatic soft-tissue sarcoma (PALETTE): a randomised, double-blind, placebo-controlled phase 3 trial[J]. Lancet, 2012, 379(9829): 1879-86. [CrossRef]
- Glade Bender J L, Lee A, Reid J M,et al. Phase I pharmacokinetic and pharmacodynamic study of pazopanib in children with soft tissue sarcoma and other refractory solid tumors: a children's oncology group phase I consortium report[J]. J Clin Oncol, 2013, 31(24): 3034-43. [CrossRef]
- Williams L T, Escobedo J A, Keating M T,et al. Signal Transduction by the Platelet-derived Growth Factor Receptor[J]. Cold Spring Harbor Symposia on Quantitative Biology, 1988, 53 Pt 1: 455-465. [CrossRef]
- Lee S B, Kolquist K A, Nichols K,et al. The EWS-WT1 translocation product induces PDGFA in desmoplastic small round-cell tumour[J]. Nature Genetics, 1997, 17(3): 309-13. [CrossRef]
- Liu J, Nau M M, Yeh J C,et al. Molecular heterogeneity and function of EWS-WT1 fusion transcripts in desmoplastic small round cell tumors[J]. Clin Cancer Res, 2000, 6(9): 3522-9.
- Vignaud J M, Marie B, Klein N,et al. The role of platelet-derived growth factor production by tumor-associated macrophages in tumor stroma formation in lung cancer[J]. Cancer Res, 1994, 54(20): 5455-63.
- Lee S B, Kolquist K A, Nichols K,et al. The EWS-WT1 translocation product induces PDGFA in desmoplastic small round-cell tumour[J]. Nat Genet, 1997, 17(3): 309-13. [CrossRef]
- Froberg K, Brown R E, Gaylord H,et al. Intra-abdominal desmoplastic small round cell tumor: immunohistochemical evidence for up-regulation of autocrine and paracrine growth factors[J]. Ann Clin Lab Sci, 1999, 29(1): 78-85.
- Negri T, Brich S, Bozzi F,et al. New transcriptional-based insights into the pathogenesis of desmoplastic small round cell tumors (DSRCTs)[J]. Oncotarget, 2017, 8(20): 32492-32504. [CrossRef]
- Zhang P J, Goldblum J R, Pawel B R,et al. PDGF-A, PDGF-Rbeta, TGFbeta3 and bone morphogenic protein-4 in desmoplastic small round cell tumors with EWS-WT1 gene fusion product and their role in stromal desmoplasia: an immunohistochemical study[J]. Mod Pathol, 2005, 18(3): 382-7. [CrossRef]
- Chao J, Budd G T, Chu P,et al. Phase II clinical trial of imatinib mesylate in therapy of KIT and/or PDGFRalpha-expressing Ewing sarcoma family of tumors and desmoplastic small round cell tumors[J]. Anticancer Res, 2010, 30(2): 547-52.
- Zhang P J, Goldblum J R, Pawel B R,et al. PDGF-A, PDGF-Rbeta, TGFbeta3 and bone morphogenic protein-4 in desmoplastic small round cell tumors with EWS-WT1 gene fusion product and their role in stromal desmoplasia: an immunohistochemical study[J]. Modern pathology, 2005(3): 18. [CrossRef]
- Adamson P C, Blaney S M, Widemann B C,et al. Pediatric phase I trial and pharmacokinetic study of the platelet-derived growth factor (PDGF) receptor pathway inhibitor SU101[J]. Cancer Chemother Pharmacol, 2004, 53(6): 482-8. [CrossRef]
- De Sanctis R, Bertuzzi A, Bisogno G,et al. Imatinib mesylate in desmoplastic small round cell tumors[J]. Future Oncol, 2017, 13(14): 1233-1237. [CrossRef]
- Bond M, Bernstein M L, Pappo A,et al. A phase II study of imatinib mesylate in children with refractory or relapsed solid tumors: a Children's Oncology Group study[J]. Pediatr Blood Cancer, 2008, 50(2): 254-8. [CrossRef]
- Apte R S, Chen D S, Ferrara N. VEGF in Signaling and Disease: Beyond Discovery and Development[J]. Cell, 2019, 176(6): 1248-1264. [CrossRef]
- Ferrara N. Vascular endothelial growth factor: basic science and clinical progress[J]. Endocr Rev, 2004, 25(4): 581-611. [CrossRef]
- Kopfstein L, Veikkola T, Djonov V G,et al. Distinct roles of vascular endothelial growth factor-D in lymphangiogenesis and metastasis[J]. Am J Pathol, 2007, 170(4): 1348-61. [CrossRef]
- Eguchi R, Kawabe J I, Wakabayashi I. VEGF-Independent Angiogenic Factors: Beyond VEGF/VEGFR2 Signaling[J]. J Vasc Res, 2022, 59(2): 78-89. [CrossRef]
- Wilhelm S M, Carter C, Tang L,et al. BAY 43-9006 exhibits broad spectrum oral antitumor activity and targets the RAF/MEK/ERK pathway and receptor tyrosine kinases involved in tumor progression and angiogenesis[J]. Cancer Res, 2004, 64(19): 7099-109. [CrossRef]
- Magnan H D, Chou T, Laquaglia M P,et al. Elevated expression of VEGFR-2 and VEGFA in desmoplastic small round cell tumor (DSRCT) and activity of bevacizumab and irinotecan in a xenograft model of DSRCT[J]. Journal of Clinical Oncology, 2009, 27(15). [CrossRef]
- Magnan H D, Price A, Chou A J,et al. A pilot trial of irinotecan, temozolomide and bevacizumab (ITB) for treatment of newly diagnosed patients with desmoplastic small round cell tumor (DSRCT)[J]. Journal of Clinical Oncology, 2017, 35(15_suppl): 11050-11050. [CrossRef]
- Loktev A, Shipley J M. Desmoplastic small round cell tumor (DSRCT): emerging therapeutic targets and future directions for potential therapies[J]. Expert Opin Ther Targets, 2020, 24(4): 281-285. [CrossRef]
- Chow L Q, Eckhardt S G. Sunitinib: from rational design to clinical efficacy[J]. J Clin Oncol, 2007, 25(7): 884-96. [CrossRef]
- Tian Y, Cheng X, Li Y. Chemotherapy combined with apatinib for the treatment of desmoplastic small round cell tumors: A case report[J]. J Cancer Res Ther, 2020, 16(5): 1177-1181. [CrossRef]
- Taylor J G T, Cheuk A T, Tsang P S,et al. Identification of FGFR4-activating mutations in human rhabdomyosarcomas that promote metastasis in xenotransplanted models[J]. J Clin Invest, 2009, 119(11): 3395-407. [CrossRef]
- Chow W A, Yee J K, Tsark W,et al. Recurrent secondary genomic alterations in desmoplastic small round cell tumors[J]. BMC Med Genet, 2020, 21(1): 101. [CrossRef]
- Hingorani P, Dinu V, Zhang X,et al. Transcriptome analysis of desmoplastic small round cell tumors identifies actionable therapeutic targets: a report from the Children's Oncology Group[J]. Sci Rep, 2020, 10(1): 12318. [CrossRef]
- Saito T, Yokotsuka M, Motoi T,et al. EWS-WT1 Chimeric Protein in Desmoplastic Small Round Cell Tumor is a Potent Transactivator of FGFR4[J]. Journal of Cancer Science & Therapy, 2012, 4: 1-6.
- Slotkin E K, Bowman A S, Levine M F,et al. Comprehensive Molecular Profiling of Desmoplastic Small Round Cell Tumor[J]. Mol Cancer Res, 2021, 19(7): 1146-1155. [CrossRef]
- Werner H, Roberts C T, Jr. The IGFI receptor gene: a molecular target for disrupted transcription factors[J]. Genes Chromosomes Cancer, 2003, 36(2): 113-20. [CrossRef]
- Subbiah V, Brown R E, Jiang Y,et al. Morphoproteomic profiling of the mammalian target of rapamycin (mTOR) signaling pathway in desmoplastic small round cell tumor (EWS/WT1), Ewing's sarcoma (EWS/FLI1) and Wilms' tumor(WT1)[J]. PLoS One, 2013, 8(7): e68985. [CrossRef]
- Werner H, Leroith D. The role of the insulin-like growth factor system in human cancer[J]. Advances in cancer research, 1996, 68: 183-223. [CrossRef]
- Werner H, Re G G, Drummond I A,et al. Increased expression of the insulin-like growth factor I receptor gene, IGF1R, in Wilms tumor is correlated with modulation of IGF1R promoter activity by the WT1 Wilms tumor gene product[J]. Proc Natl Acad Sci U S A, 1993, 90(12): 5828-32. [CrossRef]
- Karnieli E, Werner H, Rauscher F J, Iii,et al. The IGF-I Receptor Gene Promoter Is a Molecular Target for the Ewing's Sarcoma-Wilms' Tumor 1 Fusion Protein *[J]. Journal of Biological Chemistry, 1996, 271(32): 19304-19309. [CrossRef]
- Finkeltov I, Kuhn S, Glaser T,et al. Transcriptional regulation of IGF-I receptor gene expression by novel isoforms of the EWS-WT1 fusion protein[J]. Oncogene, 2002, 21(12): 1890-8. [CrossRef]
- Werner H, Idelman G, Rubinstein M,et al. A novel EWS-WT1 gene fusion product in desmoplastic small round cell tumor is a potent transactivator of the insulin-like growth factor-I receptor (IGF-IR) gene[J]. Cancer Lett, 2007, 247(1): 84-90. [CrossRef]
- Livingstone C. IGF2 and cancer[J]. Endocr Relat Cancer, 2013, 20(6): R321-39. [CrossRef]
- Kurmasheva R T, Dudkin L, Billups C,et al. The insulin-like growth factor-1 receptor-targeting antibody, CP-751,871, suppresses tumor-derived VEGF and synergizes with rapamycin in models of childhood sarcoma[J]. Cancer Res, 2009, 69(19): 7662-71. [CrossRef]
- Tap W D, Demetri G, Barnette P,et al. Phase II study of ganitumab, a fully human anti-type-1 insulin-like growth factor receptor antibody, in patients with metastatic Ewing family tumors or desmoplastic small round cell tumors[J]. J Clin Oncol, 2012, 30(15): 1849-56. [CrossRef]
- Naing A, Lorusso P, Fu S,et al. Insulin growth factor-receptor (IGF-1R) antibody cixutumumab combined with the mTOR inhibitor temsirolimus in patients with refractory Ewing's sarcoma family tumors[J]. Clin Cancer Res, 2012, 18(9): 2625-31. [CrossRef]
- Coussens L, Yang-Feng T L, Liao Y C,et al. Tyrosine kinase receptor with extensive homology to EGF receptor shares chromosomal location with neu oncogene[J]. Science, 1985, 230(4730): 1132-9. [CrossRef]
- Espinosa-Cotton M, Guo H F, Tickoo S K,et al. Identification of immunotherapy and radioimmunotherapy targets on desmoplastic small round cell tumors[J]. Front Oncol, 2023, 13: 1104693. [CrossRef]
- Zhang T, Febres-Aldana C A, Liu Z,et al. HER2 Antibody-Drug Conjugates Are Active against Desmoplastic Small Round Cell Tumor[J]. Clin Cancer Res, 2024, 30(20): 4701-4713. [CrossRef]
- Brahmi M, Vanacker H, Dufresne A,et al. High expression level of ERBB2 and efficacy of trastuzumab deruxtecan in desmoplastic small round cell tumour: a monocentric case series report[J]. ESMO Open, 2025, 10(2): 104133. [CrossRef]
- Renner M, Oleś M, Paramasivam N,et al. Multi-layered molecular profiling informs the diagnosis and targeted therapy of desmoplastic small round cell tumor[J]. Nat Commun, 2026, 17(1). [CrossRef]
- Smith R S, Odintsov I, Liu Z,et al. Novel patient-derived models of desmoplastic small round cell tumor confirm a targetable dependency on ERBB signaling[J]. Dis Model Mech, 2022, 15(1). [CrossRef]
- Yarden Y, Kuang W J, Yang-Feng T,et al. Human proto-oncogene c-kit: a new cell surface receptor tyrosine kinase for an unidentified ligand[J]. Embo j, 1987, 6(11): 3341-51. [CrossRef]
- Fine R L, Shah S S, Moulton T A,et al. Androgen and c-Kit receptors in desmoplastic small round cell tumors resistant to chemotherapy: novel targets for therapy[J]. Cancer Chemother Pharmacol, 2007, 59(4): 429-37. [CrossRef]
- Zhang P J, Goldblum J R, Pawel B R,et al. Immunophenotype of desmoplastic small round cell tumors as detected in cases with EWS-WT1 gene fusion product[J]. Mod Pathol, 2003, 16(3): 229-35. [CrossRef]
- Smithey B E, Pappo A S, Hill D A. C-kit expression in pediatric solid tumors: a comparative immunohistochemical study[J]. Am J Surg Pathol, 2002, 26(4): 486-92. [CrossRef]
- Movva S, Wen W, Chen W,et al. Multi-platform profiling of over 2000 sarcomas: identification of biomarkers and novel therapeutic targets[J]. Oncotarget, 2015, 6(14): 12234-47. [CrossRef]
- Magrath J W, Espinosa-Cotton M, Flinchum D A,et al. Desmoplastic small round cell tumor: from genomics to targets, potential paths to future therapeutics[J]. Front Cell Dev Biol, 2024, 12: 1442488. [CrossRef]
- Ogura K, Somwar R, Hmeljak J,et al. Therapeutic Potential of NTRK3 Inhibition in Desmoplastic Small Round Cell Tumor[J]. Clin Cancer Res, 2021, 27(4): 1184-1194. [CrossRef]
- Felkai L, Krencz I, Kiss D J,et al. Characterization of mTOR Activity and Metabolic Profile in Pediatric Rhabdomyosarcoma[J]. Cancers (Basel), 2020, 12(7). [CrossRef]
- Wan X, Helman L J. The biology behind mTOR inhibition in sarcoma[J]. Oncologist, 2007, 12(8): 1007-18. [CrossRef]
- Jiang Y, Subbiah V, Janku F,et al. Novel secondary somatic mutations in Ewing's sarcoma and desmoplastic small round cell tumors[J]. PLoS One, 2014, 9(8): e93676. [CrossRef]
- Tirado O M, Mateo-Lozano S, Notario V. Rapamycin induces apoptosis of JN-DSRCT-1 cells by increasing the Bax : Bcl-xL ratio through concurrent mechanisms dependent and independent of its mTOR inhibitory activity[J]. Oncogene, 2005, 24(20): 3348-57. [CrossRef]
- Dimitrakopoulou-Strauss A, Hohenberger P, Ströbel P,et al. A recent application of fluoro-18-deoxyglucose positron emission tomography, treatment monitoring with a mammalian target of rapamycin inhibitor: an example of a patient with a desmoplastic small round cell tumor[J]. Hell J Nucl Med, 2007, 10(2): 77-9.
- Thijs A M, Van Der Graaf W T, Van Herpen C M. Temsirolimus for metastatic desmoplastic small round cell tumor[J]. Pediatr Blood Cancer, 2010, 55(7): 1431-2. [CrossRef]
- Wu C C, Beird H C, Lamhamedi-Cherradi S E,et al. Multi-site desmoplastic small round cell tumors are genetically related and immune-cold[J]. NPJ Precis Oncol, 2022, 6(1): 21. [CrossRef]
- Tarek N, Hayes-Jordan A, Salvador L,et al. Recurrent desmoplastic small round cell tumor responding to an mTOR inhibitor containing regimen[J]. Pediatr Blood Cancer, 2018, 65(1). [CrossRef]
- Katz D, Azraq Y, Eleyan F,et al. Pazolimus: pazopanib plus sirolimus following progression on pazopanib, a retrospective case series analysis[J]. BMC Cancer, 2016, 16: 616. [CrossRef]
- Bulbul A. Potential therapeutic genomic alterations in desmoplastic small round blue cell tumor[J], 2017. [CrossRef]
- Lamhamedi-Cherradi S E, Maitituoheti M, Menegaz B A,et al. The androgen receptor is a therapeutic target in desmoplastic small round cell sarcoma[J]. Nat Commun, 2022, 13(1): 3057. [CrossRef]
- Michmerhuizen A R, Spratt D E, Pierce L J,et al. ARe we there yet? Understanding androgen receptor signaling in breast cancer[J]. NPJ Breast Cancer, 2020, 6: 47. [CrossRef]
- Gedminas J M, Chasse M H, Mcbrairty M,et al. Desmoplastic small round cell tumor is dependent on the EWS-WT1 transcription factor[J]. Oncogenesis, 2020, 9(4): 41. [CrossRef]
- Nicholas T R, Metcalf S A, Greulich B M,et al. Androgen signaling connects short isoform production to breakpoint formation at Ewing sarcoma breakpoint region 1[J]. NAR Cancer, 2021, 3(3): zcab033. [CrossRef]
- Liu W, Lindberg J, Sui G,et al. Identification of novel CHD1-associated collaborative alterations of genomic structure and functional assessment of CHD1 in prostate cancer[J]. Oncogene, 2012, 31(35): 3939-48. [CrossRef]
- Smith R, Liu M, Liby T,et al. Enzalutamide response in a panel of prostate cancer cell lines reveals a role for glucocorticoid receptor in enzalutamide resistant disease[J]. Sci Rep, 2020, 10(1): 21750. [CrossRef]
- Zhang X, Fang C, Zhang G,et al. Prognostic value of B7-H3 expression in patients with solid tumors: a meta-analysis[J]. Oncotarget, 2017, 8(54): 93156-93167. [CrossRef]
- Lee Y H, Martin-Orozco N, Zheng P,et al. Inhibition of the B7-H3 immune checkpoint limits tumor growth by enhancing cytotoxic lymphocyte function[J]. Cell Res, 2017, 27(8): 1034-1045. [CrossRef]
- Modak S, Gerald W, Cheung N K. Disialoganglioside GD2 and a novel tumor antigen: potential targets for immunotherapy of desmoplastic small round cell tumor[J]. Med Pediatr Oncol, 2002, 39(6): 547-51. [CrossRef]
- Modak S, Kramer K, Gultekin S H,et al. Monoclonal antibody 8H9 targets a novel cell surface antigen expressed by a wide spectrum of human solid tumors[J]. Cancer Res, 2001, 61(10): 4048-54.
- Vanarsdale T, Boshoff C, Arndt K T,et al. Molecular Pathways: Targeting the Cyclin D-CDK4/6 Axis for Cancer Treatment[J]. Clin Cancer Res, 2015, 21(13): 2905-10. [CrossRef]
- Shulman D S, Merriam P, Choy E,et al. Phase 2 trial of palbociclib and ganitumab in patients with relapsed Ewing sarcoma[J]. Cancer Med, 2023, 12(14): 15207-15216. [CrossRef]
- Hartono A B, Kang H J, Shi L,et al. Salt-Inducible Kinase 1 is a potential therapeutic target in Desmoplastic Small Round Cell Tumor[J]. Oncogenesis, 2022, 11(1): 18. [CrossRef]
- Hartono A B. The SIKness of DSRCT: Salt Inducible Kinase 1 Is a Potential Therapeutic Target in Desmoplastic Small Round Cell Tumor[D]. United States -- Louisiana: Tulane University, 2022: 116.
- Mello C A, Campos F a B, Santos T G,et al. Desmoplastic Small Round Cell Tumor: A Review of Main Molecular Abnormalities and Emerging Therapy[J]. Cancers (Basel), 2021, 13(3). [CrossRef]
- Truong D D, Magrath J W, Murgas K,et al. EWS::WT1 Isoform-Dependent Regulation of Neogenes in Desmoplastic Small Round Cell Tumors[J]. bioRxiv, 2025. [CrossRef]
- Heilig C E, Heining C, Gnutzmann E,et al. Rationale and design of the PAMSARC (pasireotide as maintenance treatment with monthly deep intramuscular injection in SSTR2/3/5-expressing synovial sarcoma and desmoplastic small round cell tumor) multicenter phase 2 trial[J]. Cancer Treat Res Commun, 2025, 45: 100986. [CrossRef]
- Anderson P M, Trucco M M, Tarapore R S,et al. Phase II Study of ONC201 in Neuroendocrine Tumors including Pheochromocytoma-Paraganglioma and Desmoplastic Small Round Cell Tumor[J]. Clin Cancer Res, 2022, 28(9): 1773-1782. [CrossRef]
- Magrath J W, Flinchum D A, Hartono A B,et al. Transcriptomic analysis identifies B-lymphocyte kinase as a therapeutic target for desmoplastic small round cell tumor cancer stem cell-like cells[J]. Oncogenesis, 2024, 13(1): 2. [CrossRef]
- Van Erp A E M, Hillebrandt-Roeffen M H S, Van Bree N,et al. Targeting the FAK-Src Complex in Desmoplastic Small Round Cell Tumors, Ewing Sarcoma, and Rhabdomyosarcoma[J]. Sarcoma, 2022, 2022: 3089424. [CrossRef]
- Hartlapp I, Hartrampf P E, Serfling S E,et al. CXCR4-Directed Imaging and Endoradiotherapy in Desmoplastic Small Round Cell Tumors[J]. J Nucl Med, 2023, 64(9): 1424-1430. [CrossRef]
- Kim J R, Moon Y J, Kwon K S,et al. Tumor infiltrating PD1-positive lymphocytes and the expression of PD-L1 predict poor prognosis of soft tissue sarcomas[J]. PLoS One, 2013, 8(12): e82870. [CrossRef]
- Van Erp A E M, Versleijen-Jonkers Y M H, Hillebrandt-Roeffen M H S,et al. Expression and clinical association of programmed cell death-1, programmed death-ligand-1 and CD8(+) lymphocytes in primary sarcomas is subtype dependent[J]. Oncotarget, 2017, 8(41): 71371-71384. [CrossRef]
- Schöffski P, Bahleda R, Wagner A J,et al. Results of an Open-label, Phase Ia/b Study of Pembrolizumab plus Olaratumab in Patients with Unresectable, Locally Advanced, or Metastatic Soft-Tissue Sarcoma[J]. Clin Cancer Res, 2023, 29(17): 3320-3328. [CrossRef]
- Sterner R C, Sterner R M. CAR-T cell therapy: current limitations and potential strategies[J]. Blood Cancer J, 2021, 11(4): 69. [CrossRef]
- Larson S M, Carrasquillo J A, Cheung N K,et al. Radioimmunotherapy of human tumours[J]. Nat Rev Cancer, 2015, 15(6): 347-60. [CrossRef]
- Modak S, Zanzonico P, Grkovski M,et al. B7H3-Directed Intraperitoneal Radioimmunotherapy With Radioiodinated Omburtamab for Desmoplastic Small Round Cell Tumor and Other Peritoneal Tumors: Results of a Phase I Study[J]. J Clin Oncol, 2020, 38(36): 4283-4291. [CrossRef]
- Dobrenkov K, Ostrovnaya I, Gu J,et al. Oncotargets GD2 and GD3 are highly expressed in sarcomas of children, adolescents, and young adults[J]. Pediatr Blood Cancer, 2016, 63(10): 1780-5. [CrossRef]
- Yankelevich M, Thakur A, Modak S,et al. Targeting GD2-positive Refractory/Resistant Neuroblastoma and Osteosarcoma with Anti- CD3 x Anti-GD2 Bispecific Antibody Armed T cells[J]. Res Sq, 2023. [CrossRef]
- Vibert J, Saulnier O, Collin C,et al. Oncogenic chimeric transcription factors drive tumor-specific transcription, processing, and translation of silent genomic regions[J]. Mol Cell, 2022, 82(13): 2458-2471.e9. [CrossRef]
- He M, Jiang H, Li S,et al. The crosstalk between DNA-damage responses and innate immunity[J]. Int Immunopharmacol, 2024, 140: 112768. [CrossRef]
- Min A, Im S A. PARP Inhibitors as Therapeutics: Beyond Modulation of PARylation[J]. Cancers (Basel), 2020, 12(2). [CrossRef]
- Devecchi A, De Cecco L, Dugo M,et al. The genomics of desmoplastic small round cell tumor reveals the deregulation of genes related to DNA damage response, epithelial-mesenchymal transition, and immune response[J]. Cancer Commun (Lond), 2018, 38(1): 70. [CrossRef]
- Van Erp A E M, Van Houdt L, Hillebrandt-Roeffen M H S,et al. Olaparib and temozolomide in desmoplastic small round cell tumors: a promising combination in vitro and in vivo[J]. J Cancer Res Clin Oncol, 2020, 146(7): 1659-1670. [CrossRef]
- Mellado-Lagarde M, Federico S M, Tinkle C,et al. PARP inhibitor combination therapy in desmoplastic small round cell tumors[J]. Journal of Clinical Oncology, 2017, 35(15_suppl): e23212-e23212. [CrossRef]
- Aune G J, Takagi K, Sordet O,et al. Von Hippel-Lindau-coupled and transcription-coupled nucleotide excision repair-dependent degradation of RNA polymerase II in response to trabectedin[J]. Clin Cancer Res, 2008, 14(20): 6449-55. [CrossRef]
- Pignochino Y, Capozzi F, D'ambrosio L,et al. PARP1 expression drives the synergistic antitumor activity of trabectedin and PARP1 inhibitors in sarcoma preclinical models[J]. Mol Cancer, 2017, 16(1): 86. [CrossRef]
- Grignani G, D'ambrosio L, Pignochino Y,et al. Trabectedin and olaparib in patients with advanced and non-resectable bone and soft-tissue sarcomas (TOMAS): an open-label, phase 1b study from the Italian Sarcoma Group[J]. Lancet Oncol, 2018, 19(10): 1360-1371. [CrossRef]
- Palmerini E, Sanfilippo R, Grignani G,et al. Transcription regulators and ultra-rare and other rare translocation-related sarcomas treated with trabectedin: A proof of principle from a post-hoc analysis[J]. Front Oncol, 2022, 12: 1042479. [CrossRef]
- Lowery C D, Dowless M, Renschler M,et al. Broad Spectrum Activity of the Checkpoint Kinase 1 Inhibitor Prexasertib as a Single Agent or Chemopotentiator Across a Range of Preclinical Pediatric Tumor Models[J]. Clin Cancer Res, 2019, 25(7): 2278-2289. [CrossRef]
- Slotkin E K, Mauguen A, Ortiz M V,et al. A phase I/II study of prexasertib in combination with irinotecan in patients with relapsed/refractory desmoplastic small round cell tumor and rhabdomyosarcoma[J]. Journal of Clinical Oncology, 2022, 40(16_suppl): 11503-11503. [CrossRef]
- Kallianpur A A, Shukla N K, Deo S V,et al. Updates on the multimodality management of desmoplastic small round cell tumor[J]. J Surg Oncol, 2012, 105(6): 617-21. [CrossRef]
- Biswas G, Laskar S, Banavali S D,et al. Desmoplastic small round cell tumor: extra abdominal and abdominal presentations and the results of treatment[J]. Indian J Cancer, 2005, 42(2): 78-84. [CrossRef]
- Hayes-Jordan A, Laquaglia M P, Modak S. Management of desmoplastic small round cell tumor[J]. Semin Pediatr Surg, 2016, 25(5): 299-304. [CrossRef]
- Zhu RX, Wang J, Zhu LF, et al. Analysis of Clinical Features and Prognostic Factors of Desmoplastic Small Round Cell Tumor[J]. Journal of Shanghai Jiao Tong University (Medical Science), 2018, 38(03): 281-287. [CrossRef]
- Osborne E M, Briere T M, Hayes-Jordan A,et al. Survival and toxicity following sequential multimodality treatment including whole abdominopelvic radiotherapy for patients with desmoplastic small round cell tumor[J]. Radiother Oncol, 2016, 119(1): 40-4. [CrossRef]
- Hayes-Jordan A A, Coakley B A, Green H L,et al. Desmoplastic Small Round Cell Tumor Treated with Cytoreductive Surgery and Hyperthermic Intraperitoneal Chemotherapy: Results of a Phase 2 Trial[J]. Ann Surg Oncol, 2018, 25(4): 872-877. [CrossRef]
- Stiles Z E, Murphy A J, Anghelescu D L,et al. Desmoplastic Small Round Cell Tumor: Long-Term Complications After Cytoreduction and Hyperthermic Intraperitoneal Chemotherapy[J]. Ann Surg Oncol, 2020, 27(1): 171-178. [CrossRef]
- Kushner B H, Laquaglia M P, Wollner N,et al. Desmoplastic small round-cell tumor: prolonged progression-free survival with aggressive multimodality therapy[J]. J Clin Oncol, 1996, 14(5): 1526-31. [CrossRef]
- Liu K X, Collins N B, Greenzang K A,et al. The use of interval-compressed chemotherapy with the addition of vincristine, irinotecan, and temozolomide for pediatric patients with newly diagnosed desmoplastic small round cell tumor[J]. Pediatr Blood Cancer, 2020, 67(10): e28559. [CrossRef]
- Yang Y, Xie L, Sun X,et al. Whole Abdominal Radiotherapy in Bone and Soft Tissue Sarcomas: Indications, Techniques, Clinical Outcomes, and Future Directions[J]. Curr Treat Options Oncol, 2026, 27(1). [CrossRef]
- Rachfal A W, Luquette M H, Brigstock D R. Expression of connective tissue growth factor (CCN2) in desmoplastic small round cell tumour[J]. J Clin Pathol, 2004, 57(4): 422-5. [CrossRef]
- Worley B S, Van Den Broeke L T, Goletz T J,et al. Antigenicity of fusion proteins from sarcoma-associated chromosomal translocations[J]. Cancer Res, 2001, 61(18): 6868-75.
- Scharnhorst V, Van Der Eb A J, Jochemsen A G. WT1 proteins: functions in growth and differentiation[J]. Gene, 2001, 273(2): 141-61. [CrossRef]
- Wisdom A J, Mowery Y M, Riedel R F,et al. Rationale and emerging strategies for immune checkpoint blockade in soft tissue sarcoma[J]. Cancer, 2018, 124(19): 3819-3829. [CrossRef]
- Lawrence M S, Stojanov P, Polak P,et al. Mutational heterogeneity in cancer and the search for new cancer-associated genes[J]. Nature, 2013, 499(7457): 214-218. [CrossRef]
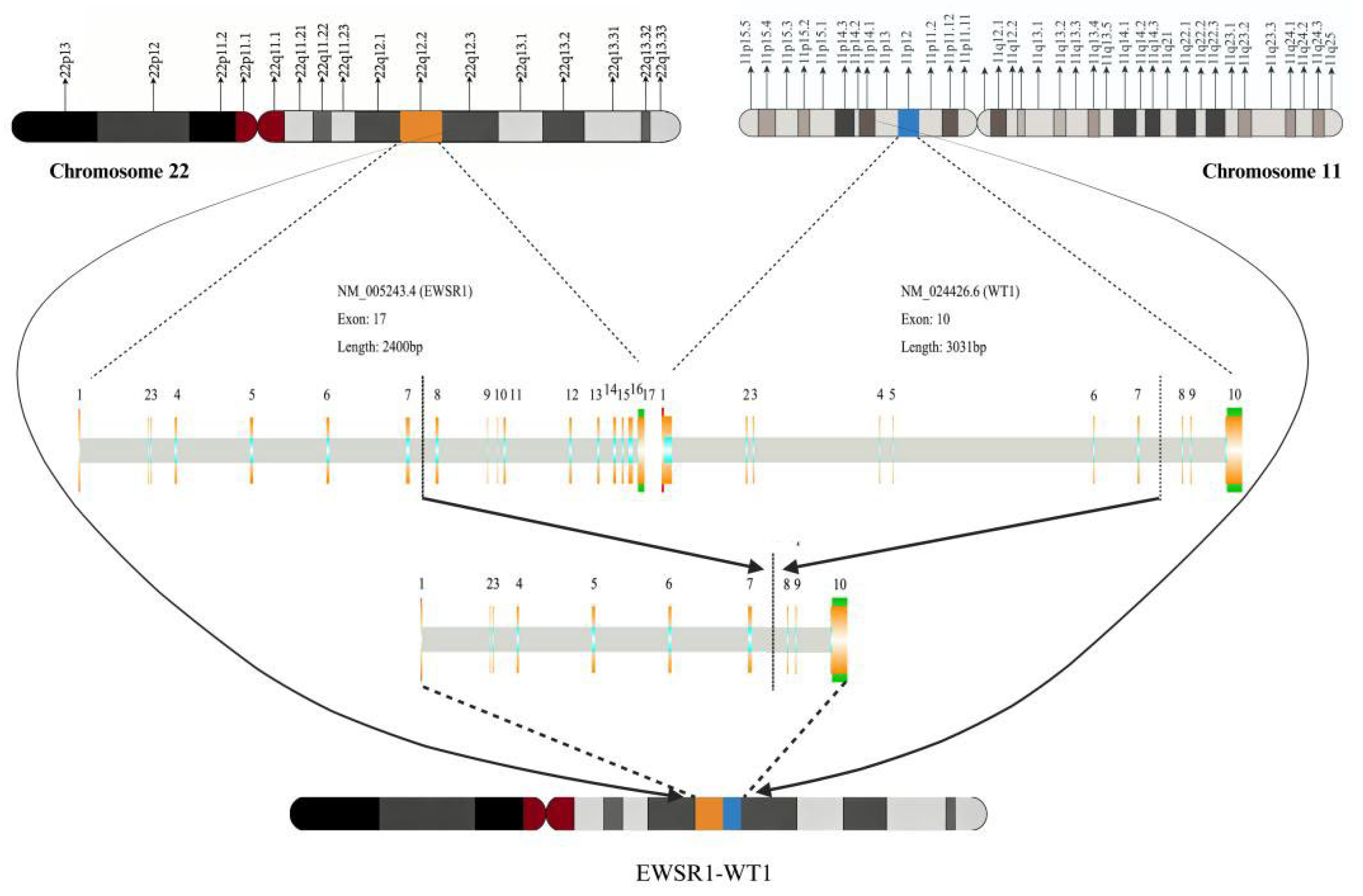
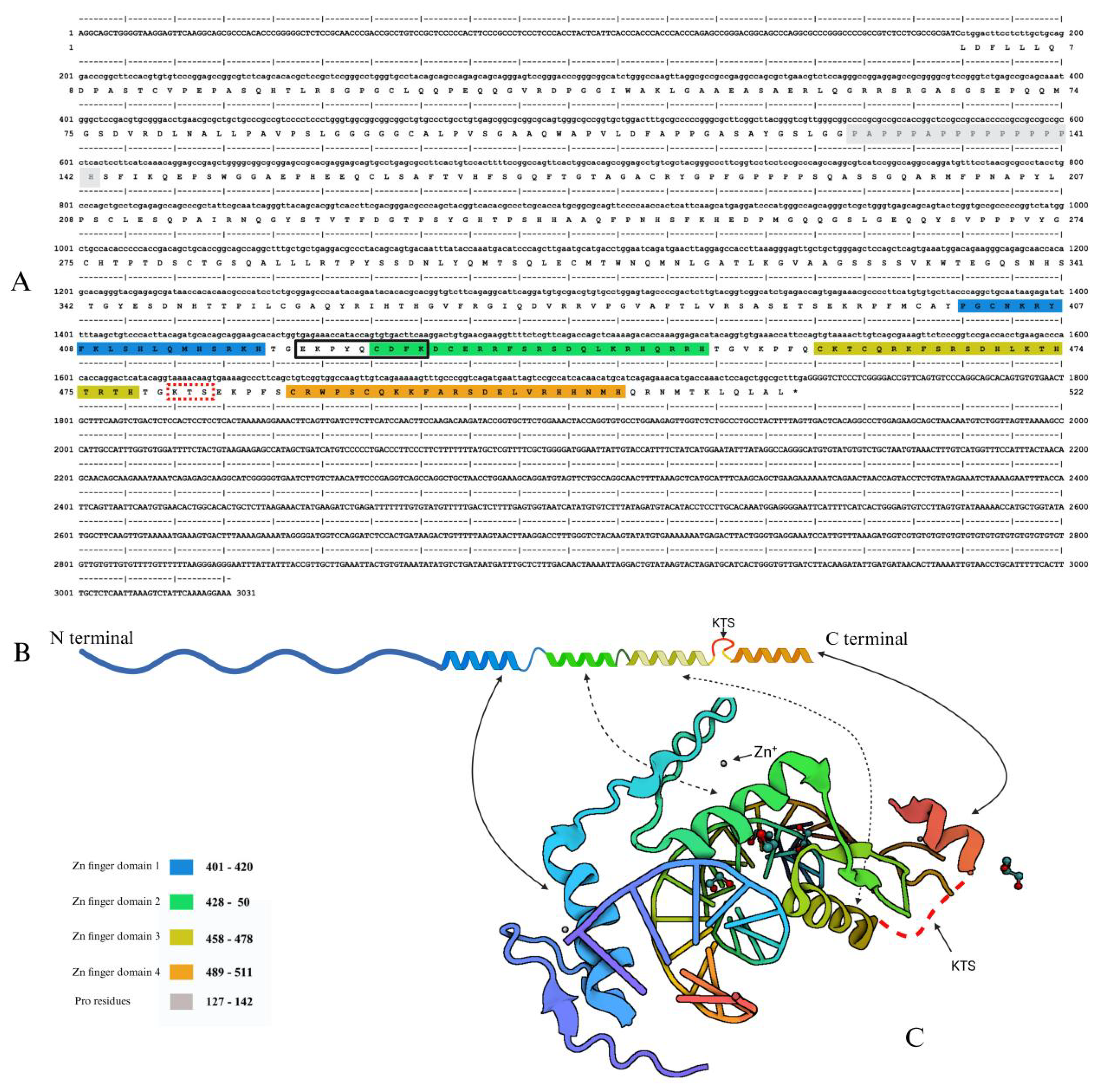
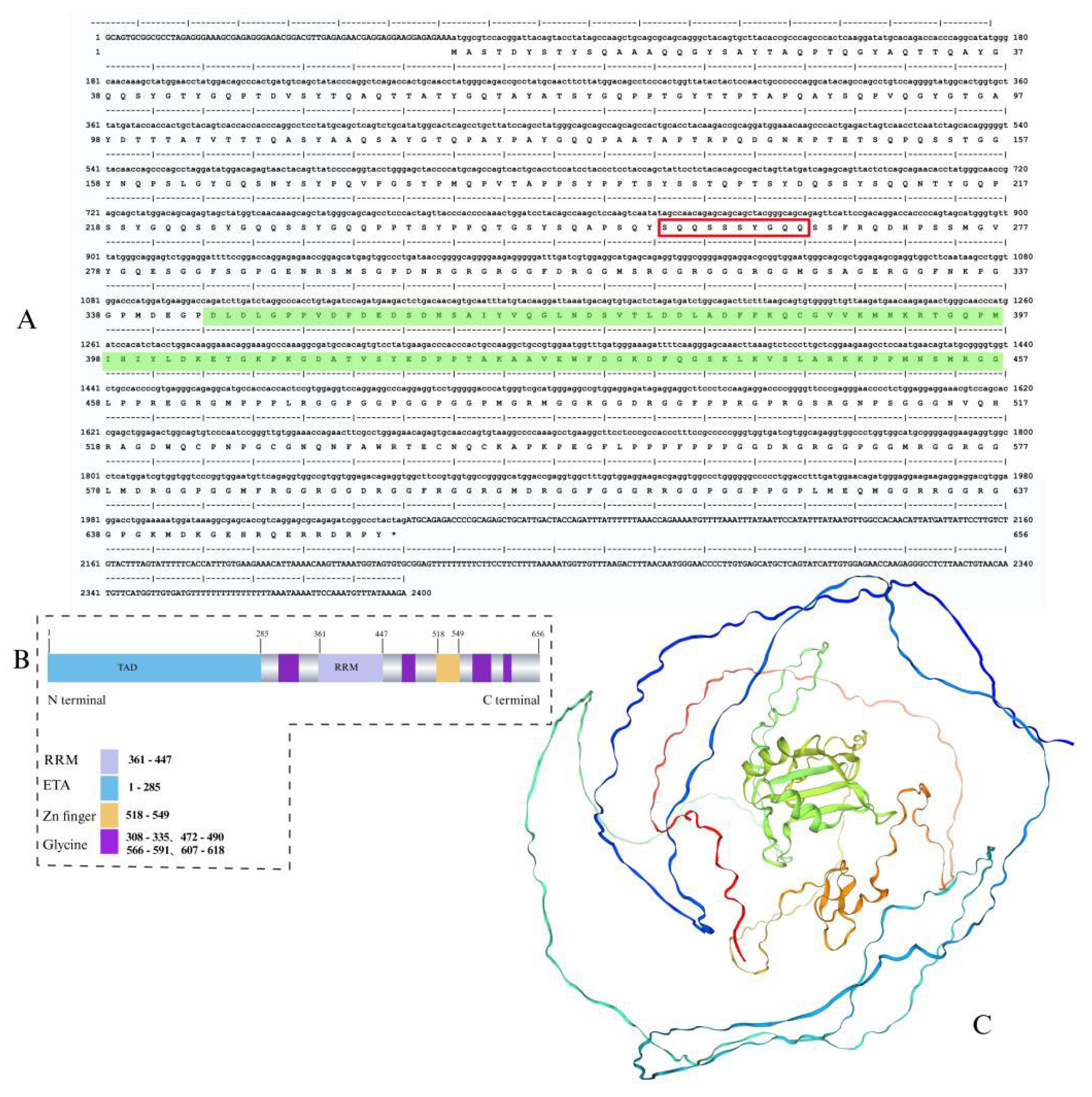
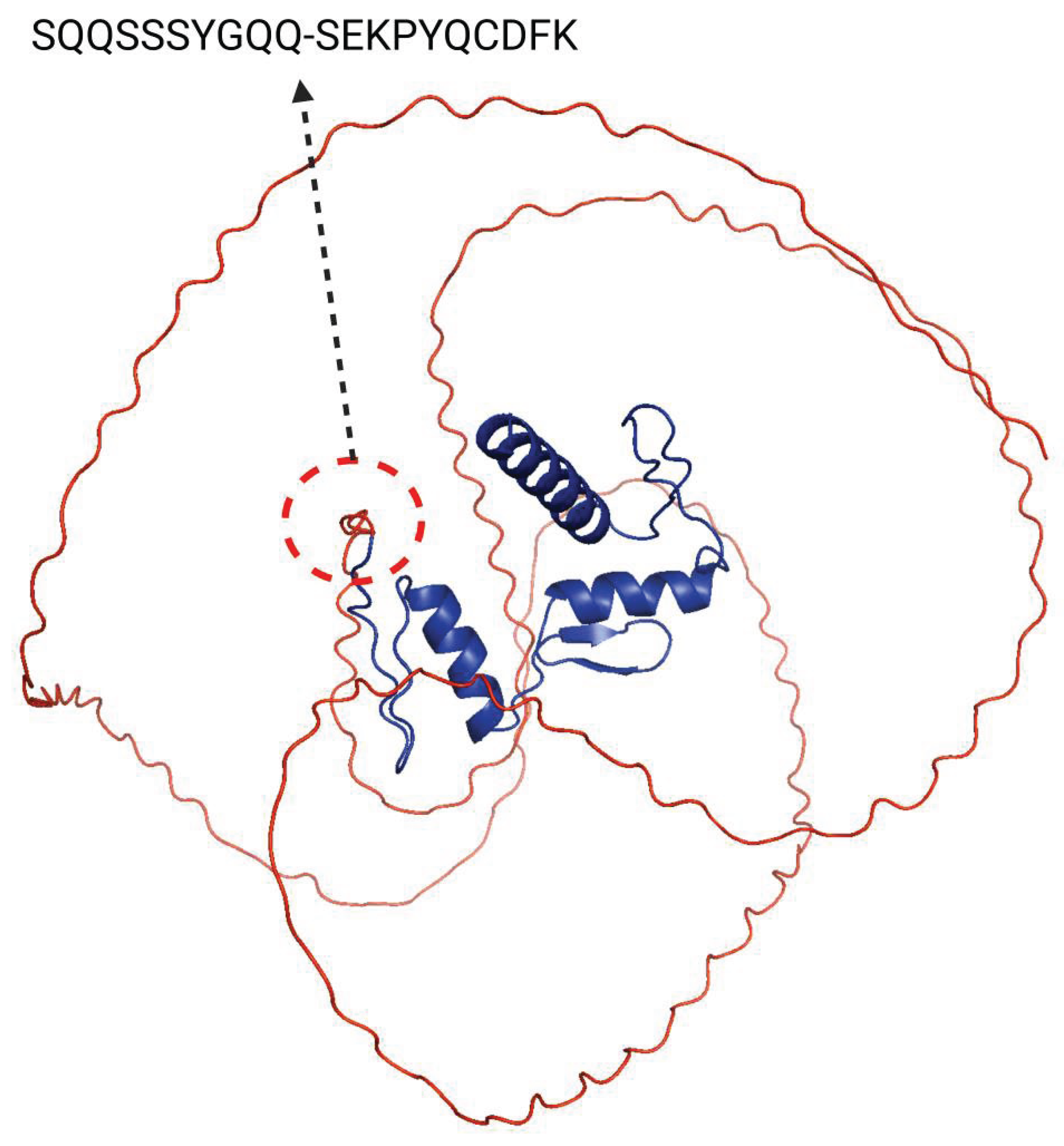
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



