Submitted:
14 April 2026
Posted:
15 April 2026
You are already at the latest version
Abstract
Keywords:
Introduction
Methods
Study Design and Setting
Study Population
SCD Diagnosis and Genotyping
Inclusion Criteria
- • Age ≤18 years at hospitalization
- • Confirmed SCD diagnosis
- • Admission to the ED for VOC
Exclusion Criteria
- • Presence of ACS at the time of admission
- • Incomplete medical records preventing outcome assessment
- • Chronic lung disease unrelated to SCD
Definitions
- • Vaso-occlusive crisis: An acute painful episode requiring hospital admission and not attributable to another identifiable cause.
- • Acute chest syndrome: Occurrence of a new pulmonary infiltrate on chest imaging associated with at least one clinical feature—fever, respiratory symptoms, hypoxemia, or thoracoabdominal pain—either during hospitalization or within 7 days following VOC admission.
Data Collection
Sample Size Calculation
Statistical Analysis
Ethical Considerations
Results
Study Population
Incidence of ACS
Predictive Factors for ACS (Table 2 and Table 3)
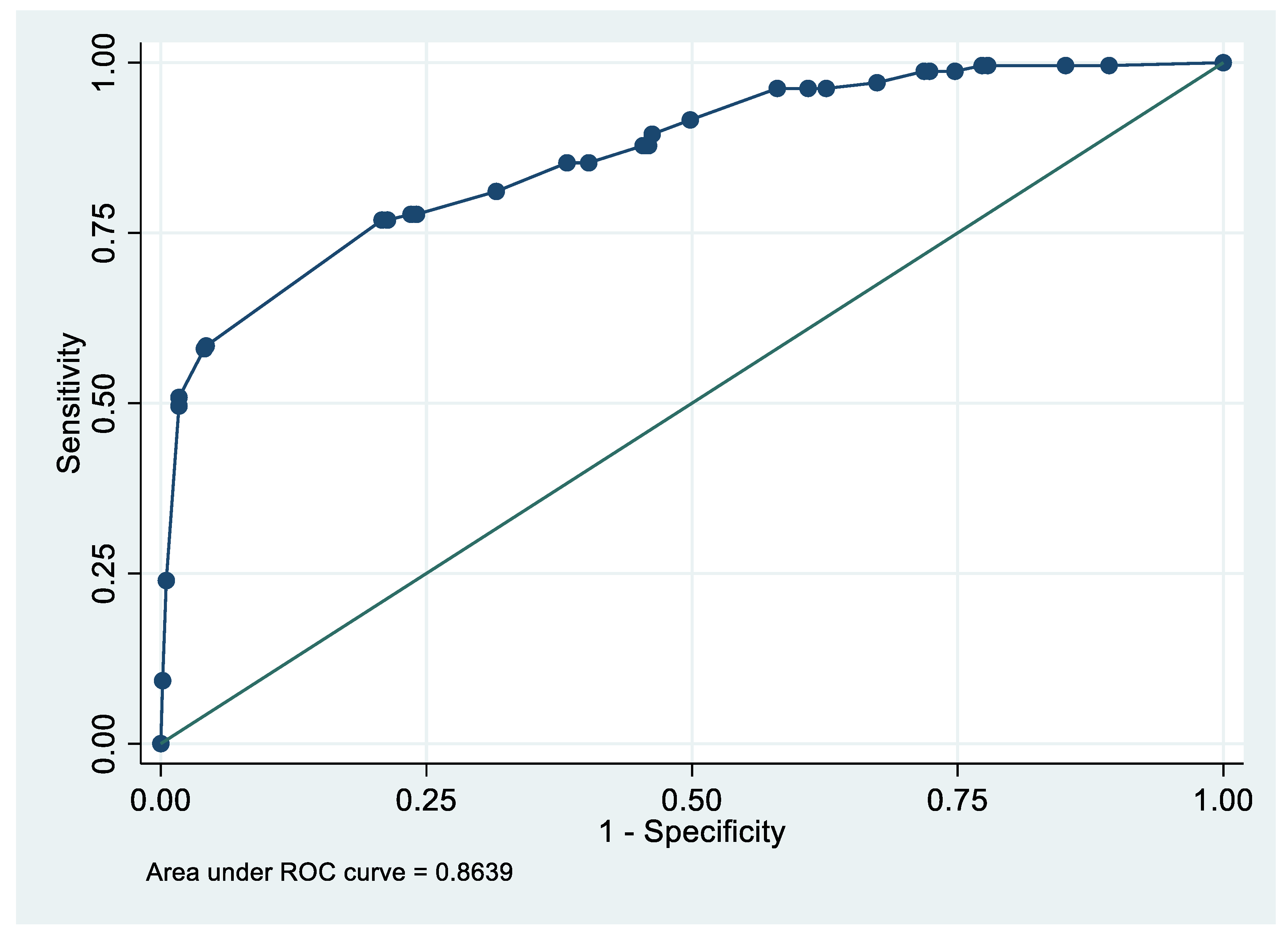
Sensitivity Analysis
- Thoracic/abdominal pain VOC: aOR 2.88 (95% CI: 1.45–5.72)
- History of ACS: aOR 2.2 (95% CI: 1.28–2.9)
- HbSS/Sβ⁰ genotype: aOR 1.91 (95% CI: 1.3–2.6)
- Hydroxyurea therapy : aOR 2.2 (95% CI: 1.6–2.6)
Discussion
Comparison with Previous Literature
Clinical Implications
- Early risk stratification: Children presenting with VOC characterized by thoracic or abdominal pain, prior ACS, or HbSS genotype should be closely monitored for early signs of pulmonary involvement.
- Preventive measures: These patients may benefit from enhanced supportive care, including oxygen monitoring, incentive spirometry, and careful fluid management.
- Therapeutic planning: Recognition of high-risk VOC episodes may guide early transfusion strategies or prompt escalation to intensive care if ACS develops rapidly.
- Hydroxyurea use was less frequent among patients who developed ACS in descriptive analyses, suggesting a potential protective effect. However, in multivariable analysis, hydroxyurea exposure was not independently associated with reduced ACS risk. This discrepancy may reflect confounding by indication, as patients receiving hydroxyurea often have more severe disease phenotypes. Therefore, these findings should be interpreted with caution.
Strengths and Limitations
Future Perspectives
Conclusion
Authorship contributions
Ethics approval statement
Patient consent statement
Permission to Reproduce Material from Other Source
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Piel, F.B.; Steinberg, M.H.; Rees, D.C. Sickle Cell Disease. N Engl J Med 2017, 376 n° 16, 1561–1573. [Google Scholar] [CrossRef] [PubMed]
- Brousse, V.; Bernaudin, F.; Melaine, A.; Goguillot, M.; Gallagher, M.; Benard, S.; Habibi, A. Severity and burden of sickle cell disease in France: a nationwide real-world study. Haematologica 108 n°9, 2476–2486. [CrossRef] [PubMed]
- Kato, G.J.; Piel, F.B.; Reid, C.D.; Gaston, M.H.; Ohene-Frempong, K.; Krishnamurti, L.; Smith, W.R.; Panepinto, J.A.; Weatherall, D.J.; Costa, F.F.; Vichinsky, E.P. Sickle cell disease. Nat Rev Dis Primers 2018, 18010. [Google Scholar] [CrossRef] [PubMed]
- Kavanagh, P.L.; Fasipe, T.A.; Wun, T. Sickle Cell Disease: A Review. JAMA 2022, 328 n°1, 57–68. [Google Scholar] [CrossRef] [PubMed]
- Meloy, P.; Rutz, D.R.; Bhambri, A. Acute Chest Syndrome. J Educ Teach Emerg Med 2023, 8 n° 1, O1–O23. [Google Scholar] [CrossRef]
- Chang, T.P.; Kriengsoontorkij, W.; Chan, L.S.; Wang, V.J. Clinical factors and incidence of acute chest syndrome or pneumonia among children with sickle cell disease presenting with a fever: a 17-year review. Pediatr Emerg Care 2013, 29 n° 7, 781–786. [Google Scholar] [CrossRef] [PubMed]
- Alghamdi, F.A.; Al-Kasim, F.; Alshhada, F.; Ghareeb, E.; Azmet, F.R.; Almudaibigh, A.; Baitalmal, L.; Alnawfal, B.; Alluqmani, R. Risk factors for acute chest syndrome among children with sickle cell anemia hospitalized for vaso-occlusive crises. Sci Rep 2024, 14 n°1, 5978. [Google Scholar] [CrossRef] [PubMed]
- Knight-Madden, J.; Lee, K.; Elana, G.; Elenga, N.; Marcheco-Teruel, B.; Keshi, N.; Etienne-Julan, M.; King, L.; Asnani, M.; Romana, M.; Hardy-Dessources, M.D. Newborn Screening for Sickle Cell Disease in the Caribbean: An Update of the Present Situation and of the Disease Prevalence. Int J Neonatal Screen 2019, 5 n°1, 5. [Google Scholar] [CrossRef] [PubMed]
- Leleu, H.; Arlet, J.B.; Habibi, A.; Etienne-Julan, M.; Khellaf, M.; Adjibi, Y.; Pirenne, F.; Pitel, M.; Granghaud, A.; Sinniah, C.; De Montalembert, M.; Galacteros, F. Epidemiology and disease burden of sickle cell disease in France: A descriptive study based on a French nationwide claim database. PLoS One 2021, 16 n°7, e0253986. [Google Scholar] [CrossRef] [PubMed]
- Djomo, C.F.; Sile, S.N.; Elenga, N. Emergency Presentations of Pediatric Sickle Cell Disease in French Guiana. Diseases 2025, 13 n°5, 142. [Google Scholar] [CrossRef] [PubMed]
- Gorphe, P.; Jannin, C. Regulatory aspects of prospective and retrospective clinical research in France in 2018. Eur Ann Otorhinolaryngol Head Neck Dis 2019, 136 n°2, 103–108. [Google Scholar] [CrossRef] [PubMed]
- Condé, M.; Bouvier, F.; Brat, R.; Ferrani, S.; Hankard, R.; Connes, P.; Dimitrov, G. Early risk factors for acute chest syndrome in sickle cell anemia: A pediatric study. PLoS One 2025, 20 n°11, e0336567. [Google Scholar] [CrossRef] [PubMed]
- Jain, S.; Bakshi, N.; Krishnamurti, L. Acute Chest Syndrome in Children with Sickle Cell Disease. Pediatr Allergy Immunol Pulmonol 2017, 30 n°4, 191–201. [Google Scholar] [CrossRef] [PubMed]
- Vichinsky, E.P.; Neumayr, L.D.; Earles, A.N.; Williams, R.; Lennette, E.T.; Dean, D.; Nickerson, B.; Orringer, E.; McKie, V.; Bellevue, R.; Daeschner, C.; Manci, E.A. Causes and outcomes of the acute chest syndrome in sickle cell disease. National Acute Chest Syndrome Study Group. N Engl J Med 2000, 342 n°25, 1855–65. [Google Scholar] [CrossRef] [PubMed]
- Creary, S.E.; Krishnamurti, L. Prodromal illness before acute chest syndrome in pediatric patients with sickle cell disease. J. Pediatr. Hematol. Oncol 2014, 36 n°6, 480–483. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.; Olave, N.; Aggarwal, S.; Oh, J.Y.; Patel, R.P.; Rahman, A.K.M.F.; Lebensburger, J.; Alishlash, A.S. Biomarkers to Differentiate Acute Chest Syndrome From Vaso-Occlusive Crisis in Children With Sickle Cell Disease. Eur J Haematol 2025, 14 n°2, 325–333. [Google Scholar] [CrossRef] [PubMed]
- Adisa, O.A.; Hu, Y.; Ghosh, S.; Aryee, D.; Osunkwo, I.; Ofori-Acquah, S.F. Association between plasma free haem and incidence of vaso-occlusive episodes and acute chest syndrome in children with sickle cell disease. Br J Haematol 2013, 162 n°5, 702–5. [Google Scholar] [CrossRef] [PubMed]
- Bartolucci, P.; Habibi, A.; Khellaf, M.; Roudot-Thoraval, F.; Melica, G.; Lascaux, A.S.; Moutereau, S.; Loric, S.; Wagner-Ballon, O.; Berkenou, J.; Santin, A.; Michel, M.; Renaud, B.; Lévy, Y.; Galactéros, F.; Godeau, B. Score Predicting Acute Chest Syndrome During Vaso-occlusive Crises in Adult Sickle-cell Disease Patients. EBioMedicine 2016, 10 10, 305–11. [Google Scholar] [CrossRef] [PubMed]
| Characteristics | Uncomplicated VOC | VOC→ACS | p | ||||
| (n=586) | (n=238) | ||||||
| Age (median, IQR) | 11 [7,8,9,10,11,12,13,14,15] | 15 [11,12,13,14,15,16,17] | <0.001 | ||||
| Female sex (%) | 325 (76) | 101 (24) | 0.001 | ||||
| HbSS/S/ß° genotype (%) | 163 (68) | 445 (76) | <0.001 | ||||
| Hydroxyurea therapy (%) | 354 (60) | 42 (18) | <0.001 | ||||
| History of acute chest syndrome (%) | 112 (19) | 122 (51) | <0.001 | ||||
| Comorbidity (%)* | 206 (35) | 104 (44) | 0.02 | ||||
| Cholecystecyomy history (%) | 152 (26) | 70 (29) | 0.3 | ||||
| Splenectomy history (%) | 68 (12) | 15 (6) | 0.02 | ||||
| Bseline hemoglobin<8 g/dl (%) | 85 (16) | 21 (10) | 0.05 | ||||
| Thoracic/abdominal pain VOC (%) | 354 (60) | 231 (97) | <0.001 | ||||
| Length of Hospital Stay>5 days (%) | 103 (17) | 34 (15) | 0.3 | ||||
| Current white blood cells (median, IQR) | 16 [11-21] | 16 [11-20] | 0.6 | ||||
| Current neutrophils (median, IQR) | 10 [7-15] | 10 [7-14] | 0.9 | ||||
| Current platelets (median, IQR) | 400 [400-400] | 400 [400-400] | |||||
| Current reticulocytes (median, IQR) | 244 [166-350] | 180 [118-270] | 0.1 | ||||
| Current LDH (median, IQR) | 650 [538-800] | 0.3 | |||||
| Current CRP (median,IQR) | 28.5 [4-128] | 71.5 [7-180] | 0.09 | ||||
| Variable | OR (95% CI) | p-value |
| Age (per year) | 2.8 (2.1–3.7) | <0.001 |
| Female sex | 0.5 (0.2–0.8) | 0.001 |
| HbSS /S/ß° genotype | 3.4 (2.3-5.1) | <0.001 |
| Thoracic or abdominal pain VOC | 18.9 (9.2-39.1) | <0.001 |
| History of ACS | 4.5 (3.2-6.2) | <0.001 |
| Hydroxyurea therapy | 7.2 (4.9-10.3) | <0.001 |
| Baseline Hb <8 g/dL | 0.6(0..4–1.0) | 0.05 |
| Comorbidity history | 1.4 (1.04-1.9) | <0.03 |
| Length of Hospital Stay | 1.7 (1.2-2.6) | 0.004 |
| Characteristics | Uncomplicated VOC | VOCà ACS | aOR (95 %CI) | p | |||
| (n=586) | (n=238) | ||||||
| Genotype Hb SS/S/ß° (%) | 163 (68) | 445 (76 | 1.8 (1.2–2.4.) | <0.001 | |||
| Hydroxyurea therapy (%) | 354 (60) | 42 (18) | 2.1 (1.6–2.5.) | <0.001 | |||
| History of acute chest syndrome (%) | 112 (19) | 122 (51) | 1.7 (1.23–2.10) | <0.001 | |||
| Thoracic/abdominal pain VOC (%) | 354 (60) | 231 (97) | 2.1 (1.3–2.8) | <0.001 | |||
| Age (years) | 0.45 (0.1-0.82)) | 0.02 | |||||
| 0-5 | 101 (17) | 8 (3) | |||||
| 05-10 years | 176 (30) | 39 (16) | |||||
| >10 | 309 (53) | 192 (80) | |||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




